Abstract
Objective
Changes in angiogenesis have been shown to contribute to cognitive decline and dementia. We aimed to identify angiogenesis blood markers associated with cognitive performance in older adults with mild cognitive impairment (MCI), remitted major depressive disorder (rMDD), or both (rMDD + MCI) who are at risk for dementia.
Method
We analyzed data from participants with MCI, rMDD, or rMDD + MCI in the Prevention of Alzheimer's Dementia with Cognitive Remediation plus Transcranial Direct Current Stimulation in Mild Cognitive Impairment and Depression study. Elastic net regression was used to select variables associated with cognitive performance among 19 angiogenesis markers and 6 covariates. Linear regressions were used to determine which of the selected angiogenesis markers were associated with cognitive performance, controlling for the selected covariates. Significant angiogenesis markers were independently analyzed without other angiogenesis markers, controlling for covariates, with subgroup analyses in those with and without rMDD.
Results
Angiogenin was the only selected marker associated with cognitive performance (
Conclusion
The association of angiogenin with cognitive performance highlights a potentially novel biological pathway that could influence cognition in older adults at risk for dementia. Angiogenin may protect against cognitive decline, opening new avenues for innovative preventive, or therapeutic interventions.
Plain Language Summary
This study aimed to identify blood markers related to angiogenesis (the process of new blood vessel formation) that could predict cognitive function (memory and thinking abilities), in older adults at risk for dementia. Angiogenesis has been linked to dementia, and understanding its associated biological factors may help in predicting cognitive decline, particularly in individuals who are at risk for dementia. Participants included adults over 50 years of age with cognitive impairment and/or a history of depression. We used the elastic net regression model to select eight out of 19 angiogenesis markers while considering five out of the six other factors (covariates) that might influence cognition, such as age, education level, apolipoprotein E genotype, diagnosis, and cardiovascular health. Among the markers studied, angiogenin, a marker that can increase angiogenesis, stood out as the only one associated with cognitive function. When considering other factors, higher levels of angiogenin were linked to better cognition. This association was present even when angiogenin was examined on its own, independent of other angiogenesis markers. Angiogenin might play a protective role in cognition in older adults at risk for dementia. Overall, the findings highlight angiogenin as a potential biomarker for cognitive health in aging adults, who were at risk with early signs of cognitive impairment and a history of depression. This suggests that angiogenin could be a target for future research into treatments aimed at preventing or slowing cognitive decline in vulnerable populations.
Introduction
Both mild cognitive impairment (MCI) and remitted major depressive disorder (rMDD) contribute to an increased risk of dementia in older adults. 1 MCI is often considered a transitional stage between normal aging and dementia, where cognitive decline becomes apparent, with no or minimal functional deficits. About 32% of those diagnosed with MCI progress to dementia within 2 years. 2 A history of major depressive disorder (MDD), even when it occurs early in life or in midlife, has been recognized as an independent risk factor for developing Alzheimer's disease (AD) and related dementias (ADRD). 3 Late-life depression has been associated with a twofold to fivefold increased risk of dementia. 4 The deleterious effect of depression may persist even after remission of MDD (rMDD, defined as being free or almost free of depressive symptoms for at least 2 months 5 ): older individuals with rMDD have a three-time higher risk of cognitive decline compared to non-rMDD controls. 6 With lifetime prevalence rates of MDD reaching 20% 7 and the prevalence of MCI in older adults being over 15%, 8 rMDD and MCI define large groups of individuals at risk for dementia. Thus, it is important to identify underlying biological factors that may contribute to brain health and cognitive performance in older individuals with MCI or rMDD.
Angiogenesis, the process of forming new blood vessels from preexisting vessels, 9 may play a critical role in the etiology of ADRD, as vascular dysfunction has been suggested as one of the earliest pathological events during the development of AD from normal aging. 10 Angiogenesis is a complex and regulated process involving different growth factors, cell adhesion molecules, and metabolites in endothelial cells. 11 In animal studies, the effects of cognitive-enhancing treatments appear to be associated with angiogenesis signaling.10,12,13 Some clinical studies have examined human angiogenesis at different stages of cognitive impairment; however, their findings have been inconsistent. For example, a study reported a similar direction of change for both plasma angiogenin (a potent angiogenic stimulator 11 ) and tissue inhibitor of matrix metalloproteinase-4 (which has been associated with indirectly inhibiting angiogenesis through matrix metalloproteinases). 14 Both of these proteins were also associated with cognitive performance assessed by the Mini-Mental State Examination (MMSE) and AD severity. 14 In another study, MMSE scores were correlated with angiogenin in AD. 15 For pre-dementia stages, plasma vascular endothelial growth factor (VEGF), a key pro-angiogenic marker, 11 was reported to be higher than controls in persons with MCI, but not in those with AD. 16 In contrast, serum VEGF has been found to be lower in individuals with amnestic MCI or AD than in controls, 17 suggesting the need for more research in individuals with MCI specifically. Angiogenesis is also of interest for cognition in rMDD: an association was found in individuals with MDD between worse cognitive performance and increased immature circulating endothelial progenitor cell counts, a sign of pro-angiogenic state. 18
Taken in toto, there is a lack of consistent clinical findings on the relationship between angiogenesis markers and cognitive performance, particularly in the pre-dementia stages, which could provide information on potential underlying biological factors. Examining blood angiogenesis markers in these populations may suggest new targets that may potentially prevent further cognitive decline or monitor the disease progression. In this context, we aimed to identify angiogenesis markers associated with cognitive performance in the pre-dementia stages of MCI, rMDD, and rMDD + MCI and to assess whether there were differences among these three diagnostic groups.
Methods
Participants
We included participants from the randomized clinical trial, Prevention of Alzheimer's Dementia with Cognitive Remediation plus Transcranial Direct Current Stimulation in Mild Cognitive Impairment and Depression (PACt-MD; NCT02386670),19,20 that had baseline blood angiogenesis marker data with no history of cancer or concomitant cancer, as angiogenesis marker levels are affected by cancer. 21 Details of the PACt-MD trial, inclusion criteria, and assessments have been described previously.19,20 Briefly, all participants provided written informed consent as approved by the local Research Ethics Board and completed detailed baseline clinical and cognitive assessments between January 2015 and August 2018. Participants were classified as rMDD (with normal cognition), MCI, or rMDD + MCI. 19 For the MCI group (without rMDD), the main eligibility criteria were age ≥ 60, meeting criteria for a diagnosis of mild neurocognitive disorder based on the Diagnostic Statistical Manual of Mental Disorders Fifth Edition (DSM-5) 5 and a Montgomery–Åsberg Depression Rating Scale (MADRS) 22 score ≤ 10. For the rMDD with or without MCI groups, the main eligibility criteria were age ≥ 65, a diagnosis of rMDD based on the Structured Clinical Interview for DSM (fourth edition, text revised), 23 a MADRS 22 score ≤ 10, and either meeting (rMDD with MCI) or not meeting (rMDD without MCI) the criteria for a diagnosis of mild neurocognitive disorder based on DSM-5. 5
Plasma Levels of Angiogenesis Markers
At baseline, blood was collected in EDTA tubes via venipuncture with fasting not required. The plasma was separated immediately after collection and aliquots were stored at −80°C. Using this blood, the following 19 markers that have been known to contribute to angiogenesis11,24–26 and have been associated with dementia or cognition in literature were measured simultaneously using the Luminex® LX200 platform (Luminex Corp., Austin, TX, USA) and a customized multiplex immunoassay from R&D/Biotechne (Minneapolis, MN, USA): angiogenin, 15 angiopoietin-1, 27 angiopoietin-2, 28 endothelial cell-specific molecule-1 (endocan), 29 endostatin, 30 e-selectin, 31 hepatocyte growth factor (HGF), 32 interleukin-33 (IL-33), 33 suppression of tumorigenicity-2 (ST2), 33 oncostatin-M, 34 platelet-derived growth factor-AA (PDGF-AA), 35 platelet-derived growth factor AB (PDGF-AB), 36 placental growth factor (PlGF), 37 tissue inhibitor of metalloproteinases-1 (TIMP-1), 38 vascular cell adhesion molecule-1 (VCAM-1), 39 VEGF,16,40 VEGF receptor-1(VEGFR1),40,41 VEGF receptor-2 (VEGFR2), 40 and VEGF receptor-3 (VEGFR3). 41 Laboratory analyses followed the manufacturer's instructions. Briefly, protein standards were homogenized and serially diluted, and protein-specific capture beads were prepared. For each test well, 25 μL of the bead mix was combined with 25 μL of sample or standard and incubated for 16–18 h. After incubation, plates were washed, and 25 μL of detection antibody was added for 2 h at room temperature. Following this, 50 μL of streptavidin was added, and plates were incubated for 30 additional minutes. After a final wash, wells were resuspended in 100 μL of fluid drive buffer and read on the LX200® system with Exponent® software (Luminex Corp.). Data were analyzed using Belysa® 1.2.2 software (Merck Millipore, Darmstadt, Germany), with results expressed in pg/mL. The detection limits were as follows: angiogenin (3.17 pg/mL), angiopoietin-1 (9.43 pg/mL), angiopoietin-2 (17.1 pg/mL), endocan/ESM-1 (1.08 pg/mL), HGF (1.0 pg/mL), IL-33 (1.8 pg/mL), oncostatin M/OSM (44.3 pg/mL), PDGF-AA (0.747 pg/mL), PlGF (1.9 pg/mL), e-selectin/CD62E (18.8 pg/mL), TIMP-1 (3.42 pg/mL), VEGFR1/Flt-1 (5.68 pg/mL), VEGFR2/KDR/Flk-1 (7.4 pg/mL), VEGFR3/Flt-4 (2.7 pg/mL), endothelin-1 (1.31 pg/mL), PDGF-AB (3.28 pg/mL), ST2/IL-33R (29.9 pg/mL), VCAM-1/CD106 (238 pg/mL), and VEGF-A (0.99 pg/mL).
ApoE Genotyping
Apolipoprotein E (
Cognitive Measures
A composite cognitive score was created using results from a comprehensive neuropsychological battery, as described previously.
19
Briefly,
Statistical Analysis
Data were analyzed using R version 4.4.1.
Based on visual inspection, angiogenesis marker levels (pg/mL) were not normally distributed, so they were log-transformed to better approximate a normal distribution; as five of the markers (counts) had non-detectable values with zeros: IL-33 (98), VEGF (80), PIGF (8), PDGF-AB (1), and VEGFR3 (1), these values were imputed using half of the minimum value
43
for each marker prior to transformation. A priori covariates included age,44,45 diagnosis,
46
gender,
47
highest level of education,
47
Results
Participant Characteristics
Out of the 418 randomized PACt-MD participants,
20
308 participants who had blood angiogenesis marker data and no indication of cancer were included in this analysis. This subset of participants did not differ from the overall sample in age (
Demographic, clinical, and cognitive characteristics are summarized in Table 1. Age, gender, education, comorbidities, vascular risk factors, and
Demographic, Clinical, and Cognitive Characteristics.
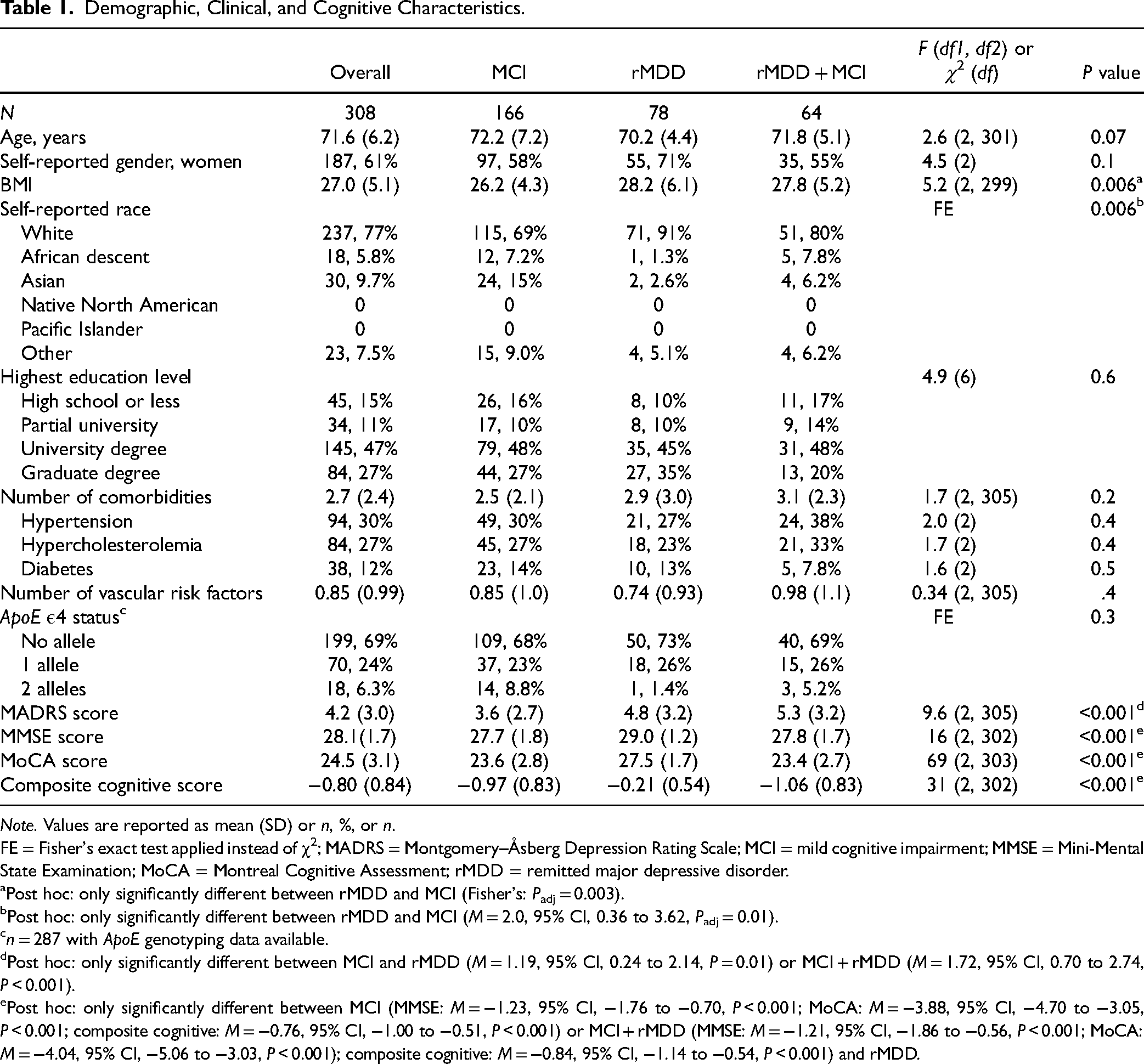
FE = Fisher's exact test applied instead of χ2; MADRS = Montgomery–Åsberg Depression Rating Scale; MCI = mild cognitive impairment; MMSE = Mini-Mental State Examination; MoCA = Montreal Cognitive Assessment; rMDD = remitted major depressive disorder.
Post hoc: only significantly different between rMDD and MCI (Fisher's:
Post hoc: only significantly different between rMDD and MCI (
Post hoc: only significantly different between MCI and rMDD (
Post hoc: only significantly different between MCI (MMSE:
Angiogenesis Markers and Cognitive Performance
The log-transformed values of angiogenesis markers are shown in Figure S1. Among the 19 angiogenesis markers and the six covariates for cognitive performance, elastic net regression identified the following relevant variables (non-zero coefficients): angiogenin (0.13), endocan (0.04), e-selectin (−0.02), IL-33 (0.01), oncostatin M (−0.28), PDGF-AB (0.04), ST2 (−0.15), TIMP-1 (0.10), VCAM-1 (−0.02), age (−0.03), education (0.18), diagnosis (0.03),
These nine angiogenesis markers and five covariates were analyzed in a linear model for cognitive performance (
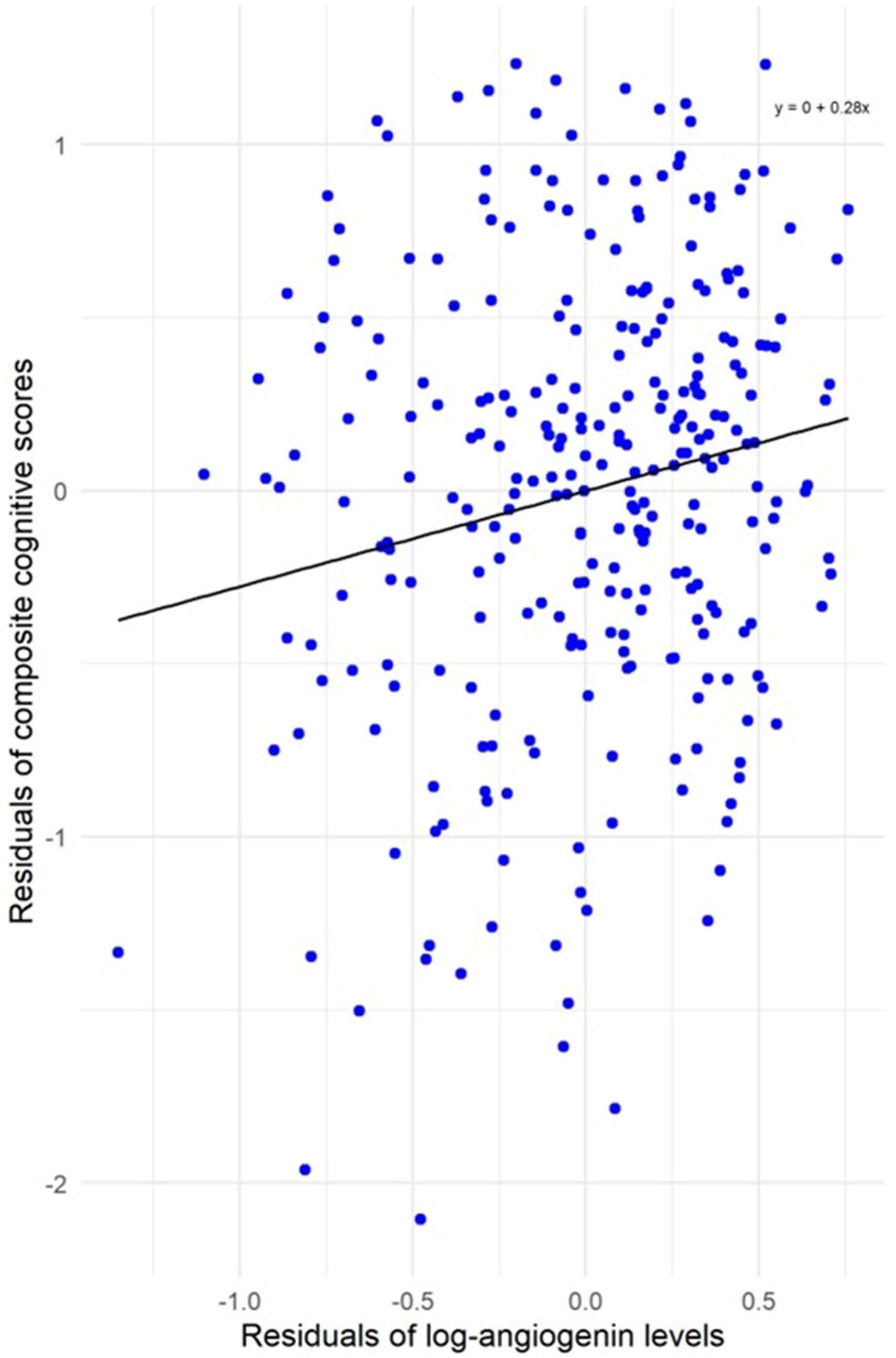
Partial regression plot of log angiogenin levels (residuals from its regression against other angiogenesis markers and covariates) and composite cognitive scores (residuals from its regression against other angiogenesis markers and covariates).
Multiple Linear Regression With Relevant Variables for Cognition.
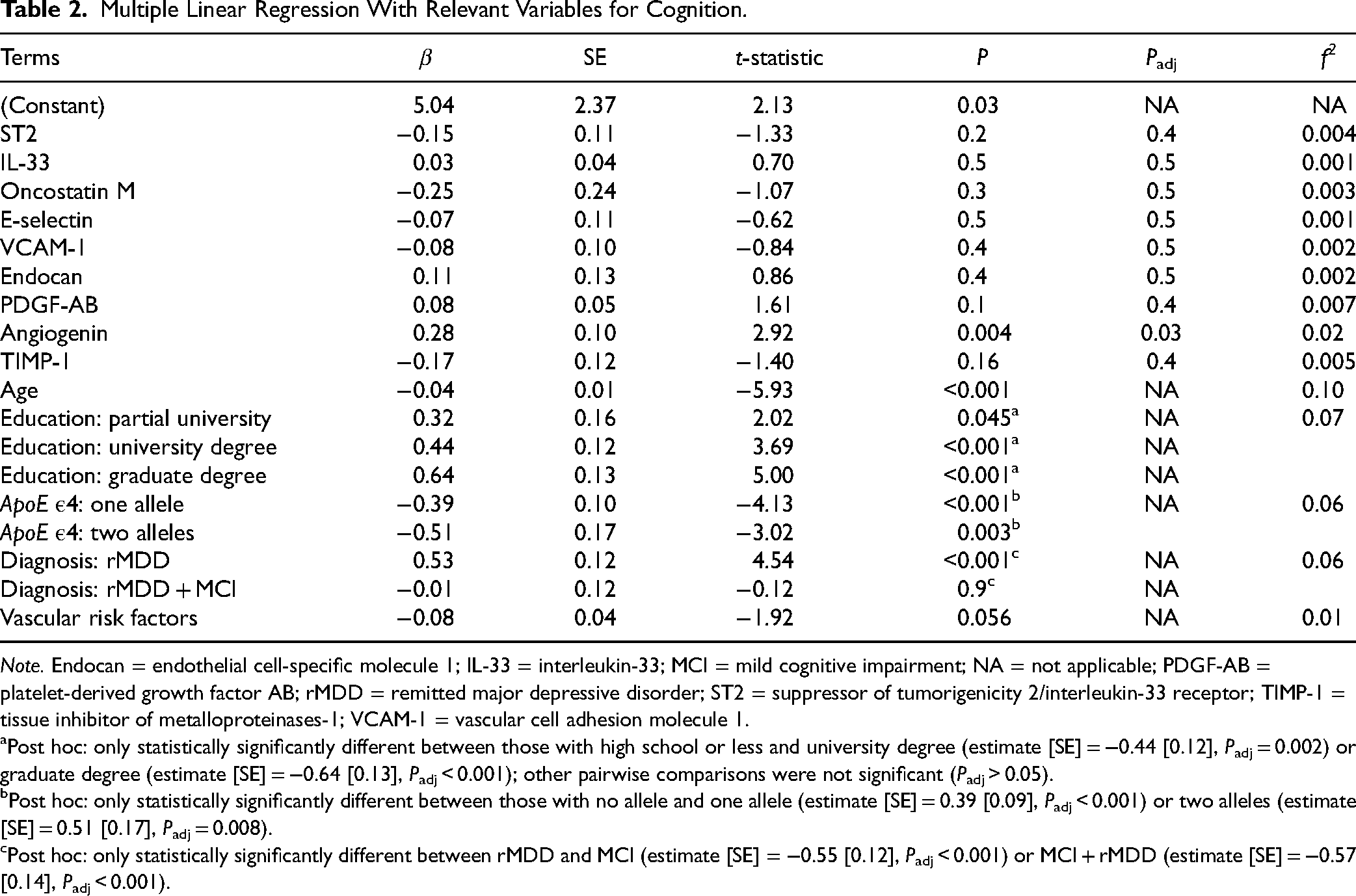
Post hoc: only statistically significantly different between those with high school or less and university degree (estimate [SE] = −0.44 [0.12],
Post hoc: only statistically significantly different between those with no allele and one allele (estimate [SE] = 0.39 [0.09],
Post hoc: only statistically significantly different between rMDD and MCI (estimate [SE] = −0.55 [0.12],
Angiogenin and Cognitive Performance
In the model excluding other angiogenesis markers and controlling for the same covariates (
Multiple Linear Regression With Only Angiogenin and Covariates for Cognitive Performance.
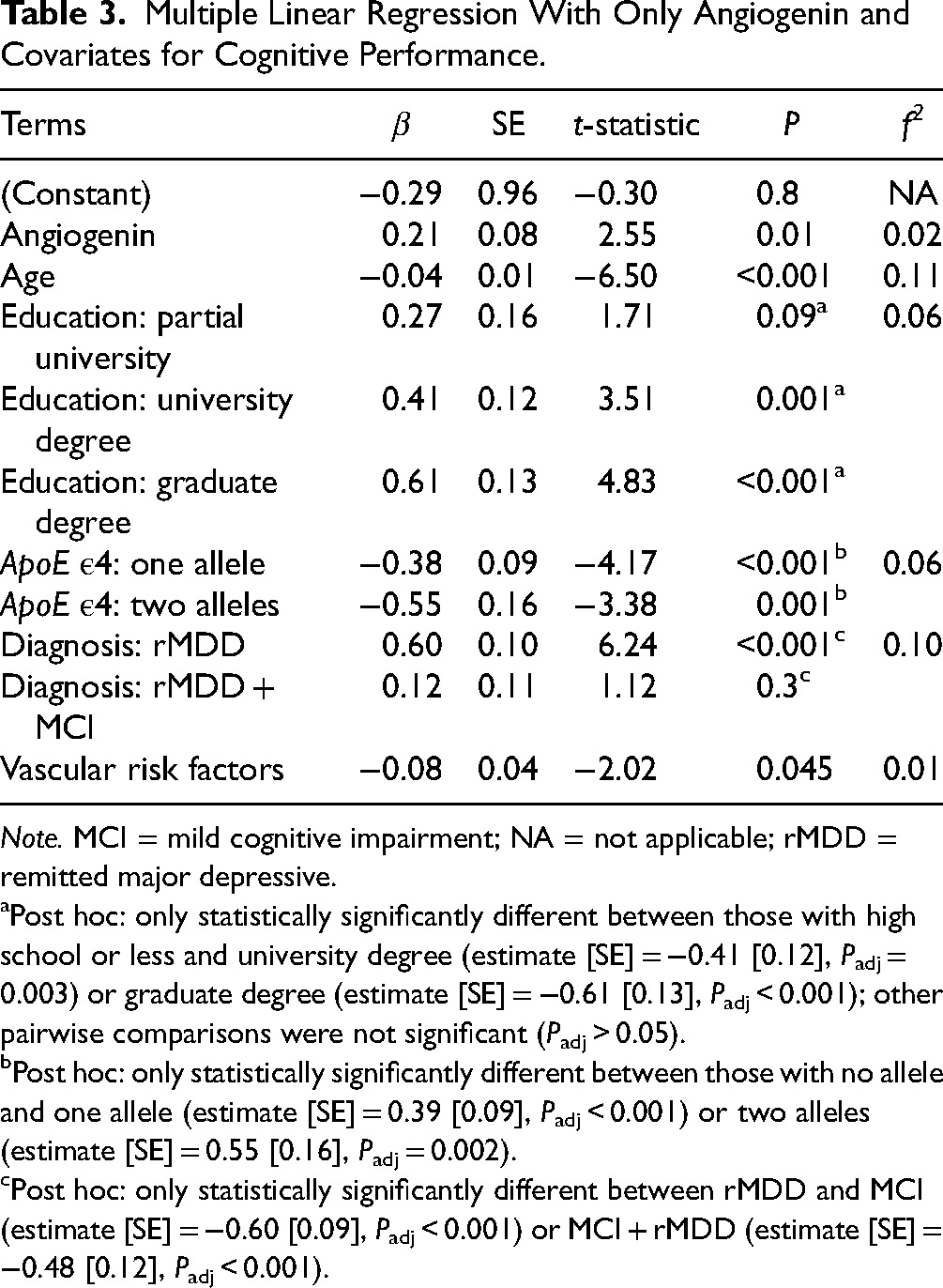
Post hoc: only statistically significantly different between those with high school or less and university degree (estimate [SE] = −0.41 [0.12],
Post hoc: only statistically significantly different between those with no allele and one allele (estimate [SE] = 0.39 [0.09],
Post hoc: only statistically significantly different between rMDD and MCI (estimate [SE] = −0.60 [0.09],
Angiogenin and Different Clinical Characteristics
No differences in angiogenin were observed between gender (
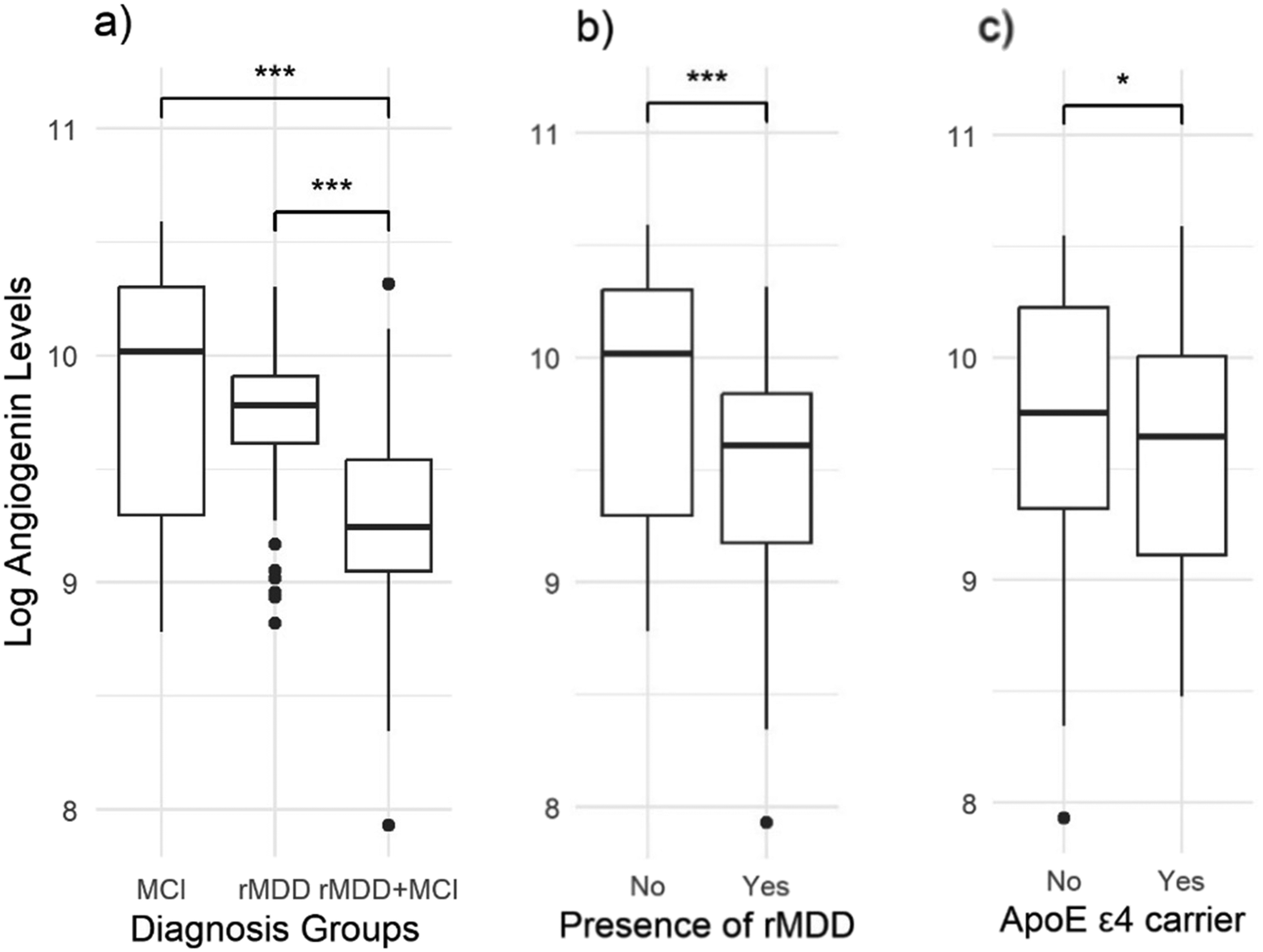
Boxplots of log angiogenesis levels in subgroups by (a) diagnosis, (b) presence of rMDD, (c)
Discussion
In this study of older adults with MCI, rMDD, or both, angiogenin, a pro-angiogenic factor was associated with cognitive performance when controlling for well-established covariates including age,44,45 diagnosis,
46
education,
47
Previous literature has not found a consistent relationship between angiogenin and cognitive performance. For example, the relationship between angiogenin and cognition has been assessed in AD with conflicting findings;15,52 with a small study (AD:
The exact mechanisms linking angiogenesis and cognition have not been elucidated. In AD models, amyloid beta can trigger angiogenesis leading to hypervascularity and blood–brain barrier leakiness. Yet, hyper-induction of angiogenesis could ameliorate cognitive deficits and reduce AD-related neuropaththologies. 11 In support of this potentially beneficial effect, a deficiency in angiogenesis has been linked to cognitive impairment.58,59 Angiogenesis in AD has been thought to occur as a compensatory mechanism for impaired cerebral blood flow,60,61 further supporting angiogenesis’ suggested neuroprotective effects. Angiogenin belongs to the ribonuclease A superfamily, and it works with actin on endothelial and smooth muscle cell surfaces to stimulate plasmin catalytic activity, promoting angiogenesis. 11 By complexing with fibulin-1, it can enhance the proliferation, migration, and stabilization of the new vessel walls. 11 It contributes to vascularization in both healthy and disease states. 11 Thus, the positive association found between angiogenin and cognitive performance in our study aligns with the existing literature in animal studies connecting angiogenesis to neuroprotection.62,63
Angiogenesis has been linked to neurogenesis in animal and in vitro models. For example, in ischemic mice, increased angiogenin in the neurogenic subventricular zone due to physical exercise was associated with the migration of derived neuroblasts, linking angiogenin to the post-stroke neurogenesis processes. 64 Similarly, human angiogenin has been associated with neurite extension and pathfinding and survival of motor neurons. 65 In vitro, human angiogenin has been shown to prevent cell death due to oxidative stress and play a role in cell migration. 66 Vascular disease, while interacting with other pathological processes, such as inflammation and angiogenesis, 61 can lead to changes in brain function associated with depression, such as dorsal hypometabolism and ventral hypermetabolism; 67 this connection is consistent with the relationship between cognitive performance and angiogenin found in the subgroup with rMDD. On the other hand, older men with higher serum levels of endostatin, an anti-angiogenic protein, had greater odds of depression in a large cross-sectional study; they predicted that the probability of depression could be increased with serum endostatin levels to as high as 20–25% in older men, 68 suggesting that the relationship between angiogenic balance and depression is complex. Angiogenesis is a multi-faceted process potentially contributing to both depression and cognition.
This analysis has some limitations. First and foremost, it has a cross-sectional design, which prevents us to make any causal interpretation of the results. Future longitudinal studies will need to assess the trajectory of angiogenesis marker and cognition over time, which will provide more information to better understand the relationship between angiogenesis and cognition. While the MADRS scores for all participants were lower than 10, the mean scores were 1–2 points higher in the rMDD and rMDD + MCI groups than in the MCI group. While we cannot rule out the residual effects of depressive symptoms on cognition,
69
the MADRS scores of the two rMDD groups were typically within the non-depressed range (i.e., 0–6) and a 2-point difference is well below the 6-point minimal clinically important difference for the MADRS.
70
Due to the small sample size, the rMDD and rMDD + MCI groups could not be compared in subgroup analysis; the
Conclusion
Our results provide some preliminary support for angiogenin as a biomarker associated with cognitive impairment. If replicated, these results suggest that interventions that promote angiogenesis may offer a novel approach to prevent or slow cognitive decline in individuals with rMDD, MCI, or both who are at risk for dementia. Further research is needed to explore angiogenesis longitudinally, and specifically angiogenin, to assess its potential as a therapeutic target for cognitive decline.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437251337627 - Supplemental material for Blood Angiogenesis Markers and Cognition in Older Adults at Risk for Dementia: Marqueurs sanguins de l’angiogenèse et cognition chez les personnes âgées à risque de démence
Supplemental material, sj-docx-1-cpa-10.1177_07067437251337627 for Blood Angiogenesis Markers and Cognition in Older Adults at Risk for Dementia: Marqueurs sanguins de l’angiogenèse et cognition chez les personnes âgées à risque de démence by Bing Xin Song, PhD, Erica Vieira, PhD, Damien Gallagher, MD, Breno S. Diniz, MD, PhD, Corinne E. Fischer, MD, Alastair J. Flint, MD, Nathan Herrmann, MD, Linda Mah, MD, MHSc, Benoit H. Mulsant, MD, MS, Tarek K. Rajji, MD, Clement Ma, PhD, Krista L. Lanctôt, PhD and on behalf of the PACt-MD Study Group in The Canadian Journal of Psychiatry
Footnotes
Acknowledgments
PACt-MD Study Group: Benoit H. Mulsant, MD, MS (principal investigator); Tarek K. Rajji, MD (co-PI; site PI, Centre for Addiction and Mental Health, lead, neurostimulation and neurophysiology); Nathan Herrmann, MD (co-PI; site PI, Sunnybrook Health Sciences Centre), Bruce G. Pollock, MD, PhD (co-PI); Daniel Blumberger, MD, MSc (co-investigator); Christopher Bowie, PhD, C.Psych (co-investigator; lead, cognitive remediation and neuropsychology); Meryl Butters, PhD (xonsultant, neuropsychology); Corinne Fischer, MD (co-investigator; site PI, St. Michael's Hospital); Alastair Flint, MD (co-investigator; site PI, University Health Network); Angela Golas, MD (lead, CSF); Ariel Graff, MD (lead, neurochemistry); James L. Kennedy, MD (lead, genetics); Sanjeev Kumar, MD (co-investigator); Krista Lanctot, PhD, (site PI, Sunnybrook Health Sciences Centre), Lillian Lourenco, MPH (study co-manager), Linda Mah, MD, MHS (co-investigator; site PI, Baycrest Health Sciences); Shima Ovaysikia, MA (study co-manager); Mark Rapoport, MD (co-investigator); Kevin Thorpe, MSc (biostatistician); Nicolaas P.L.G. Verhoeff, MD, PhD (co-investigator); Aristotle Voineskos, MD, PhD (lead, neuroimaging). We also acknowledge the contribution of Kathleen Bingham, MD; Lina Chiuccariello, PhD; Tiffany Chow, MD; Pallavi Dham, MD; Breno Diniz, MD, PhD; Dielle Miranda, Carmela Tartaglia, MD; and the PACt-MD Research Staff. Genotyping was performed by the CAMH Biobank and Molecular Core Facility (previously CAMH Microarray Facility) and funded by the Discovery Fund.
Data Availability
The data generated and analyzed in the current study is available from the corresponding author and the PACt-MD Study Group upon request.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KLL has received consulting fees from Boehringer Ingelheim, Bristol Meyers Squibb, Eisai Co. Ltd., Exciva, Ironshore Pharmaceuticals, H Lundbeck A/S, Novo Nordisk, Otsuka and Praxis Therapeutics
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has been made possible by Brain Canada through the Canada Brain Research Fund, with the financial support of Health Canada and the Chagnon Family and the CAMH Discovery Fund. BXS has received funding from the Ontario Graduate Scholarship and CAN-TAP-TALENT Doctoral Award supported by the Canadian Institutes of Health Research (CIHR). KLL holds the Bernick Chair in Geriatric Psychopharmacology and has received grants from CIHR (PJT 183584, PJ2 179753, CNA 163902, PJT153079, PJ8 159823, and PJ8 169698), Alzheimer's Drug Discovery Foundation (GC-201808-2016354), Alzheimer's Association (PTCG-20–700751, PTC18-543823), and Weston Brain Institute (CT190002). CEF has received funding from Novo Nordisk, ADDF, NIH, NIA, CCNA, CIHR, and the Hilary and Galen Weston Foundation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
