Abstract
There is a variable and often prolonged period between the occurrence of the first cognitive symptoms and the development of the full clinical syndrome of dementia [1], [2]. The focus of this study is this predementia period, which has been referred to by various terms such as ‘amnestic mild cognitive impairment’ (aMCI) and ‘cognitive impairment no dementia’ (CIND). Amnestic MCI has the most currency and is defined as performance at 1.5 SD below age norms on a memory test, subjective memory complaints, intact general cognition and not meeting criteria for dementia [3]. Its reliance on subjective and objective memory impairment alone, however, limits it to possibly being a precursor of Alzheimer's disease but not the other dementias.
Until now, CIND has not been strictly defined using a neuropsychological cut-point and has been characterized by difficulty with memory and/or other areas of cognition not sufficiently severe to meet criteria for dementia [4]. ‘Cognitive impairment no dementia’ differs from other predementia definitions, such as aMCI, as it is not defined to be exclusively a prelude to Alzheimer's disease (AD), but includes individuals with vascular brain changes and other known causes of cognitive impairment [5]. The high prevalence of cerebrovascular disease among the elderly and the contribution of vascular factors in AD and dementia with Lewy bodies (DLB) [6], [7] supports use of this broader definitions when studying predementia in a population [8].
These pre-dementia syndromes have been the subject of intense research recently as earlier diagnosis may allow for treatment which could delay the conversion to dementia. Considerable controversy exists whether it is possible to define a pre-dementia syndrome, and if so, which definition to use [9].
A number of studies have reported the prevalence of CIND in older populations. The Italian Longitudinal Study of Ageing reported a prevalence of CIND of 10.7% in subjects between 65 and 84 years [10]. The Canadian Study of Health and Ageing reported a prevalence of 16.8% in subjects above 65 years [5]. In the Kungsholmen project, a prevalence of CIND of 14.7% in subjects over 75 years was detected using the Mini-Mental State Exam (MMSE) [11]. The Indianapolis Longitudinal Study of Ageing found CIND in 23.4% of subjects above 65 years [12]. In the Sydney Older Persons Study, preclinical syndromes (cognitive impairment with Alzheimer, vascular, Parkinsonian or mixed features) were found to affect 55.7% of subjects over 75 years [13].
The narrower criteria for aMCI have resulted in lower reported population rates. The Personnes Agees QUID (PAQUID) reported a prevalence of 2.8% [14], the Kupio population study found a prevalence of 5.3% [15], the North Karelia Project reported a prevalence of 6.1% [16] and the Eugeria Project a prevalence of 3.2% [9].
Annual conversion rates in longitudinal population studies of CIND to dementia range from 5.8% to 9.4% [13], [17] and from aMCI to AD ranging from 3.7 to 8.3% [9], [14]. Reported predictors of CIND include older age [10], lower education [10], previous stroke [10], heart failure [10], apolipoprotein E e4 [18] and raised cholesterol [19].
This study aimed to determine the prevalence of CIND and aMCI in a population survey in Sydney, Australia and to investigate risk factors for CIND. We focused on people aged 70–79 to balance the higher prevalence with age than would be found in a younger sample against complicating comorbidity if an even older population were to be studied. Potential predictive factors for CIND investigated included age, gender, education, occupation and cardiovascular risk factors. We define CIND as a test performance 1.5 SDs (or more) below age-adjusted and education-adjusted (where available) norms on any cognitive measure in the test battery. This broad definition will allow detection of individuals at risk of developing different dementia types.
Method
The study was approved by the research ethics committee of the South-eastern Sydney Area Health Service. The potential sample comprised 600 people between 70 and 79 years, residing in the Sutherland Shire, Sydney, whose names were randomly selected from the electoral roll, a public document maintained by the Australian Electoral Commission. The Sutherland Shire was chosen as a geographically convenient area which has a high proportion of elderly residents who are mostly native English speakers. A letter describing the study was sent to the first 400 potential subjects, that is those with an available contact telephone number and who were not known to be institutionalized. The letter contained a refusal slip and a prepaid return envelope, with which recipients could decline participation in the study. Those who did not return their refusal slip were contacted by telephone by a researcher (RE or LFL) and screened to determine consent and eligibility. If verbal informed consent was obtained, subjects were interviewed by telephone. The interview included questions on demographics, subjective ratings of memory and of physical and emotional health and medical conditions. Subjects were included if they were aged 70–79 years, living in the community, had an informant with whom they had at least weekly contact and had adequate eyesight, hearing and English language ability sufficient for standard psychometric testing. Subjects were excluded if they had a previous diagnosis of dementia, bipolar disorder, Parkinson's disease, multiple sclerosis, motor neurone disease, a developmental disability, central nervous system inflammation, progressive malignancy, psychotic symptoms or a diagnosis of schizophrenia or an alcohol or drug dependency. Subjects were also excluded if they had any medical or psychological conditions that may have prevented them from completing assessments. If subjects withdrew consent at any point, no further contact was made.
Psychometric assessment
One in three eligible subjects were randomly selected by computer for psychometric assessment. A trained research psychologist (RE) performed assessments in the subjects' homes. Written informed consent was obtained from all subjects who underwent psychometric testing and from their informants. Detailed demographics, current medication, alcohol usage and vascular risk factors were recorded. The Mini-Mental State Examination (MMSE) [20] was administered as a screening tool; subjects scoring ≤ 24 were considered to have possible dementia and were excluded.
The neuropsychological battery consisted of tests assessing five cognitive domains: (i) attention/speed of information processing, Trail Making Test A [21]; (ii) memory and learning, Logical Memory, Wechsler Memory Scale–III [22], Visual Reproduction, Wechsler Memory Scale-Revised [23], Rey Auditory Verbal Learning Test (RAVLT) [24], [25]; (iii) language, the Boston Naming Test [26], Category Fluency [27]; (iv) visuo-spatial/construction, Block Design, Wechsler Adult Intelligence Scale III [28]; and executive functioning, Controlled Oral Word Association Test (COWAT) [29], Trail Making Test B [21]. The National Adult Reading Test (NART) [30] was administered to estimate premorbid general intellectual functioning. All tests have well documented validity, reliability and age-corrected normative scores for the elderly. Subjects also completed the Geriatric Depression Scale (GDS) [31] and the ‘state’ section of the State-Trait Anxiety Inventory (STAI-S) [32].
The tests selected for the study sample examined a broad range of cognitive functions and have sufficient sensitivity to detect the early changes in cognition associated with Alzheimer's disease, vascular dementia and dementia with Lewy bodies, which are the most common dementia diagnoses in this age group [33–36]. Reduced performance on these cognitive measures is associated with an increased risk of developing dementia as shown by retrospective and prospective longitudinal studies [37–44].
Informants completed the short Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE) regarding the subject, a 16-item valid screening instrument for dementia [45]. The informant rates cognitive and functional changes over the last 10 years on a 5-point scale from 1 (much improved) to 5 (much worse).
Definitions
‘Cognitive impairment no dementia’ was defined as performance 1.5 SDs below age-corrected (and education where available) norms on at least one test in the neuropsychological battery and MMSE >24. Amnestic mild cognitive impairment was defined as performance 1.5 SDs below age-corrected norms on at least one memory test and memory self-rated as fair, poor or bad and MMSE >24. All other subjects were considered to be cognitively normal.
Data analysis
Data were analyzed using SPSS V 10.0 (SPSS Inc, 2000). Differences between subjects with CIND and those with normal cognition were examined on variables previously found to distinguish between cognitively impaired or demented subjects and non-impaired subjects. These differences were analyzed using t, χ2 and Mann–Whitney U-tests, as appropriate. Then age, gender and variables found to differ between groups were entered into a logistic regression with CIND status as the dependent variable, using forward conditional selection. All p-values quoted are two-tailed; the p-value required for significance after Bonferroni correction for 17 multiple comparisons between CIND and cognitively normal subjects is 0.002.
Results
Details of recruitment, screening and assessment are presented in Fig. 1. Eligible subjects had a mean age of 74.47 years (± 2.71) and education of 10.58 years (± 2.80). Sixty-eight (51.9%) were male. There were no significant differences between those who were eligible and those excluded on gender (χ2 = 2.744, p = 0.140), age (t = −1.894, p = 0.060) and years of education (t = 0.832, p = 0.407). Forty-three (32.8%) subjects were randomly selected for detailed neuropsychological assessment but one subject withdrew consent prior to the assessment.
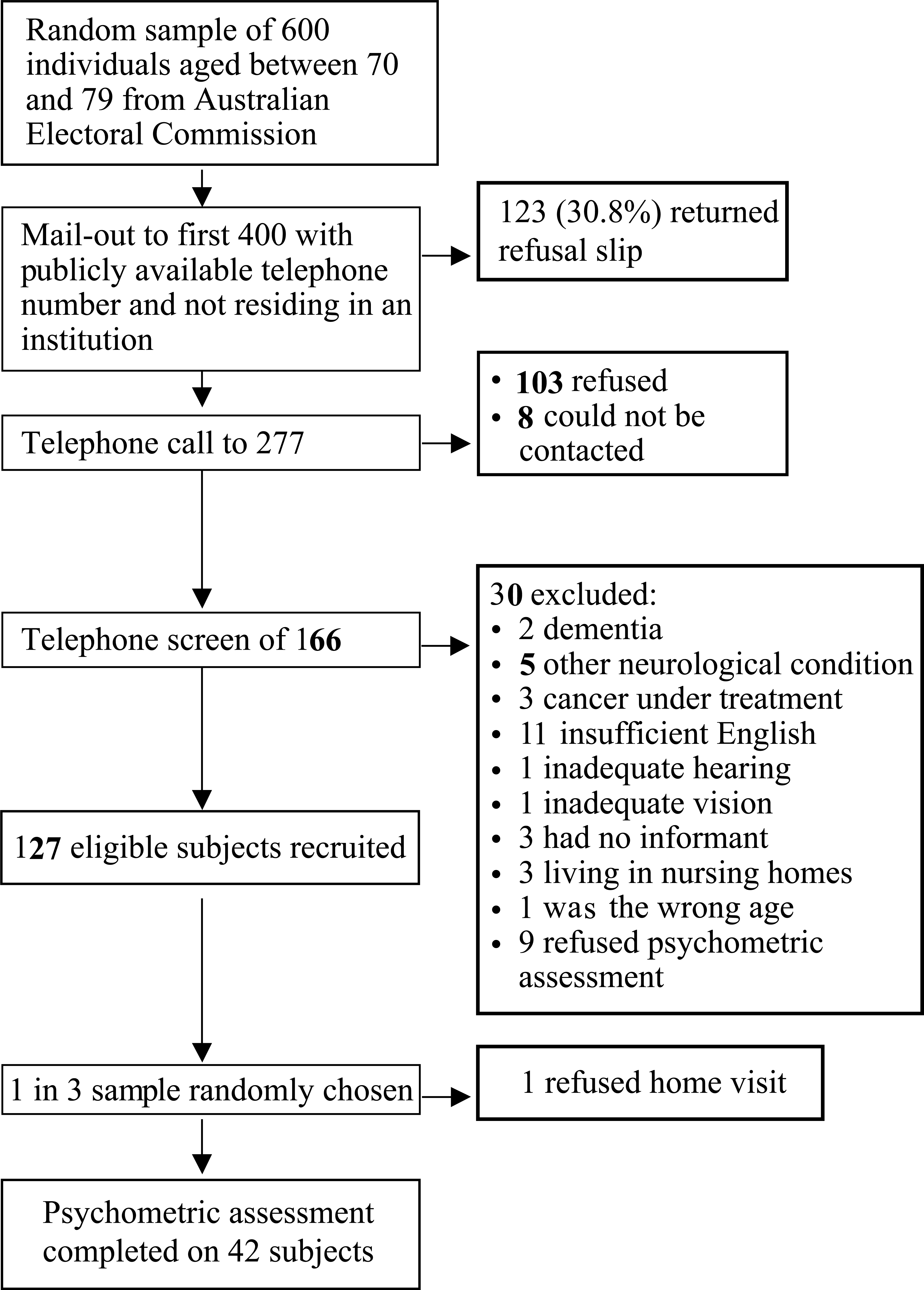
Subject recruitment, screening and assessment
Of the 42 subjects who underwent psychometric testing, 25 were male (59.5%), their mean age was 74.38 years (± 2.47) and they had 11.26 years (± 3.20) of education. There were no significant differences between subjects who received an in-home assessment and the remaining eligible subjects who were not assessed, on age (t = −0.245 p = 0.807), years of education (t = 1.968, p = 0.051) and gender (χ2 = 1.436, p = 0.264).
One (2.4%) had possible dementia, 27 (64.3%) were cognitively normal and 14 (33.3%) subjects met our criteria for CIND, of whom four (9.8%) also met criteria for aMCI.
The MMSE scores for subjects with CIND (mean =28.00 ± 1.52) were significantly lower than those for cognitively normal subjects (mean = 29.04 ± 0.90) (t = −2.350 p = 0.030). There was a significant difference between the IQCODE scores for the CIND group (mean = 3.281 ± 0.34) and cognitively normal subjects (mean = 2.969 ± 0.47) (t = 2.135, p = 0.040). IQCODEs were not available for three subjects; all subjects had a suitable informant prior to home assessment but one informant withdrew consent and two questionnaires were not returned. There were no significant differences between the GDS scores of CIND (mean = 3.07 ± 3.02) and cognitively normal subjects (mean = 1.84 ± 1.43) (t = 1.436, p = 0.170). Similarly, the STAI scores of CIND (36.79 ± 7.22) and cognitively normal subjects (33.15 ± 8.13) were not significantly different (t = 1.399, p = 0.170). After Bonferroni correction for multiple comparisons, none of these differences remained significant.
Table 1 reports a comparison of the CIND and cognitively normal groups on demographic variables and potential risk factors. Subjects with CIND were older, had lower ranking occupations and were less likely to be currently working than those classified as cognitively normal. Logistic regression analysis was conducted with ‘age’, ‘gender’, ‘occupation ranking’ and ‘still working’ as the independent variables. ‘Still working’ was significantly related to CIND status (β = 1.928, p = 0.046). Since current unemployment could be a risk factor for or consequence of CIND, a second logistic regression was conducted with only ‘age’ ‘gender’ and ‘occupation ranking’ as the independent variables. The significant predictors of CIND status were ‘age’ (β = 0.704, p = 0.040) and ‘occupation ranking’ (β = 0.535, p = 0.024).
Demographic data and possible risk factors for cognitive impairment no dementia (CIND) and cognitively normal subjects (n = 41)
Table 2 presents the number of subjects meeting different criteria for cognitive impairment and self-reported memory functioning.
Number and percentage of subjects meeting different criteria for cognitive impairment without dementia (n = 42)
Discussion
We have presented a pilot investigation to determine the prevalence of CIND and aMCI in a representative community sample of elderly individuals. The noteworthy finding from our study is the very high prevalence of CIND (33.3%), being higher than that reported in most other published studies, but lower than in the Sydney Older Persons Study, possibly because they recruited older subjects [13]. This is most likely due to the broad and inclusive definition of CIND we purposely used in our study. The rates are very different if other, and narrower, definitions are used. If we apply the criteria of self-report of fair or poorer memory and performance 1.5 below norms on a memory test (the criterion for aMCI by Petersen et al. [46]), only 9.5% of subjects would be classified as impaired. Half the subjects meeting criteria for CIND were impaired in a cognitive domain other than memory.
A second explanation for the discrepant prevalence rates is the variable age range in the different studies, although this only partially accounts for the variance. The Italian Longitudinal Study on Ageing reported population-adjusted prevalence of CIND of 8.3% in subjects 70–74 years and 16.4% in subjects 75–79 years [47]. The Indianapolis site from the Indianapolis-Ibadan Dementia Study reported prevalence of CIND in community-dwelling subjects of 19.2% in subjects 65–74 and of 27.6% in subjects 75–84 years [48]. Our CIND rate is lower than that reported in the Sydney Older Persons study: 55.7% in subjects over 75 years [13].
Another possible explanation for the high prevalence rate in our study is selection bias, since only one in three potential subjects agreed and were eligible to participate. We consider this to be unlikely as those not included did not differ from those who participated on some key variables, and we know from previous research that individuals who are impaired are less likely to participate in such studies [49]. As regards the participation rate, other larger studies have obtained much higher participation rates: Sydney Older Persons Study 73%, Italian Longitudinal Study of Ageing 83%, Indianapolis Study of Health and Ageing 85%. This project was described as specifically studying memory and cognition in the elderly and was not subsumed under a study of ageing as is the case with some other epidemiological studies and was not associated with any prior community publicity campaign.
The use of a broad and inclusive definition of CIND has some merit. Since there is no one universally accepted definition of a predementia impairment of cognition and the various definitions remain to be validated, a broad definition is a reasonable starting point for a refining process to form the basis for a larger and longitudinal study. Moreover, recent studies examining neuropsychological predictors of dementia provide support for broadening the criteria to include deficits in cognitive domains other than memory [44], [50].
Our classification of CIND and non-cognitively impaired controls is supported by differences (uncorrected) between groups on MMSE score, informantrated change (the IQCODE) and self-rated emotional health and memory. Differences on cognition were not associated with differences in physical health (indicated by both self-rated physical health, a good summary of physical health [51], and number of medications), nor depression or state anxiety during assessment.
Our study was not adequately powered to examine a large number of correlates of CIND. The cross-sectional correlates and possible determinants of CIND identified in the regression analyses (older age, lower ranked occupations and not currently working) are consistent with the literature on predementia and dementia. It is well established that cognitive impairment increases with age [52]. Our CIND sample had an equal proportion of men and women, but the normal group (63% vs. 37%) had more men. This is noteworthy, even though the differences were not statistically significant in our small sample. It is generally accepted that women have higher rates of Alzheimer's disease [53] and the published literature suggests that female gender is a risk factor for cognitive decline in old age [54].
Our data show that subjects with CIND were more likely to be either not working or in lower previous best occupation. The cross-sectional nature of our study does not permit the determination of the direction of this relationship, that is, whether lower occupation predisposes an individual to CIND or the latter leads to early retirement or decrement in occupational status. We favour the former explanation based on the already considerable evidence in the literature [55]. The importance of the status ranking of a subject's ‘best’ occupation may reflect a range of influential factors that are associated with high socioeconomic status such as better diet [56] or health care [57], greater intellectual activity [58] and spare-time activity [59] that may have a protective effect on cognition.
In conclusion, we report a high rate of CIND in a Sydney community sample of 70–79-year-olds. Despite the limitations, the study points to an important health problem that will need to be confronted in the near future. Our results suggest that subjective memory and emotional complaints and informant reports of change may be useful clinically in detecting CIND. Large cohort longitudinal studies are necessary to establish the true extent, pattern of progression and more importantly, specific predictors of conversion to different dementia types in subjects with CIND. A practical and valid definition of mild cognitive impairment is then likely to emerge. This is necessary if the prevention of dementia is our goal.
Footnotes
Acknowledgements
We thank all participants for their time.
