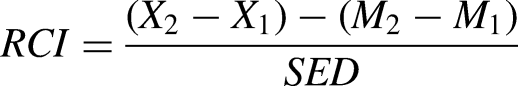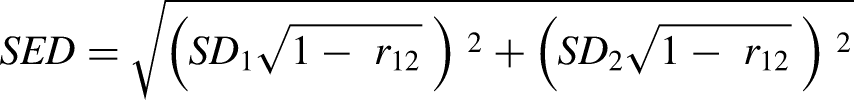Abstract
Background
Late-life depression (LLD) is often accompanied by cognitive impairment, which may persist despite antidepressant treatment. Repetitive transcranial magnetic stimulation (rTMS) is an efficacious treatment for depression, with potential benefits on cognitive functioning. However, research on cognitive effects is inconclusive, relatively sparse in LLD, and predominantly focused on group-level cognitive changes. This study aimed to explore individual-level cognitive changes following rTMS treatment in patients with LLD.
Method
Data were analyzed from 153 patients with LLD from the FOUR-D study (ClinicalTrials.gov identifier: NCT02998580) who received bilateral standard rTMS or theta burst stimulation (TBS) targeting the dorsolateral prefrontal cortex (DLPFC). Cognitive function was assessed pre- and post-treatment using measures of executive function, information processing speed, and learning and memory. Reliable change indices, adjusted for practice effects and test-retest reliability, were employed to evaluate individual-level cognitive changes. Chi-square tests examined if proportions of cognitive improvers differed from expected proportions.
Results
Cognitive performance from baseline to end of treatment remained stable for most patients. Reliably improved performance was observed in 0.0% to 20.0% of participants across cognitive measures, while worsened performance was observed in 0.0% to 2.7%. A small but significant proportion (20.0%) of participants showed improvement in verbal learning.
Conclusions
Bilateral standard rTMS or TBS of the DLPFC in LLD yielded no substantial cognitive enhancing effects, although a small proportion showed improved verbal learning after treatment. Importantly, both interventions were cognitively safe with relatively stable performance across time. Future research is needed to explore approaches to enhance the cognitive benefits of standard rTMS and TBS in patients with LLD.
Introduction
Late-life depression (LLD) is often accompanied by cognitive impairment, particularly in the domains of executive functioning, information processing speed, and learning and memory. 1 These impairments significantly reduce quality of life and increase the risk of neurodegenerative disorders such as Alzheimer's disease and related dementias. 2 Antidepressant medications are widely used in treating LLD, yet tend to be only partially beneficial for many patients who still have significant depressive symptoms. 3 Importantly, cognitive impairments often persist even when there is improvement in depressive symptoms and severity.4,5 While a recent meta-analysis suggested potential improvements in learning and memory, mediated by alleviation of depressive symptoms, 6 the overall effects of antidepressants on cognitive function in LLD remain controversial. 7 This highlights the need for interventions that can effectively address both depressive and cognitive symptoms in LLD.
Repetitive transcranial magnetic stimulation (rTMS) of the dorsolateral prefrontal cortex (DLPFC) is a well-established treatment for patients with treatment-resistant depression (TRD). The standard approach involves high-frequency rTMS (10 Hz) over the left DLPFC, and/or low-frequency rTMS (1 Hz) over the right DLPFC. 8 Another form of rTMS, theta burst stimulation (TBS), replicates natural brain oscillatory activity through patterned pulse trains, and is delivered as intermittent (iTBS) or continuous (cTBS) bursts over the left or right DLPFC, respectively. 8 Evidence indicates that TBS is comparably effective to standard rTMS for depression. 9 While high-frequency rTMS and iTBS are thought to exert excitatory effects on neuronal activity, low-frequency rTMS and cTBS are believed to be inhibitory, although these effects show substantial inter-individual variability. 10 In contrast to electroconvulsive therapy (ECT), rTMS is recognized for its cognitive safety 11 and potential to improve certain cognitive functions in TRD. 12 The DLPFC is a key node within the cognitive control network, 13 which is thought to subserve executive functions, such as response inhibition, set-shifting, and planning. rTMS uses focused magnetic field pulses to modify neural activity at the stimulation site and can induce structural and functional connectivity changes in distributed brain networks, 14 including the cognitive control network. 15 By targeting the DLPFC, rTMS may have the potential to improve executive function in patients with depression.
Cognitive improvements following rTMS have been observed in younger adults with depression across various domains including learning and memory,16–18 attention, 19 executive function19–24 and psychomotor processing speed.22,25,26 However, findings have been inconsistent and large meta-analyses have not demonstrated robust cognitive effects, 27 with only modest effects observed in specific cognitive tasks. 28 Research on cognitive effects of rTMS in LLD is limited, despite potential differences within this age group. Some rTMS studies in LLD have reported improvements in attention and immediate memory, 29 executive function,30,31 verbal fluency and visuospatial abilities, 32 and global cognitive function. 33 However, other studies found no significant changes in cognitive function after treatment.34–36
Studies investigating cognitive changes following rTMS have primarily relied on group-level analyses, which may mask changes at the individual (i.e., patient) level. Practice-adjusted reliable change indices (RCIPE) can be used to detect improved or worsened cognitive performance in individuals beyond measurement error and practice effects, 37 which can address this limitation. In this context, we investigated individual-level cognitive changes associated with a course of bilateral standard rTMS or TBS to the DLPFC in patients with LLD using an RCIPE methodology. We hypothesized that bilateral rTMS/TBS treatment would enhance cognitive function, particularly executive function. Additionally, we explored potential predictors of cognitive improvement and whether cognitive changes varied across different baseline cognitive subgroups.
Methods
Participants
Cognitive data was analyzed from the FOUR-D study, a randomized, non-inferiority trial comparing bilateral TBS to bilateral standard rTMS in patients with LLD (ClinicalTrials.gov identifier: NCT02998580). 9 The study received approval from the CAMH Research Ethics Board and enrolled outpatients age 60 and older with Major Depressive Disorder. Detailed inclusion and exclusion criteria have been previously reported. 9 In brief, participants were required to show non-response to at least one antidepressant trial at an adequate dosage and duration, or intolerance to at least two antidepressants, and have a Montgomery Åsberg Depression Rating Scale (MADRS) 38 total score ≥ 18. Prior to their participation in the study, all participants provided written informed consent.
Cognitive Assessment
Trained personnel conducted the following cognitive tests at baseline and post-treatment:
The NIH Toolbox Flanker Inhibitory Control and Attention Test assessed executive function, specifically selective attention and inhibitory control.
39
Participants were instructed to respond to a central stimulus while ignoring similar surrounding stimuli. Scores were computed based on accuracy and reaction times and then converted into age-adjusted standard scores (M = 100, SD = 15). The Delis-Kaplan Executive Function System (D-KEFS) Color-Word Interference test (CWI)
40
was conducted including four conditions: The two baseline conditions Color Naming (Condition 1) and Word Reading (Condition 2) were used as measures of information processing speed. Executive functioning was evaluated using the Interference condition (Condition 3), measuring response inhibition, and the Inhibition/Switching condition (Condition 4), measuring both inhibition and cognitive flexibility. Raw scores (i.e., time taken to complete the task) for all four conditions were then converted into age-adjusted scaled scores (M = 10, SD = 3). The California Verbal Learning Test – second edition (CVLT-II)
41
was used to assess verbal learning and memory. Participants were presented with a list of 16 semantically related words over 5 learning trials, with verbal learning scores derived from the total number of correctly recalled words over these trials. Subsequently, a short-delay free recall assessed the number of correctly recalled words after an interference list. This was followed by a long-delay free recall after a 20-min interval. Raw CVLT-II scores were converted into normative T- and z-scores adjusted for age and reported sex.
Additional cognitive measures included the Montreal Cognitive Assessment (MoCA version 2),
42
which evaluated overall global cognitive function. The Test of Premorbid Functioning (TOPF),
43
a word reading test, was used to estimate premorbid IQ at baseline.
Clinical Assessment
Clinical assessments included the MADRS to assess depressive symptom severity, the Cumulative Illness Rating Scale-Geriatrics (CIRS-G) 44 to evaluate burden of physical illness, and the Brief Symptom Inventory (BSI)–Anxiety Subscale (BSI-Anxiety). 45 Additional clinical variables encompassed age at first onset of depression, duration of current depressive episode, details regarding current medications, and adequacy of current and previous antidepressant medications as quantified using the Antidepressant Treatment History Form (ATHF). 46
Procedures
Participants were randomized to receive either standard sequential bilateral rTMS starting with right-sided 1 Hz stimulation (10 min for 600 pulses), followed by left-sided 10 Hz stimulation (37.5 min for 3000 pulses, 4 s on, 26 s off) or sequential bilateral TBS starting with right-sided cTBS (40 s for 600 pulses in 50 Hz triplet bursts, repeated at 5 Hz) followed by left-sided iTBS (3 min 9 s for 600 pulses in 50 Hz triplet bursts, repeated at 5 Hz, 2 s on, 8 s off). All treatments were administered at 120% resting motor threshold to the DLPFC and delivered 5 days a week over 4 weeks, totaling 20 treatment sessions. If remission was not achieved after the first 20 sessions, 10 additional sessions over the following 2 weeks were offered.
Statistical Analysis
This analysis included all participants with available cognitive data at both time points for at least one cognitive measure to allow for the calculation of change scores. Baseline demographic and clinical characteristics of the sample were summarized using means and standard deviations (SD) for continuous variables, and frequencies for categorical variables. To account for measurement error and practice effects inherent in repeated neuropsychological assessments, we used RCIPE
37
because of its demonstrated usefulness in assessing cognitive change in older adults compared to alternative change score methods.
47
RCIPE were computed by subtracting the mean practice effect (derived from a normative sample of the respective test) from the individuals’ observed discrepancy score (time 2−time 1), divided by the standard error of the difference (SED):
For cognitive tests with a significant proportion of participants showing a “reliable improvement,” further exploratory analyses examined demographic and clinical predictors of cognitive improvement using binary logistic regression with backward elimination method based on Akaike's Information Criterion. Baseline performance of the respective test was entered as a covariate, and potential predictive variables included sex, age, years of education, estimated premorbid IQ, baseline MADRS, baseline BSI-A, ATHF score, duration of current depressive episode, benzodiazepine use (y/n), treatment allocation and number of rTMS treatments delivered. To assess potential associations with effects of rTMS on depressive symptoms, change in MADRS total score (post-treatment MADRS score−baseline MADRS score), response (reduction ≥ 50% from baseline in MADRS score), and remission (MADRS score ≤ 10 at end of treatment) were included as additional predictors. Additionally, Spearman's rank correlation between the RCI z-score and MADRS change score was calculated.
To investigate how cognitive changes vary depending on an individual's initial cognitive profile, we examined reliable changes across three distinct baseline cognitive subgroups. These subgroups were identified in a previous study using a k-means clustering approach based on baseline cognitive test scores from the current sample. 53 Specifically, the cluster analysis revealed 3 distinct cognitive subgroups labeled as “Cognitively Intact” (n = 84), “Cognitively Diminished” (n = 25), and “Impaired Memory” (n = 39). We examined whether RCI z-scores differed significantly across these three cognitive subgroups using ANOVA. We also used chi-square tests to examine whether proportions of participants showing reliable improvement, no change, or decline differed across the cognitive subgroups. A false discovery rate (FDR) correction was applied to account for multiple tests across cognitive measures and p-values < 0.05 were considered statistically significant. All statistical analyses were conducted using R (version 4.1.2).
Results
Out of 172 randomized participants, 8 participants did not complete treatment, and another 11 participants did not undergo post-treatment cognitive assessment, resulting in the inclusion of 153 participants in this analysis who were assessed at both time points on at least one cognitive measure (mean [SD] age, 66.7 [6.0] years; 56% female, Table 1). There were no significant differences in demographic or clinical variables between participants lost to follow-up and those who underwent both assessments. However, participants who did not complete treatment or undergo post-treatment cognitive assessment showed significantly worse performance at baseline on the CVLT-II Total Trials 1–5 (t(19.81) = 3.68, p = 0.001), the CVLT-II Short-Delay Free Recall (t(19.05) = 2.73, p = 0.013), and the CVLT-II Long-Delay Free Recall (t(18.44) = 3.12, p = 0.006).
Baseline Demographic and Clinical Characteristics of the Study Sample.
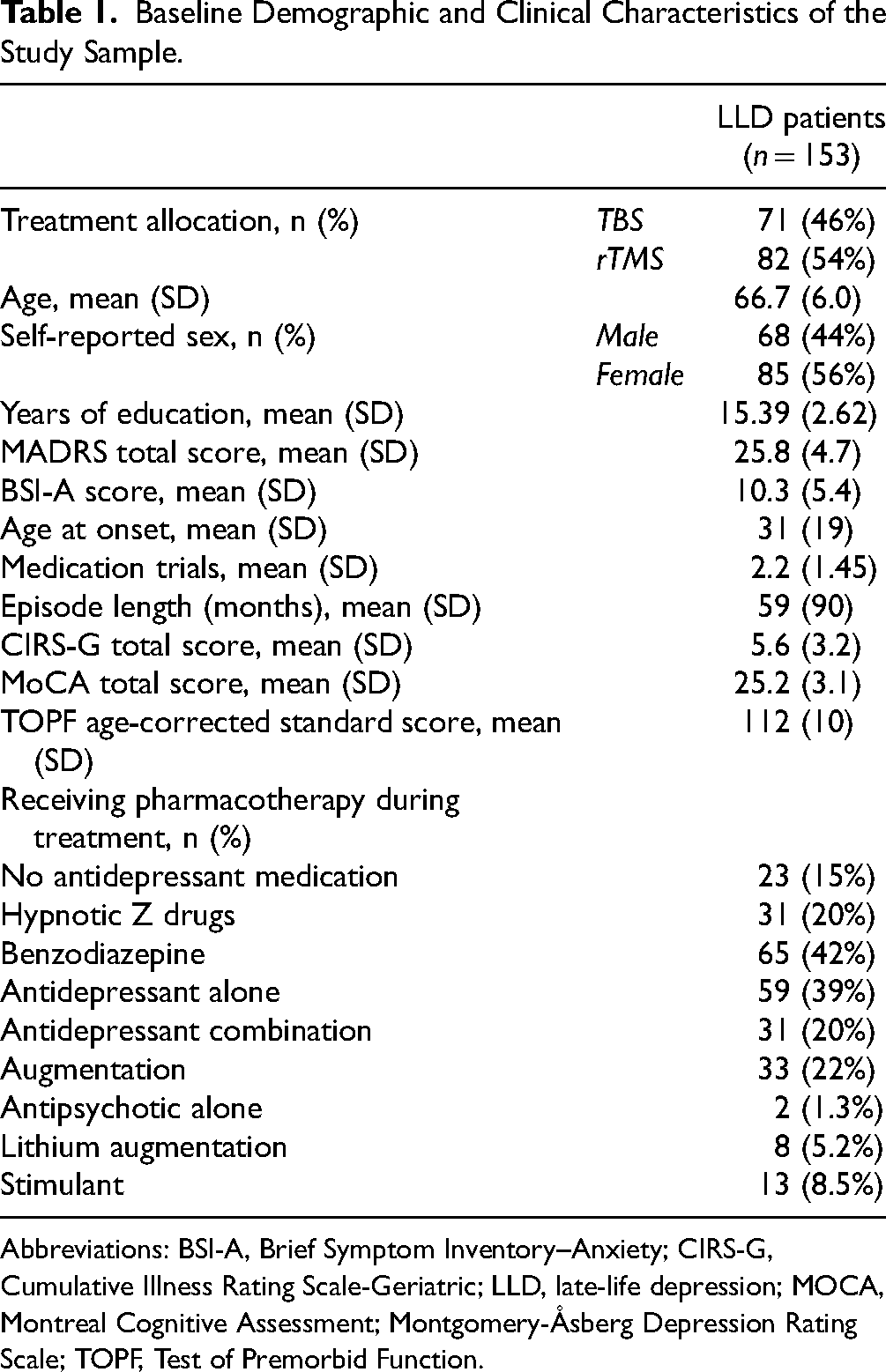
Abbreviations: BSI-A, Brief Symptom Inventory–Anxiety; CIRS-G, Cumulative Illness Rating Scale-Geriatric; LLD, late-life depression; MOCA, Montreal Cognitive Assessment; Montgomery-Åsberg Depression Rating Scale; TOPF, Test of Premorbid Function.
Practice-Adjusted Reliable Change Index
Most participants showed no reliable change in performance on cognitive measures (Table 2). The percentage of participants showing a reliable improvement ranged across cognitive measures from 0.0% (on the DKEFS-CWI Inhibition Task) to 20.0% (on the CVLT-II Total Trials 1–5). Overall, 25.5% (39/153) of participants showed a reliable improvement in at least one cognitive measure. Conversely, the percentage of participants showing a reliable worsened performance ranged from 0.0% (on the DKEFS-CWI Inhibition Task) to 2.7% (on the DKEFS-CWI Color Task) (Table 2). Distributions of RCI z-scores for each cognitive measure are depicted in Figure 1. Notably, for most cognitive measures, the percentage of participants exhibiting a reliable increase or decrease in performance fell within the expected 5% range based on the distribution of RCIs. This was exceeded only by the percentage of participants who improved in verbal learning on the CVLT-II Total Trials 1–5, where 30 out of 150 participants (20.0%) had reliably improved performance. This difference between the observed percentage with the expected percentage was significant (χ2(2) = 48.4, p < 0.001).
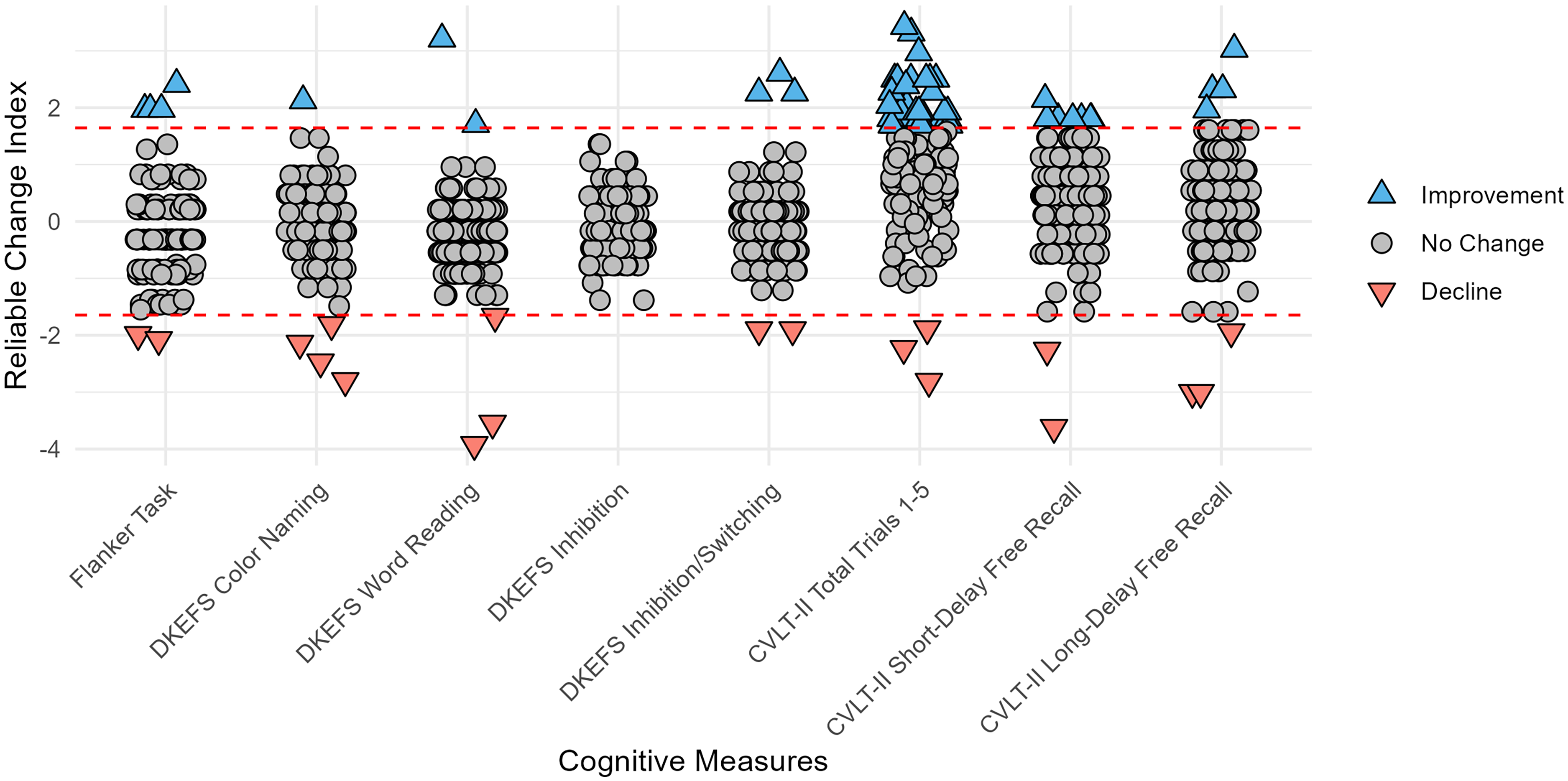
Distribution of RCIPE z-scores. Dashed lines indicate 90% confidence interval. Abbreviations: CVLT-II, California Verbal Learning Test – second edition; DKEFS, Delis-Kaplan Executive Function System.
Cognitive Scores at Baseline and End of Treatment and Frequency (Percentage) of Participants Showing Reliable Cognitive Changes Using the RCIPE Method With a 90% Confidence Interval.
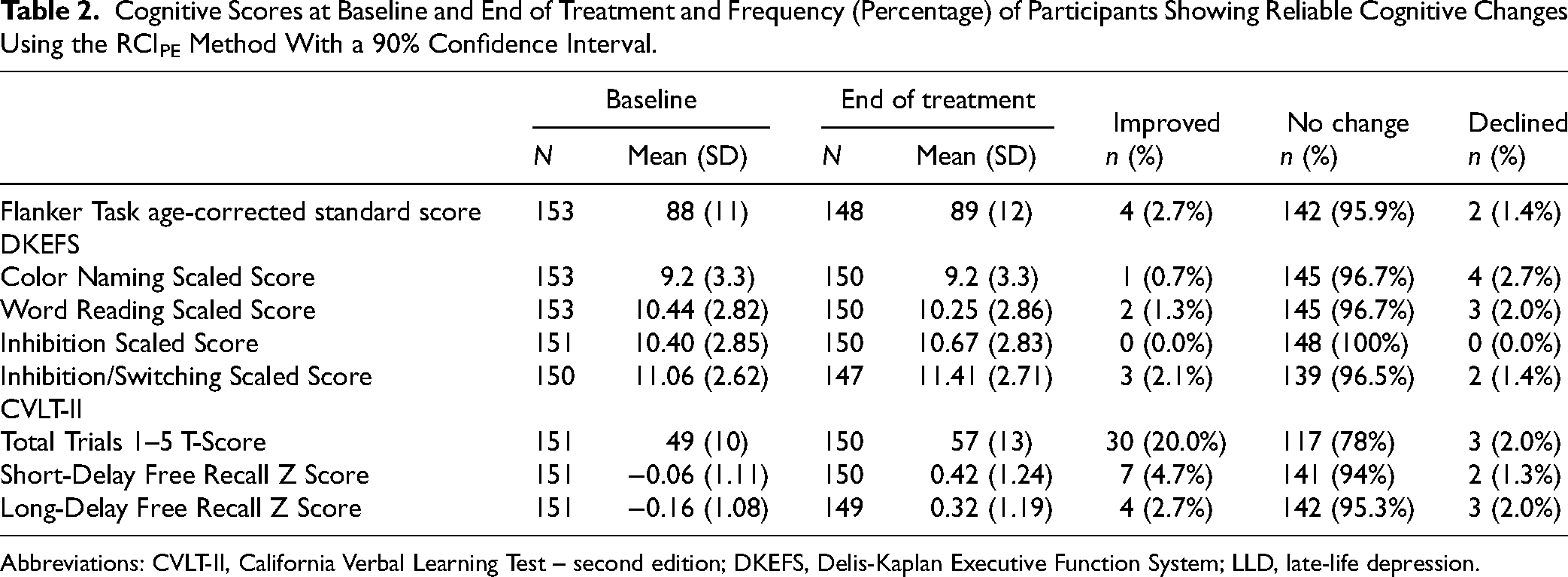
Abbreviations: CVLT-II, California Verbal Learning Test – second edition; DKEFS, Delis-Kaplan Executive Function System; LLD, late-life depression.
Exploratory Prediction Analysis
A binary logistic regression analysis using backward elimination was conducted to determine significant predictors of a reliable improvement on the CVLT-II Total Trials 1–5. The final model retained three significant predictors. A lower baseline performance on this measure (p = 0.01, OR = 0.94, CI 0.90–0.98), a higher premorbid IQ (p = 0.01, OR = 1.07, CI 1.02–1.13), and being a responder to rTMS/TBS treatment (p = 0.04, OR = 2.48, CI 1.05–6.00) were associated with higher odds of showing a reliable improvement on the CVLT-II Total Trials 1–5. No other variable significantly predicted reliable improvement CVLT-II Total Trials 1–5, including treatment allocation (i.e., standard rTMS vs TBS). There was a weak but significant correlation between the RCI z-scores of the CVLT-II Total Trials 1–5 and the MADRS change score (ρ = −0.23, p = 0.005).
Baseline Cognitive Subgroups
After FDR-correction, no significant differences in RCI z-scores were found across the three baseline cognitive subgroups in any cognitive measure, nor in percentages of participants showing reliable improvement, no change, or worsened performance (Table 3).
Mean RCI z-Scores and Proportions of Participants Showing Reliable Improvement, No Change, or Decline for Three Baseline Cognitive Subgroups.
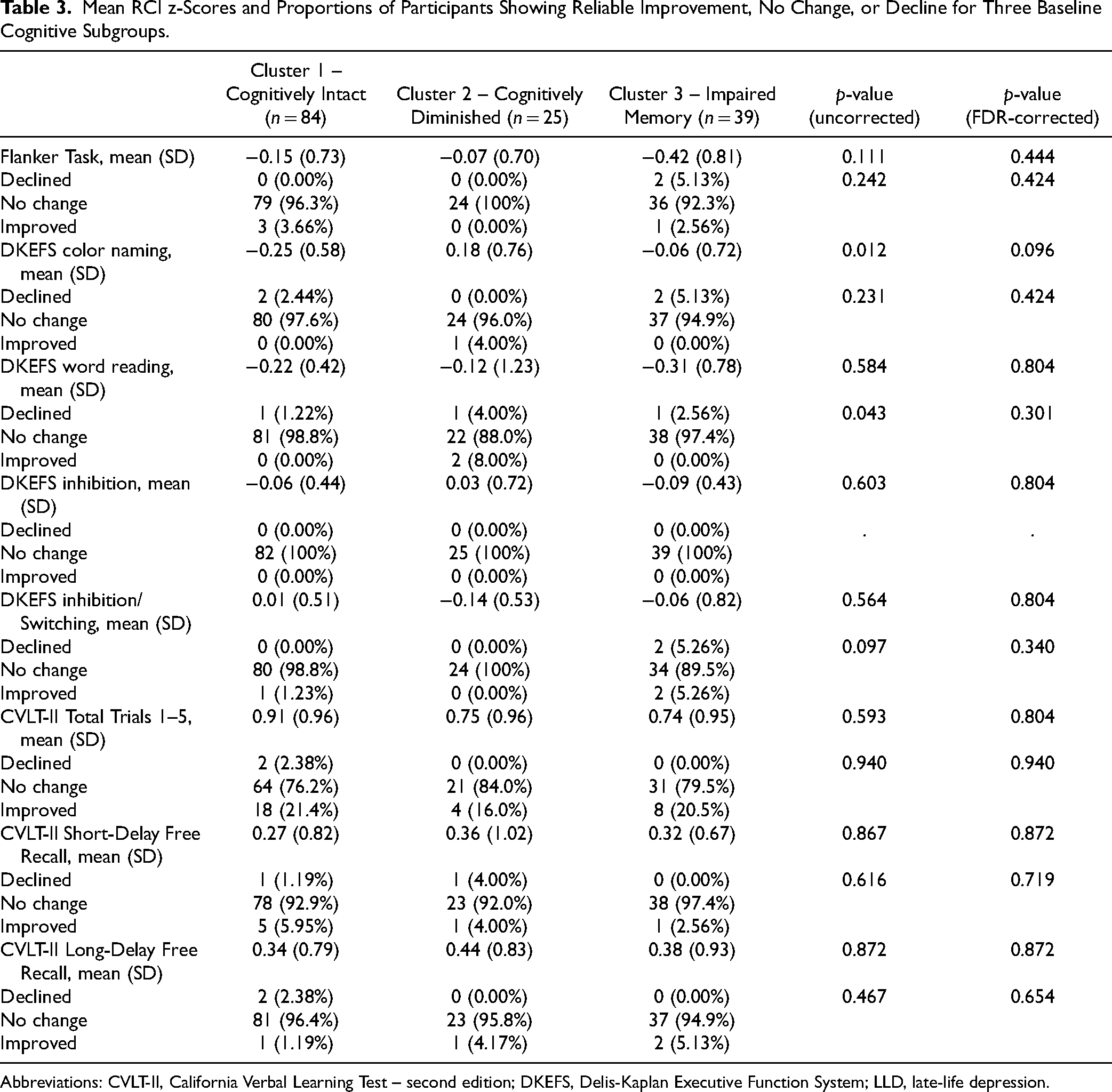
Abbreviations: CVLT-II, California Verbal Learning Test – second edition; DKEFS, Delis-Kaplan Executive Function System; LLD, late-life depression.
Discussion
Despite clear evidence for the therapeutic effects of standard rTMS and TBS in LLD, their effects on cognition remain unclear. In this study, we investigated the cognitive outcomes from the FOUR-D trial, a recent non-inferiority trial that compared bilateral standard rTMS with bilateral TBS to the DLPFC in patients with LLD. RCIPE were used to effectively control for measurement error and practice effects of repeated neuropsychological testing. Overall, our findings indicated that bilateral standard rTMS or TBS to the DLPFC had limited effects on cognition across the domains of executive function and information processing speed. A small but statistically significant proportion of participants (i.e., 20%) experienced reliable improvements in verbal learning, which was associated with a higher estimated premorbid IQ and being a responder to rTMS treatment.
The literature on adults across the lifespan reveal mixed previous results regarding cognitive improvements after rTMS. For example, a transdiagnostic meta-analysis including depression reported positive rTMS effects on working memory, but found no effects on other cognitive domains. 54 Another review of 31 RCTs reported positive effects on executive function and attention in some adults with depression. 55 In the context of TRD, a review of 22 studies found a trend supporting that rTMS improves cognitive performance, although some studies reported negative findings. 12 In line with our results, a meta-analysis of 30 RCTs of various neuropsychiatric disorders concluded that, overall, rTMS has no robust cognitive enhancing effects. 27 However, a subsequent meta-analysis from the same authors focused on depression and revealed task-dependent improvements in aspects of executive function (i.e., observed on the TMT A and B), suggesting that measures used in different studies may not always capture the specific cognitive effects of rTMS. 28
Contrary to our hypothesis, we observed no significant improvements in executive function among patients with LLD. Some research has suggested that executive function benefits of rTMS may be more pronounced in patients (and particularly older adults) with underlying executive dysfunction.22,35,54 While a previous meta-analysis found no significant age-related effects of rTMS on executive function in depression, 35 a qualitative review of the literature suggested that, compared to younger adults, older adults with depression experience more notable improvements in executive function. 35 In an open-label study with older adults with depression and co-existing executive dysfunction, bilateral iTBS led to significant improvements in executive function, 30 though this finding was not replicated in the subsequent randomized sham-controlled trial. 34 Similarly, other studies of rTMS, including deep TMS, found no improvements in executive function in LLD.36,56,57 Taken together, these findings suggest that DLPFC-rTMS may have no or weak beneficial impacts on executive function. This lack of a substantial effect may be due to the focus on treating depression rather than executive dysfunction, as well as the relatively short treatment duration and assessment periods used in these studies.
Although only a minority of participants exhibited reliable improvements in verbal learning, this aligns with several previous studies in older 29 and younger adults16,17,58 with depression. Notably, improvement in verbal learning was predicted by a higher premorbid IQ which is indicative of greater cognitive reserve and may facilitate cognitive recovery or improvement after rTMS. Moreover, a reliable improvement in verbal learning was more likely in patients who responded to treatment (i.e., showed a reduction ≥ 50% from baseline in MADRS score). Previous research across various treatments, including pharmacological and psychological interventions, has linked improvements in learning and memory to reductions in depressive symptoms. 6 This association has also been observed in prior rTMS studies,17,36 suggesting that improvements in verbal learning may be secondary to improvement in mood. However, the relationship between improvement in mood and cognition following rTMS remains inconsistent across cognitive domains. Some studies have linked cognitive improvements to reductions in depressive symptoms,22,35 while others found cognitive benefits independent of mood improvement,28,31,59,60 suggesting divergent mechanisms underlying depressive symptoms and cognitive function. Treatment allocation—whether participants received standard bilateral rTMS or bilateral TBS—did not significantly predict improvements in verbal learning. TBS is believed to induce more robust changes in synaptic plasticity compared to standard rTMS, as it more closely replicates the brain's natural firing patterns. 61 In healthy adults, iTBS has demonstrated greater cognitive enhancement, particularly in working memory and executive function, when compared to high-frequency rTMS. 62 However, both treatments showed similar effectiveness in improving depressive symptoms, 9 which aligns with the lack of superior cognitive results from TBS in this study.
While our study observed no robust cognitive improvement after rTMS, we found no evidence of significantly worsened performance on any cognitive measure. Across all measures, the percentage of participants who experienced a reliable cognitive decline ranged from 0% to 2.7%, which is below the 5% expected to be due solely to measurement error. These findings reinforce the extensive literature demonstrating the cognitive safety of rTMS, 55 supporting its use in treating LLD without the risk of cognitive decline. This makes rTMS preferable to other treatments (e.g., ECT, antipsychotics), which may have cognitive side effects.
It is important to note that rTMS primarily aimed to alleviate depressive symptoms. To address both depression and cognitive impairment in LLD, rTMS treatment may need to be designed accordingly. Similar to optimizing the site of stimulation to enhance therapeutic effects for depressive symptoms, identifying optimal targets within the DLPFC that modulate cognition (e.g., through neuroimaging) could be beneficial.63,64 Exploring alternative target sites beyond the DLPFC, 65 such as the dorsomedial prefrontal cortex,65,66 or augmenting DLPFC stimulation with other targets, 67 may also enhance rTMS cognitive effects. Recent research indicates that combining rTMS with transcranial direct current stimulation (tDCS), compared to rTMS alone, significantly enhances cognitive function and reduces clinical symptoms in younger adults with depression. 68 Another promising strategy involves combining rTMS with cognitive training, which improved global cognition across diverse clinical populations. 69
This study has some limitations. First, since the current data was drawn from a non-inferiority trial that compared two types of rTMS, there was no sham-TMS control group. To account for repeated assessments and practice effects, we calculated RCIPE using published normative data, which was derived from adults without clinical diagnoses, and for the CVLT-II, only raw scores uncorrected for age were available. 50 Given that age and clinical diagnosis affect the magnitude of practice effects, 70 the cognitive benefits of rTMS treatment may be underestimated in older adults with depression because the practice effects may be different compared to the normative sample. Second, randomized participants who did not complete treatment or follow-up cognitive assessments had lower baseline cognitive performance. This limits generalizability of our results and could have biased our findings. Moreover, the high education level and predominantly white demographic of our participants may limit the generalizability of our findings to other older adult populations.
In conclusion, our study demonstrated limited cognitive benefits after bilateral standard rTMS or bilateral TBS for the treatment of LLD, with only a small proportion of participants showing reliable improvement in verbal learning. Future research examining cognitive effects of rTMS should include a clinically similar comparator group undergoing repeat cognitive assessment and evaluate cognitive changes both at group and individual levels. Comprehensive assessments should evaluate changes in specific cognitive tests, and measures of various aspects of executive functions like response inhibition, working memory, and cognitive flexibility should be included. Additionally, research should focus on adjunctive strategies and/or refined neuroanatomical targets to optimize the cognitive benefits of rTMS to effectively address both depressive and cognitive symptoms in LLD.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SMM reports research support from the National Institutes of Health (NIH). He is a consultant to Pearson, receives teaching honoraria from Duke University School of Medicine, and receives royalties from Guilford Press, Inc. LM has received research funding from the Alzheimer's Society of Canada, Centre for Aging and Brain Health Innovation, Ontario Ministry of Health and Long-Term Care, and Brainsway, Ltd. TKR has received research support from Brain Canada, Brain and Behavior Research Foundation, BrightFocus Foundation, Canada Foundation for Innovation, Canada Research Chair, Canadian Institutes of Health Research, Centre for Aging and Brain Health Innovation, National Institutes of Health, Ontario Ministry of Health and Long-Term Care, Ontario Ministry of Research and Innovation, and the Weston Brain Institute. TKR also received for an investigator-initiated study in-kind equipment support from Newronika, and in-kind research online accounts from Scientific Brain Training Pro, and participated in 2021 and 2022 in an advisory activity for Biogen Canada Inc. TKR is also an inventor on the United States Provisional Patent No. 17/396,030 that describes cell-based assays and kits for assessing serum cholinergic receptor activity. SMN reports research and salary support from the Sunnybrook Health Sciences Centre Foundation, Harquail Centre for Neuromodulation; research support from the Brain and Behaviour Research Foundation Young Investigator Grant, Labatt Family Network Discovery Program and the Donald T Stuss Young Investigator Research Innovation Award. JD reports research support from NIH, CIHR, Brain Canada, Ontario Brain Institute, the Klarman Family Foundation, the Arrell Family Foundation, and the Buchan Family Foundation, in-kind equipment support for investigator-initiated trials from MagVenture, is an advisor for BrainCheck, Arc Health Partners and Salience Neuro Health, and is a co-founder of Ampa Health. YN received a grant in aid for scientific research from the Japan Society for the Promotion of Science; research grants from Japan Agency for Medical Research and Development; investigator-initiated clinical study grants from Teijin Pharma and Inter Reha Co; receives research grants from Japan Health Foundation, Meiji Yasuda Mental Health Foundation, Mitsui Life Social Welfare Foundation, Takeda Science Foundation, SENSHIN Medical Research Foundation, Health Science Center Foundation, Mochida Memorial Foundation for Medical and Pharmaceutical Research, Taiju Life SocialWelfare Foundation, and Daiichi Sankyo Scholarship Donation Program; speaker's honoraria from Dainippon Sumitomo Pharma, Mochida Pharmaceutical Co, Yoshitomiyakuhin Corporation, Qol Co, Teijin Pharma, and Takeda Pharmaceuticals within the past 5 years; and equipment in-kind support for an investigator-initiated study from Magventure, Inter Reha Co, Brainbox, and Miyuki Giken Co. ZJD has received research and equipment in-kind support for an investigator-initiated study through Brainsway Inc and Magventure Inc. He also currently serves on the scientific advisory board for Brainsway Inc. His work has been supported by the National Institutes of Mental Health (NIMH), the Canadian Institutes of Health Research (CIHR), Brain Canada and the Temerty Family, Grant and Kreutzcamp Family Foundations. BM holds and receives support from the Labatt Family Chair in Biology of Depression in Late-Life Adults at the University of Toronto. He currently receives or has received within the past three years research support from Brain Canada, the Canadian Institutes of Health Research, the CAMH Foundation, the Patient-Centered Outcomes Research Institute (PCORI), the US National Institute of Health (NIH), Capital Solution Design LLC (software used in a study founded by CAMH Foundation), and HAPPYneuron (software used in a study founded by Brain Canada). DMB receives research support from CIHR, NIH, PCORI, the Wellcome Trust, Brain Canada and the Temerty Family through the Centre for Addiction and Mental Health Foundation and the Campbell Family Research Institute. He received research support and in-kind equipment support from Brainsway Ltd. for an investigator-initiated study. He was the site principal investigator for 3 sponsor-initiated studies for Brainsway Ltd. He also receives in-kind equipment support from MagVenture for investigator-initiated research. He received medication supplies for an investigator-initiated trial from Indivior. He is a scientific adviser for Sooma Oy Medical.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research (grant PJT-148840).