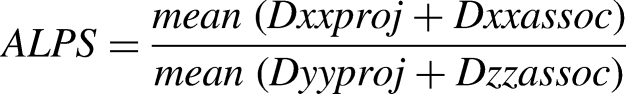Abstract
Objective
The pathophysiological mechanisms influencing psychosis spectrum disorders are largely unknown. The glymphatic system, which is a brain waste clearance pathway, has recently been implicated in its pathophysiology and has also been shown to be disrupted in various neurodegenerative and vascular diseases. Initial studies examining the glymphatic system in psychosis spectrum disorders have reported disruptions, but the findings have been confounded by medication effects as they included antipsychotic-treated patients. In this study, we used diffusion tensor imaging analysis along the perivascular space (DTI-ALPS) as a technique to measure the functionality of the glymphatic system in a sample of antipsychotic-minimally exposed patients with psychosis spectrum disorders and healthy controls.
Methods
The study included 13 antipsychotic-minimally exposed (2 weeks antipsychotic exposure in the past 3 months/lifetime) patients with psychosis spectrum disorders and 114 healthy controls. We quantified water diffusion metrics along the x-, y-, and z-axes in both projection and association fibres to derive the DTI-ALPS index, a proxy for glymphatic activity. Between-group differences were analyzed using two-way ANCOVA controlling for age and sex. Partial correlations were used to assess the association between the ALPS index and clinical variables.
Results
Analyses revealed that antipsychotic-minimally exposed psychosis spectrum disorder patients had a lower DTI-ALPS index value than healthy controls in both hemispheres and the whole brain (all P < 0.005). Significant differences were also observed between the x and y projections/associations between patients and healthy controls (P < 0.001). Furthermore, we did not find any significant correlations (all P > 0.05) between the DTI-ALPS index with age, body mass index, symptomatology, and metabolic parameters.
Conclusion
This study shows that the glymphatic system is dysregulated in antipsychotic-minimally exposed patients with psychosis spectrum disorders. Understanding the mechanisms that influence the glymphatic system may help to understand the pathophysiology of psychosis spectrum disorders as proper waste clearance is needed for normal brain functioning.
Introduction
Psychosis spectrum disorders (PSDs) are a group of debilitating mental illnesses characterized by hallucinations, delusions, and cognitive impairment. 1 PSDs have a prevalence rate of 4.6 per 1,000 people and represent a huge economic burden to society, including a lowering of quality of life.2–4 It is largely unknown which mechanisms or pathways contribute to the pathophysiology of PSDs; however, a range of genetic and environmental factors are hypothesized to play a role in psychosis development, 5 resulting in abnormal brain structure and functionality.6,7
The glymphatic system (GS) is a major waste clearance pathway that removes soluble proteins and metabolites from the central nervous system while also aiding in the distribution of glucose. 8 Aquaporin-4 (AQP4) are water channels that are integral to this waste system; mice without these channels show reduced solute clearance. 9 There is evidence that the GS is dysregulated in PSDs, with a schizophrenia-like behaviour mouse model showing a higher expression of AQP4 channels in the prefrontal cortex and hippocampus. 10 Additionally, patients with PSDs have been shown to have genetic polymorphisms in AQP4 compared to healthy controls (HCs). 11 There is also recent evidence showing that the GS is impaired in patients with schizophrenia spectrum disorders (SSDs), but these studies included patients with chronic SSDs who have received antipsychotic (AP) treatment.12,13
Structural imaging studies have shown that grey matter volume (GMV) increased in the frontal and temporal regions with mid-to-long term AP-use (10 months–4 years),14,15 while a decrease in GMV within the same regions was observed with short-term use (8 weeks).16–18 In patients with first-episode psychosis (FEP), task-based and resting-state functional imaging studies have reported that AP treatment can normalize functional dysconnectivity involving the frontal and temporal regions.18–20 As such, AP usage represents a potentially important confounding factor in this field of study.
Dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) has been used to study the GS in humans; however, it requires the injection of a gadolinium-based contrast agent.21–23 There is also evidence of gadolinium brain depositions after repeated administration, though, this has not been associated with harmful effects. 24 A commonly used alternative to DCE-MRI is diffusion tensor imaging (DTI), a non-invasive imaging modality. DTI is a technique that assesses white matter tracts in the brain by measuring fractional anisotropy (FA), an indication of overall diffusion of water with more organized tracts providing higher directionality. 25 Previous research in FEP patients shows that white matter integrity is disrupted. 26 Diffusion tensor imaging analysis along the perivascular space (DTI-ALPS) is a supplement technique that measures the functionality of the GS and has been validated using DCE-MRI 27 with good intra-observer agreement. 28 A lower DTI-ALPS index indicates a reduction in GS clearance and has also been observed in neuropsychiatric illnesses such as Alzheimer's disease (AD) 29 and Parkinson's disease (PD). 30
With evidence suggesting that there is an interplay between the GS and PSDs, we aimed to investigate this association in AP-minimally exposed patients with PSDs to eliminate the potential effects of treatment with APs on the GS. We hypothesized that patients with PSDs will have a lower DTI-ALPS index when compared to HCs, which would suggest that patients have impaired GS function. In an exploratory analysis, we also investigated whether DTI-ALPS was associated with BMI and age.
Methodology
Participants
This study is a secondary analysis of data collected from two different centres. Antipsychotic-naïve (AP-naïve) patients were recruited at the Center for Addiction and Mental Health (CAMH) in Toronto, Canada (REB #060/2014). The HC dataset was taken from the University of California, Los Angeles Consortium for Neuropsychiatric Phenomic LA5c Study via OpenNeuro, accession number ds000030.23. The HC inclusion criteria are reported elsewhere. 31 AP-minimally exposed participants had to meet the following criteria: (i) age between 12 and 35 years; (ii) received AP treatment for less than 2 weeks in their lifetime, or they had not received AP treatment for more than 2 weeks within the past 3 months; and (iii) DSM-V diagnosis (SSDs, mood disorder with psychotic features, or other developmental disorders with psychotic symptoms requiring treatment with APs. The exclusion criteria were as follows: (i) AP treatment for more than 2 weeks within the preceding 3 months; (ii) concomitant medications for the treatment of lipids, glucose, or weight; (iii) pregnancy; (iv) active or previous eating disorder; (v) a major medical or surgical event within the preceding 3 months; (vi) acute suicidal risk. Both studies were reviewed and approved by their respective institutional Research Ethics Board, and all participants provided written informed consent.
The study involved 13 AP-minimally exposed patients and 123 healthy controls. During visual inspection of Diffusion Weighted Imaging (DWI) data, 8 healthy controls had corrupted volumes and/or ghost images in their DWI images, making their data not suitable for further processing. However, no such issues were observed in the AP-minimally exposed patient data. The final sample for analysis consisted of 13 AP-minimally exposed patients and 115 HCs.
MRI Acquisition Parameters
The DWI images of HCs were acquired using a Siemens Trio 3T MRI scanner. The DWI scan parameters used were as follows: slice thickness of 2 mm, 64 directions, TR/TE of 9,000/93 ms, flip angle of 90°, matrix of 96 × 96, axial slices, and b-values of 0 and 1,000 s/mm2. On the other hand, the data of the AP-minimally exposed patients was acquired using a Siemens Skyra 3T MRI, with the following DWI parameters: slice thickness of 2 mm, 64 directions, TR/TE of 12,300/91 ms, flip angle of 90°, matrix of 120 × 120, axial slices, and b-values of 0 and 1,000 s/mm2.
DTI Processing
The 4D DTI DICOM files were converted to NIFTI format using the MRIcroGL's terminal command “dcm2niix”. An in-house bash script with the DTI images as input involved the use of FSL (https://fsl.fmrib.ox.ac.uk/fsl/fslwiki/FSL) and MRtrix3 (https://www.mrtrix.org/) commands. To prepare the DTI images, artefact correction was implemented using the Marchenko–Pastur Principal Component Analysis (MP-PCA) denoising algorithm and Gibbs-unringing with MRtrix3 command line tools “dwisenoise” and “mrdegibbs”. The susceptibility-induced distortion corrections, eddy currents, and movements were accomplished with FSL command line tools “topup” and “eddy”. With the help of the FSL command line tool “dtifit”, we generated the FA map and x-, y-, and z-axes diffusivity maps for all participants. 29
Each individual's FA map was co-registered to the JHU-ICBM-FA template, and the transformation matrix was applied to all the diffusivity maps using the FSL command line tool “flirt”. The superior corona radiata (SCR) and the superior longitudinal fasciculus (SLF) were recognized as the projection and association fibres at the level of the lateral ventricle body, respectively, based on the JHU-ICBM-DTI-81-white-matter Labeled Atlas, a comprehensive atlas of white matter tracts. The bilateral SCR and SLF areas were defined as spheres with a 5 mm diameter, and these ROIs were applied to the diffusivity maps of all participants. The JHU-ICBM-FA template-based ROIs’ centres were defined as follows: Right SCR (64, 110, 99), Right SLF (51, 110, 99), Left SCR (116, 110, 99), and Left SLF (128, 110, 99). 29 The bilateral SLF and SCR diffusivity values were automatically computed along the x-, y-, and z-axes (i.e., Dxx, Dyy, and Dzz) for the ALPS calculation.
ALPS Index Calculation
The ALPS index is calculated by taking the average of bilateral ALPS indexes, which is determined by the ratio of the mean of x-axis diffusivity in the area of projection fibres (Dxxproj) and x-axis diffusivity in the area of association fibres (Dxxassoc), to the mean of the y-axis diffusivity in the area of projection fibres (Dyyproj) and z-axis diffusivity in the area of association fibres (Dzzassoc).
29
This formula is given below:
Statistical Analysis
All statistical analyses were conducted using IBM SPSS Statistics software (version 25, SPSS, Inc., Chicago, IL, USA). The Shapiro–Wilk test was used to determine if the data were normally distributed. Tukey's method was used to remove extreme outliers from the analysis. These were defined as values outside the range of the 3rd quartile + 3 times the interquartile range and the 1st quartile – 3 times the interquartile range. Only extreme outliers were removed, while milder outliers that may be characteristics of the distribution were retained. This method aided in avoiding a false impression of the population. A sub-analysis was also conducted to match AP-minimally exposed patients with HCs according to potentially confounding variables, such as age and number of smoking years. Partial eta-squared (ηp2) was calculated as an effect size measure. Two-sample independent t-tests or the Mann–Whitney U test was used to compare separate groups, depending on the nature of the continuous variables. Additionally, exploratory partial correlation analyses were done to determine the relationship between the ALPS index and various clinical parameters, while making necessary adjustments for age and sex. P values < 0.05 (2-sided) were considered statistically significant.
Results
Clinical Characteristics
Table 1 presents the demographic and clinical characteristics of each group of the sample. One HC individual was deemed an outlier, which left 114 HCs and 13 AP-minimally exposed patients for the final analysis. The AP-minimally exposed patients were found to be significantly younger (t = 6.42 P < 0.001) and had a lower number of smoking years (t = 4.59, P < 0.001) when compared to HCs. As such, all subsequent analyses were also conducted on a sub-sample matched on these variables.
Demographic and Clinical Characteristics of All Participants.
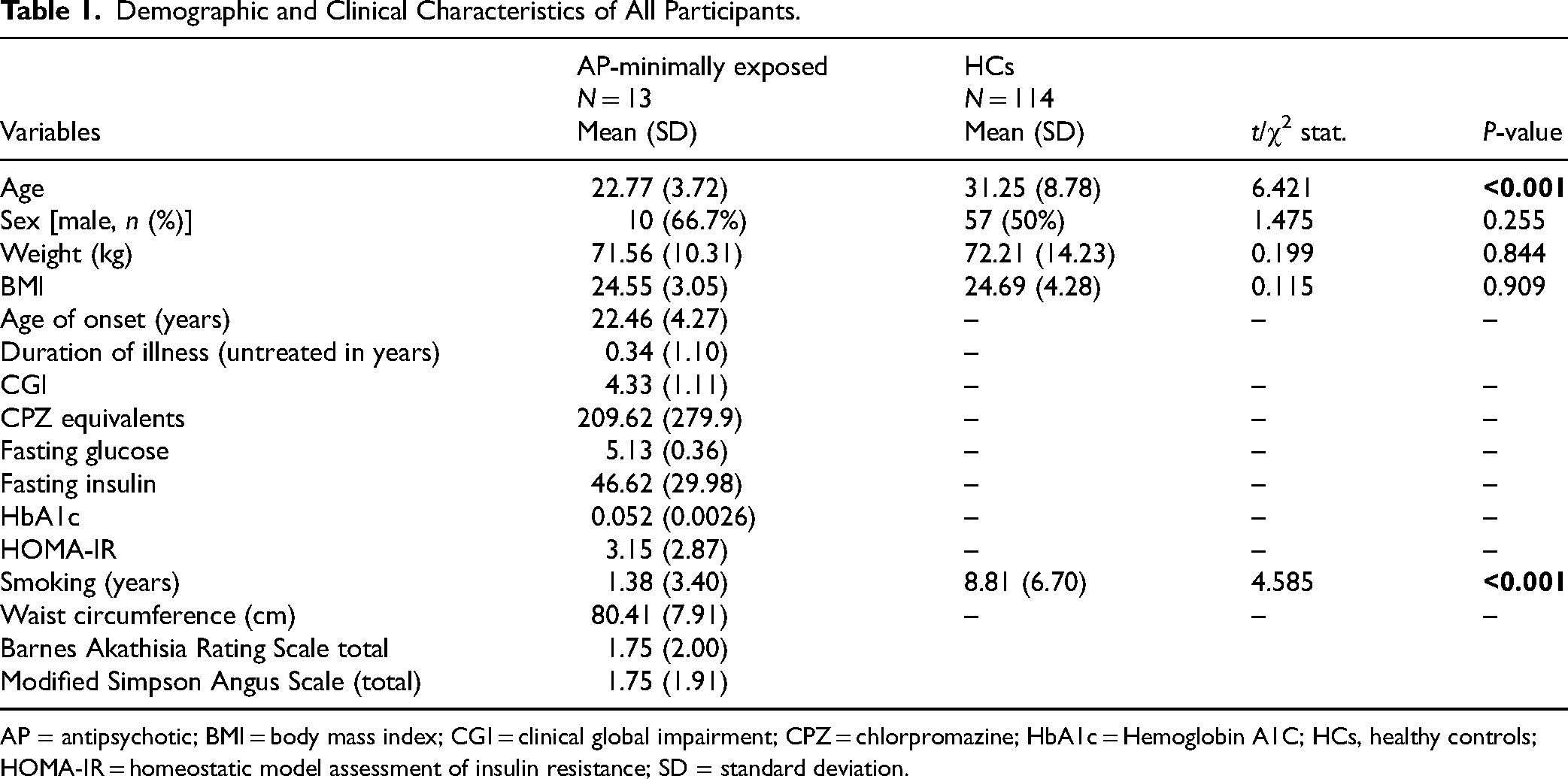
AP = antipsychotic; BMI = body mass index; CGI = clinical global impairment; CPZ = chlorpromazine; HbA1c = Hemoglobin A1C; HCs, healthy controls; HOMA-IR = homeostatic model assessment of insulin resistance; SD = standard deviation.
Group Comparison of ALPS
As shown in Table 2 and Figure 1, AP-minimally exposed patients had a significantly lower DTI-ALPS index of the left and right hemispheres and the whole brain compared to HCs, even after adjusting for age and sex (Falps_L = 11.09, P = 0.001; Falps_R = 10.92, P = 0.001; Falps = 13.06, P < 0.001). The effect sizes for the hemispheres and whole brain are: left (ηp2 = 0.112), right (ηp2 = 0.134) and whole brain (ηp2 = 0.142).12,13
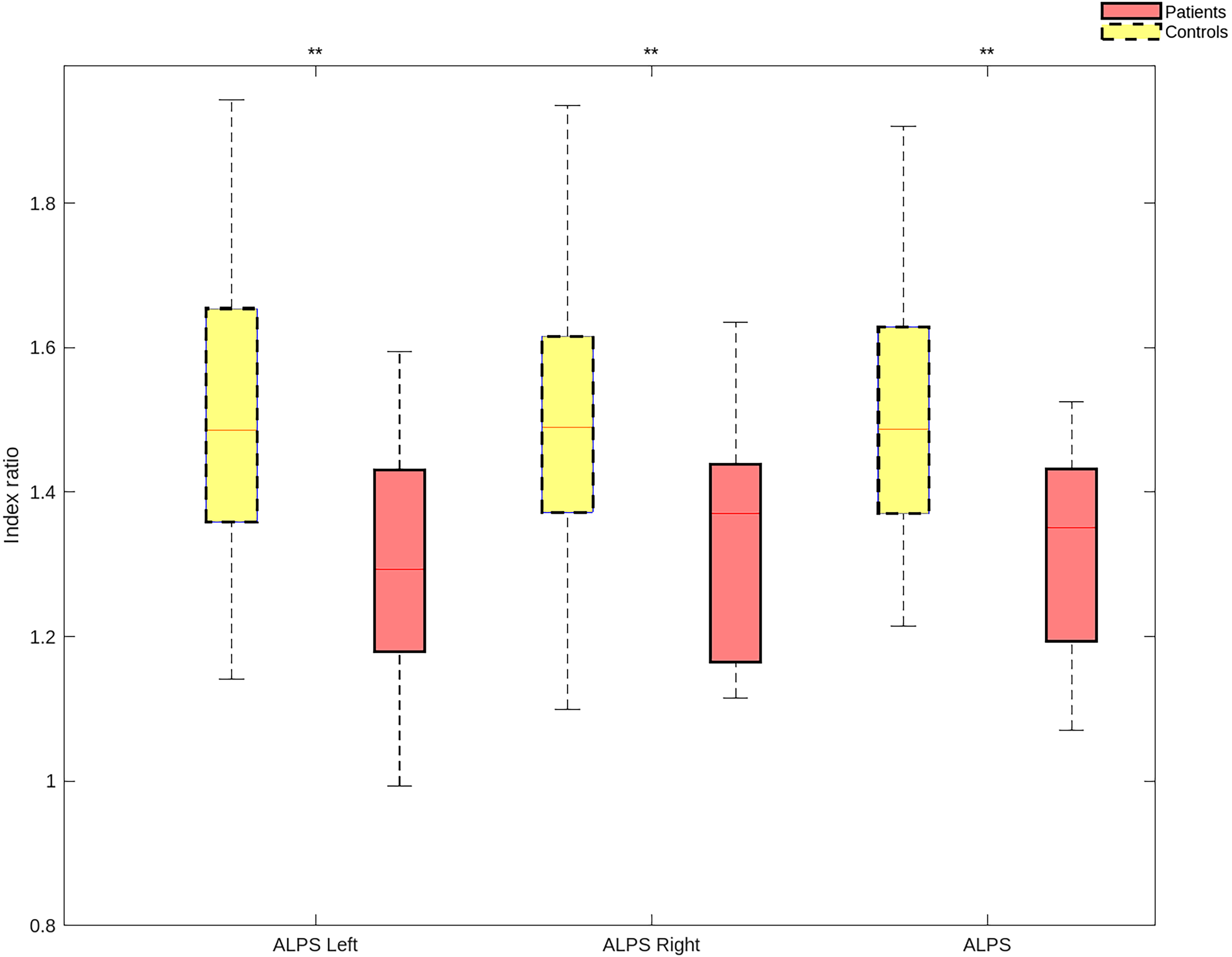
Comparisons of DTI-ALPS index between AP-minimally exposed and HC groups in the left/right hemisphere and whole brain. **P < 0.005. Abbreviation: AP = antipsychotic; DTI-ALPS = diffusion tensor image analysis along the perivascular space; HCs = healthy controls.
Group Comparisons of ALPS Index Between AP-Minimally Exposed Patients and Healthy Controls.
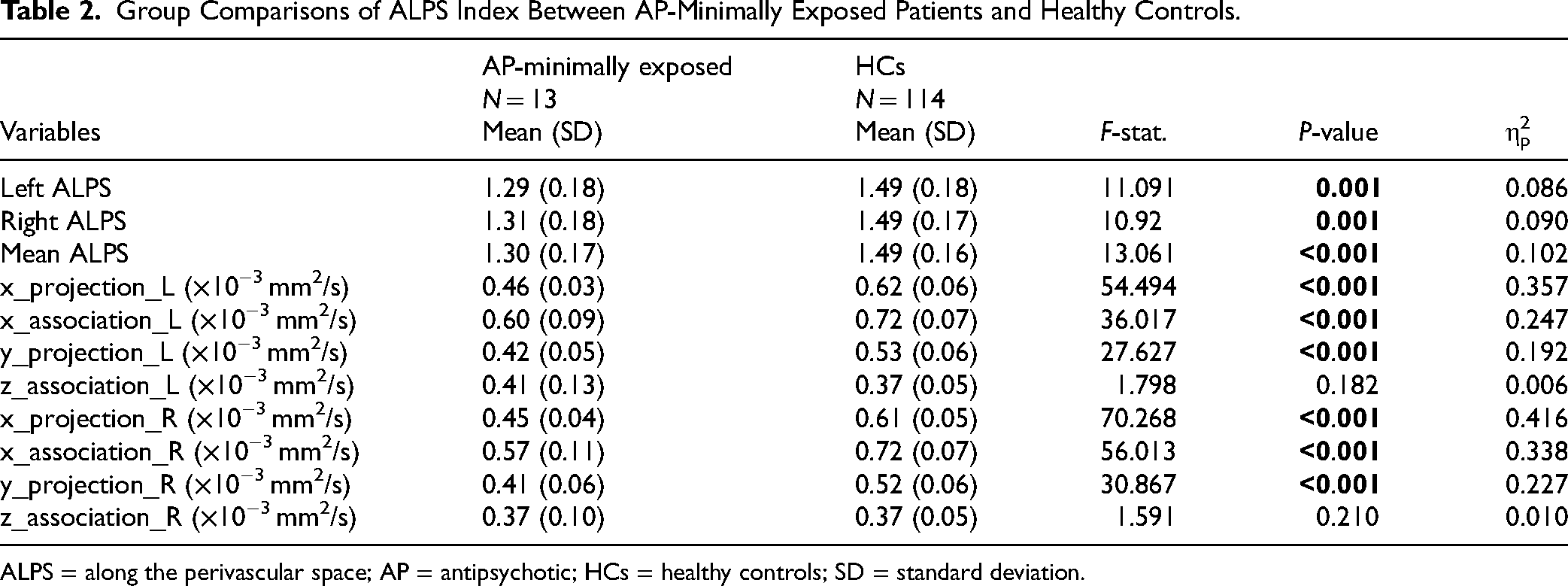
ALPS = along the perivascular space; AP = antipsychotic; HCs = healthy controls; SD = standard deviation.
Similarly, the diffusivities along the x-axis of both projection and association fibres, in both the left and right hemispheres, were also lower in AP-minimally exposed patients than in HCs (Fproj_L = 54.49, P < 0.001; Fproj_R = 70.268, P < 0.001; Fassoc_L = 36.02, P < 0.001; Fassoc_R = 56.01, P < 0.001). In addition, the diffusivities along the y-axis of projection fibres in both the left and right hemispheres were lower in AP-minimally exposed patients when compared to HCs (Fproj_L = 27.63, P < 0.001; Fproj_R = 30.87, P < 0.001). However, the diffusivities of association fibres along the z-axis were not significant in the left or right hemisphere (Fassoc_L = 1.80, P = 0.182; Fassoc_R = 1.59, P = 0.210).
Sub-Group Comparison of ALPS by Matched Age and Smoking
In the sub-analysis matching for age and smoking status, the DTI-ALPS index of the left and right hemispheres and the whole brain was significantly lower among the AP-minimally exposed patients compared to the HCs, as shown in Table 3 (Falps_L = 10.062, P = 0.004; Falps_R = 12.237, P = 0.002; Falps = 13.54, P = 0.001). The effect sizes for the hemispheres and whole brain are: left (ηp2 = 0.295), right (ηp2 = 0.338) and whole brain (ηp2 = 0.361).
Sub-Group Comparisons of ALPS index Between AP-Minimally Exposed Patients and Healthy Controls After Matching for age and Smoking Years.
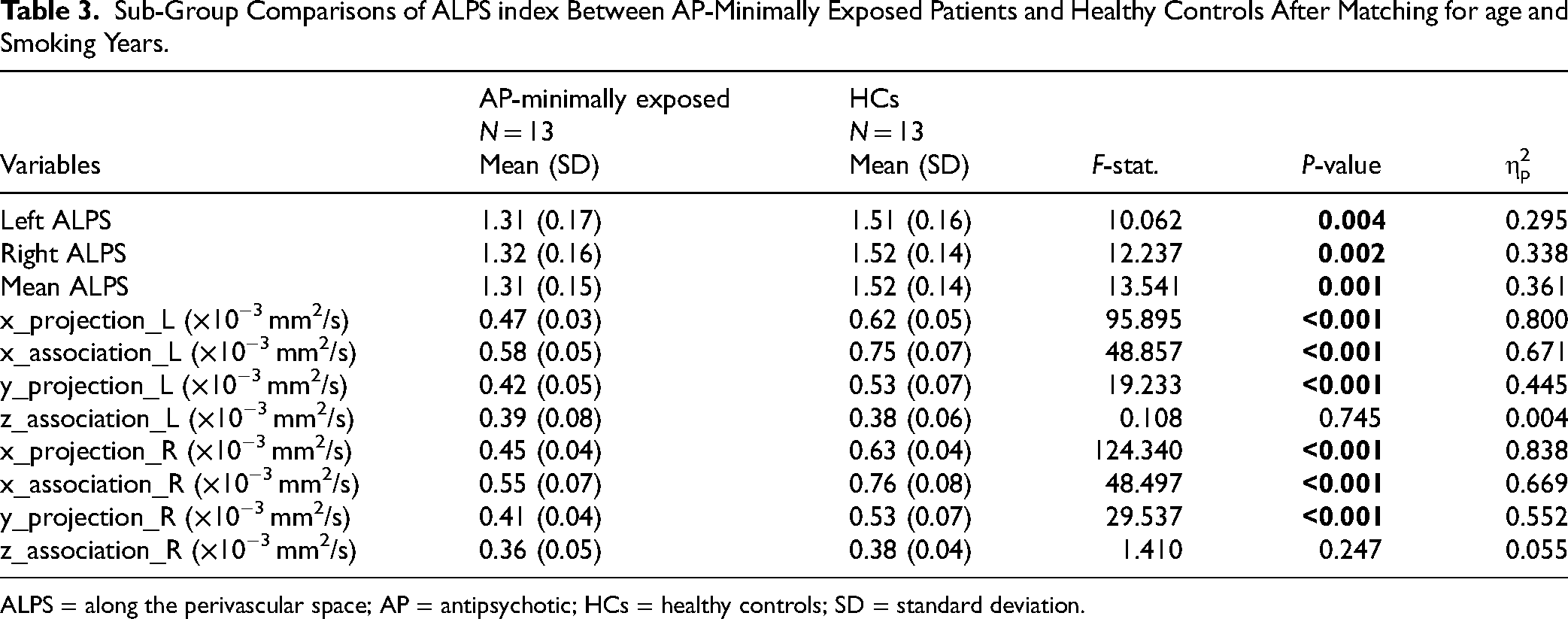
ALPS = along the perivascular space; AP = antipsychotic; HCs = healthy controls; SD = standard deviation.
The projection and association fibres along the x-axis direction was found to be significantly lower in both the left and right hemispheres in the AP-minimally exposed group compared to HCs, (Fproj_L = 95.895, P < 0.001; Fproj_R = 124.340, P < 0.001; Fassoc_L = 48.857, P < 0.001; Fassoc_R = 48.497, P < 0.001). Additionally, the y-axis projection fibres in the left and right hemispheres were lower in AP-minimally exposed patients when compared to HCs (Fproj_L = 19.233, P < 0.001; Fproj_R = 29.537, P < 0.001). However, we did not find significant differences along the z-axis fibres in the left or right hemisphere (Fassoc_L = 0.108, P = 0.745; Fassoc_R = 1.410, P = 0.247).
Association Between ALPS and Clinical Variables
No significant correlations were identified between demographic, clinical and metabolic parameters, and the ALPS indices in both the full and patient population, as shown in Table 4. For our exploratory objectives, we observed a non-significant negative correlation between age/BMI and the ALPS index in the whole brain.
Association Between Mean Total ALPS Index and Clinical Variables in AP-Minimally Exposed PSD Patients and Healthy Controls.

AP = antipsychotic; BARS = Barnes Akathisia Rating Scale; CGI = Clinical Global Impairment; BMI = body mass index; BARS = Barnes-Akathisia Ratings Scale; CPZ = chlorpromazine; HbA1c = hemoglobin A1C; HOMA-IR = homeostatic model assessment of insulin resistance.
111 for HCs; b 12 for AP-minimally exposed.
Discussion
This study investigated GS functioning in AP-minimally exposed PSD patients using the DTI-ALPS index, a non-invasive diffusion-based technique. Our findings revealed a decrease in glymphatic function, as suggested by lower ALPS indices in AP-minimally exposed patients compared to HCs. Additionally, we observed a significant decrease in diffusivities along the projection and association fibres in both the left and right hemispheres of the brain in the X and Y-axis directions, but not in the Z-axis direction. Lastly, we found no significant associations between clinical measures and ALPS indices.
Presently, this is the first study investigating the GS in an AP-minimally exposed PSD population using DTI-derived data. Interestingly, the findings are in line with recently published work examining patients further in the illness course.12,13 A recent study by Abdolizadeh et al. (2024) showed that individuals with SSDs exhibit lower DTI-ALPS indices in both hemispheres and the whole brain. Similarly, Tu et al. (2024) reported that individuals with SSDs show decreased ALPS indices in both the left and right hemispheres as well as the whole brain when compared to healthy controls. However, both studies focused on chronically AP-treated patients with schizophrenia, and most antipsychotics are associated with impaired metabolism, even in the early stages of psychosis. 32 Thus, this study extends previous findings by addressing, in part, whether AP treatment is a confounding factor for GS functioning in the earlier studies. In the main analysis, we found similar effect sizes as that of previous studies. However, after matching for age and smoking, we found higher effect sizes than both studies, suggesting that GS dysfunction may be present in PSDs prior to AP exposure, and that this is independent of smoking status, which is higher in chronically ill patients with schizophrenia compared to the general population.
The GS encompasses three key physiological processes: (1) the entry of cerebrospinal fluid (CSF) from the subarachnoid space into the periarterial space, known as CSF influx; (2) the exchange process, referred to as AQP4-dependent, which takes place between the periarterial CSF and the parenchymal interstitial fluid (ISF), and (3) the efflux of brain waste products through the perivenous pathway. 9 Recent studies using animal models have revealed that older mice or aging rats display diminished glymphatic influx activity, a loss of perivascular AQP4 polarization, and reduced CSF–ISF exchange activity.33,34 Previous studies indicate that the brains of people with chronic SSDs seem to exhibit an accelerated aging process compared to their actual age, in contrast to the general population. 35 This makes it challenging to interpret impaired GS functioning in these individuals. However, our findings validate the existence of abnormal glymphatic function in the initial stages of the illness. Besides age, obesity has negatively affected lymphatic transport and node structure. 36 Previous research has indicated a link between obesity, pro-inflammatory states, perivascular inflammation, oxidative stress, and compromised mitochondrial function. 37 These obesity-related factors may potentially impede the glymphatic system pathway by disrupting convective flow and CSF-to-ISF turnover. 38 Moreover, the absence of a significant association between age, BMI, and DTI-ALPS in our study is probably due to the small sample size and the narrow age and BMI range of the participants. Thus, confirming the impacts of psychosis on overall GS functioning, eliminating the potential confounding effects of age and higher BMI on GS. For further understanding, future research should explore these associations in PSDs with a larger sample size.
While the mechanisms through which the GS may be impaired in patients with PSDs remain unclear, sleep disturbances have been identified as a potential contributor to GS dysfunction.39,40 Supporting this notion, previous research has revealed that individuals at ultra-high risk of psychosis have impaired sleep–wake functioning, which might result in compromised callosal microstructure. 41 Consequently, this sleep disruption may be influencing the potential GS contributing factors such as reduced arterial pulsation, 42 enlarged perivascular space, 43 astrocytic dysfunction, 44 and altered expression of AQP-4 channels. 9 Moreover, a recent study has demonstrated that during slow-wave sleep or under anaesthesia, glymphatic inflow is higher than during wakefulness,45,46 and the clearance rate of beta-amyloid (Aβ) increases 2-fold during sleep. 47 Furthermore, a recent study suggested that patients with major depressive disorder (MDD) suffering from sleep disturbances might experience GS dysfunction. This impaired GS is possibly due to a decrease in AQP4-IR astrocyte coverage in the orbitofrontal cortex, which could impact cerebral blood circulation, metabolism, and synaptic adjustment. 48 As evidence of this, earlier research has established a notable association between sleep disturbances and dysfunction in the glymphatic system in the older general population and PD.49,50 This underscores the importance of addressing potential confounders like sleep disruptions in treating psychosis. 51
A key aspect of glymphatic clearance is its reliance on sleep state, with increased glymphatic function observed during N3 (slow-wave sleep) and with some anaesthetic treatments.45,52 Additionally, studies show that sleep disruptions are common among individuals predisposed to psychosis, often appearing early in illness progression. 53 Since the GS works most efficiently during deep sleep; sleep disturbances or disorders could hinder the GS’ ability to clear neurotoxic waste. 54 The link between N3 sleep and the brain waste egress may be due to a decrease in central noradrenergic tone, which leads to a higher volume fraction of interstitial fluid and, results in enhanced waste removal. 45 Therefore, the role of adequate sleep in promoting glymphatic waste elimination has become a new focus for maintaining brain health.
One possible pathway by which GS dysfunction may be linked to PSDs is through oxidative stress and neuroinflammation. 48 GS dysfunction may increase oxidative stress by impaired clearance of reactive oxygen species (ROS), leading to its accumulation. 48 Previous research has shown that ROS can activate microglial inflammasomes and inflammatory cytokines; this can increase neuroinflammation, which has been linked to PSDs.55–57 Additionally, GS dysfunction has been observed in various neurodegenerative diseases, including MDD, 48 AD, 29 cerebral small vessel disease, 54 PD,49,50 and SSDs.12,13 This implies that a dysfunctional GS could be a common denominator across neuropsychiatric disorders.
Our research has revealed that the AP-minimally exposed individuals show a decreased diffusion coefficient in the X-direction of the projection and association fibre regions when compared to HCs. Our findings align with a recently published study conducted on chronic SSDs.12,13 This decrease in projection fibres may contribute to deficits in the integration of sensory and motor functions, which are often observed in individuals with SSD and possibly within early stages of psychosis.58,59 Furthermore, previous findings showed that FEP patients have abnormalities in the superior longitudinal fasciculus (SLF) fibres, including elevated crossing fibres and altered anisotropy, which suggest a possible link between DTI X-axis association fibres and psychosis, implying that higher-order cognitive functions, such as executive control, are impaired. 60 Besides, the lower Y-axis projection fibres in the AP-minimally exposed patients may indicate changed convergence in fronto-striatal wiring in FEP, which could impact cognitive functions, such as cognitive control and metacognition. 61 In summary, these findings are important as comprehending the specific changes in projection and association fibres can enhance our knowledge about the neural pathophysiology in PSDs.
Our study findings should be interpreted with caution in light of a few limitations. First, our patient population was significantly younger than the HCs, and this can impact the results as the GS is affected by age. Second, our study includes a small sample size (N = 13) in the AP-minimally exposed group, which may limit the generalizability of the findings. Future studies should include a larger sample size. Third, the scans occurred while the patients were awake, and there is evidence that glymphatic clearance is heightened during sleep. 45 As such, implementing DTI-ALPS scans while asleep may provide a clearer picture of the association between PSDs and the GS. Fourth, we did not collect information on sleep disturbances on our patients, and there is evidence showing that patients with PSDs often suffer from sleep disturbances. 62 Fifth, we did not use the gold standard approach of MRI-based tracers to measure glymphatic function in humans 63 ; however, previous research has demonstrated a strong correlation with glymphatic clearance function, as determined by glymphatic MRI following intrathecal gadolinium administration. 27 Lastly, the datasets were obtained from two different centres using similar, yet different, scanning protocols. However, DTI-ALPS is a ratio, and differences in scanning parameters would be expected to impact both parts of the equation equally.
Conclusion
Overall, this study demonstrates that AP-minimally treated patients with PSDs have a lower DTI-ALPS index compared to HCs. This indicates that patients have a lower waste clearance in the GS as proper brain functioning may require a normal waste clearance. Future research should include a larger sample size and longitudinal components to identify the impact of APs on the GS. Understanding the pathways and/or mechanisms related to this lower waste clearance may help uncover novel pathophysiological markers of PSDs.
Footnotes
Data Availability
Data analyzed in this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
Remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. SMA has received honoraria from HLS Therapeutics and Boehringer Ingelheim outside the submitted work. MKH has received honoraria from Alkermes and grants from Merck outside the submitted work. GR has received grants from HLS Therapeutics.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the American Association of Child and Adolescent Psychiatry Pilot Research Award, supported by Pfizer to AC. AGG is currently supported by the Canadian Institutes of Health Research (CIHR), CAMH Foundation, Temerty-Tanz-TDRA Seed Fund for Research on Depression and Dementia at the University of Toronto, and Ontario AHSC AFP Innovation Fund (MOHLTC). SMA is supported in part by an Academic Scholars Award from the Department of Psychiatry, University of Toronto and the CAMH Discovery Fund.