Abstract
Introduction
Amphetamine-type stimulants (ATSs) are related to significant harm worldwide, with limited effective pharmacological treatments for ATS use disorder (ATSUD). Modafinil has been explored as a potential treatment for ATSUD. This systematic review and meta-analysis (PROSPERO ID: CRD42023388487) aimed to evaluate the efficacy and safety of modafinil for the treatment of ATSUD.
Methods
A comprehensive search of major indexing sources and trial registries, from inception to search date, was conducted on February 15, 2023, and updated on October 31, 2023. Eligible studies were randomized placebo-controlled trials (RCTs) of modafinil in individuals meeting the criteria for the Diagnostic and Statistical Manual of Mental Disorders, fourth and fifth editions, diagnoses of ATSUD. Eligible studies were assessed for risk of bias, using the Cochrane Risk of Bias tool. The primary outcome included the effect of modafinil on ATS use. Secondary outcomes included retention in treatment, ATS craving, treatment discontinuation due to adverse events (AEs), and serious AEs. Subgroup analysis by modafinil dose was conducted where appropriate. Risk ratio (RR) or Peto's odds ratio (OR) was calculated for the meta-analysis of dichotomous variables and standardized mean difference (SMD) was calculated for the random-effect meta-analysis of continuous variables.
Results
Five RCTs (N = 451 participants) were included. Modafinil did not significantly impact ATS use (RR = 0.99; 95% CI, 0.97 to 1.02; p = 0.655), retention in treatment (RR = 1.02; 95% CI, 0.91 to 1.14; p = 0.799), ATS craving (SMD = −0.36; 95% CI, −1.19 to 0.47; p = 0.398), or treatment discontinuation due to AEs (Peto's OR = 0.48; 95% CI, 0.20 to 1.14; p = 0.100). These results were consistent across subgroup analyses. More episodes of serious AEs were reported in the modafinil group than in the placebo group, at higher doses (Peto's OR = 4.80; 95% CI, 1.18 to 19.56, p = 0.029).
Conclusion
There is currently no evidence suggesting that modafinil has a statistically significant effect on efficacy outcomes in populations with ATSUD. Continued research into effective treatments and harm reduction strategies for ATSUD is essential.
Introduction
Amphetamine-type stimulants (ATSs), such as amphetamine and methamphetamine, are psychoactive substances associated with significant harm worldwide. Most recent drug seizure data have suggested that the manufacture, trafficking, and use of these substances are expanding beyond traditional markets, and spreading across the globe. 1 The continued use of ATS, specifically non-regulated drugs such as “crystal meth” or “speed,” has been associated with a wide range of physical and mental health-related harms, including cardiovascular and renal dysfunction, 2 increased risk for viral hepatitis and HIV infection,3,4 as well as psychosis, depression, and cognitive deficits. 5 While those of the male sex remain more likely to consume ATS, such as methamphetamine, compared to those of the female sex, ATS use has increased across demographic groups. 6 More specifically, methamphetamine use disorder without injection has increased in heterosexual men and women, gay/bisexual men, lesbian/bisexual women, non-Hispanic Whites, African Americans, Asians, Hispanics, and other Pacific Islanders, demonstrating that people at increased risk of ATS use have diversified rapidly. 6 The rate of drug overdose deaths related to non-pharmaceutical forms of ATS use has been increasing rapidly over the past 10 years in the United States. 7 Between 2019 and 2020, there was a 50% increase in drug overdose deaths involving ATS and other psychostimulants. 8 These numbers underscore the current challenges faced by ATS users and by health-care providers trying to support them, given the limited availability of evidence-based treatments for ATS use disorders (ATSUDs). 9
Presently, the only evidence-based management options available for the treatment of ATSUD are psychosocial approaches, with the best evidence for contingency management strategies and psychotherapies including cognitive behavioural therapy and motivational interviewing. 10 While these options can be effective for some, they are also time- and resource-intensive, are not readily available in many settings, and may not be sufficient for a significant proportion of treatment-seeking ATS users. 11 Only 20% of individuals with a stimulant use disorder (STUD) were said to have received specialized treatment in the United States in 2018. 12 Some have suggested that such low rates may be due, in part, to the lack of pharmacological options for the management of ATSUD. 13 Although a wide range of pharmacological options have been tested, there are currently no recommended pharmacological treatments for ATSUD. 14
Among the medications recently studied for ATSUD is modafinil, a wakefulness-promoting agent, that is currently approved for narcolepsy, shift work sleep disorder, and obstructive sleep apnea. 15 While the exact mechanism of action is unknown, modafinil seems to have stimulant-like effects through the reduction of dopamine uptake via dopamine transporter blockade.16,17 Moreover, the literature also suggests that it has an acceptable pharmacological and safety profile. 18 However, the first randomized-controlled studies examining modafinil in ATSUD populations had negative results.19,20 The effects of modafinil on outcomes in ATSUD have previously been reviewed in other meta-analyses and systematic reviews.13,21-23 Overall, these studies have tentatively concluded that modafinil is generally not effective for increasing abstinence in this population, with meta-analyses showing statistically insignificant results.13,21,23 There are two major limitations with these previous reviews. First, to our knowledge, there has yet to be a systematic review and meta-analysis specifically focusing on the effect of modafinil on a wide range of outcomes; published works have typically pooled modafinil data with the data of other psychostimulants.13,21 Second, these same studies reported the effect of modafinil in both cocaine-using and ATS-using populations.13,21 These are important limitations considering the unique pharmacological action of modafinil (i.e., compared to psychostimulants), and since the ATSUD population differs from the cocaine use disorder population on a range of socio-demographic and clinical dimensions (e.g., ethnicity, sexual conduct, and alcohol consumption). 24
As such, in this study, we aimed to systematically review randomized placebo-controlled trials (RCTs) of pharmacological interventions of modafinil in individuals with ATSUD, as well as conduct a meta-analysis on pooled data from these trials. Our main outcome of interest was the effect of modafinil on ATS use as measured by urine drug analysis. Additionally, we reviewed and analysed the evidence for the effect of modafinil on retention in treatment, ATS craving, treatment discontinuation due to adverse events (AEs), and serious AEs (SAEs) reported in these trials. Moreover, we conducted a subgroup analysis by modafinil dosing (200 mg vs. 400 mg daily).
Methods
Study Conceptualization and Registration
A preliminary search for identification of the outcomes of interest and confirmation of study availability for candidate outcomes was conducted in December 2022. The study protocol was first registered on PROSPERO (CRD42023388487) (see Supplemental Material 1) on January 19, 2023. The study was conducted and reports were prepared following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines25,26 (see Supplemental Material 1 for PRISMA checklists).
Search Methods
The original literature search was run on February 15, 2023, using a strategy designed by an experienced librarian (DZ). This search was then updated, on October 31, 2023, as part of another systematic review with similar inclusion and exclusion criteria (PROSPERO: CRD42023473768). As recommended by the PRESS checklist, this search strategy was then reviewed by another librarian. 27 Our search covered topics in the domains of ATSUD, modafinil and clinical trial, and was conducted in the following databases: MEDLINE (Ovid), CINAHL (EBSCOhost), PsycINFO (Ovid), EBM Reviews (Ovid), EMBASE (Ovid), and Web of Science. A search was also completed in the following clinical trial registries: clinicaltrials.gov, the International Clinical Trials Registry Platform. Finally, Google Scholar was also examined for the first 50 consecutive search records. There were no time/date restrictions and filters in the search process and all above-mentioned databases were searched from inception to the last available update on the search date. The references of included primary studies, systematic reviews, and meta-analyses were also screened to find additional candidate studies. The complete search strategy for this study is reported in Supplemental Material 1.
Study Screening and Selection
All identified RCTs of modafinil in individuals with ATSUD were included, whether published or unpublished. Other criteria of inclusion were: diagnosis of ATSUD following the Diagnostic and Statistical Manual of Mental Disorders-IV (DSM-IV) or DSM-5 for STUD, the use of modafinil as a treatment for ATSUD, and articles written in English, French, or Spanish. Individuals of any sex and gender, and of any age, were included in this study. Studies were excluded if they used a different form of clinical trials, such as open-label trials, human laboratory studies or conducted in a controlled environment, and animal studies, or if they were reviews, letters, editorials, and/or case reports. Studies were also removed if they excluded participants who were using ATSs during the study. Finally, trials with <10 participants in either the intervention or placebo arm were also excluded.
Duplicates were first removed in Endnote by DZ, using the method described by Bramer. 28 Search results were then uploaded to DistillerSR (Version 2.35). Study titles and abstracts were first independently screened by two authors (LE and HB), and conflicts were resolved through mutual consensus. The full texts of the remaining studies were then examined for eligibility by the same authors, according to the above-described eligibility criteria. Conflicts in study eligibility assessment were resolved through mutual discussions. The remaining disagreements were resolved through a consensus via consultation with the study supervisor (DJA).
Outcomes
A set of 9 target outcomes was initially described in the study protocol uploaded to the PROSPERO registry: ATS use as determined by urine analysis (UA), retention in treatment, ATS craving, treatment discontinuation due to AEs and SAEs, self-reported ATS use, addiction severity, treatment compliance, and depression symptom severity. These outcomes were selected based on the relevancy of outcomes, as well as what was thought to be available after preliminary searches. After data extraction, the available outcomes for meta-analysis were: ATS use as determined by UA, retention in treatment, ATS craving, and treatment discontinuation due to AEs and SAEs. The main outcome of interest was ATS use by UA. ATS use by UA was defined as a positive qualitative or quantitative UA for amphetamine and/or methamphetamine throughout the treatment phase of the study. UA results were pooled as the proportion of positive UAs by treatment arm. We included four secondary outcomes. Retention in treatment was measured via the proportion of participants completing the treatment course. ATS craving was measured by scales such as the visual analogue scale (VAS) or Brief Substance Craving Scale. 29 The mean and standard deviation (SD) of the last visit (treatment end) were included for data pooling. Treatment discontinuation due to AE was defined as the proportion of participants who discontinued treatment because of any AEs. Finally, SAE was defined as the number of SAEs in the studies, as reported and measured by the study authors.
Data Extraction
Data were independently extracted from the included studies by two study authors (LE and CM) using the spreadsheet included in (Supplemental Material 2). Authors of included studies were contacted if data were not directly extractable from the study report/article, not available online via data repositories, or if certain clarifications were required (see Supplemental Material 1 for outcomes of communications with authors). When appropriate, the WebPlotDigitizer V4.6 was used to extract data from study graphs. For the main outcome (ATS use), whenever it was possible, the non-imputed results extracted from the studies or obtained from the authors were imputed with positive UA results for the missing values. If the SD values for continuous variables were not accessible, they were imputed by the mean SD values from other studies (with the same measure and scale) included in this meta-analysis.
Risk of Bias and Evidence Certainty Assessment
Risk of bias assessment was conducted independently by two study authors (LE and HB), using the Risk of Bias Assessment tool (RoB 2) and Excel tool (Cochrane Collaboration). 30 Risk of Bias figures were produced using the robvis software. 31 Conflicts were resolved via mutual agreement or discussion with the study supervisor (DJA). The certainty of evidence was assessed by two authors independently (LE and HS). Authors followed the Grading of Recommendations, Assessment, Development and Evaluation (GRADE), 32 and conflicts were resolved via mutual agreement or discussion with the study supervisor (DJA).
Data Analysis
Random-effect (via inverse variance method) meta-analysis was run for any outcome with two or more poolable results, with p-values of ≤0.05 being considered statistically significant. For outcomes with a dichotomous variable, a risk ratio (RR) and a 95% confidence interval (CI) were calculated, whereas standardized mean differences (SMDs) were calculated for outcomes with continuous measures, as well as 95% CI. For study outcomes with rare events (<5%), we removed studies with zero values in both arms and calculated odds ratios (ORs) using the Peto's method, with a fixed effect model. All analyses were performed using the metapackage 33 in Rv4.3.1 (R Core Team). Subgroup analyses by trial medication dose (200 mg vs. 400 mg) were also conducted. While other subgroup analyses were proposed in the original proposal (such as by psychiatric diagnoses), these were not possible due to a lack of available data across studies or outcomes. For the main outcome (ATS use), a sensitivity analysis using originally extracted results, either non-imputed or imputed for missing values (missing UA as positive), was conducted.
Results
Search and Study Selection Results
The screening process is summarized in a PRISMA flowchart (Figure 1). We initially identified 1764 records via the search methods described above. A total of 1454 records were screened after the removal of 310 duplicates. After the abstract screening, 41 records were sought for full-text retrieval, but only 36 full reports were available and assessed for eligibility by the study authors. Among these, 5 studies met the full criteria to be included in the systematic review and meta-analysis. The most common reasons for exclusion were: wrong study design (23), duplicate report/registry of the same study population (6), and wrong article type (2). The updated search yielded no additional studies. See Supplemental Material 1, for full reasons for exclusion.
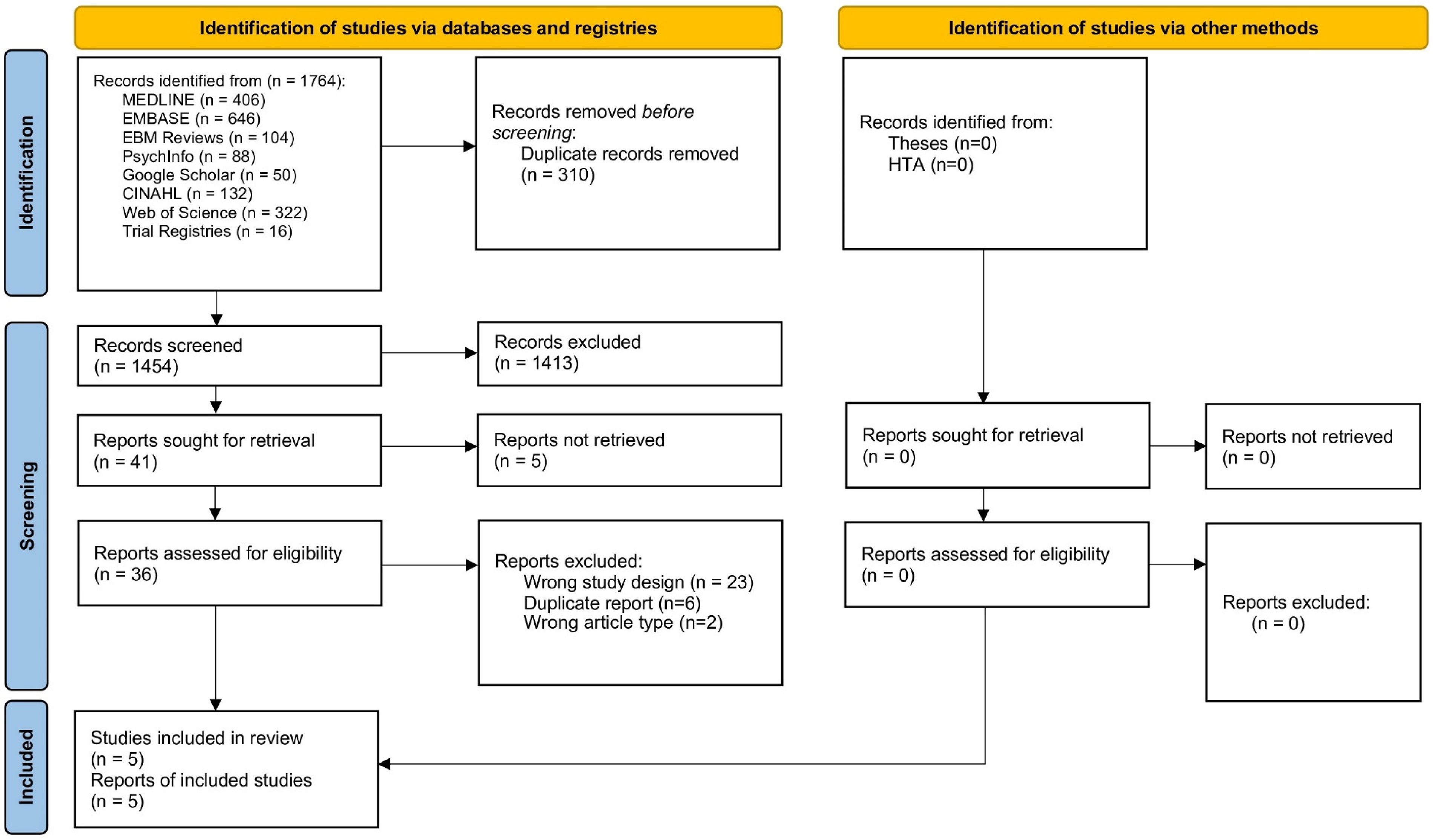
Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) 2020 flow diagram for results of search, screening, and eligibility assessment of studies.
Study Characteristics
Characteristics of studies and participants are described in Table 1. Five RCTs,19,20,34-36 including 1 unpublished work, 36 were included in this study. The total number of participants across studies was 451. The randomized treatment duration ranged from 4 weeks 36 to 12 weeks.20,34,35 All of the individuals in the included studies were aged 18 or older. The mean age ranged from 29.5 to 39.0 years and from 32.5% to 100% of participants were males (Table 1). Three studies included some form of cognitive behavioural intervention,19,20,34 and 1 study 20 also included contingency management as an additional intervention. Two trials19,35 studied daily doses of 200 mg of modafinil, with another 2 examining daily doses of 400 mg,20,36 and 1 study examining both 200 mg and 400 mg per day of modafinil. 34 Methamphetamine was the primary stimulant used by participants in all studies, with all studies including individuals with methamphetamine use disorder. One study 35 also included individuals with amphetamine use disorder.
Characteristics of the Included Studies.
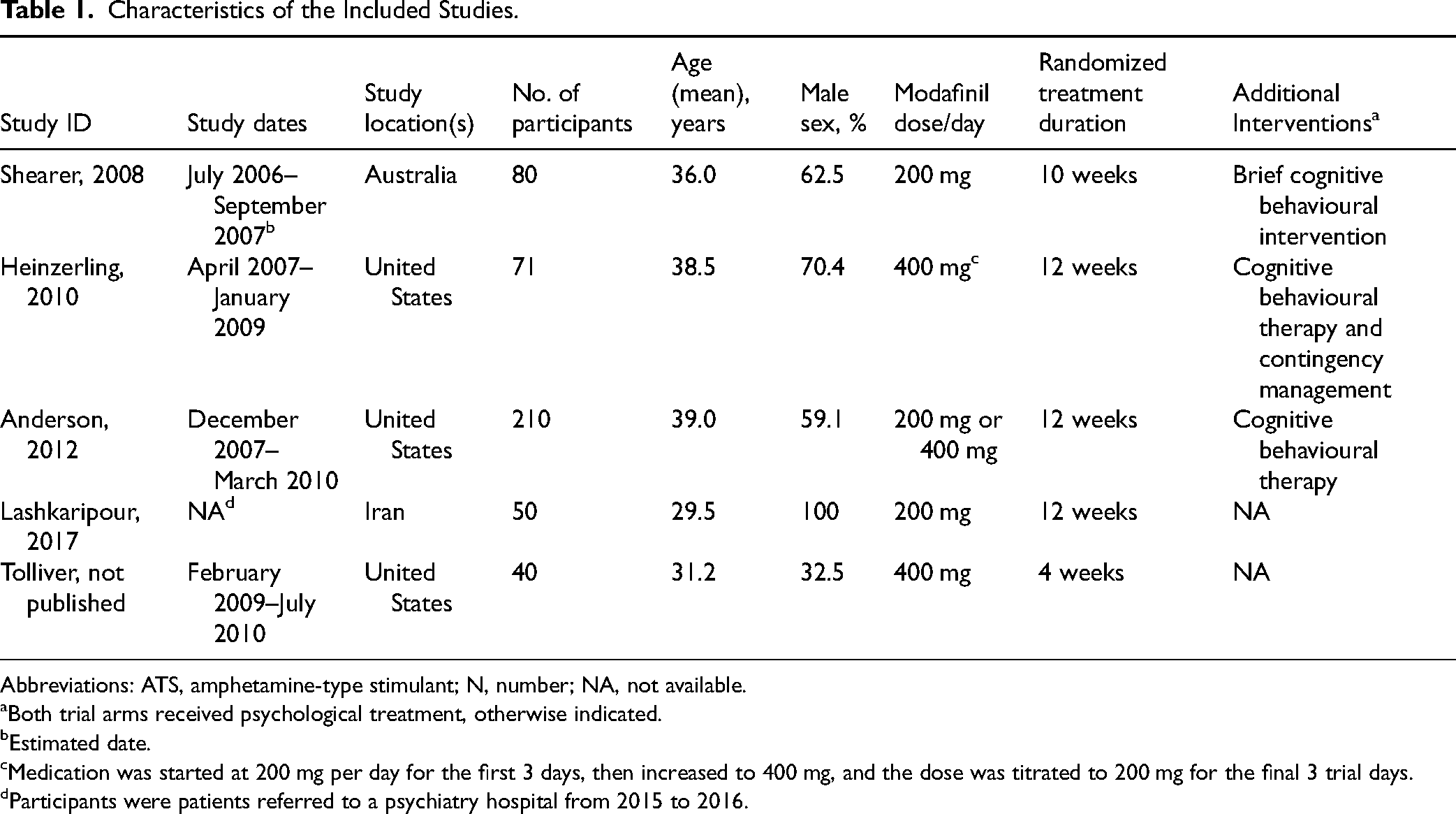
Abbreviations: ATS, amphetamine-type stimulant; N, number; NA, not available.
Both trial arms received psychological treatment, otherwise indicated.
Estimated date.
Medication was started at 200 mg per day for the first 3 days, then increased to 400 mg, and the dose was titrated to 200 mg for the final 3 trial days.
Participants were patients referred to a psychiatry hospital from 2015 to 2016.
Selected Outcomes
We included 5 outcomes in the systematic review and meta-analysis, all of which had data available for pooling in 2 or more studies: ATS use, retention in treatment, ATS craving, SAEs, and treatment discontinuation due to AEs. See Table 2 for an overview of the included outcomes and approaches used to analyze them in this meta-analysis. Due to data unavailability, the following outcomes were removed from the study: self-reported ATS use, addiction severity, and severity of depressive symptoms. Another pre-specified outcome, treatment compliance, was dropped because the pooling of available results was not possible.
Overview of outcomes assessed in the meta-analysis by the included studies and their analysis approach. a
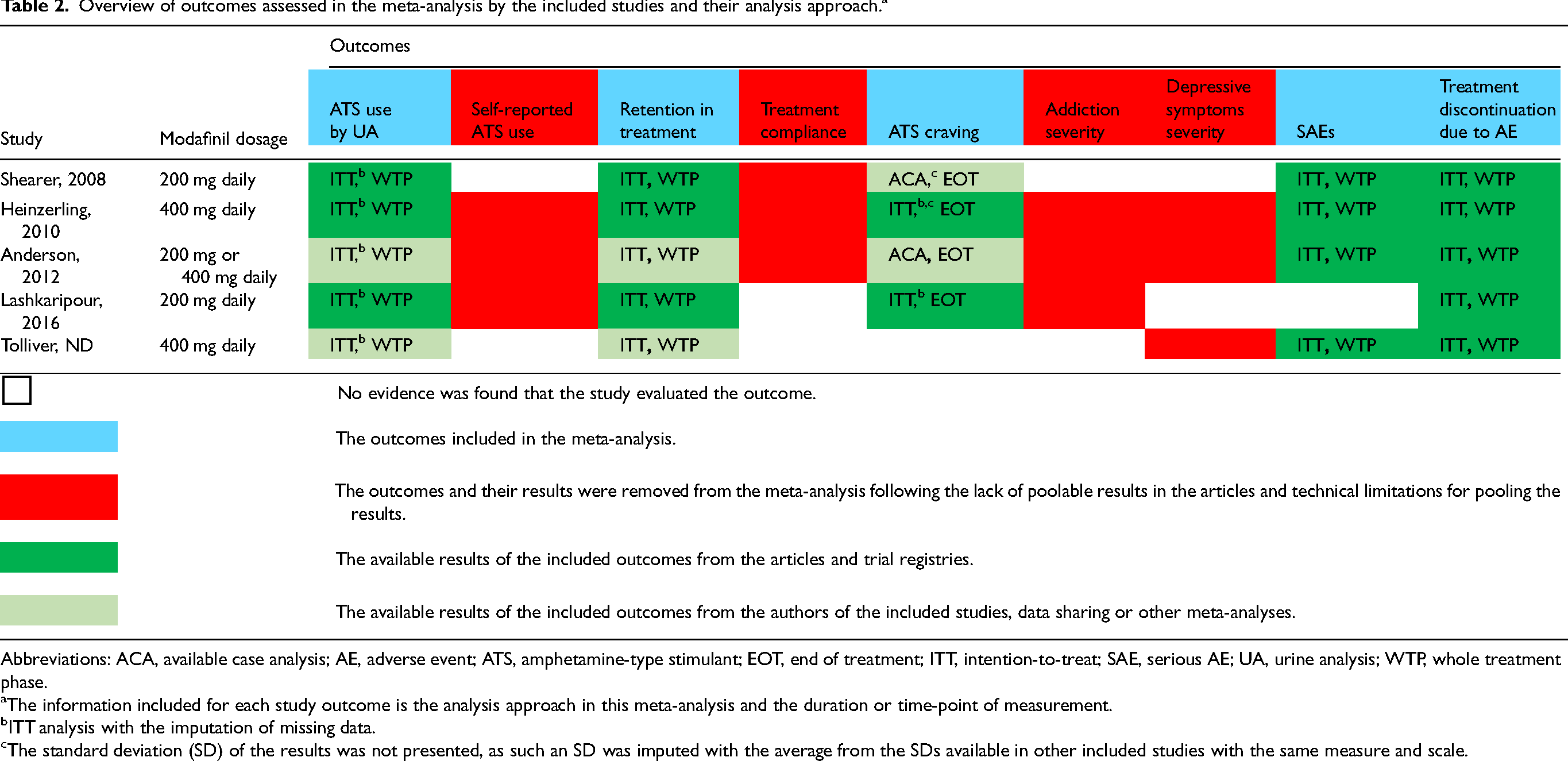
Abbreviations: ACA, available case analysis; AE, adverse event; ATS, amphetamine-type stimulant; EOT, end of treatment; ITT, intention-to-treat; SAE, serious AE; UA, urine analysis; WTP, whole treatment phase.
The information included for each study outcome is the analysis approach in this meta-analysis and the duration or time-point of measurement.
ITT analysis with the imputation of missing data.
The standard deviation (SD) of the results was not presented, as such an SD was imputed with the average from the SDs available in other included studies with the same measure and scale.
Effects of Intervention
Main Outcome
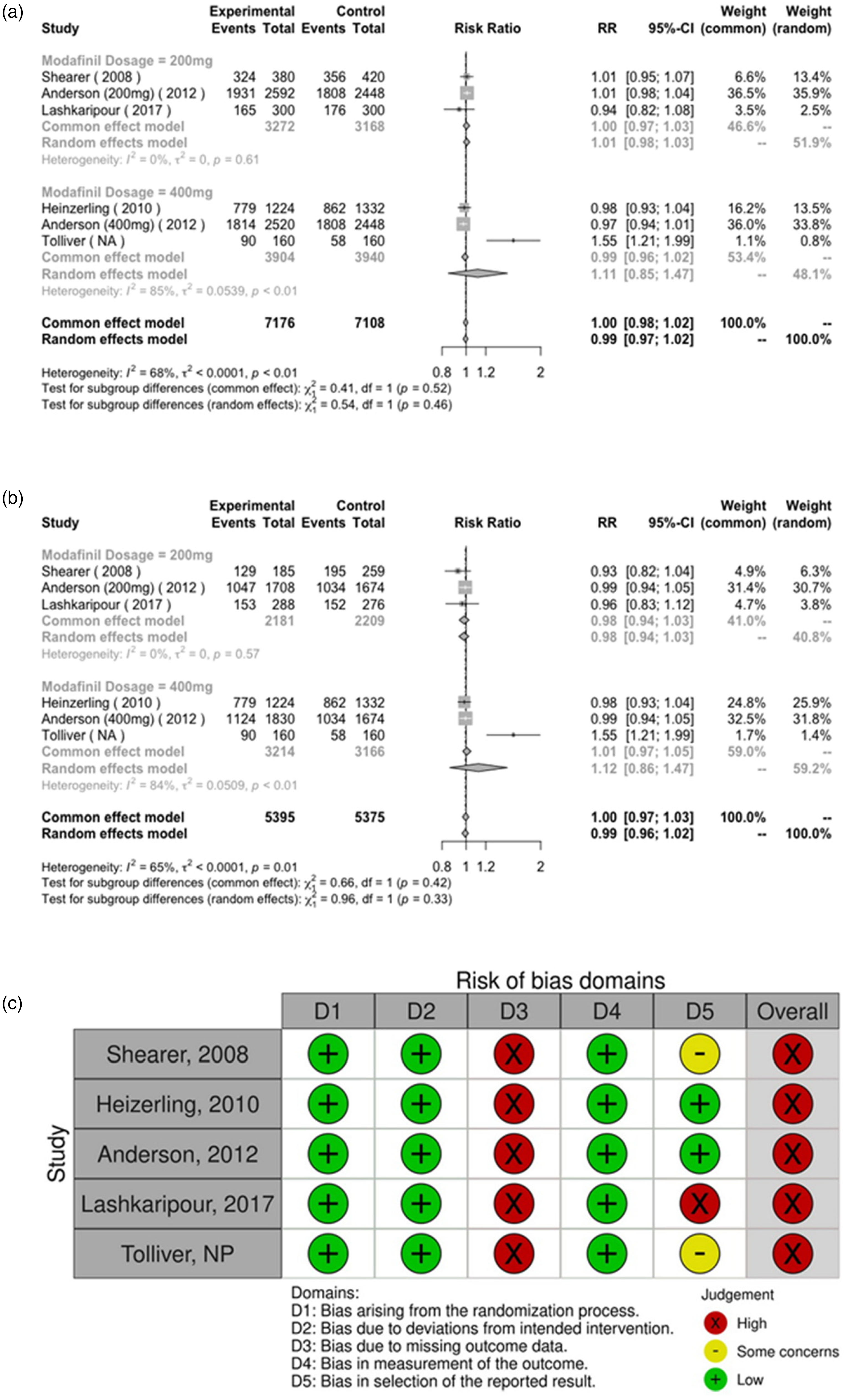
(a) Forest plot of the main analysis; (b) Forest plot of the sensitivity analysis, and (c) risk of bias assessment for amphetamine-type stimulant use by assessment of urine analysis.
Secondary Outcomes
Main results of the meta-analysis for the secondary outcomes (outcomes other than ATS use). a
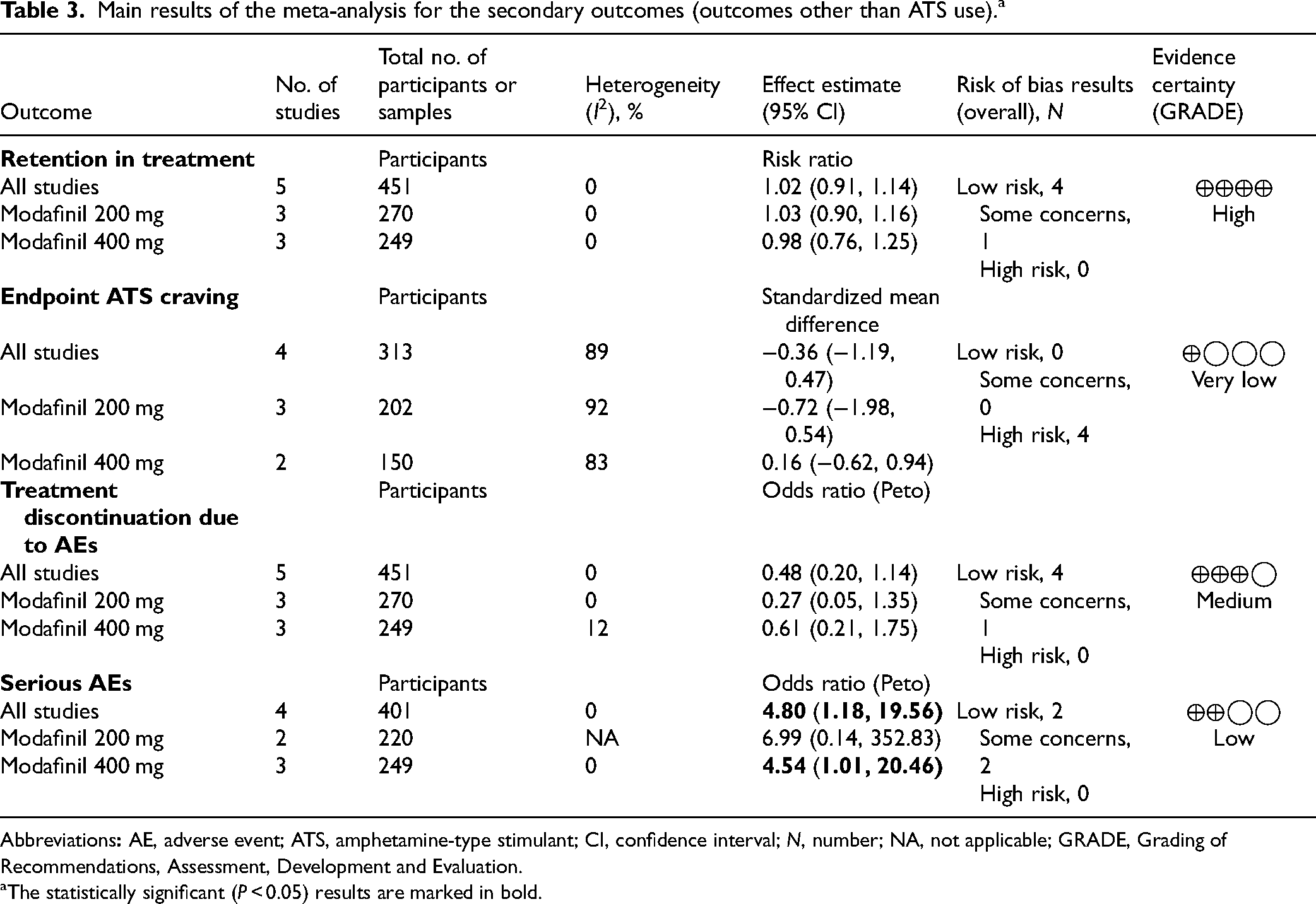
Abbreviations
The statistically significant (P < 0.05) results are marked in bold.
Discussion
In this study, we examined the effects of modafinil on various outcomes in adults with ATSUD by pooling data from available RCTs. Our meta-analysis reveals that modafinil does not appear to exert significant effects on multiple outcomes relevant to ATSUD management, including ATS use, retention in treatment, ATS craving, and treatment discontinuation due to AEs. Additionally, participants assigned to the modafinil group, especially at higher doses, experienced more SAEs. These results were consistent across sensitivity and sub-group analyses. To our knowledge, this is the first meta-analysis specifically assessing modafinil's effects on a broad range of outcomes in this specific population. Previous meta-analyses have either focused on a limited number of outcomes in stimulant-using populations (ATS and cocaine),13,23 or pooled the effects of various psychostimulant medications together. 21 Furthermore, we incorporated data from two studies not included in prior reviews.35,36 Overall, our findings suggest that, based on the available evidence, modafinil is not associated with positive outcomes in the management of ATSUD.
Our conclusions are bolstered by the low heterogeneity in 3 of the 5 outcomes of interest. This pattern could be partially explained by some of the similarities in the demographic characteristics of the various study populations. For example, 4 of the 5 studies,19,20,34,35 had a majority of male participants, and the mean ages across all studies were also quite similar. However, it was not possible to examine if these descriptive characteristics influenced our outcomes of interest, due to data unavailability. It should be highlighted that 1 study, 36 with a majority of female participants, is also the only study in which the main outcome, ATS use measured by UA, has a RR which favours placebo. A potential reason for this result may be due to the relatively short study period (4 weeks). The result may also be suggestive that a certain sex-related effect exists, the above-mentioned study was underpowered (N = 40) and also is the only study with unpublished results, which in itself could be a risk of bias. Moreover, all of the studies included for this outcome were rated as having a high risk of bias. This is primarily due to the domain 3 of RoB2, bias due to missing outcome data, being judged as having a high risk of bias for each study. Missingness is a major problem, particularly in substance use clinical research.37,38 It has been suggested that missing data, in the context of substance use research, may represent potential information with real-world or clinical implications. 38 Specifically, with regard to clinical outcomes such as drug use measured by UA, data are often labelled as missing not at random, considering the potential implications of a positive drug test.37,39 Such patterns of missingness influence the potential risk of bias and limit the quality of evidence in both primary and secondary (meta-analysis) research.
The significant heterogeneity in our primary outcome, ATS use measured by UA, could possibly be explained by the varying frequency of measurements of UA across studies (ranging from once a week to 3 times a week), as well as possible differences in measurement techniques (immunoassay versus mass spectroscopy). With regard to the outcome of ATS craving, the exhibited high heterogeneity, was possibly due to varying craving scales used across studies, and time frames questioned (i.e., craving in the past 24 h vs. craving in the past week). The study location also varied across studies, with 3 studies taking place in the United States,20,34,36 1 study taking place in Australia, 19 and 1 in Iran. 35 While this could also be considered a possible source of heterogeneity between studies, for all outcomes, it should be noted that all studies were conducted in an outpatient setting, at University or State funded hospitals. Other possible sources of heterogeneity between studies, such as the method of ATS administration, socioeconomic status, or even comorbid psychiatric symptoms, could not be considered as these data were not available in the examined studies.
The small CIs across the four non-significant findings further reinforce modafinil's lack of positive impact on ATSUD outcomes. In contrast, a recent meta-analysis has demonstrated that prescription psychostimulants—such as methylphenidate and dextroamphetamine—have a significant effect on various outcomes in an ATSUD population. 40 While modafinil's mechanism is yet to be completely elucidated, there is evidence to suggest that its mechanisms are distinct from that of “typical” stimulants (methylphenidate and amphetamines). 41
The only statistically significant finding was the significantly higher odds of reported SAEs in the modafinil arms, particularly at higher doses. SAEs reported in the studies were suicidal ideation by drug overdose, chest pain, and bacterial infection.20,34 While none of the studies reported that the SAEs were directly related to the study medication,20,34 the finding of increased odds of SAE is congruent with previous studies indicating that modafinil intake, especially at higher doses, is associated with increased AEs and SAEs.18,42,43 Preliminary evidence suggests that longer modafinil usage may correlate with a larger number of reported AEs. 44 Considering the lack of evidence for significant effects on the other clinical outcomes examined in this study, it may therefore be prudent to avoid longer usage and higher doses of modafinil in this particular at-risk population.
Examining the ATSUD population as distinct from other STUD, such as cocaine use disorder, is imperative given the significant differences between these disorders. These populations notably have distinct clinical and socio-demographic profiles.24,45 For example, methamphetamine-using populations reportedly exhibit more severe positive psychotic symptoms compared to cocaine users. 45 Moreover, in a recent draft guidance document, the Food and Drug Administration recommended that in conducting trials for STUD, one should differentiate between specific substance subgroups (i.e., amphetamines, methamphetamines, and cocaine). 46 One should also consider the distinct mechanisms of action of cocaine and ATS. A recent meta-analysis highlighted substantial reductions in dopamine transport availability in ATS users compared to cocaine users, 47 suggesting different underlying mechanisms between chronic ATS and cocaine use. Mechanistic disparities, evident in their differential impacts on dopamine, serotonin, and norepinephrine transport,48-51 are important when considering “dopamine-replacement” models of care. If consistent reductions in dopamine transporter availability are present in this population, this hints that strong inhibition of dopamine reuptake inhibitor, or by other pathways (e.g., increasing release) may be more apt for managing ATSUD compared to cocaine use disorder. This notion is further supported by meta-analytic findings associating higher doses of psychostimulants with better outcomes in the ATSUD population.13,40 These discrepancies potentially imply that the pharmacological management of ATSUD may differ from that of the cocaine use disorder population. However, the most recent ATSUD management guidelines are largely based on cocaine use disorder trials, 14 which underscores the necessity for ATSUD-focused research.
Our study delineates several strengths and limitations that warrant discussion. A primary strength lies in the rigorous methodological framework adhered to, following established PRISMA guidelines for systematic reviews and meta-analyses, thereby enhancing the reliability and validity of our findings. Moreover, focusing on a distinct ATSUD population addresses a significant literature gap, offering a more accurate understanding of modafinil's efficacy in this demographic. In contrast, the short-term trial designs give us little insight into modafinil's long-term efficacy and safety for ATSUD. However, the lack of effectiveness of treatment after more than 2 months suggests that longer-term studies may be unnecessary. The high-risk bias in some outcomes also challenges the interpretability of our results. The relatively small number of included studies and the modest sample size could potentially limit the generalizability of the findings. Moreover, we were unable to study many outcomes initially identified in the study protocol, such as self-reported ATS use, addiction severity and depressive symptom severity, due to the unavailability of data. It should be noted that raw data were not made available to us for most studies. This limited conduct of certain sub-group analyses on demographic or other variables of interest, such as analyses by sex or age, or method of ATS use. Furthermore, the high risk of bias across the study outcomes makes it difficult to assess the certainty of the results in the reported studies. Our study was also limited by our inability to study the effects of modafinil as a harm-reduction option (i.e., improved quality of life and improved psycho-social functioning) or substance-use reduction in this population. While these would have been very relevant given the current movement towards harm and substance-use reduction strategies in the management of substance-use disorders overall, it was impossible to study in our context as outcomes such as these were not examined in the identified studies. Finally, the lack of data on psychiatric comorbidities and concurrent substance use may have influenced the study outcomes, making it difficult to draw firm conclusions about modafinil's efficacy across different sub-groups within the ATSUD population.
Conclusion
This systematic review and meta-analysis aimed to critically examine the efficacy and safety of modafinil as a therapeutic intervention for individuals with ATSUD. Our findings demonstrate that there is currently no evidence suggesting that modafinil has a statistically significant effect on a variety of efficacy outcomes in populations with ATSUD. The lack of effective pharmacological interventions for ATSUD remains a significant challenge, emphasizing the critical need for further well-designed, long-term RCTs while testing other potential molecules and interventions. Our study underscores the importance of not extrapolating findings from cocaine-using populations to those with ATSUD, as the pharmacological management may significantly differ between these groups. Future research efforts should be directed towards understanding the unique physiological and psychological challenges faced by individuals with ATSUD, which in turn could inform the development of more effective and safer pharmacological interventions. These endeavours are imperative for advancing the clinical management of ATSUD, ultimately aiding in reducing the morbidity and mortality in ATSUD populations.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437241262967 - Supplemental material for Efficacy and Safety of Modafinil for Treatment of Amphetamine-Type Stimulant Use Disorder: A Systematic Review and Meta-Analysis of Randomized Placebo-Controlled Trials: Efficacité et innocuité du modafinil pour le traitement des troubles liés à l'usage de stimulants de type amphétamine : revue systématique et méta-analyse d'essais randomisés contrôlés par placebo
Supplemental material, sj-docx-1-cpa-10.1177_07067437241262967 for Efficacy and Safety of Modafinil for Treatment of Amphetamine-Type Stimulant Use Disorder: A Systematic Review and Meta-Analysis of Randomized Placebo-Controlled Trials: Efficacité et innocuité du modafinil pour le traitement des troubles liés à l'usage de stimulants de type amphétamine : revue systématique et méta-analyse d'essais randomisés contrôlés par placebo by Laurent Elkrief, Heidar Sharafi, Hamzah Bakouni, Christina McAnulty, Gabriel Bastien, Simon Dubreucq, Nicolas Garel, Annie Trépanier, Daniela Ziegler and Didier Jutras-Aswad in The Canadian Journal of Psychiatry
Footnotes
Acknowledgements
We would like to express our kindest gratitude to James Shearer and Brian Tolliver for providing us with the information and results of their studies.
Author contributions
Conceptualization: LE, HS, and DJA; methodology: LE, HS, HB, CM, GB, DZ, and DJA; formal analysis: LE and HS; investigation: LE, HS, HB, GB, and CM; writing—original draft: LE, HS, HB, CM, GB, NG, AT, SD, and DJA; writing—review and editing: LE, HS, HB, CM, GB, NG, SD, AT, DZ, and DJA; supervision: DJA; project administration: LE, HS, and DJA; funding acquisition: DJA.
Data Access Statement
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LE is a shareholder and employee of OneCare Inc., which is a biotechnology mental health company. OneCare Inc., biotechnologies’ work does not relate to the contents of the present article. DJA received study materials from Cardiol Therapeutics and Exka for clinical trials not related to the present article (2022–2024). The other authors have no potential conflicts to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This meta-analysis was supported by the Canadian Institutes of Health Research (CIHR) (grant number REN-181675), Université de Montréal and Centre de recherche du Centre Hospitalier de l’Université de Montréal (CRCHUM). The study sponsor had no role in study conceptualization, data acquisition and analysis, interpretation of results, and manuscript drafting and editing. GB reports a Doctoral Award from the Fonds de recherche du Québec—Santé (FRQS); DJA is supported by a research career award from the FRQS.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
