Abstract
Background
Schizophrenia spectrum disorders (SSDs) are a group of psychiatric disorders characterized by positive and negative symptoms as well as cognitive impairment that can significantly affect daily functioning.
Method
We reviewed evidence-based strategies for improving cognitive function in patients with SSDs, focusing on the Canadian landscape.
Results
Although antipsychotic medications can address the positive symptoms of SSDs, cognitive symptoms often persist, causing functional impairment and reduced quality of life. Moreover, cognitive function in patients with SSDs is infrequently assessed in clinical practice, and evidence-based recommendations for addressing cognitive impairment in people living with schizophrenia are limited. While cognitive remediation (CR) can improve several domains of cognitive function, most individuals with SSDs are currently not offered such an intervention. While the development of implementation strategies for CR is underway, available and emerging pharmacological treatments may help overcome the limited capacity for psychosocial approaches. Furthermore, combining pharmacological with non-pharmacological interventions may improve outcomes compared to pharmacotherapy or CR alone.
Conclusion
This review highlights the challenges and discusses the potential solutions related to the assessment and management of cognitive impairment to help mental health-care practitioners better manage cognitive impairment and improve daily functioning in individuals with SSDs.
Plain Language Summary Title
Improving Thinking Skills in People With Schizophrenia: A Focus on Canada
Plain Language Summary
Schizophrenia spectrum disorders (SSDs) are mental health conditions that can cause a mix of symptoms making everyday life difficult for people. For example, some people might experience positive symptoms like seeing and hearing things that are not there, having false believes, or feeling suspicious. Others might experience negative symptoms like isolating from social interactions, having trouble showing emotion, or finding it hard to connect with others. Finally, they might also have trouble with thinking skills, like paying attention, planning and organizing, remembering things, or understanding social cues. While some medicines can help with some of the symptoms, they often do not help with improving thinking skills. In addition, these thinking problems are not checked often enough in patients with SSDs. In fact, there are not many proven ways to help with these issues. One method that can help with thinking skills is called cognitive remediation (CR), but it is not offered to most people with SSDs. There are ongoing efforts to make CR more available. It is also possible that CR may be combined with new medicines to make it work better than using just one treatment. This article looks at ways to improve thinking skills in people with SSDs, with a focus on what is happening in Canada. We present the challenges and possible solutions for checking and managing thinking problems, to help health-care workers better care for people with SSDs.
Introduction
Schizophrenia spectrum and other psychotic disorders (SSDs) are a group of potentially enduring psychiatric disorders characterized by positive symptoms (hallucinations, delusions, suspiciousness), negative symptoms (social withdrawal, diminished emotional expression, poor rapport, lack of spontaneity), and cognitive impairments (attention, processing speed, executive functions, psychomotor speed, memory, and social cognition) that can significantly impact daily functioning (Figure 1).1,2 According to national data, 1% of Canadians aged 10 years and older were diagnosed with schizophrenia between 2016 and 2017. 3 SSDs are associated with increased all-cause mortality and increased use of health-care services, posing a considerable challenge to the Canadian health-care system. 3
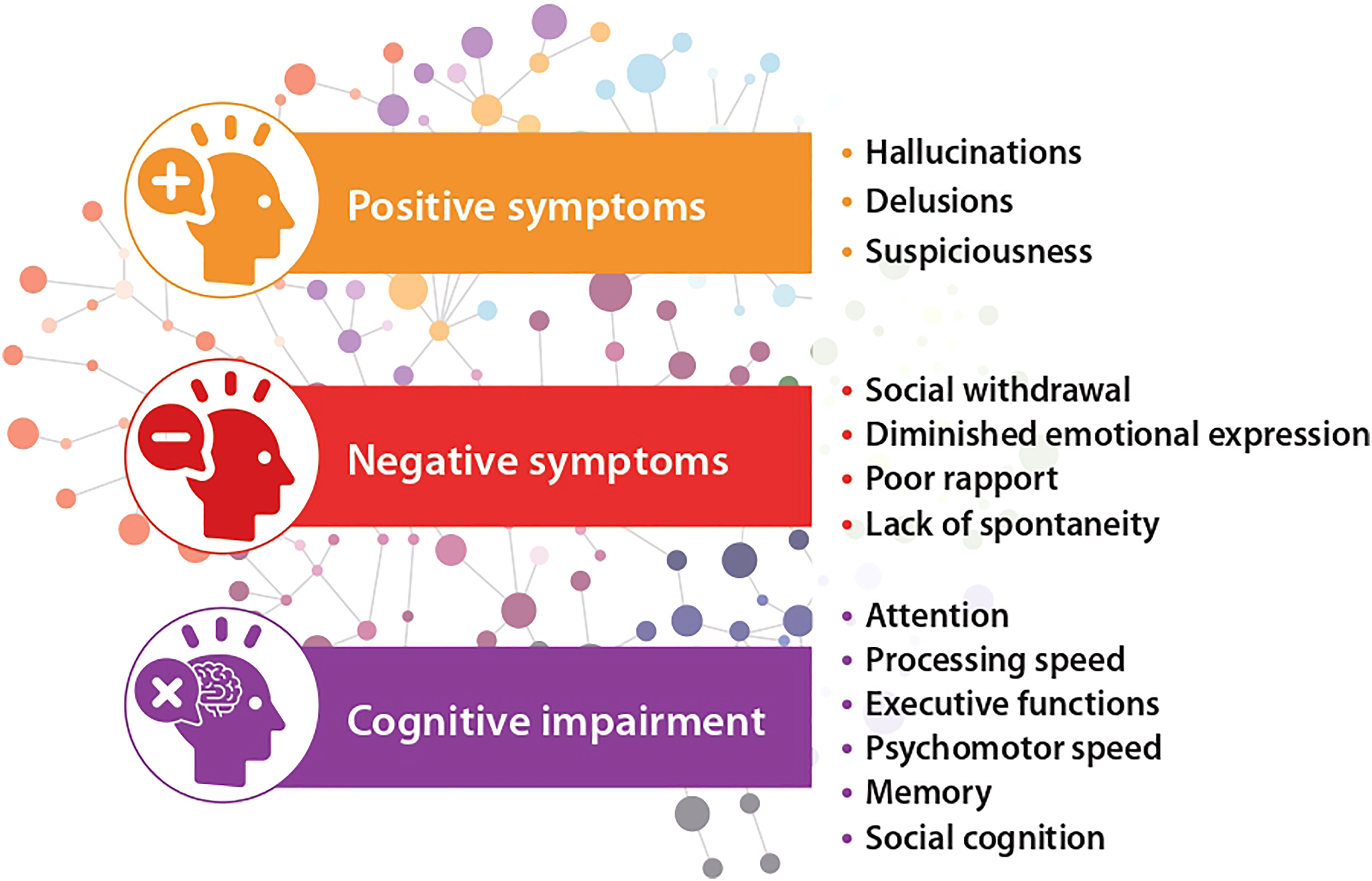
Positive, negative, and cognitive symptoms of schizophrenia spectrum disorders.
Kraepelin's concept of dementia praecox, which is characterized by a persistent cognitive impairment early in life marked by rapid cognitive decline and disruption in attention and memory, played a key role in distinguishing schizophrenia from other psychiatric disorders. 4 Cognitive impairment is associated with functional impairment across different phases of schizophrenia 5 and negatively affects the quality of life (QOL).5,6 Although antipsychotic treatment can help address the positive symptoms of SSDs, cognitive impairment often persists, posing significant barriers to recovery. 7 Cognitive impairment should be a treatment priority for people with psychosis; 8 however, cognition is infrequently assessed in clinical practice, and improving cognitive function in patients with SSDs is often not a priority perhaps because clinicians have few or no treatments to offer. 9 The Canadian guidelines for the management of schizophrenia have not been updated since 2017,10,11 and evidence-based recommendations for addressing cognitive impairment in people living with SSDs are limited. The European Psychiatric Association has published guidelines for the assessment and treatment of cognitive impairment in schizophrenia,12,13 but these guidelines are not tailored to the Canadian health-care system.
In this article, we review evidence-based approaches for improving cognitive function in patients with SSDs. We discuss challenges and potential solutions related to the assessment and treatment of cognitive impairment. This comprehensive overview of evidence-based best practices aims to help Canadian psychiatrists, psychologists, and other mental health practitioners better manage cognitive impairment in patients with SSDs.
Prevalence and Mechanisms of Cognitive Impairment in Patients with SSDs
A Canadian study measuring cognitive impairment in 121 patients with schizophrenia showed that most (85%) participants had at least mild cognitive impairment, the extent of which was correlated with the severity of illness and negative symptoms. 14 Cognitive impairments typically begin early, sometimes before an SSD diagnosis is established, and do not improve spontaneously.15,16 It affects non-social cognitive domains (processing speed, working memory, problem solving) and social cognition domains (emotion processing, social perception, and mentalizing). 17 When combined, these deficiencies are thought to affect functional outcomes, such as community functioning (social activities, work, independent living) and ability to acquire skills. While there are speculations on the potential role of social isolation and psychological factors in mediating cognitive changes, data remain scarce.17,18
Similarly, the neurobiological mechanisms underlying cognitive impairment in SSDs are not fully understood. The neurodevelopmental hypothesis of schizophrenia suggests that cognitive impairments may be related to neurodevelopmental abnormalities as cognitive deficits are often present before the onset of psychosis. 19
Hypofunction of glutamate transmission in the cortex and excessive synaptic pruning have been associated with cognitive impairment and negative symptoms in SSDs. 20 Dysregulation of the dopamine system has also been implicated. 21 Excessive dopamine activity in brain regions, such as the striatum, may contribute to cognitive deficits, particularly in tasks requiring cognitive flexibility and response inhibition. 22 Furthermore, studies have shown reduced muscarinic receptor binding in the prefrontal cortex of individuals with schizophrenia, suggesting a role for the dysfunction of cholinergic neurotransmission. 23 Oxidative stress, inflammation, and imbalances in neurotrophic factors, such as brain-derived neurotrophic factor (BDNF), have also been associated with cognitive impairment in SSDs.24,25
Alterations in functional networks and neural synchronization have also been implicated in cognitive impairment in individuals with SSDs.26–29 Alterations in functional connectivity within and between the default mode and cognitive control networks have been identified as a basis for impairments in schizophrenia.26,27 Evidence suggests that synchronous oscillations may underlie cognitive functions, such as object perception, selective attention, and working memory, and may be impaired in patients with schizophrenia. 28 Comparisons of functional connectivity networks between individuals with schizophrenia and healthy adults further support the role of network connections in cognitive control. 29
Assessment of Cognitive Function in Patients with SSDs
Importance of Cognitive Assessment and Commonly Used Tests
Assessing cognitive function in patients with SSDs is essential for identifying areas of strength and impairment and understanding the impact of cognitive impairment on real-life functioning. By identifying and assessing cognitive impairment, clinicians can develop targeted interventions, implement measurement-based care, and monitor treatment response to improve the level of functioning and overall well-being of patients with SSDs.
Various standardized neuropsychological tests are recommended to assess cognitive domains in individuals with SSDs, 30 including the Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery (MCCB) 31 and Brief Assessment of Cognition in Schizophrenia (BACS) 32 (Table 1). Interview-based measures, such as the Schizophrenia Cognition Rating Scale (SCoRS), can be used to evaluate cognitive abilities related to daily functioning. 34 Subjective cognitive measures, such as the Subjective Scale to Investigate Cognition in Schizophrenia (SSTICS), do not always correlate highly with objective assessments. 36 Although useful, standardized neuropsychological tests and interview-based assessments have many limitations, including high costs, limited availability, and the need for trained specialists to administer them. Newer digital tools, such as smartphone-based cognitive assessments, have shown promise in monitoring cognitive function and may help overcome some limitations of standardized neuropsychological tests.39,40
Commonly Used Neuropsychological and Interview-based Tests for Assessing Cognitive Function.a

Note. BACS = Brief Assessment of Cognition in Schizophrenia; CANTAB = Cambridge Neuropsychological Test Automated Battery; MATRICS = Measurement and Treatment Research to Improve Cognition in Schizophrenia; MCCB = MATRICS Consensus Cognitive Battery; SCoRS = Schizophrenia Cognition Rating Scale; SSTICS = Subjective Scale to Investigate Cognition in Schizophrenia.
This is not an exhaustive list.
The Diagnostic and Statistical Manual (DSM-5-TR) recognizes cognition as an essential domain for evaluation during diagnostic assessments. It recommends a systematic assessment of the neurocognitive domains identified by the MATRICS initiative at all stages of the disorder, including individuals at risk for psychosis, and emphasizes the importance of early assessment in the development of effective interventions. 30 The importance of documenting cognitive impairment with neuropsychological testing for patients with a first episode of psychosis and for those with poor response to treatment is highlighted in the Canadian guidelines. 1 Standardized neurocognitive tests, such as the MCCB, can be administered by any appropriately trained mental health professional. However, in Canada, it is recommended that neurocognitive test results be interpreted by a neuropsychologist or a clinical psychologist.
Challenges and Potential Solutions
A comprehensive neuropsychological assessment of cognitive performance is time-consuming and requires multiple tests and specialized expertise (Table 2). Most mental health professionals do not have the appropriate training to undertake these and often rely on subjective self-report scales, which may not accurately capture objective cognitive difficulties. 50 In addition, resource availability may limit access to cognitive function assessments. A workflow that supports the adoption of cognitive function assessments in routine care for people with SSDs could increase systematic evaluations. The implementation of brief cognitive tests or screening tools (e.g., BACS) at intake may help start a conversation about patients’ functional goals. 51
Key Challenges and Potential Solutions for Improving Cognitive Health in Individuals with SSDs.

Note. BACS = Brief Assessment of Cognition in Schizophrenia; CDSS = Calgary Depression Scale for Schizophrenia; CR = cognitive remediation therapy; DWP = Dealing with Psychosis Toolkit; SCIP = Screen for Cognitive Impairment in Psychiatry; SSDs = schizophrenia spectrum disorders.
The advent of new pharmacological and non-pharmacological interventions to enhance cognition in SSDs may provide an impetus to change this clinical gap and improve the quality of care. Health-care teams must work closely with patients and family members to emphasize the importance of enhancing cognitive function in relation to recovery goals. Adopting language, such as “cognitive health,” instead of specific psychosis symptoms may help patients become more receptive to conversations about cognitive impairment. These conversations may include questions about the level of functionality for current daily activities and the patient's recovery and personal goals (e.g., living independently, obtaining employment, or returning to school), which should be at the centre of disease management. Using goal-related questions and cognitive screening results will help clinicians develop a more personalized measurement-based management approach. An example of this approach is Dealing with Psychosis Toolkit (DWP), 41 a collaborative initiative of the British Columbia Ministry of Health and Fraser Health to engage with individuals with SSDs, help them set goals and take appropriate steps toward these goals, and monitor their progress.
Non-Pharmacological Strategies for Improving Cognitive Function in Patients with SSDs
Principles and Efficacy of Psychoeducation
Psychoeducation has an important role in the management of cognitive impairment in patients with SSDs and in empowering patients and their families to better understand the causes of functional difficulties. 52 Providing patients with information about their illness, its impact on cognitive function, and available treatment options can enhance their understanding and motivation to engage in cognitive remediation (CR). 52 Educating patients about the relationship between cognitive impairment and functional outcomes also helps them recognize the importance of addressing cognitive impairment in their treatment plan. 53 By empowering patients with knowledge about their illness, psychoeducation contributes to better cognitive outcomes related to social skills, work functioning, social performance, and overall well-being.54,55 Psychoeducation can also benefit family members as it reduces caregiver burden and improves communication. 56
Principles and Efficacy of CR
Non-pharmacological interventions designed to directly address cognitive impairment, such as CR, offer additional avenues to enhance cognitive function in patients with SSDs. CR is an evidence-based approach involving structured exercises that aim to improve various cognitive domains and can be tailored to individual needs.57,58 Several CR platforms and programs are widely used across various clinical populations. CR leads to small-to-moderate improvements in several domains of cognition and a small improvement in functioning in individuals with schizophrenia.59,60 People using CR report improvements in their attention, problem solving, memory, motivation, social engagement, communication skills, independence, and daily life activities. 59 CR has been found to enhance verbal memory and overall cognitive function in at-risk individuals. 61 Clinical trials have demonstrated that CR can lead to meaningful improvements in various domains of cognitive function in patients with schizophrenia, including global cognitive performance, attention, verbal memory, functional capacity, negative symptoms, and self-rated QOL. 62 Additionally, intensive auditory CR enhanced verbal memory and overall cognitive function in at-risk individuals. 63
Neurocognitive and social-CR treatments aiming to support individuals to adopt strategies to compensate for cognitive difficulties have proven effective in improving learning, understanding of social situations, and functioning in youth at high risk for psychosis. 64 Self-efficacy training can help individuals increase overall competency, 65 and occupational therapy and supported employment can help individuals learn new skills. 66 Psychotherapy may help improve multiple domains of cognitive function in individuals with schizophrenia, 67 but larger and well-controlled studies are needed to confirm the ability of psychotherapy to improve cognition in patients with SSDs.
The Canadian Agency for Drugs and Technologies in Health (CADTH) conducted a qualitative review to determine experiences with accessing CR, expectations of CR, and experiences engaging with CR among people living with schizophrenia. 68 It identified that the presence of an encouraging, empathetic, and friendly clinician is key to the beneficial views of CR. 68 Well-received characteristics of CR include self-paced tasks that gradually increased in difficulty and tasks involving the use of technology and playing games. 68 Integrative CR programs that include a discussion group to help apply acquired cognitive skills to everyday life appear to be more effective in improving global cognition and verbal memory. 59 Additionally, the programs that include strategy coaching are more effective in enhancing verbal and visual learning and episodic memory. 59 Several initiatives in Canada69,70 are currently taking place to develop implementation strategies for CR.
Usefulness of Lifestyle Modifications
Lifestyle modifications, including regular physical exercise, adequate sleep, and a healthy diet, may have beneficial effects on cognitive health. 71 Controlled trials have shown significant positive effects of physical exercise on the global cognitive performance, working memory, attention, vigilance, and social cognition. The amount of exercise was correlated with larger cognitive gains, particularly when supervised by a trained professional. 72 Engaging in exercise improves cardiovascular health, neuroplasticity, and neurotrophic factors in the brain. 73 However, further research is needed to determine the optimal type, duration, and intensity of exercise for improving cognitive function in individuals with SSDs.
Canadian Guidelines on Non-Pharmacological Interventions for SSDs
According to the Canadian guidelines on non-pharmacological interventions for SSDs, all patients must be offered non-pharmacological interventions, if available, and CR may be considered for individuals with persisting cognitive impairment. 10 According to the guidelines, education about the nature and treatment of schizophrenia, in combination with other treatments, is an integral part of disease management. 10 In addition, psychoeducation, problem solving, and communication skills should be part of the training offered to all family members. 10
Barriers to Adoption of Non-Pharmacological Strategies
Despite promising findings regarding the effects of various education and CR approaches, access to even basic disease education interventions must be improved. 10 Organizational change and a collaborative approach may aid in the integration of psychoeducation into SSD care (Table 2). Psychoeducation must ideally be tailored to the stage of the disease and patient characteristics to fully realize its potential. 74 In the early stages of the disease, psychoeducation should include discussion about the illness, its symptoms, and available non-pharmacological and pharmacological approaches for managing schizophrenia symptoms and cognitive impairment and preventing relapse. 75 In later stages, psychoeducation should address issues, such as managing co-occurring medical conditions, maintaining adherence to treatments, and managing residual or persistent symptoms using coping strategies. 76 Tailoring psychoeducation to patient characteristics should consider individual differences in cognitive abilities, social support networks, and cultural backgrounds. For example, individuals with schizophrenia and borderline intellectual disability may benefit from psychoeducational groups that are creatively delivered and adapted to their specific needs. 77 However, adopting an individualized psychoeducation approach can be challenging. A potential solution to increasing adoption of self-management approaches and optimizing the effective delivery of psychoeducation for patients and their families is to train psychiatrists and their teams on when and how to initiate disease education and how to continue to motivate patients. The Health Quality Ontario quality standard 42 can be used to advocate for the delivery of self-management training for adults with SSDs and their families.
CR is well validated and recommended in the Canadian guidelines; 10 however, determining the optimal duration and CR characteristics is challenging. Appropriate delivery of CR is crucial, 57 and many clinicians lack adequate training or capacity to effectively deliver it. Although prioritizing individuals who would benefit the most from CR could help address the limited capacity and staffing, it remains unclear which patients are more likely to benefit. 78 Providing extensive training and coaching to various mental health professionals, including occupational therapists and social workers, could help increase the capacity for CR. A stepped-care model that includes psychoeducation for all patients as a first step, engagement in group CR as the second step, and individual CR as the most intensive step could help overcome limited capacity and staffing. Psychoeducation can also be offered to patients waiting to undergo CR. Providing training for family members on supporting individuals with cognitive impairment could also help improve cognitive health and overcome some of those challenges.
Patients with SSDs face barriers to adopting lifestyle modifications, including a lack of awareness about the benefits of physical exercise, adequate sleep, and a healthy diet, as well as the absence of necessary skills to implement these modifications. 43 The high prevalence of obesity and metabolic syndrome in individuals with SSDs, poor eating habits, and insufficient physical activity pose an additional obstacle to the success of lifestyle modifications. 44 A comprehensive and multidisciplinary approach is needed to increase awareness of the importance of lifestyle modifications among patients with SSDs and equip them with the necessary skills to implement these changes. 43 Education on lifestyle modification must be provided to all individuals with SSDs, with a focus on addressing existing metabolic abnormalities and monitoring progress to ensure effectiveness. 45 Patient referral to a sleep specialist or dietician may help improve the specificity and implementation of lifestyle modifications.
Pharmacological Strategies for Improving Cognitive Function in Patients with SSDs
Usefulness of Pharmacological Interventions
Pharmacological interventions can be used to manage some of the symptoms of schizophrenia, and the Canadian guidelines recommend antipsychotic medications considering the potential benefits and side effects of each drug. 11 However, these guidelines lack specific guidance on the use of antipsychotics for cognitive symptoms, 11 and there are currently no medications approved in Canada for the treatment of cognitive impairment in schizophrenia. In fact, the current pharmacological treatments alone provide minimal improvements in cognitive performance48,79,80 despite suggestions that some second-generation antipsychotics (SGAs), such as clozapine, olanzapine, quetiapine, and risperidone, may exert potential beneficial effects (or less negative effects) on cognition as compared to first-generation antipsychotic medications.81–83
Adjunctive pharmacotherapy with cholinergic agents may provide moderate improvements in some domains of cognitive function in patients with SSDs. 84 Xanomeline–trospium (KarXT) is an emerging treatment for schizophrenia that combines the muscarinic receptor agonist xanomeline with the peripherally restricted muscarinic antagonist trospium to minimize peripheral cholinergic effects.85,86 Clinical trials have shown that treatment with KarXT improves cognitive function in patients with schizophrenia-associated cognitive impairment. 87 However, evidence on the effects of KarXT on cognitive function is derived from a single post-hoc analysis, and only participants with the worst cognitive functioning at recruitment had an improvement compared to placebo. 87 KarXT was recently submitted for FDA approval for schizophrenia. 88
Iclepertin (BI 425809) is a potent and selective GlyT-1 inhibitor being developed for the treatment of cognitive impairment associated with schizophrenia. 89 A phase-2 study demonstrated that add-on treatment with iclepertin at doses of 10 mg and 25 mg is safe, well tolerated, and improves cognition in patients with schizophrenia on stable antipsychotic treatment. 90 Three phase-3 trials are ongoing to confirm the effects of iclepertin on cognition and daily functioning in individuals with schizophrenia. 91
Barriers to Improving Cognitive Function in Individuals with SSDs Using Pharmacological Interventions
Thus far, studies on the cognitive effects of antipsychotics are inconclusive because many studies were non-randomized or non-controlled trials, and their results are not easy to interpret. 92 Patient concerns that medications cause cognitive decline are another barrier to cognitive improvement with pharmacotherapy. It is important to limit the use of anticholinergic and benzodiazepine medications as their long-term use can negatively influence cognitive outcomes.93,94 Anticholinergic burden assessment tools, such as the Drug Burden Index, 49 can be useful in mitigating anticholinergic burden in patients with SSDs.
Many individuals with SSDs self-medicate with substances, including cannabis and alcohol, which can pose challenges in the effective management of cognitive impairment. 95 Clinicians should make efforts to non-judgementally open lines of communication regarding the use of these products and, if needed and possible, refer patients to a specialist for substance use treatment. Furthermore, there is controversy regarding the use of stimulants to improve attention. 96 A small case series showed that stimulants may help reduce clozapine-induced sedation in patients with treatment-resistant schizophrenia. 97 However, evidence from large placebo-controlled trials is lacking, and clinicians should consider the risk-benefit (e.g., risk of psychosis) before prescribing stimulants.
Future Perspectives
With more evidence-based treatments becoming available for SSDs, it is vital to prioritize interventions that improve cognitive functioning and continue to develop more effective treatments. Further research is needed to explore the long-term effects of currently available strategies for improving cognitive function and to identify and validate biomarkers that predict response to cognition-targeting interventions. Future research efforts should focus on understanding the role of neurotransmitters in cognitive function in individuals with SSDs, as well as on developing novel pharmacological agents that target specific cognitive domains.
More research is also needed to refine existing non-pharmacological approaches and further explore promising ones to optimize cognitive health. As studies on CR are heterogeneous in terms of populations and program components, future studies are needed to determine the optimal duration of CR and the program characteristics that would benefit the most patients. Individualizing CR and other non-pharmacological treatments to deliver the optimal approach for each patient might also lead to better outcomes.
Despite strong evidence of the efficacy of CR, most people in Canada with SSDs are not offered or cannot access CR, underscoring a major implementation issue for psychosocial approaches. Investing in capacity building and training care providers to deliver non-pharmacological interventions will improve outcomes and reduce costs for the health-care system. Increasing staffing representation from specialized allied health disciplines and providing specialized training to clinicians who are most suited for CR delivery (e.g., neuropsychologists and occupational therapists) would be more beneficial than training health-care providers who do not have the foundational knowledge and training related to cognitive assessment and intervention. Development of interventions that can be implemented virtually can facilitate access to CR in remote or limited-resource settings. It may also be helpful in assessing the impact of current and future provincial initiatives in supporting these strategies and interventions.
Moreover, innovative coping strategies or self-management approaches, such as immersive virtual reality and mindfulness-based techniques, have shown promise in the treatment of cognitive impairment in individuals with schizophrenia.98–100 Future studies are warranted to validate the usefulness of novel technologies improving current non-pharmacological treatments and assess the feasibility of their implementation. The role of administrative teams in the adoption of novel technologies for the management of cognitive impairment should also be explored.
Cognitive impairment significantly affects daily functioning and QOL in patients with SSDs. Addressing cognitive impairment is critical for optimizing treatment outcomes and enhancing functional outcomes and QOL. By using reliable tools to comprehensively measure cognitive impairment, clinicians can identify and implement evidence-based treatment options and develop targeted and personalized interventions to improve the overall well-being of patients with SSDs. Tailored comprehensive treatment with a combination of pharmacological and non-pharmacological interventions can help individuals with SSDs achieve better cognitive health and functional outcomes.
Footnotes
Acknowledgments
All authors significantly contributed to the conceptualization, drafting, and revision of the manuscript. The authors extend their gratitude to Christos Evangelou and STA Healthcare Communications (Montréal, Canada) for their assistance with writing and editing.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ML reports grants from Otsuka Lundbeck Alliance, personal fees from Otsuka Canada, personal fees from Lundbeck Canada, grants and personal fees from Janssen, grants from Roche, and personal fees from Boehringer Ingelheim, all outside the submitted work. SG reports research grant support from the Canadian Institute of Health Research (CIHR), Le Fond de Recherche du Québec en Santé (FRQS), and Emerging Research Innovators in Mental Health (e-RIMH), consultation fees from Boehringer Ingelheim Canada, and being a junior board member at the Schizophrenia International Research Society, all outside the submitted work. TR reports research grant support from Boehringer Ingelheim, SyneuRx International, and TEVA and speaker/consultation fees from AbbVie, Boehringer Ingelheim, HLS Therapeutics, Newron, Otsuka, Sunovion, and TEVA, all outside the submitted work. HEM reports consultation fees from Boehringer Ingelheim outside of the submitted work. HCM reports research grant support from AIFred, SyneuRx, and the Montreal General Hospital Foundation and speaker/consultation fees from AbbVie, Boehringer Ingelheim, HLS Therapeutics, Lundbeck, Janssen, Newron, Otsuka, Sunovion, and TEVA, all outside the submitted work. TU reports consulting fees from Mental Health Commission of Canada, honoraria from Boehringer Ingelheim, and being director and board member of Telus Fund and Scientific Advisor for LongTermGevity, all outside the submitted work. MB reports consulting fees and honoraria from Boehringer Ingelheim and consulting fees from and interests in Best Psychological Services, all outside the submitted work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Assistance with the writing and editing of this manuscript was funded by Boehringer Ingelheim.
