Abstract
Objective
In recent years, the relationship between nutrition and mental health has gained considerable interest. We identified, synthesized, and appraised all meta-analyses of randomized controlled trials (RCTs) and observational studies reporting on the efficacy of dietary patterns and nutrient supplements in the prevention and treatment of mental disorders in children and adolescents.
Methods
Systematic research in MEDLINE, PsycINFO, Scopus, and Cochrane Database of Systematic Reviews was completed on 8 January 2024.
Results
Our research found 24 meta-analyses: 14 on RCTs, 8 on observational studies, and 2 combining both. Emerging evidence suggests that omega-3, in particular eicosapentaenoic acid, and Vitamin D may have adjunctive benefits in the treatment of attention deficit hyperactivity disorder (ADHD), while no evidence was found for autism spectrum disorder (ASD). Observational data also indicated that prenatal folic acid supplementation (>400 μg daily) was associated with a reduced risk of ASD in offspring. In terms of dietary habits, several meta-analyses of observational data revealed that healthy dietary patterns (rich in fruits, vegetables, and fibre, low in saturated fats) during the prenatal period, childhood, and adolescence were linked to a significantly reduced risk of internalizing disorders and externalizing disorders. Conversely, unhealthy dietary habits (high in sugars, saturated animal fats, and industrial foods, low in fruits, vegetables, and fibre) are associated with an elevated risk of these mental health issues. However, the number of available studies on dietary interventions for the treatment of depression, ASD, and ADHD was limited, and the results obtained were either nonsignificant or contradictory.
Conclusion
Our findings emphasize the need to establish clear causal relationships between dietary habits and the risk of mental illness in children and adolescents. Moreover, further investigation of the benefits observed with some nutrient supplements (such as omega-3 and vitamin D for ADHD) through larger-scale RCTs is imperative to establish more robust conclusions.
Plain Language Summary
We investigated the link between nutrition and mental health in children and adolescents through a meta-review of 24 relevant meta-analyses. Emerging evidence suggests potential benefits of Omega-3 and Vitamin D in treating ADHD, while no evidence supports their effectiveness in ASD. Observational data also indicate that prenatal folic acid supplementation may lower ASD risk. Healthy dietary patterns reduce the risk of internalizing and externalizing disorders, whereas unhealthy habits elevate the risk. Limited studies on dietary interventions for depression, ASD, and ADHD provide inconclusive results. In summary, our results emphasize the need to clearly understand the cause-and-effect relationships between dietary habits and mental health risks in young individuals. Larger-scale randomized controlled trials are essential for confirming the observed benefits of nutrient supplements such as omega-3 and vitamin D in treating ADHD and for forming more reliable conclusions.
Introduction
In recent years, the relationship between diet and mental health has gained considerable interest.1–3 The International Society For Nutritional Psychiatry Research (ISNPR), founded in 2013 reflects emerging research on nutritional approaches in the prevention and treatment of mental disorders, and the importance of conducting high-quality research in this field. 4
Changes in the food habits of children and adolescents over the past century, driven by economic and social development, have resulted in an unbalanced diet. 1 Unhealthy eating patterns, characterized by consumption of processed foods high in sugar, saturated fats, and refined carbohydrates, along with malnutrition, are major risk factors for noncommunicable diseases such as childhood obesity, cardiovascular and respiratory diseases, and diabetes.5–7 Furthermore, these chronic diseases, which are strongly associated with lifestyle, are highly comorbid with mental disorders such as depression, anxiety, sleep disorders, and eating disorders.1,6
Diet directly impacts brain development and function across all age groups, starting from the first 1,000 days, which include 270 days of pregnancy and the initial 2 years of life. 8 This period of vulnerability is characterized by rapid brain growth,9–11 involving processes such as neurogenesis, axonal and dendritic growth, synaptogenesis, cell death, synaptic pruning, myelination, and gliogenesis. 12 The rapidly growing brain during critical developmental phases is particularly prone to nutrient deficiencies, including protein, energy, specific fats, folic acid, vitamin A, iron, zinc, copper, iodine, selenium, and choline.13,14 Notably, epidemiological studies of populations exposed to famine such as the Dutch Hunger Winter of 1944 to 1945 or the Chinese famine of 1959 to 1961 have shown that moderate to severe nutritional deficiencies during pregnancy and early childhood could increase the risk of mental disorders in later adulthood, 15 such as schizophrenia, 16 schizoid personality disorder,17,18 or affective disorders. 19
Along with regular food intake, nutrients can also be consumed in supplement form. Supplements are commonly used for several purposes: (a) to complement an insufficient diet or address nutrient deficiencies, especially during pregnancy to support the child's brain development 13 ; (b) to provide nutrients at higher doses than typically found in regular diets for potential physiological benefits; (c) to offer nutrients in more easily absorbable forms, suitable for individuals with genetic variations or health issues affecting nutrient absorption. 20
There is a growing interest in using dietary patterns and nutrient supplements in the prevention and treatment of mental disorders, driven by advances in our understanding of the neurobiological basis underlying them. This emerging research suggests that certain nutrients or dietary patterns could influence mental health including during the sensitive stages of development, via different biological pathways such as gut microbiota, oxidative stress, inflammation, hypothalamus–pituitary adrenal axis, mitochondrial dysfunction, neuroplasticity, or epigenetics. 21 For instance, diet and pre/probiotics influence the composition and diversity of the gut microbiota and can modulate brain structure and function via anti-inflammatory effects mediated by the microbial metabolites of dietary fibre and polyphenols. 22 This implies that certain dietary patterns and pre/probiotic supplements might offer promising new therapeutic options that should be explored further. 20
Despite the available research findings, the link between diet and children's behaviour is complex, with many conflicting studies.2,23,24 To our knowledge, the broader role of dietary factors, across the spectrum of paediatric mental disorders, has yet to be established. Some meta-reviews of meta-analyses about the role of nutrition (diet and nutrient supplements) have been published in the adult population,20,25 but none in the paediatric population. Recognizing and investigating this area in both research and routine clinical practice is crucial for advancing our approach to managing mental disorders in this age group.
The present paper aims to establish the current evidence regarding the efficacy of dietary factors, including eating habits and dietary supplements, in both preventing and treating psychiatric disorders in children and adolescents. To do this, we identified, synthesized, and appraised all available data from meta-analyses of randomized controlled trials (RCTs) and observational studies examining mental health outcomes for all dietary factors across various paediatric mental disorders.
Methods and Materials
This meta-review aimed to systematically gather the most recent high-quality evidence regarding the role of “dietary factors” in preventing and treating mental disorders in children and adolescents. The literature search (Supplemental Appendix 1) was performed with the assistance of an academic health sciences librarian (Valérie Durieux) and followed the Peer Review of Electronic Search Strategies (PRESS) method. 26 Additionally, our reporting adhered to the PRISMA statement 27 for comprehensive and transparent reporting, and we followed a preregistered protocol (PROSPERO: CRD42023390196).
Literature Search Strategy
All references were exported and managed using Endnote x9.3.3. The systematic search was conducted using Ovid MEDLINE®, PsycINFO, Scopus, and Cochrane Database of Systematic Reviews, from inception until 8 January 2024. We have expanded our search to include all languages except for Chinese and Japanese.
Eligibility Criteria
This meta-review included previously published meta-analyses. The eligibility criteria for the included meta-analyses were organized following the participants, interventions, comparisons, outcomes, and study design reporting structure.
Participants
- -
Comparisons
-
Interventions
- -
Outcomes
-
-
Study Design
- -
To prevent potential bias resulting from overlap in our meta-analysis selection, we calculated the corrected covered area (CCA) and utilized a Graphical Representation of Overlap for Overviews (GROOVE) using the freely available tool developed by Bracchiglione et al. 28
We systematically assessed the degree of overlap between each pair of meta-analyses, categorizing it as follows: slight overlap (<5%), moderate overlap (>5% and <10%), high overlap (10% to <15%), and very high overlap (≥15%). 28
Where meta-analyses analyses with a very high overlap (≥15%), the most recently updated meta-analysis was used. Older meta-analyses, providing unique insights such as a larger study pool or specific subgroup analyses, were used as secondary analyses.
Data Extraction
Two authors (MT and MR) independently screened titles and abstracts of all publications that were obtained by the search strategy. Full texts identified in the first step were by one author (MT) and then verified by another author (MR). If differences in opinion existed, they were resolved until the two authors agreed with each other. The remaining disagreements were discussed with a third author (HN) until a consensus was reached.
Results of the eligible meta-analysis were extracted narratively to present the association of dietary factors (nutritional supplementation or dietary pattern) and the risk of child mental illness, as well as the effects of nutritional interventions on psychiatric outcomes in the paediatric population.
When associations between dietary factors and child mental disorders were quantified as categorical variables, these were extracted as odds ratios (ORs) or relative risks (RRs) with their confidence intervals (CI) to 95%.
When data on effect size were quantified as a continuous variable (i.e., effect size on psychiatric symptoms), these were extracted as the standardized mean difference (SMDs) (Cohen's d or Hedges’g), and their 95% CIs. Following conventional interpretations, SMDs were categorized as negligible (<0.2), small (0.2–0.4), moderate (0.4–0.8), or large (>0.8).20,25 Some meta-analyses did not provide SMDs but weighted mean differences or raw mean differences, and we also extracted these data.
For all meta-analyses, data on the degree of between-study heterogeneity (quantified as I2 values) were also extracted and categorized as low (I2 < 25%), moderate (I2 = 25% to 50%) or high (I2 > 50%).20,25
Quality Assessment of Included Studies
Authors (MT and MR) independently assessed the methodological quality of the included meta-analyses using “A Measurement Tool to Assess Systematic Reviews” Version 2 (AMSTAR-2), 29 allowing for the consideration of specific methodological biases in the results. An overall confidence rating in the results of the review was categorized as high, moderate, low, or critically low, following the method defined by Shea et al. 29 The details of this process are provided in Supplemental Appendix 2.
Results
We identified 625 potential records from databases. After the removal of duplicates, 431 studies were screened from which 56 full-text documents were reviewed, and 24 studies were included; see Figure 1 and Supplemental Appendix 3 for details of papers excluded.
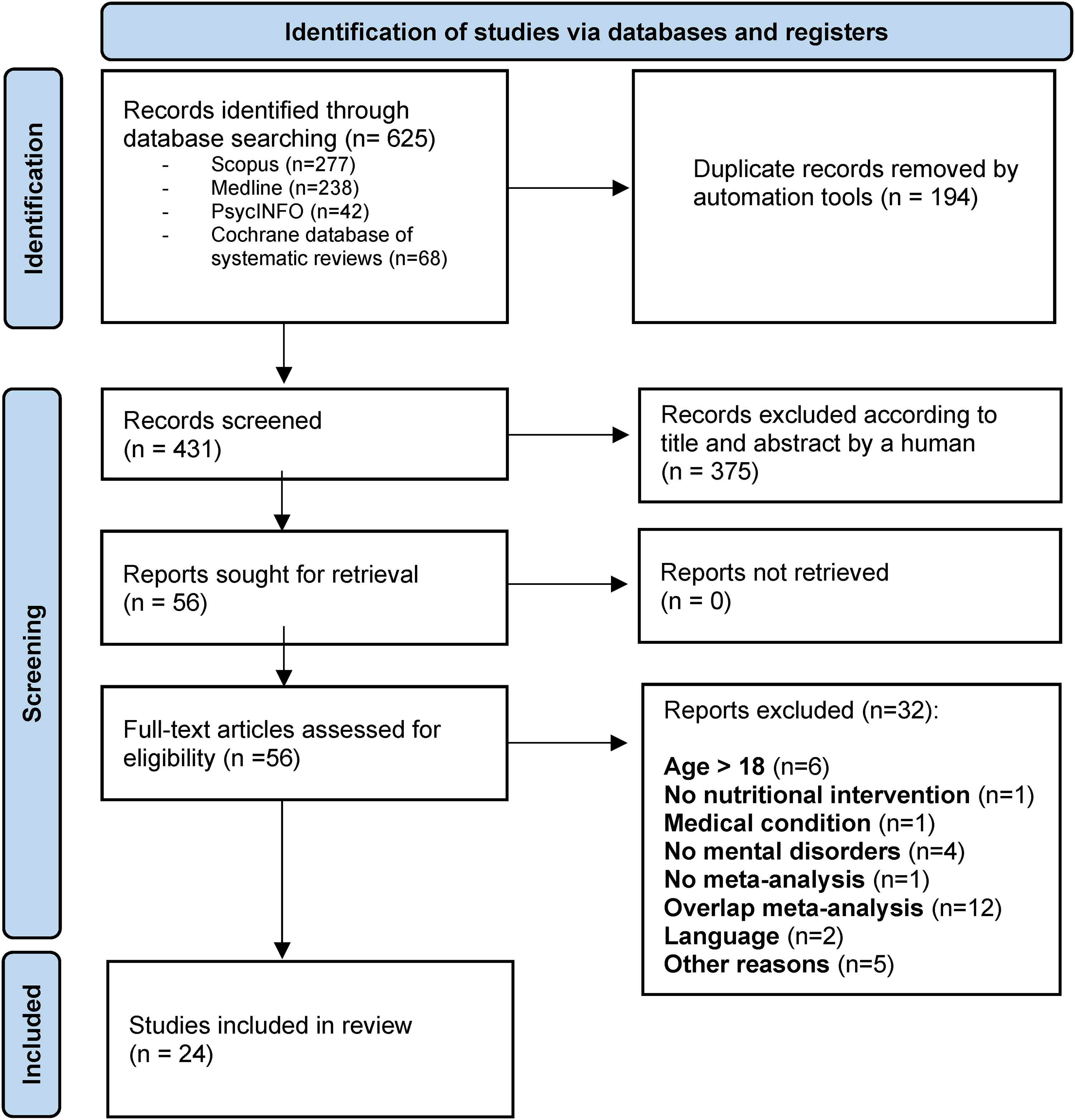
PRISMA 2020 flow diagram.
Characteristics and Methodological Quality of the Included Meta-Analyses
Study characteristics are presented in Tables 1 to 3. Out of the 24 included meta-analyses, 8 were meta-analyses of observational studies, 14 were meta-analyses of RCTs, and 2 were meta-analyses including both observational studies and RCTs.
Diet and Risk of Paediatric Mental Disorders.

Note. ADHD = attention deficit hyperactivity disorder; DASH = the dietary approaches to stop hypertension score; E-DII = energy-adjusted dietary inflammatory index score; FVI = fruit and vegetable intake; k = number of studies included in the meta-analysis; n = sample size; NR = not reported; NIH = quality of the study evaluated by the National Institutes of Health's Quality Assessment Tool for Systematic Reviews and Meta-Analyses (good: 7 or 8; fair: 4–6; poor: 0–3); OR = odds ratio; RCT = randomized controlled trial; FFQ = food frequency questionnaire.
*The details regarding the establishment of certainty of the evidence according to AMSTAR-2 are provided in Supplemental Appendix 3.
The association between sugar and soft drink consumption and the risk of ADHD symptoms was provided based on the random-effects model.
Effect of Dietary Interventions in Paediatric Mental Disorders.
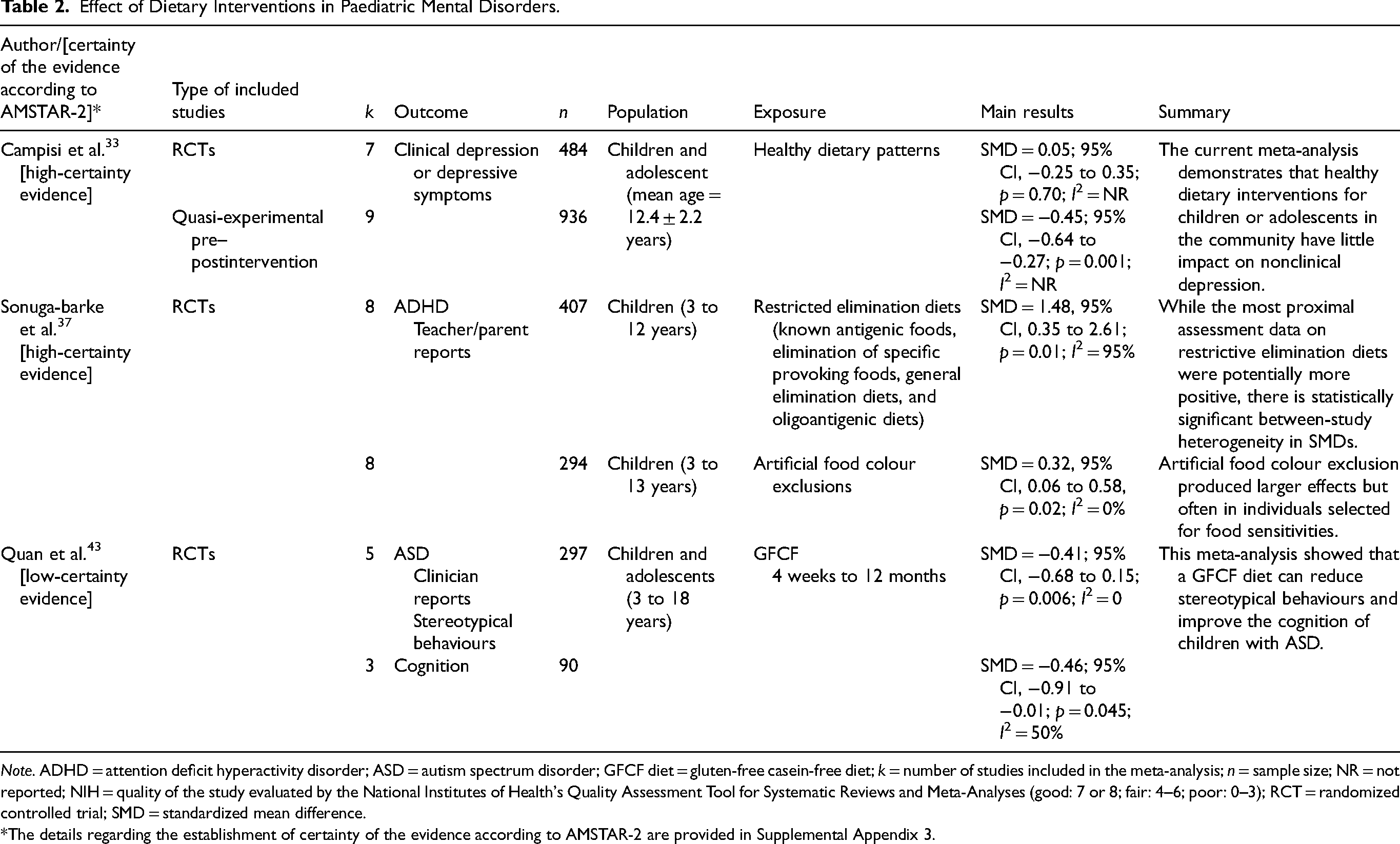
Note. ADHD = attention deficit hyperactivity disorder; ASD = autism spectrum disorder; GFCF diet = gluten-free casein-free diet; k = number of studies included in the meta-analysis; n = sample size; NR = not reported; NIH = quality of the study evaluated by the National Institutes of Health's Quality Assessment Tool for Systematic Reviews and Meta-Analyses (good: 7 or 8; fair: 4–6; poor: 0–3); RCT = randomized controlled trial; SMD = standardized mean difference.
*The details regarding the establishment of certainty of the evidence according to AMSTAR-2 are provided in Supplemental Appendix 3.
Effects of Nutrient Supplements in Paediatric Mental Disorders.
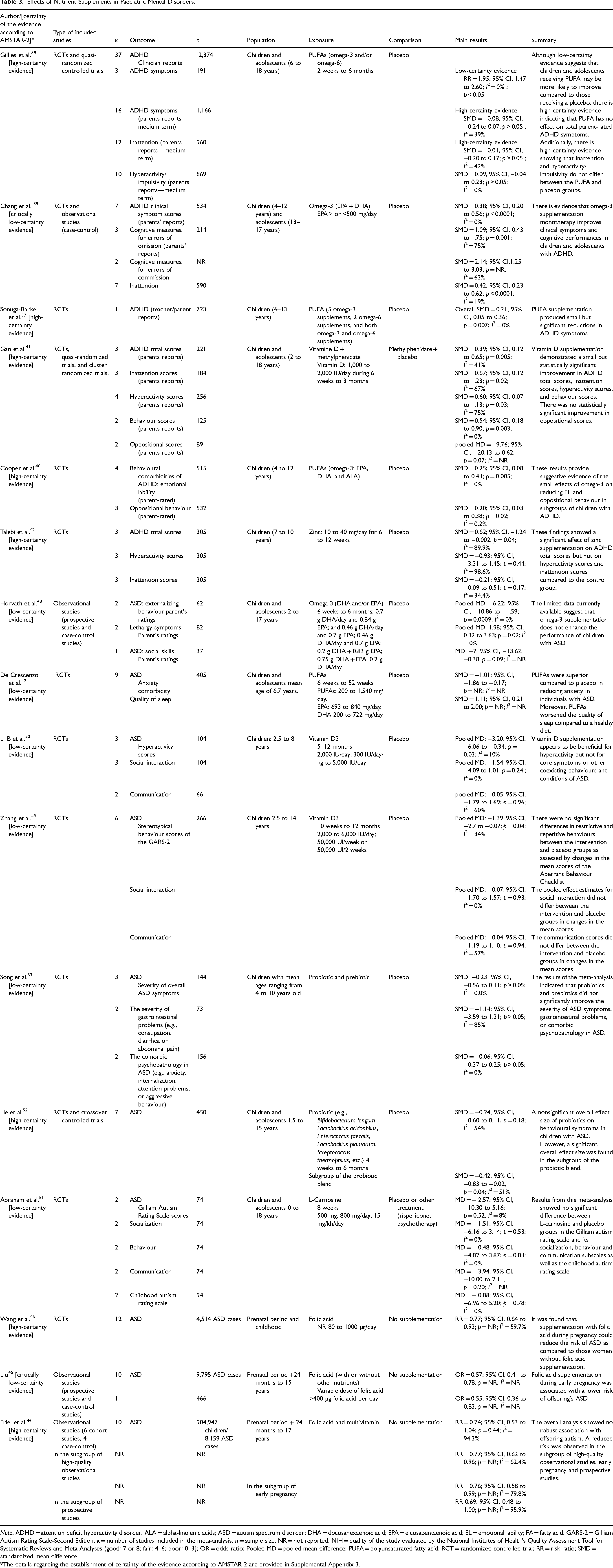
Note. ADHD = attention deficit hyperactivity disorder; ALA = alpha-linolenic acids; ASD = autism spectrum disorder; DHA = docosahexaenoic acid; EPA = eicosapentaenoic acid; EL = emotional lability; FA = fatty acid; GARS-2 = Gilliam Autism Rating Scale-Second Edition; k = number of studies included in the meta-analysis; n = sample size; NR = not reported; NIH = quality of the study evaluated by the National Institutes of Health's Quality Assessment Tool for Systematic Reviews and Meta-Analyses (good: 7 or 8; fair: 4–6; poor: 0–3); OR = odds ratio; Pooled MD = pooled mean difference; PUFA = polyunsaturated fatty acid; RCT = randomized controlled trial; RR = risk ratio; SMD = standardized mean difference.
*The details regarding the establishment of certainty of the evidence according to AMSTAR-2 are provided in Supplemental Appendix 3.
All data regarding specific psychiatric conditions or psychiatric symptoms considered in this meta-review include depressive disorders, internalizing disorders, externalizing disorders, ADHD, ASD, and behavioural comorbidities such as emotional lability (EL), aggression, oppositional behaviour and cognitive outcomes.
Sixteen meta-analyses examined nutritional supplementation including PUFAs, vitamins, minerals, amino acid supplements and pre/probiotics. These data were based on nutritional supplementation administered as an adjuvant treatment to conventional therapy such as a specific class of psychotropics (e.g., methylphenidate in ADHD) or as a monotherapy. On the other hand, 9 meta-analyses assessed dietary patterns or specific diets including healthy dietary patterns and unhealthy diet (n = 6); restriction diet and artificial food colour exclusion (n = 1); GFCF diet (n = 1), and sugar and soft drink consumption (n = 1).
Figure 2 depicts the CCA and GROOVE specific to each identified psychiatric disorder after excluding studies with a very high overlap (≥15%). This provides a representation of the degree of overlap among different meta-analyses. Meta-analyses with a high degree of overlap (≥15%) that were excluded are detailed in Supplemental Appendices 3 and 4.
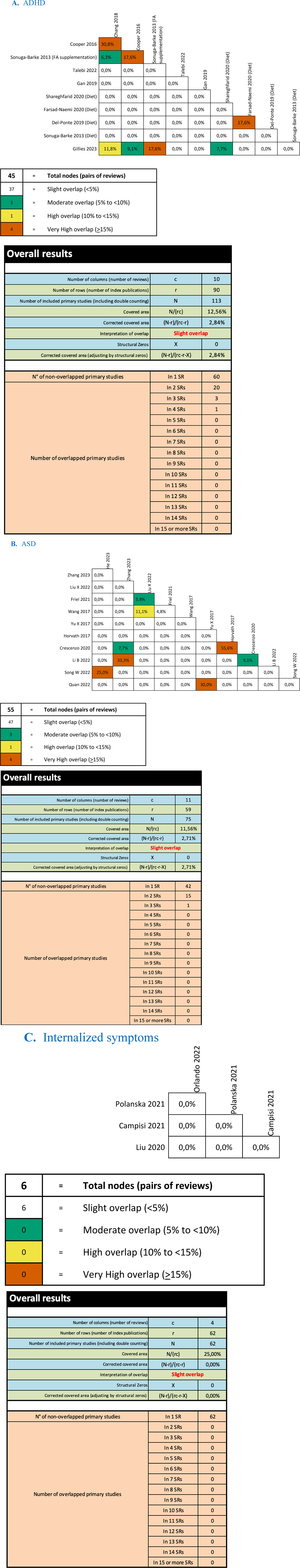
CCA and GROOVE for included meta-analysis.
The CCA reveals slight overlap, specifically 2.84% for ADHD, 2.71% for ASD, and 0% for internalized symptoms.
The quality assessment of the meta-analyses included using AMSTAR-2 is presented in Tables 1 to 3 and Supplemental Appendix 2. The overall confidence rating categorized 10 studies as “high,” 8 studies as “low,” 4 with a “moderate” rating, and 2 classified as “critically low.”
Nineteen of the 24 included meta-analyses were published between 2019 and 2023.
Results of Syntheses
Detailed study findings are presented in Tables 1 to 3.
Internalizing and/or Externalizing Disorders
During prenatal period, higher maternal DASH scores, indicating better dietary quality, are associated with a lower risk of depressive and anxiety symptoms (k = 4; n = 11,817; OR = 0.97; 95% CI, 0.95 to 0.99; I2 = 24%), aggressive behaviour symptoms (k = 4; n = 11,829; OR = 0.97, 95% CI, 0.94 to 0.99; I2 = 61%), and ADHD symptoms (k = 4; n = 11,829; OR = 0.97; 95% CI, 0.95 to 0.98; I2 = 2%) in children. Conversely, a proinflammatory diet was associated with an increased risk of depression and anxiety symptoms (k = 4; n = 11,817; OR = 1.07, 95% CI, 1.03 to 1.11; I2 = 0%), aggressive behaviour symptoms (k = 4; n = 11,829; OR = 1.07; 95% CI: 1.02 to 1.13; I2 = 24%), and ADHD symptoms (k = 4; n = 11,829; OR = 1.07; 95% CI: 1.01 to 1.13; I2 = 33%). 30
During childhood and adolescence, meta-analyses of observational studies with a total number of participants enrolled ranging from 73,726 to 116,546, found that healthy dietary patterns were negatively associated with internalizing symptoms (anxiety and emotional problems) (k = 31; pooled effect size = −0.09; 95% CI, −0.12 to 0.06; p < 0.001; I2 = 96.777) and particularly depressive symptoms (k = 19; pooled effect size = −0.13; 95% CI, −0.18 to −0.08; p < 0.001; I2 = 94.944). However, unhealthy dietary patterns were positively associated with internalizing (anxiety and emotional problems) (k = 25; pooled effect size = 0.10; 95% CI, 0.06 to 0.14; p = 0.05; I2 = 97.928) and depressive symptoms (k = 13; pooled effect size = 0.11; 95% CI, 0.05 to 0.17; p = 0.001; I2 = 98.175). 31 A meta-analysis of cross-sectional studies indicated that dietary fruit and vegetable of <5 times/day (versus higher) was associated with an increased risk of depressive symptoms (k = 25; n = 65,267; OR = 1.10; 95% CI, 1.02 to 1.18; p = 0.028; I2 = 38.34%). 32
Attention Deficit Hyperactivity Disorder (ADHD)
A meta-analysis of 6 RCTs, involving a total of 8,129 participants, revealed that a healthy dietary pattern, rich in vegetables, fruits, legumes, and fish, decreased the odds of ADHD by up to 37% (k = 5; n = 8,816; OR = 0.63; 95% CI, 0.41 to 0.96; I2 = 46.5%). Conversely, a “Western diet” comprising red meat, refined grains, processed meats, and hydrogenated fat (k = 2; n = 3,243; OR = 1.92; 95% CI, 1.13 to 3.26; p = 0.016, I2 = 0.0%), as well as a “junk food” pattern consisting of sweetened beverages and desserts (k = 5; OR = 1.51; 95% CI, 1.06 to 2.16; p = 0.024, I2 = 48.7%), were found to increase the risk of ADHD. 34
Similar effects were observed in a pooled analysis, indicating a significantly decreased risk of ADHD (OR = 0.65; 95% CI, 0.44 to 0.97; p < 0.05). The effects remained consistent after stratifying the studies by design (cohort/case-control or cross-sectional) (k = 11; OR = 0.59; 95% CI, 0.46 to 0.74; I2 = 73%), by continent (Europe or Asia/Oceania) (k = 11; OR = 0.59; 95% CI, 0.46 to 0.74; I2 = 73%), and sample size (≥1000 or <1000) (k = 11; OR = 0.66; 95% CI, 0.60 to 0.72; I2 = 73%). Conversely, an unhealthy dietary pattern can increase the risk of ADHD (OR = 1.41; 95% CI, 1.15 to 1.74). The effects remained consistent after stratifying the studies by design (cohort/case-control or cross-sectional) (k = 14; OR = 1.41; 95% CI, 1.25 to 1.58; I2 = 86.2%), by continent (Europe or Asia/Oceania) (k = 13; OR = 1.41; 95% CI, 1.25 to 1.58; I2 = 86.2%), and sample size (≥1000 or <1000) (k = 13; OR = 1.41; 95% CI, 1.25 to 1.58; I2 = 86.2%). 35
Another meta-analysis indicated a positive relationship between overall sugar and sugar-sweetened beverages consumption and the risk of ADHD symptoms (k = 8; n = 25,945; pooled effect size: 1.22; 95% CI, 1.04 to 1.42, p = 0.01, I2 = 81.9%). 36
A subsequent meta-analysis, incorporating 11 trials (5 on omega-3 supplements, 2 on omega-6 supplements, and 4 on both omega-3 and omega-6 supplements), showed small but significant reductions in ADHD symptoms (SMD = 0.21; 95% CI, 0.05 to 0.36; p = 0.007; I2 = 0%). 37
More specifically, omega-3 supplements demonstrated a significant reduction in both inattention (k = 7; n = 590; SMD = 0.42; 95% CI, 0.23 to 0.62; p < 0.0001; I2 = 19%) and total ADHD score (k = 7; n = 534; SMD = 0.38; 95% CI, 0.20 to 0.56; p < 0.0001; I2 = 0%). These effects were also found for the total ADHD score to be significant in subgroup analyses that separately tested studies with eicosapentaenoic acid (EPA) dosage of 500 mg/day or greater, as well as studies with an EPA dosage <500 mg (k = 7; n = 590; SMD = 0.42; 95% CI, 0.23 to 0.62; p < 0.0001; I2 = 19%). Moreover, for hyperactivity symptoms, studies with an EPA dosage of 500 mg per day or greater showed a significant effect (k = 3; n = 277; SMD: 0.81; 95% CI, 0.12 to 1.49; p = 0.02; I2 = 87%). 39
Regarding behavioural comorbidities, studies have shown that omega-3 supplementation can lead to small improvements in measures of EL and oppositional behaviour. Indeed, subgroup analyses of higher quality studies and those meeting strict inclusion criteria have revealed significant reductions in parent-rated EL (k = 4; n = 515; SMD = 0.25; 95% CI, 0.08 to 0.43; p = 0.005; I2 = 0%) and oppositional behaviour (k = 3; n = 532; SMD = 0.20; 95% CI, 0.03 to 0.38; p = 0.02; I2 = 0.2%) when omega-3 supplementation is used. 40
A meta-analysis of RCTs was conducted to assess the effects of vitamin D supplementation (1000 to 2000 IU/day during 6 weeks to 3 months) as an adjunctive therapy to methylphenidate on ADHD symptoms in children. The results indicated that vitamin D supplementation led to a small but statistically significant improvement in ADHD total scores (k = 3; n = 221; SMD = 0.39; 95% CI, 0.12 to 0.65; p = 0.005; I2 = 41%), inattention scores (k = 3; n = 185; SMD = 0.67; 95% CI, 0.12 to 1.23; I2 = 67%; p = 0.02), hyperactivity scores (k = 4; n = 256; SMD = 0.60; 95% CI, 0.07 to 1.13; I2 = 75%; p = 0.03), and behaviour scores (k = 2; n = 125; SMD = 0.54; 95% CI, 0.18 to 0.90; p = 0.003; I2 = 0%). However, no statistically significant improvement was observed in oppositional scores (k = 2; n = 89; pooled mean difference [MD] = −9.76; 95% CI, −20.13 to 0.62; p = 0.07). 41
In a meta-analysis of RCTs involving school-aged children with ADHD, zinc supplementation was found to significantly reduce ADHD total scores (k = 3; n = 305; SMD = 0.62; 95% CI, 0.002 to 1.24; p = 0.04; I2 = 89.9%). However, zinc supplementation did not show a significant effect on hyperactivity scores (k = 3; n = 305; SMD = −0.93; 95% CI, −3.31 to 1.45; p = 0.44; I2 = 98.6%) or inattention scores (k = 3; n = 305; SMD = −0.21; 95% CI, −0.51 to 0.09; p = 0.17; I2 = 34.4%) compared to the control group. 42
Autism Spectrum Disorder (ASD)
More specifically, folic acid supplementation during early pregnancy was associated with a lower risk of offspring's ASD (k = 10; n = 9,795 ASD cases; OR = 0.57; 95% CI, 0.41 to 0.78). The consumption of a daily amount of at least 400 μg folic acid from dietary sources and supplements, was associated with a reduced risk of offspring ASD (k = 1; n = 466; OR = 0.55; 95% CI, 0.36 to 0.83). 45
Supplementation with folic acid during pregnancy could reduce the risk of ASD in children (k = 12; n = 4,514 ASD cases; RR = 0.77; 95% CI, 0.64 to 0.93; I2 = 59.7%), irrespective of ethnicity, when compared to women who did not supplement with folic acid. Notably, significant associations were observed in Asian, European, and American populations. 46
In children and adolescents with ASD, supplementation with PUFAs (omega-3 and/or omega-6) was found to be superior to placebo or a healthy diet in reducing anxiety comorbidity (k = 9; n = 405; SMD = 1.01; 95% CI, 0.17 to 1.86; very low certainty of the evidence). However, it should be noted that PUFAs were associated with worsened quality of sleep when compared to a healthy diet (SMD = −1.11; 95% CI, −2.00 to −0.21; very low certainty of evidence). Additionally, PUFAs did not show superiority over placebo in reducing aggression, hyperactivity, adaptive functioning, irritability, restricted and repetitive interests and behaviours, and communication in individuals with ASD. 47
Similarly, supplementation with omega-3 (docosahexaenoic acid [DHA] and/or EPA) does not any improvement in the performance of children with ASD compared to placebo, based on findings from 5 RCTs including 183 participants. However, parent's ratings indicated a significant improvement in lethargy symptoms (k = 2; n = 82; pooled MD: 1.98; 95% CI, 0.32 to 3.63; p = 0.02; I2 = 0%), but worsening of externalizing behaviour (k = 2; n = 62; pooled MD: −6.22; 95% CI, −10.86 to −1.59; p = 0.0009; I2 = 0%) and social skills (k = 1; n = 37; pooled MD: −7; 95% CI, −13.62 to −0.38; p = 0.09) in the omega-3 group compared to the placebo group. 48
Vitamin D supplementation showed no significant differences in restrictive and repetitive behaviours between the intervention and placebo groups (k = 6; n = 266; pooled MD: −1.39; 95% CI, −2.7 to −0.07; p = 0.04; I2 = 34%). Furthermore, vitamin D did not demonstrate improvement in other core ASD symptoms such as social interaction (pooled MD: −0.07; 95% CI, −1.70 to 1.57; p = 0.93; I2 = 0%) and communication (pooled MD: −0.04; 95% CI, −1.19 to 1.10; p = 0.94; I2 = 57). 49 However, Vitamin D supplementation indicated a small but significant improvement in coexisting hyperactivity scores (k = 3; n = 104; pooled MD: 3.20; 95% CI, 0.34 to 6.06; p = 0.03; I2 = 10%). 50
Supplementation with L-carnosine for 8 weeks did not show any significant difference compared to the placebo groups in the Gilliam Autism Rating Scale scores (k = 2; n = 74; MD = −2.57; 95% CI, −10.30 to 5.16; p = 0.52; I2 = 8%), including its socialization (k = 2; n = 74; MD = −1.51; 95% CI, −6.16 to 3.14; p = 0.53; I2 = 0%), behaviour (k = 2; n = 74; MD = −0.48; 95% CI, −4.82 to 3.87; p = 0.83; I2 = 0%), and communication (k = 2; n = 74; MD = −3.94; 95% CI, −10.00 to 2.11; p = 0.20) subscales, as well as the childhood autism rating scale (k = 2; n = 94; MD = −0.88; 95% CI, −6.96 to 5.20; p = 0.78; I2 = 0%). 51
Regarding probiotics (e.g., Bifidobacterium longum, Lactobacillus acidophilus, Enterococcus faecalis, etc.), our study identified a meta-analysis of RCTs that showed no significant improvement in the behavioural symptoms in children with ASD (k = 3; n = 450; SMD = −0.24; 95% CI, −0.60 to 0.11; p = 0.18; I2 = 54%). However, a significant overall effect size was found in the subgroup of the probiotic blend (SMD = −0.42; 95% CI, −0.83 to −0.02; p = 0.04; I2 = 51%). 52 Another meta-analysis revealed that prebiotics and probiotics (e.g., Streptococcus thermophilus, Lactobacillus plantarum, Bifidobacterium infantis, etc.) did not show a significant improvement in the severity of gastrointestinal problems (e.g., constipation, diarrhea, or abdominal pain) (k = 2; n = 73; SMD = −1.14; 95% CI, −3.59 to 1.31; p > 0.05; I2 = 85%) or the comorbid psychopathology in ASD (e.g., anxiety, internalization, attention problems, or aggressive behaviour) (k = 2; n = 156; SMD = −0.06; 95% CI, −0.37 to 0.25; p > 0.05; I2 = 0%). 53
Discussion
This meta-review aggregated and evaluated all the recent evidence from meta-analyses of RCTs and observational studies examining the efficacy of dietary patterns and nutritional supplements in the prevention and treatment of mental disorders in children and adolescents.
In the field of nutritional psychiatry in children and adolescents, the most extensively studied mental disorders are ASD,43–53 followed by ADHD.34–42 These results are consistent with the observed increase in the prevalence of these disorders.54,55
Several meta-analyses of observational data have shown that healthy dietary patterns during the prenatal period, childhood and adolescence are associated with a significantly reduced risk of certain mental disorders including internalizing disorders and externalizing disorders, whereas the opposite is observed for unhealthy dietary habits.30–32,34–36 The included studies in these meta-analyses attempted to control for the impact of important confounding factors, such as participant sex, unfavourable socio-economic status, obesity, physical inactivity, or family conflicts.30,32,34–36 However, prospective links with diagnosed mental disorders were not established.
Dietary Interventions
In depression, dietary interventions showed promise in reducing depressive symptoms in some studies, but RCTs did not consistently support their efficacy in the paediatric population. 31 It is important to note that the included studies focused on promoting healthy habits in overweight or obese children. As a result, the generalizability of these findings to individuals of normal weight is limited. Also, they differ from adult research where dietary interventions had a small but significant effect on reducing depressive symptoms. 56
Restriction diets eliminating synthetic food colours appeared to significantly reduce ADHD symptoms but often in individuals selected for food sensitivities. 37
Among children with ASD, the dietary interventions identified in our paper are limited to the GFCF diet. While this diet may lead to improved cognition and reduced stereotypical behaviours, 43 it is important to note that further research with larger samples is necessary.
Nutrient Supplements
Regarding the role of dietary supplements in the prevention of paediatric mental disorders, ASD has been most widely studied. Prenatal folic acid (>400 μg daily) and multivitamin supplementation have shown potential in reducing the risk of ASD in children,44–47 especially in high-quality observational and prospective studies.44,45
Vitamin D supplementation in ASD, as revealed in 2 meta-analyses, showed no improvements in core autism symptoms. Both meta-analyses included RCTs with small sample sizes, making it difficult to draw definitive conclusions.49,50
Additionally, probiotic and prebiotic supplementation did not demonstrate a significant effect on ASD symptoms in a recent meta-analysis of 7 RCTs, though more extensive trials are necessary. 52
PUFAs have been the most widely evaluated dietary supplement in the treatment of paediatric mental disorders, particularly in the context of ADHD.37–40 Omega-3 supplementation with high-EPA formulas (>500 mg/day) for 3 months was identified as potentially beneficial for hyperactivity symptoms and comorbidities in ADHD, including EL or oppositional behaviour in high-quality meta-analyses of RCTs.39,40 Moreover, vitamin D supplementation (400 to 2000 IU/day) as an adjunct to methylphenidate appeared effective in treating ADHD symptoms, 41 while zinc supplementation may also have beneficial effects. 42
Strengths and Limitations
Our data should be considered in the light of some limitations. Firstly, meta-analyses of observational studies included in our work may show associations between nutrition and mental health but establishing a causal relationship can be challenging due to the observational nature of these studies and the possibility of residual confounding. Intervention studies, using an overall diet strategy, are a robust way to support the causal roles of dietary patterns in the onset of any mental illness. 2 However, meta-analyses of intervention trials are often limited to nutritional supplementation, and studies corresponding to dietary patterns are scarce. Moreover, studies evaluating the impact of nutritional supplementation in the treatment of mental diseases often lack data on serum levels of the nutrients studied before and after treatment. This can make it difficult to establish a clear link between supplementation and therapeutic response.
Moreover, many of the outcomes included in this meta-review exhibited significant heterogeneity between the included studies or were based on a small number of samplers. This may limit the generalization of the results of the meta-review and make it difficult to draw definitive conclusions. Finally, the execution of a meta-analysis was not possible due to the diversity of extracted data (OR, RR, SMD, and pooled mean difference).
Despite these limitations, our study has notable strengths. It is the first comprehensive review providing a complete overview of available evidence in the field of nutritional psychiatry in the paediatric population. Our approach to include the results of several meta-analyses facilitates a thorough examination of the available evidence. Furthermore, to mitigate bias related to the overlap of included meta-analyses, we minimized the overall CCA by excluding the majority of meta-analyses with significant overlap (≥15%). This approach reduced overlap for each studied disorder, allowing for a more accurate assessment of our data. Additionally, we systematically assessed the risk of bias for each included meta-analysis using the AMSTAR tool, enhancing the reliability and validity of our findings.
Future Perspective
A potential next step in this field of research is to shift from a categorical diagnostic approach to a dimensional one with a transdiagnostic perspective such as the Research Domain Criteria approach framework. 57 For example, research can be conducted to understand how specific components of the diet, such as nutrients, antioxidants, omega-3 fatty acids, etc., may modulate emotional responses, cognitive processes, and stress responses, and influence psychiatric symptoms. This future research should be reported to the ISNPR to promote research in the field of nutritional medicine for child and adolescent psychiatry.
Conclusion
Our results emphasize the necessity of establishing clear causal relationships between dietary habits and the risk of mental illness through interventional studies relying on comprehensive dietary strategies, such as the Mediterranean diet or a healthy diet, particularly in the paediatric population. Furthermore, emerging evidence suggests that omega-3 and vitamin D may be beneficial for ADHD in children and adolescents. However, it is essential to interpret these results with caution, and conducting larger-scale RCTs is imperative to establish more robust conclusions.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437241248070 - Supplemental material for The Role of Dietary Patterns and Nutritional Supplements in the Management of Mental Disorders in Children and Adolescents: An Umbrella Review of Meta-Analyses: Le rôle des habitudes alimentaires et des suppléments nutritionnels dans la prise en charge des troubles mentaux chez les enfants et les adolescents : une méta-revue de méta-analyses
Supplemental material, sj-docx-1-cpa-10.1177_07067437241248070 for The Role of Dietary Patterns and Nutritional Supplements in the Management of Mental Disorders in Children and Adolescents: An Umbrella Review of Meta-Analyses: Le rôle des habitudes alimentaires et des suppléments nutritionnels dans la prise en charge des troubles mentaux chez les enfants et les adolescents : une méta-revue de méta-analyses by Maria Talib, Majda Rachdi, Anna Papazova and Hélène Nicolis in The Canadian Journal of Psychiatry
Footnotes
Acknowledgments
We express our sincere appreciation to Valérie Durieux for her assistance in conducting the literature search. Additionally, we extend our thanks to Professor Stamatios Nicolis for reviewing the English language of the present article.
Data Availability
Data and materials associated with this rapid review are available upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
