Abstract
Objective
This study represents the inaugural attempt to systematically review and analyse the efficacy of bright light therapy on depression among women experiencing major depressive disorder or depressive symptoms during the perinatal period, encompassing its efficacy on depression scores, remission rates, and response rates.
Methods
We searched 10 databases for randomized controlled trials examining bright light therapy's efficacy on perinatal depression up to January 2024. Data extraction was performed independently by 2 investigators. The Cochrane Handbook guidelines appraised the study quality, and the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) approach assessed evidence certainty.
Results
We incorporated 6 studies, encompassing 151 participants. When contrasted with dim light therapy, bright light therapy did not significantly alter depression scores (standard mean difference = −0.29, 95% confidence interval [CI], −0.62 to 0.04, P = 0.08, I² = 34%) or response rates (risk ratio [RR] = 1.56, 95% CI, 0.98 to 2.49, P = 0.06, I² = 0%) in women experiencing perinatal depression. Conversely, bright light therapy was associated with a substantial increase in remission rates (RR = 2.63, 95% CI, 1.29 to 5.38, P = 0.008, I² = 2%).
Conclusion
Bright light therapy did not show efficacy in treating perinatal depression in terms of depression scores and response rates. However, regarding the remission rate, bright light did show efficacy compared to control conditions. Due to the limited sample size in the included studies, type II err or may occur. To obtain more conclusive evidence, future studies must employ larger sample sizes.
Background
Perinatal depression, which is characterized by a persistent depressed mood and diminished interest in activities, 1 encompasses both antenatal and postpartum depression. A systematic review of the literature up to April 2021 revealed that perinatal depression prevalence rates stand at 24.7% in upper-middle-income countries, 25.5% in lower-middle-income countries, and 20.7% in low-income nations. 2 The prevalence of perinatal depression varies but remains high across countries. Kumar 3 reported that the differences in prevalence between low- and middle-income countries are attributed to increased social support in low-income countries. Perinatal depression poses a significant risk to both maternal and infant health.4–6 Studies have demonstrated that affected mothers are at an increased risk of developing pre-eclampsia and suicidal ideation.7,8 Furthermore, the progeny of depressed mothers are at a heightened risk of several adverse outcomes, including preterm birth, inhibited postnatal growth, obesity, deficient social skills, and frequent episodes of diarrhea. 9
Management strategies for perinatal depression are bifurcated into pharmacological and nonpharmacological therapies. However, the use of antidepressants during pregnancy has the potential to adversely affect both maternal and infant health. The most commonly used antidepressants during pregnancy are selective serotonin reuptake inhibitors (SSRIs) and tricyclic drugs. 10 Numerous studies have shown that exposure to SSRIs in pregnant women increases the risk of postpartum haemorrhage, 11 persistent pulmonary hypertension in newborns,12–14 and potential neuroanatomical changes in children, 15 and exposure to tricyclic drugs in pregnant women may increase the risk of pre-eclampsia and postpartum bleeding.11,16 Consequently, only a minority of pregnant women with depression are amenable to pharmacotherapy. 17 Contrarily, nonpharmacological interventions are generally met with greater acceptance among this demographic. 18
Bright light therapy, a nonpharmacological modality, involves the administration of bright fluorescent light from a specialized apparatus for a designated time period. Originally established for seasonal affective disorder treatment, bright light therapy has since been validated for nonseasonal depressive disorders as well.19,20 Although the mechanism of action of bright light therapy remains incompletely understood, several hypotheses have been proposed. The main hypotheses suggest that bright light therapy may influence mood through the retinal pathway and indirectly regulate mood by normalizing sleep patterns and circadian rhythms.21,22
While bright light therapy is a recommended first- or second-line treatment for major depressive disorder in clinical guidelines, 23 its efficacy for perinatal depression remains controversial. For instance, Donmez et al. 24 reported a significant impact of bright light therapy on perinatal depression, while Bais et al. 25 found the efficacy to be nonsignificant. This divergence underscores the necessity for a systematic synthesis of the available evidence. Although a meta-analysis of the efficacy of bright light therapy on depression scores in pregnant and postpartum women was published in 2023, 26 none of the meta-analyses examined the efficacy of bright light therapy on depression in women experiencing major depressive disorder or depressive symptoms during the perinatal period. Therefore, our study is the first systematic review and meta-analysis of randomized controlled trials to assess the impact of bright light therapy on perinatal depression in women experiencing major depressive disorder or depressive symptoms during the perinatal period, encompassing its efficacy on depression scores, remission rates, and response rates.
Methods
We registered this systematic review and meta-analysis in INPLASY (https://inplasy.com/) before initiating this study. The INPLASY registration number was INPLASY2022110007.
Search Strategy
A comprehensive search was conducted across 10 databases: CINAHL, PubMed, Embase, Web of Science, Cochrane Library, Clinical Trials, China National Knowledge Infrastructure, VIP Database, Wanfang Database, and Sinomed. The search aimed to identify studies assessing the impact of bright light therapy on perinatal depression up to January 2024. Searches were executed independently by 2 reviewers, with any uncertainties resolved by consultation with a third reviewer to finalize the search outcomes. The search strategy was designed to combine MeSH terms (depression, pregnancy, and phototherapy) and corresponding free words. The exhaustive search strategy is elaborated in Supplemental Appendix A (Tables A.1–A.5).
Inclusion and Exclusion Criteria
Studies were included if they met the following criteria: (a) participants were pregnant or postpartum women diagnosed with perinatal depression according to standardized criteria (e.g., DSM-IV, DSM-5, and ICD-10) or assessed via clinician-rated instruments (e.g., Structured Interview Guide for the Hamilton Depression Rating Scale-Seasonal Affective Disorders Version [SIGH-SAD], 29-Item Structured Interview Guide for the Hamilton Depression Rating Scale with Atypical Depression Supplement [SIGH-ADS-29]) or self-report scale (e.g., Edinburgh Postnatal Depression Scale [EPDS]); (b) bright light therapy as the only or main treatment method, if there are adjuvant therapies (such as antidepressants or sleep deprivation), the control group and intervention group should be treated equally; (c) include a placebo control, such as dim light, negative air ions, or implement bright light therapy with the same intensity and duration at different times of the day as the control group; (d) depression scores were measured both pretreatment and posttreatment; (e) the methodological design was a randomized controlled trial.
Exclusion criteria encompassed studies with (a) mixed populations of unipolar and bipolar patients where data specific to unipolar patients could not be isolated; (b) abstracts, review articles, conferences, and articles published in non-English or non-Chinese.
Study Selection and Data Extraction
Two extractors independently extracted article information, which included characteristics of the included studies (authors, year of publication, country, diagnostic criteria, and sample size), characteristics of intervention programs in the control group and intervention group (control method, light intensity, and duration) and outcomes (outcome indicators, side effects, depression scores, response rate, and remission rate).
Quality Assessment
Two reviewers independently assessed the quality of the included studies using the Cochrane Handbook. This assessment scrutinized potential biases across 7 domains: selection bias (including random sequence generation and allocation concealment), performance bias, detection bias, attrition bias, reporting bias, and other biases. In case of discrepancies in evaluation, these were resolved through discussion with a third reviewer.
Evidence quality was gauged according to the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) methodology, taking into account the risk of bias, inconsistency, indirectness, imprecision, and publication bias. This process categorized the certainty of evidence into four levels: high, moderate, low, and very low. Finally, the certainty of the evidence was evaluated independently by 2 reviewers and, if controversial, was discussed with a third reviewer to produce the final result.
Statistical Analysis
The efficacy of bright light therapy in treating perinatal depression, characterized by major depressive disorder or depressive symptomatology, was quantitatively analysed by examining primary and secondary outcomes. Primary outcomes were delineated by changes in depression scores, assessed using validated scales such as SIGH-SAD, 27 SIGH-ADS-29, 28 and EPDS. 29 Where multiple assessment tools were utilized, selection of the outcome measure was informed by the quality of the scale and the extent of heterogeneity across scales. Priority was given to SIGH-SAD or SIGH-ADS-29 due to their structured and comprehensive approach to assessing perinatal depression, offering clear criteria for depressive symptoms.
Secondary outcomes encompassed: (a) remission rates, defined as the proportion of participants achieving scores indicative of remission upon treatment completion (SIGH-SAD < 8; SIGH-ADS-29 ≤ 8; and EPDS < 12); (b) response rate, delineated as the proportion of participants experiencing a clinically significant improvement in depression scores (≥50% reduction) at the end of treatment.
Data analysis was conducted utilizing Review Manager (RevMan) 5.4. Effect sizes for dichotomous outcomes (response rate and remission rate) were expressed as risk ratios (RRs). Effect sizes for continuous-type variables (e.g., depression score) were expressed as mean difference (MD) or standardized MD (SMD). SMD was chosen when the measurement tools for the result were different, and MD was selected when the measurement tools for the result were the same. All effect sizes were calculated using point estimates and 95% confidence intervals (CIs), and P < 0.05 were considered statistically significant. Heterogeneity was examined using Cochran's Q test and quantified by Higgins’ I² statistic, with P < 0.1 indicating notable heterogeneity. Degrees of heterogeneity were categorized as low (I² < 25%), moderate (25% ≤ I² < 50%), or high (I² ≥ 50%). 30 A random effects model was employed in the presence of significant heterogeneity (P ≤ 0.1, I² ≥ 50%), whereas a fixed effects model was applied otherwise. Sources of heterogeneity were explored via subgroup analyses, and the robustness of the results was assessed through sensitivity analyses.
Results
Search Results
An initial search across 10 databases yielded 538 articles that were potentially relevant. Screening of titles and abstracts resulted in the exclusion of 311 articles due to irrelevance to the predetermined topics, interventions, or study designs. Further examination of full texts led to the exclusion of 28 articles that did not fulfil the specified intervention or study design requirements. Six studies ultimately conformed to the stringent inclusion criteria and were incorporated into this systematic review and meta-analysis24,25,31–34 (Figure 1).
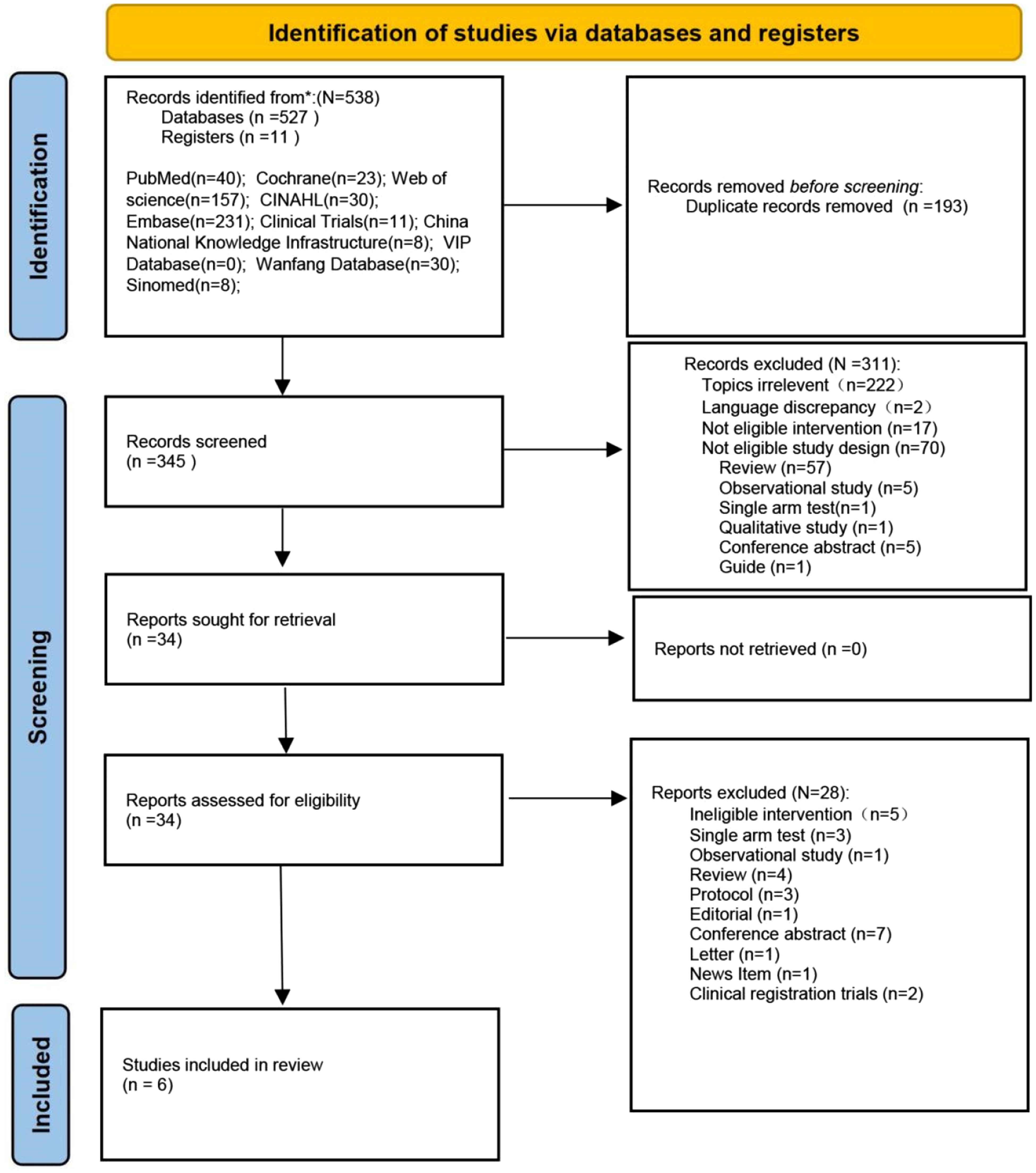
Flowchart of literature identification and selection.
Characteristics of the Included Studies
Table 1 delineates the study characteristics, encapsulating participant demographics, interventions, control conditions, and measurement instruments. The sample sizes across these studies varied, encompassing 8 to 56 participants. Geographically, 2 studies were undertaken in Switzerland,31,32 with the remainder conducted in Turkey, 24 the Netherlands, 25 Canada, 33 and the United States. 34 The temporal focus of the studies varied, with 3 addressing the prenatal period,25,31,34 1 addressing the postnatal period, 33 and 2 spanning the perinatal phase.24,32 The assessment of depression was conducted using an array of scales, including SIGH-SAD,25,33,34 EPDS,24,25,32,33 Hamilton Rating Scale for Depression (HAM-D),24,25,31 SIGH-ADS-29, 31 and the Montgomery–Asberg Depression Rating Scale (MADRS). 24
Characteristics of the Included Studies.
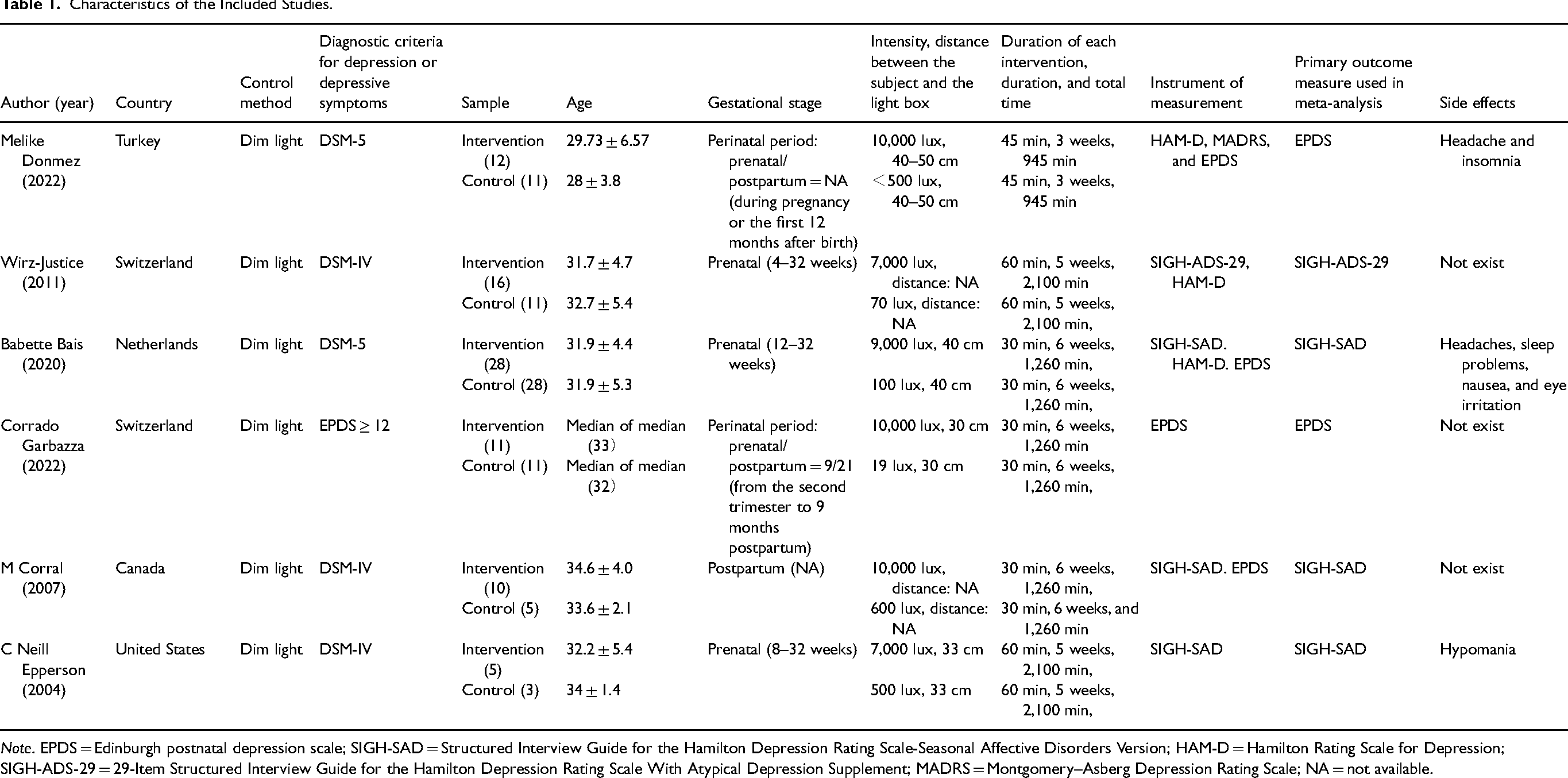
Note. EPDS = Edinburgh postnatal depression scale; SIGH-SAD = Structured Interview Guide for the Hamilton Depression Rating Scale-Seasonal Affective Disorders Version; HAM-D = Hamilton Rating Scale for Depression; SIGH-ADS-29 = 29-Item Structured Interview Guide for the Hamilton Depression Rating Scale With Atypical Depression Supplement; MADRS = Montgomery–Asberg Depression Rating Scale; NA = not available.
Variations were noted in the parameters of the bright light therapy across studies, including light intensity (10,000 lux, 9,000 lux, and 7,000 lux), exposure duration (30, 45, 60, and 75 min per day), and intervention span (3, 5, and 6 weeks).
Risk of Bias
The risk of bias assessment is visualized in Figure 2. Within this assessment, 2 studies were identified as having a high risk of performance bias; one because only the participants were blinded, and the other because only the investigators were blinded.32,33 Additionally, 1 study was deemed to have a high risk of attrition bias attributable to incomplete outcome data reporting. 34 Two other studies were classified as high risk for other biases: one received funding from a commercial entity, Philips, 32 while the other included a sample divergent from the protocol. 25
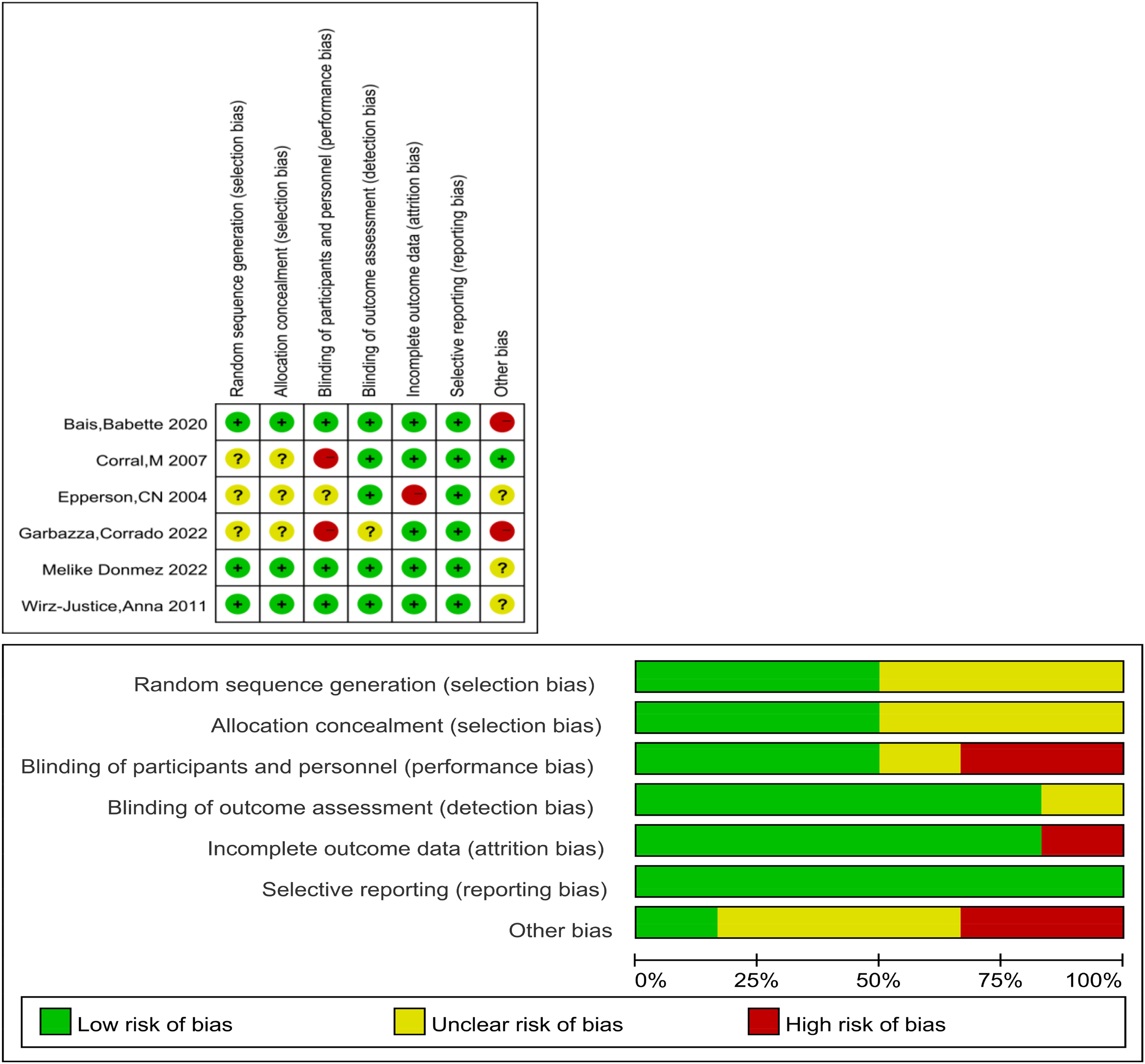
The result of quality assessment.
Quality of Evidence
Based on the GRADE rating system, the evidence quality for the 3 specified outcomes (depression score differences, remission rates, and response rates) was deemed low. The rationale for downgrading the quality included limited sample sizes, broad confidence intervals, and a lack of double-blinding in the methodology. The details of the quality of evidence assessment are shown in Supplemental Appendix Table A6.
Overall Effect Analysis
All 6 studies provided depression scores and were incorporated into the meta-analysis (Figure 3). Given the diversity of measurement tools used, results were aggregated using SMD. The heterogeneity test indicated minimal variance, allowing for the application of a fixed effects model. The analysis revealed no significant difference in the change in depression scores between the intervention and control groups (SMD = −0.29, 95% CI, −0.62 to 0.04, P = 0.08).
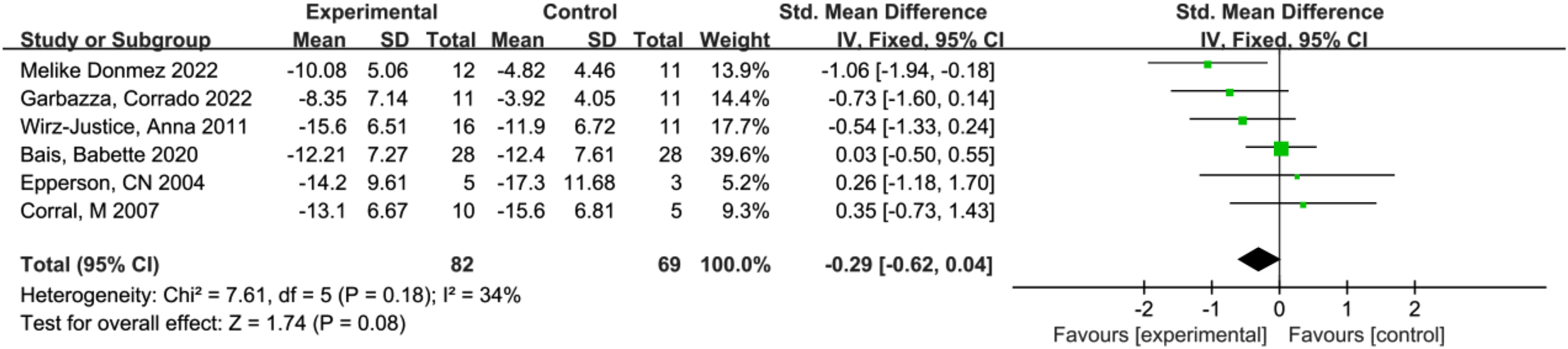
Forest plot of postintervention treatment effect sizes for scores of depressive symptoms.
Regarding the secondary outcomes, remission rates were addressed in 4 studies.24,31–33 Supplemental Figure A1 shows that bright light therapy significantly improved remission rates in perinatal depression compared to controls (RR = 2.63, 95% CI, 1.29 to 5.38, P = 0.008).
Moreover, the response rates to bright light therapy in perinatal depression were reported in 5 studies.24,31–34 According to Supplemental Figure A2, there was no statistically significant enhancement in response rates for perinatal depression with bright light therapy when compared to control groups (RR = 1.56, 95% CI, 0.98 to 2.49, P = 0.06).
Subgroup Analysis
Subgroup analyses were conducted to examine the potential influence of intervention duration, outcome measures, and gestational stages on the variation in depression scores, as presented in Table 2.
Subgroup Analyses.
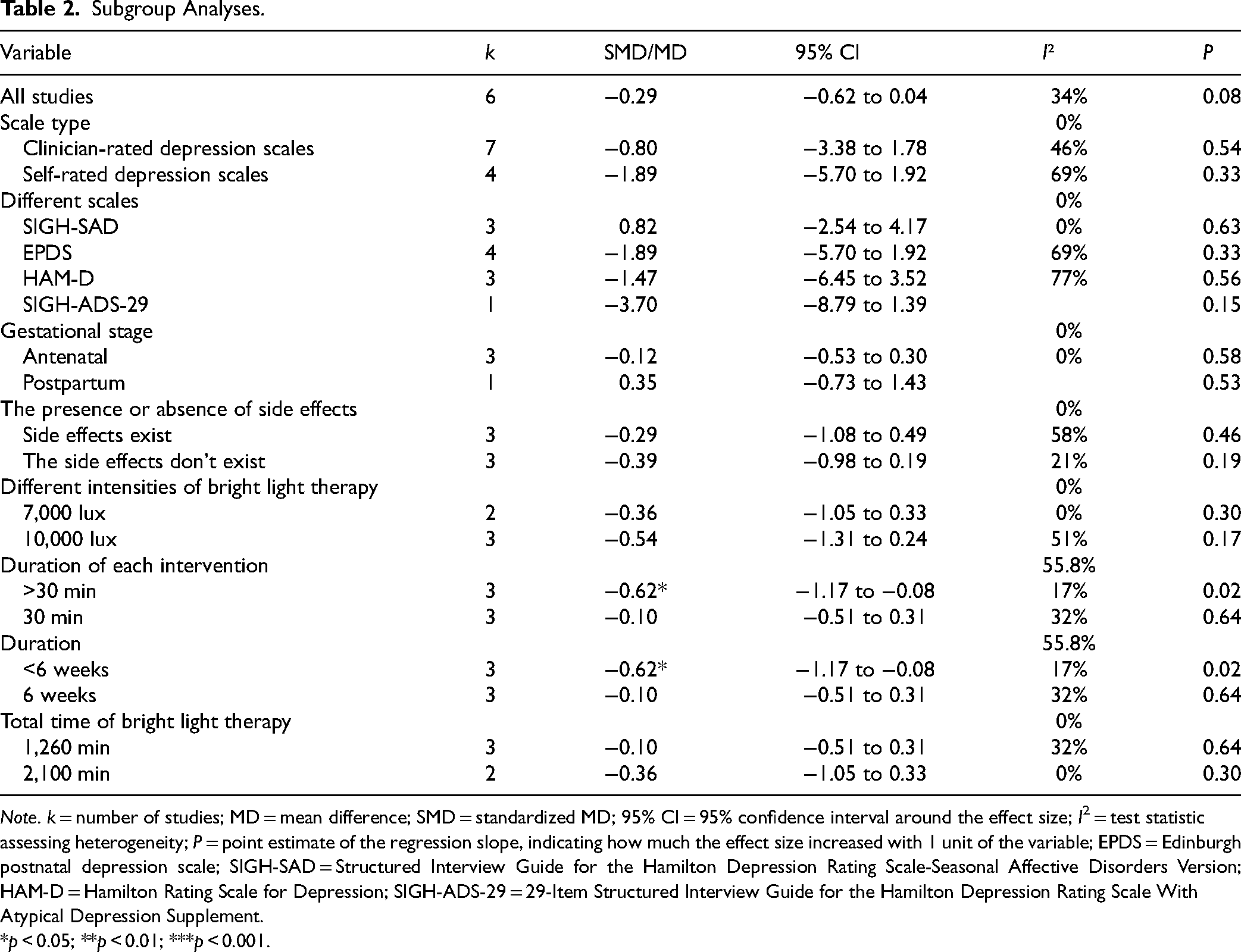
Note. k = number of studies; MD = mean difference; SMD = standardized MD; 95% CI = 95% confidence interval around the effect size; I2 = test statistic assessing heterogeneity; P = point estimate of the regression slope, indicating how much the effect size increased with 1 unit of the variable; EPDS = Edinburgh postnatal depression scale; SIGH-SAD = Structured Interview Guide for the Hamilton Depression Rating Scale-Seasonal Affective Disorders Version; HAM-D = Hamilton Rating Scale for Depression; SIGH-ADS-29 = 29-Item Structured Interview Guide for the Hamilton Depression Rating Scale With Atypical Depression Supplement. *p < 0.05; **p < 0.01; ***p < 0.001.
Scales
According to the types of scales, scales were divided into clinician-rated depression scales and self-rated depression scales. The results showed that there was no significant difference in the change of perinatal depression scores between bright light and dim light whether using clinician-rated depression scales or self-rated depression scales: clinician-rated depression scales24,25,31,33,34 (SMD = −0.80, 95% CI, −3.38 to 1.78, P = 0.54); self-rated depression scales24,25,32,33 (SMD = −1.89, 95% CI, −5.70 to 1.92, P = 0.33).
When evaluating different measurement instruments, there were no statistically significant disparities in perinatal depression score reduction between the bright light therapy and dim light therapy: SIGH-SAD25,33,34 (MD = 0.82, 95% CI, −2.54 to 4.17, P = 0.63); EPDS24,25,32,33 (MD = −1.89, 95% CI, −5.70 to 1.92, P = 0.33; HAM-D24,25,31 (MD = −1.47, 95% CI, −6.45 to 3.52, P = 0.56); SIGH-ADS-29 (MD = −3.70, 95% CI, −8.79 to 1.39, P = 0.15). 31
Gestational stage
Subgroup analyses across different gestational stages indicated an absence of significant differences in perinatal depression score improvement between bright light therapy and dim light therapy. During pregnancy,25,31,34 the SMD was −0.12 (95% CI, −0.53 to 0.30; P = 0.58), and in the postpartum phase, 33 the SMD was 0.35 (95% CI, −0.73 to 1.43; P = 0.53).
Adverse events
Subgroup analyses based on the occurrence of side effects revealed no significant differences in perinatal depression score improvements between bright light therapy and dim light therapy groups, regardless of side effects’ presence: with side effects24,25,34 (SMD = 0.29, 95% CI, −1.08 to 0.49, P = 0.46); without side effects33,31,32 (SMD = −0.39, 95% CI, −0.98 to 0.19, P = 0.19).
Different intensity of bright light therapy
Analyses of different intensities of bright light therapy demonstrated a statistically insignificant variance in the reduction of perinatal depression scores in comparison to dim light therapy: 7,000 lux31,34 (SMD = −0.36, 95% CI, −1.05 to 0.33, P = 0.30); 10,000 lux24,32,33 (SMD = −0.54, 95% CI, −1.13 to 0.24, P = 0.17).
Duration of each intervention
Interventions exceeding 30 min in duration yielded a statistically significant reduction in perinatal depression scores for the bright light therapy group compared to the placebo groups24,31,34 (SMD = −0.62, 95% CI, −1.17 to −0.08, P = 0.02). Conversely, interventions lasting 30 min did not result in a statistically significant difference in perinatal depression scores between the 2 groups25,32,33 (SMD = −0.10, 95% CI, −0.51 to 0.31, P = 0.64).
Duration
For a duration <6 weeks,24,31,34 bright light therapy demonstrated significant efficacy in reducing perinatal depression scores compared to dim light therapy (SMD = −0.62, 95% CI, −1.17 to −0.08, P = 0.02). However, at a treatment duration of exactly 6 weeks,25,32,33 no statistical significance was observed in score reduction between bright light therapy and dim light therapy (SMD = −0.10, 95% CI, −0.51 to 0.31, P = 0.64).
Total time of bright light therapy
Varying total duration of bright light therapy did not produce a statistically significant difference in perinatal depression scores as compared to dim light therapy: 1260 min of treatment25,32,33 (SMD = −0.10, 95% CI, −0.51 to 0.31, P = 0.64); 2100 min31,34 (SMD = −0.36, 95% CI, −1.05 to 0.33, P = 0.30).
Sensitivity Analysis
Sensitivity analyses were conducted on depression score outcomes to evaluate the robustness of the findings. These analyses entailed the exclusion of individual studies sequentially, followed by aggregation of the effect sizes from the remaining studies. Both aggregated and original results indicated that light therapy did not have a statistically significant impact on the change in depressive scores in perinatal women. Additionally, sensitivity analyses incorporating different analytical models (random effects or fixed effects) corroborated the lack of efficacy of bright light therapy in ameliorating depressive symptoms.
Adherence and Adverse Events
Patient adherence is pivotal to therapeutic outcomes and consequently informs the appraisal of bright light therapy's efficacy in perinatal depression. In the studies reviewed, 3 failed to delineate the adherence rates of the intervention and control groups, precluding a meta-analytical synthesis of adherence data. Therefore, a qualitative overview of adherence was undertaken. Adherence monitoring was conducted weekly in 2 studies25,33 and daily in 3 others.24,31,34 Four studies captured adherence via self-report,24,25,33,34 while 1 utilized assessment scales (mood and alertness ratings, along with sleep and light therapy logs). 31 Predominant reasons cited for suboptimal adherence were symptom resolution, 25 nonresponsiveness, 34 and the demanding nature of the therapy.32,34
Adverse events were evaluated in 6 studies, with 4 outlining the methodologies employed to detect these effects, such as questionnaire administration (Young Mania Rating Scale); 24 self-report version of the Systematic Assessment for Treatment Emergent Effects31,32) and periodic verbal inquiries. 25 During the intervention phase, all 6 studies investigated the adverse events, with 3 studies indicating their presence and 2 studies documenting severe adverse events. Epperson et al.'s study revealed that a participant in the bright light intervention group experienced a severe side effect: hypomania. 34 Donmez et al. 24 reported that 1 participant in the intervention group terminated the intervention owing to headaches. Furthermore, Donmez et al. 24 reported the occurrences of minor headaches and insomnia among participants, while Bais et al. 25 observed minor adverse events such as headaches, sleep problems, nausea, and eye irritation in participants during the intervention phase. Owing to the lack of clarity regarding the groups in which these side effects arose, an analytical assessment of side effects as an endpoint was unattainable.
Discussion
This systematic review and meta-analysis represents the inaugural comprehensive evaluation of randomized controlled trials assessing the efficacy of bright light therapy in the treatment of depression among perinatal women experiencing major depressive disorder or depressive symptoms during the perinatal period, in terms of depression scores, remission rates, and response rates.
Our result indicated that bright light therapy did not demonstrate efficacy in ameliorating depression scores in perinatal women. In contrast, Li et al. 26 meta-analysis revealed a significant improvement in depression scores among women during pregnancy and postpartum with bright light therapy. The disparity in results may be attributed to 3 primary factors. The first factor is the variation in the included literature resulting from different research objectives. Our study aimed to assess the efficacy of bright light therapy for depression, including depression scores, remission rates, and response rates, in perinatal women diagnosed with major depressive disorder or experiencing depressive symptoms. However, Li et al.'s study aimed to examine the efficacy of bright light therapy on depression scores among pregnant and postpartum women. This divergence leads to the exclusion of Verma et al.'s and Liu et al.'s studies from our analysis, whereas Li et al.'s study incorporates these 2 studies. The second factor is the variation in the included literature due to heterogeneity. Li et al.'s meta-analysis excluded Wirz-Justice et al. 31 study due to high heterogeneity, while our study included Wirz-Justice et al.'s study. The third main factor may be attributed to the utilization of different scales in the results analysis. For instance, in the study by Bais et al., the measurement tools for depression encompassed SIGH-SAD, HAM-D, and EPDS. For our study, we selected SIGH-SAD as the final outcome measurement tool based on its comprehensiveness and structured format. However, Li et al. opted for the EPDS scale in their analysis.
In addition, our results showed no significant change in response rates compared to the control group. Nevertheless, an improved remission rate was observed, indicating some therapeutic benefit. The mixed results may be attributed to the limitations of the included studies. Firstly, the variability in light intensity and intervention duration across studies necessitates a cautious interpretation of the data. Secondly, the limited sample size poses a risk of type II error. Thirdly, given the absence of a clear threshold for the light effect at present, the potential beneficial impacts of dim light therapy as a control group cannot be disregarded. To derive more definitive conclusions, future research should aim for standardization in light therapy protocols, increased sample sizes, and refined study designs.
The subgroup analysis examining the relationship between side effects and perinatal depression scores indicated no significant disparities between the intervention and control groups, irrespective of the occurrence of side effects during the intervention period. Although the results were consistent between the side-effect and no-side-effect groups, the validity of these findings is constrained by the small number of studies included in the subgroup analysis. It is noted that side effects led to treatment discontinuation in 4 participants across 2 studies, suggesting an impact on treatment adherence and, consequently, on therapeutic efficacy.24,25 Given the propensity for physiological changes induced by side effects to be perceived negatively by individuals, the onset of such side effects could precipitate depressive symptoms, thereby attenuating the benefits of bright light therapy. 35 Minimizing side effects is thus essential to the success of bright light therapy. Research indicates that lower light intensities are associated with reduced side effects and that administering light therapy during the evening may result in fewer adverse outcomes compared to morning sessions.36,37 Special consideration should be given to patients on lithium therapy due to their increased susceptibility to the side effects of bright light therapy. 38 Future research on bright light therapy for perinatal depression should prioritize the minimization of side effects to enhance therapeutic efficacy.
When conducting subgroup analyses on the intensity of bright light therapy utilizing a random effects model, no significant differences were noted when compared with placebo. Nevertheless, the heterogeneity of intensity at 51% suggests a threshold where the random effects model converges with the fixed effects model. To ascertain the robustness of these results, the application of a fixed effect model revealed that an intensity of 10,000 lux was efficacious in ameliorating symptoms of perinatal depression. The Canadian consensus guidelines corroborate the recommendation of a 10,000 lux intensity for a duration of 30 min. 39 This variability in model outcomes underlines the instability of the therapeutic efficacy in our study and highlights the necessity for further investigation into the influence of varying light intensities on perinatal depression.
Interventions exceeding 30 min in duration did not demonstrate a significant impact of bright light therapy, whereas sessions precisely 30 min evidenced a significant therapeutic efficacy. Bright light therapy for durations shorter than 6 weeks proved to be more efficacious than placebo in mitigating symptoms of perinatal depression. Previous meta-analytic findings suggest no substantial benefit of bright light therapy for periods ≥4 weeks over shorter durations of 1 to 3 weeks, though all durations assessed indicated the therapy's efficacy for depression. 39 Moreover, the onset of therapeutic efficacy of bright light therapy on depressive symptoms has been observed within 2 to 3 weeks of commencement. 40 Thus, the efficacy of bright light therapy may accumulate and saturate over time, and subsequent treatments may reduce its therapeutic efficacy. Furthermore, the convergence of the intervention duration being <6 weeks across 3 studies included in this research, which is the same as the articles stipulating a daily intervention period of 30 min, complicates the attribution of improvement in perinatal depression to specific factors. Future research is warranted to meticulously investigate the impact of intervention timing.
Given that all studies reviewed herein conducted interventions in the morning, no subgroup analysis regarding the timing of the intervention was performed. Parry et al.’s 41 study indicated that women experiencing depression during pregnancy demonstrated an advanced plasma melatonin phase, whereas those with postpartum depression exhibited delayed phases. This suggests that bright light at night, by altering circadian rhythm, may be more efficacious for depression during pregnancy, while morning exposure periodically advances melatonin rhythm, potentially proving more efficacious for postpartum depression. 42 Subsequent research can be conducted to verify the efficacy of bright light therapy by selecting the appropriate time for bright light therapy based on specific situations.
Strengths and Limitations
Our study possesses several strengths. Primarily, it synthesizes extant studies concerning bright light therapy for perinatal depression, yielding a comprehensive aggregation of results. Additionally, subgroup analyses were performed, examining the impact of variables such as intervention duration, outcome measures, and gestational periods, thereby facilitating a nuanced understanding of these factors on therapeutic outcomes. Finally, the quality of the evidence was thoroughly assessed, allowing for a more objective appraisal of the results.
Nevertheless, our study has some limitations that should be taken into consideration. It is restricted to Chinese and English language publications, potentially overlooking pertinent studies in other languages. In addition, the sample size was relatively small, comprising only 151 subjects, which could increase the risk of type II error occurrence and potentially affect the generalizability of the research findings. Moreover, studies have shown that bright light therapy can be integrated with wake therapy and pharmacotherapy, potentially augmenting their therapeutic impact and extending their efficacious duration.43–45 However, since bright light therapy was not combined with other treatments in the included studies, the efficacy of bright light therapy in combination therapy was missing in our study. Ultimately, due to the absence of a definitive threshold indicating a positive light effect at present, it is not possible to entirely dismiss the potential beneficial impact of dim light therapy. In summary, these limitations necessitate a cautious interpretation of the study's outcomes.
Conclusions
Bright light therapy did not significantly outperform dim light therapy in reducing depression scores or enhancing the response rate among women with perinatal depression; however, it did significantly improve remission rates. According to the results of our research, there is no persuasive evidence to support the absolute efficacy of bright light therapy in treating perinatal depression. Additionally, the limited sample size in the included studies may lead to type II error, ultimately impacting the generalizability of the results. Nevertheless, considering the fewer observed side effects and higher compliance rate, bright light therapy may be regarded as a safe and feasible option. Future research should aim to conduct larger-scale, high-quality randomized controlled trials to further elucidate and affirm the efficacy of bright light therapy for this population.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437241248051 - Supplemental material for Effect of Bright Light Therapy on Perinatal Depression: A Systematic Review and Meta-Analysis: Effet de la luminothérapie sur la dépression périnatale: une revue systématique et une méta-analyse
Supplemental material, sj-docx-1-cpa-10.1177_07067437241248051 for Effect of Bright Light Therapy on Perinatal Depression: A Systematic Review and Meta-Analysis: Effet de la luminothérapie sur la dépression périnatale: une revue systématique et une méta-analyse by Yujia Chen, Jing Zhao, Jiarun Wang, Li Peng, Zhongxiang Cai, Zhijie Zou and Xiaoli Chen in The Canadian Journal of Psychiatry
Footnotes
Acknowledgements
The authors would like to thank Dr. Xiaoli Chen and Dr. Zhijie Zou for their input and assistance in this study.
Availability of Data
The data used in this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The Fundamental Research Funds for the Central Universities (grant number 2021PT073).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
