Abstract
Objective
This study aimed to evaluate the effectiveness of flexible take-home dosing of buprenorphine/naloxone (BUP/NX) and methadone standard model of care in reducing depressive symptoms in people with prescription-type opioid use disorder (POUD). This trial also evaluated whether improvements in depressive symptoms were mediated by opioid use.
Methods
Analyzed data came from the OPTIMA study (clinicaltrials.gov identifier: NCT03033732), a pragmatic randomised controlled trial comparing flexible take-home dosing of BUP/NX and methadone standard model of care for reducing opioid use in people with POUD. A total of 272 participants were recruited in four Canadian provinces. Participants were randomised 1:1 to BUP/NX or methadone. After treatment induction, past two-week opioid use was measured using the Timeline Followback every two weeks for a total of 24 weeks. Depressive symptoms were measured with the Beck Depression Inventory at baseline, weeks 12 and 24.
Results
Both BUP/NX and methadone significantly reduced depressive symptoms at week 12 (aβ ± SE = −3.167 ± 1.233;
Conclusions
BUP/NX and methadone showed similar effectiveness in decreasing comorbid depressive symptoms in people with POUD. This effect was partially explained by a reduction in opioid use. As both treatments seem equally effective, clinicians are encouraged to tailor the selection of OAT to patients’ needs and characteristics.
Introduction
Between 47 and 60 million individuals use opioids for nonmedical reasons, with the highest rate being in North America.1,2 Among people with prescription-type opioid use disorder (POUD), depression is the most common psychiatric comorbidity, with an estimated current prevalence between 9% and 61%.3,4 Opioid use disorder is also associated with an increased risk of developing subsequent depression that persists for decades. 5 Traditional treatments for depression are challenging to implement in OUD populations. Indeed, pharmacological interactions between some antidepressants and OATs can lead to reduced bioavailability and effectiveness6,7 and increased cardiotoxicity. 8 Moreover, there is only a little evidence of antidepressants effectiveness in people with OUD as these populations have classically been excluded from such trials. Psychotherapy may also be difficult to deliver to people with OUD, as they often experience unstable living conditions, cognitive impairments, or limited financial resources. 9
Two OATs – buprenorphine/naloxone (BUP/NX) and methadone – have shown beneficial impacts on depressive symptomatology. BUP/NX can significantly improve depressive symptoms, independently of drug use, or withdrawal symptoms.10,17 A recent open-label clinical trial evaluating BUP/NX for treatment-resistant depression in people with opioid use disorder (OUD) reported a significant reduction in depressive mood 48 h after BUP/NX initiation. 18 Karp and colleagues also found a significant decline in depression severity during BUP/NX treatment and a sudden increase when treatment was discontinued. 1
Methadone may also improve depressive symptoms, although studies are relatively limited. In a randomised controlled trial comparing methadone in combination with fluoxetine to methadone alone, both treatment arms significantly reduced depressive symptoms compared to baseline. 20 Improvements were unrelated to drug use, and the addition of fluoxetine did not confer additional improvements. 20 Another study found that methadone can quickly alleviate depressive states associated with opioid withdrawal. 21 A recent meta-analysis on the effect of methadone on depression among people with OUD found significant reductions in depressive symptoms following methadone initiation. 22
BUP/NX is thought to have a superior antidepressant profile than methadone due to its antagonist action on kappa opioid receptors.10,15,18,23 BUP/NX also interacts with serotoninergic and hypothalamic–pituitary–adrenal systems,10,19 two systems involved in the pathogenesis and maintenance of depression. To our knowledge, the only study that compared the effectiveness of methadone and buprenorphine (without naloxone) in decreasing depressive symptoms found that both treatments improved depression in heroin-dependent subjects. Baseline depressive symptoms and treatment retention were predictive of symptoms at three months for participants receiving methadone but not for those receiving buprenorphine. These results show that buprenorphine can effectively alleviate depressive symptoms regardless of depression severity and may have a longer term effect. Therefore, although study conclusions were limited by its small sample size, the authors hypothesised that buprenorphine might have a differential impact on depressive symptoms and recommended further trials to evaluate this hypothesis. 24
Since this latter study published in 2004, no recent data have been published on the antidepressant properties of OATs in people treated for OUD. However, trends in opioid use have changed drastically since the 2000s, switching from mainly heroin use to higher potency prescription opioids. 25 Indeed, from 1999 to 2011, prescription opioid misuse increased between two- and five-fold. 26 It remains unclear whether studies conducted in samples with heroin use disorder can be extrapolated to POUD populations.
Considering that depression is highly prevalent in people with POUD, and that depression has been reported to be the strongest predictor of OATs outcomes, 27 new robust data on the antidepressant properties of OATs in POUD are urgently needed.
This secondary analysis evaluated the differential effectiveness of BUP/NX and methadone models of care in decreasing depressive symptomatology in Canadians with POUD. We further investigated whether decreases in depressive symptoms were mediated by the number of days of opioid use in the past two weeks. We hypothesised that flexible take-home dosing of BUP/NX would be superior to the methadone standard model of care in decreasing depressive symptomatology.
Methods
Study Design
Our study is a secondary analysis of data drawn from the OPTIMA study, a pan-Canadian, two-arm, open-label, multicentric, pragmatic randomised controlled trial designed to compare flexible take-home dosing of BUP/NX to the methadone standard model of care in treating POUD. OPTIMA's methodology and main outcomes have been published in detail elsewhere.28,29 This study was conducted in full conformity with the international Good Clinical Practice guidelines from the International Conference on Harmonisation (Declaration of Helsinki), Canadian (Tri-Council Policy Statement, Health Canada division 5), provincial, and institutional guidelines. All study sites’ institutional ethics review boards have approved this study, and all participants signed an informed consent after study procedures had been fully explained. The secondary analysis plan was not registered and has not been reviewed by ethics review boards, other than through the approval of the main trial.
Participants
A total of 272 participants were randomised. The main inclusion criteria were being aged between 18 and 64 years, having a POUD diagnosis (DSM-5 criteria), and being of non-childbearing potential or using contraception. Exclusion criteria included having any preexisting physiological or severe psychiatric conditions or using medication precluding safe participation, self-reporting using heroin as the most frequent opioid in the 30 days prior to enrollment, currently receiving OAT, having pain requiring opioid treatment, having a pending legal action preventing study completion, or being pregnant, breastfeeding, or planning to conceive during the study period. We excluded from analyses participants who did not provide any BDI-II data throughout the study.
Procedures
This study was led by the Canadian Research Initiative in Substance Misuse (CRISM) at the following sites: The Rapid Access Addiction Clinic (Vancouver, BC); the Portland Hotel Society Clinic (Vancouver, BC); the Opioid Dependency Program Clinic (Edmonton, AB); the Opioid Dependency Program Clinic (Calgary, AB); the Centre for Addiction and Mental Health (Toronto, ON); the Ontario Addiction Treatment Centre (Sudbury, ON); the Centre hospitalier de l’Université de Montréal (Montréal, QC); and the Centre de Recherche et d’Aide pour Narcomane (Montréal, QC). Eligible participants completed baseline assessments and were randomised in a 1:1 ratio to receive methadone or BUP/NX. Assigned OAT was initiated in accordance with local guidelines and Health Canada-approved product monographs. Follow-up visits took place every two weeks during which questionnaires were administered and urine samples were collected. Participants received a $40 compensation after each study visit, for a maximum of $560. For more details on procedures, refer to our previous publication.28,29
Measurements
Our main outcome was depressive symptoms, as measured by the Beck Depression Inventory – Second Version (BDI-II) at baseline, week 12, and week 24. The BDI-II questionnaire consists of 21 self-reported items resulting in a total score between 0 and 63, with higher scores representing more severe symptomatology. Opioid use was quantified using the Timeline Follow-Back (TLFB) every two weeks for the 24-week trial duration. For these analyses, we only included days of opioid use in the past two weeks prior to baseline, week 12, and week 24 visits.
Statistical Analyses
Primary Analyses
We used a repeated measure generalised linear model (rmGLM) to estimate the impact of OAT group on depressive symptoms using the
Post-hoc Analyses
We conducted
Handling of Missing Data
Main analyses were performed using collected data only. Participants with incomplete observations for the BDI-II (e.g., did not answer item 15) were considered as missing for this time point. For
Sensitivity Analyses
We performed per-protocol sensitivity analyses including 1) participants initiating the assigned treatment and 2) participants still actively in the study and having a positive urine drug screening to the assigned treatment at week 24 (i.e., retained participants). Given that recruitment and data collection were impacted by the COVID-19 pandemic, we also performed sensitivity analyses removing data collected after March 24, 2020.
Results
Participants
Figure 1 shows the CONSORT flow diagram (modified from Jutras-Aswad et al.
29
). A total of 272 eligible participants were randomised to methadone (
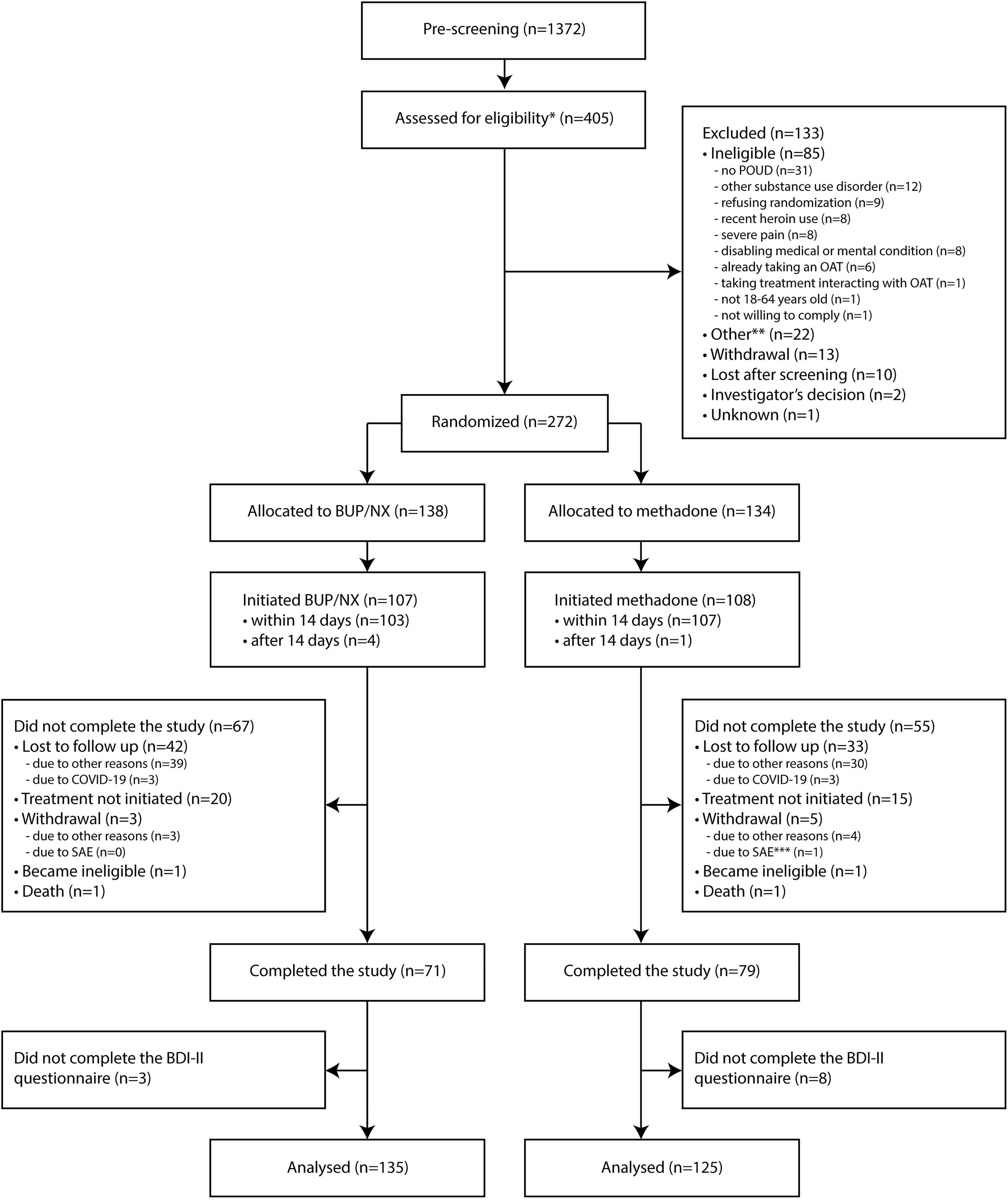
CONSORT flow diagram illustrating pre-screening, screening, randomising, and analysis with the number of participants for each step. *Thirteen participants were screened twice but are counted only once in the flowchart. **Other reasons for exclusions were unspecified (
Demographic and Characteristics of Analyzed Participants.
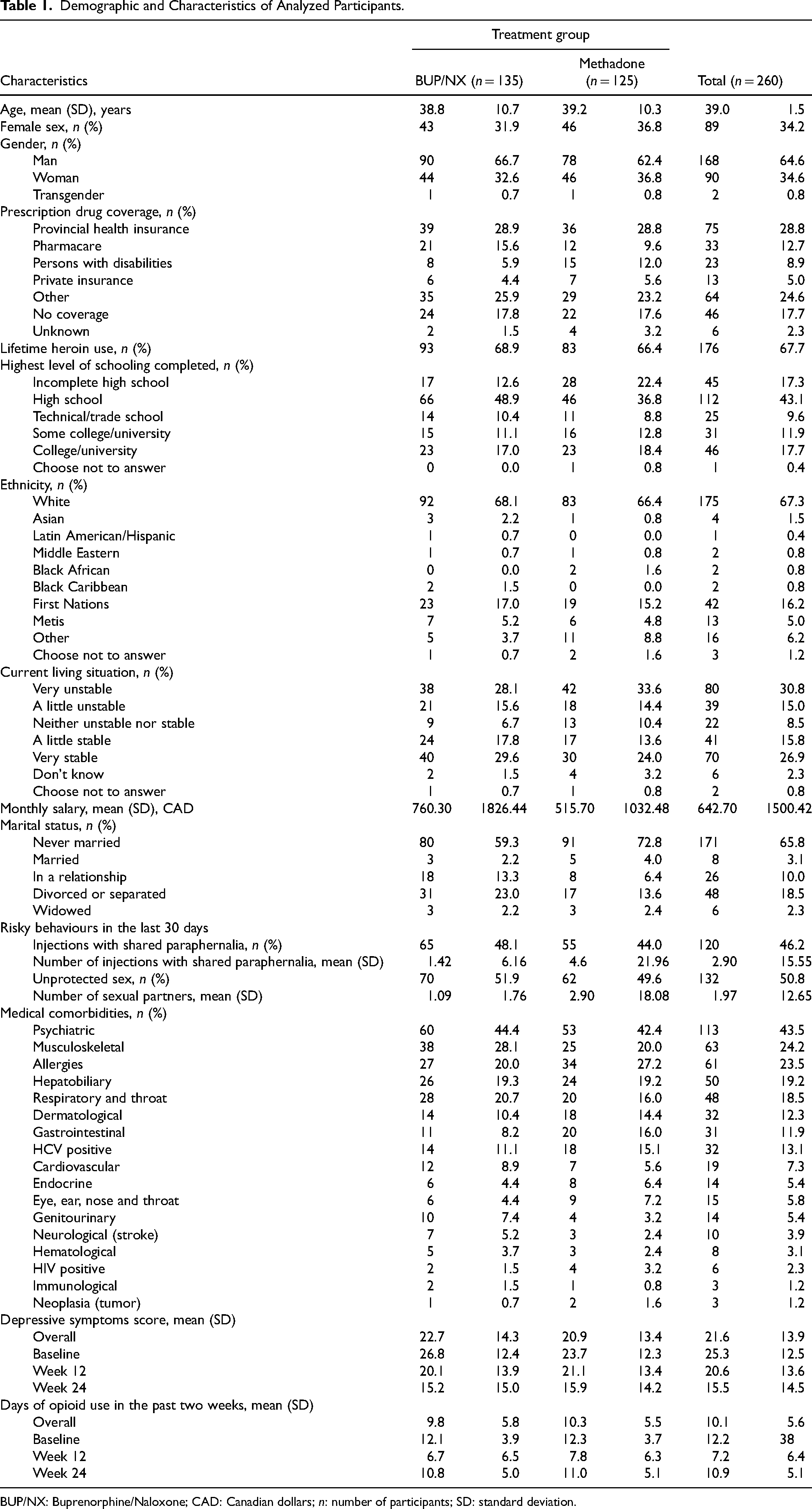
BUP/NX: Buprenorphine/Naloxone; CAD: Canadian dollars;
Depressive Symptoms Scores
Mean depressive symptoms scores in both methadone and BUP/NX groups throughout study visits are reported in Table 1 and illustrated in Figure 2. Baseline average depressive symptoms scores (±SD) were similar between treatments, with 23.66 (±12.26) for methadone and 26.84 (±12.44) for BUP/NX, indicating moderate depression severity. For methadone, mean depressive symptoms (±SD) showed a constant decrease, reaching 21.11 (±13.35) at week 12 and 15.87 (±14.17) at week 24. The same pattern was observed with BUP/NX with a mean depressive symptom (±SD) of 20.08 (±13.92) at week 12 and 15.17 (±14.99) at week 24.
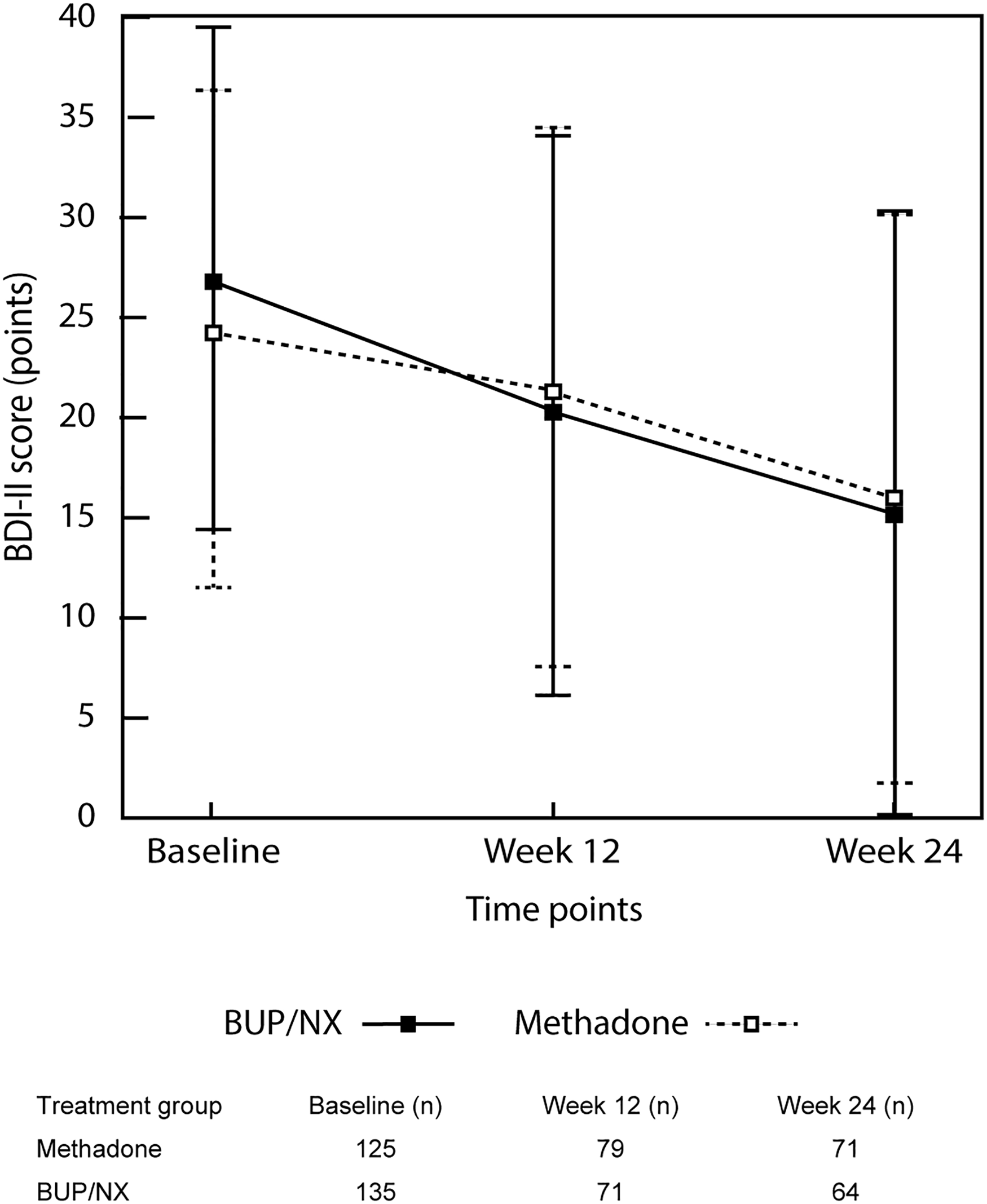
Mean (±SD) depressive symptoms scores across study visits for methadone and buprenorphine/naloxone treatment groups. Baseline depressive scores were similar between treatment groups. Depressive symptoms showed a constant decrease throughout the study period. BDI-II, Beck Depression Inventory – Second Edition.
Main Results
Table 2 reports the results of the adjusted rmGLM models. Participants receiving methadone had less severe overall depressive symptoms compared to BUP/NX (aβ ± standard error [SE] = 3.298 ± 1.604;
Results from the Repeated Measured Generalised Linear Model for Depressive Symptoms.
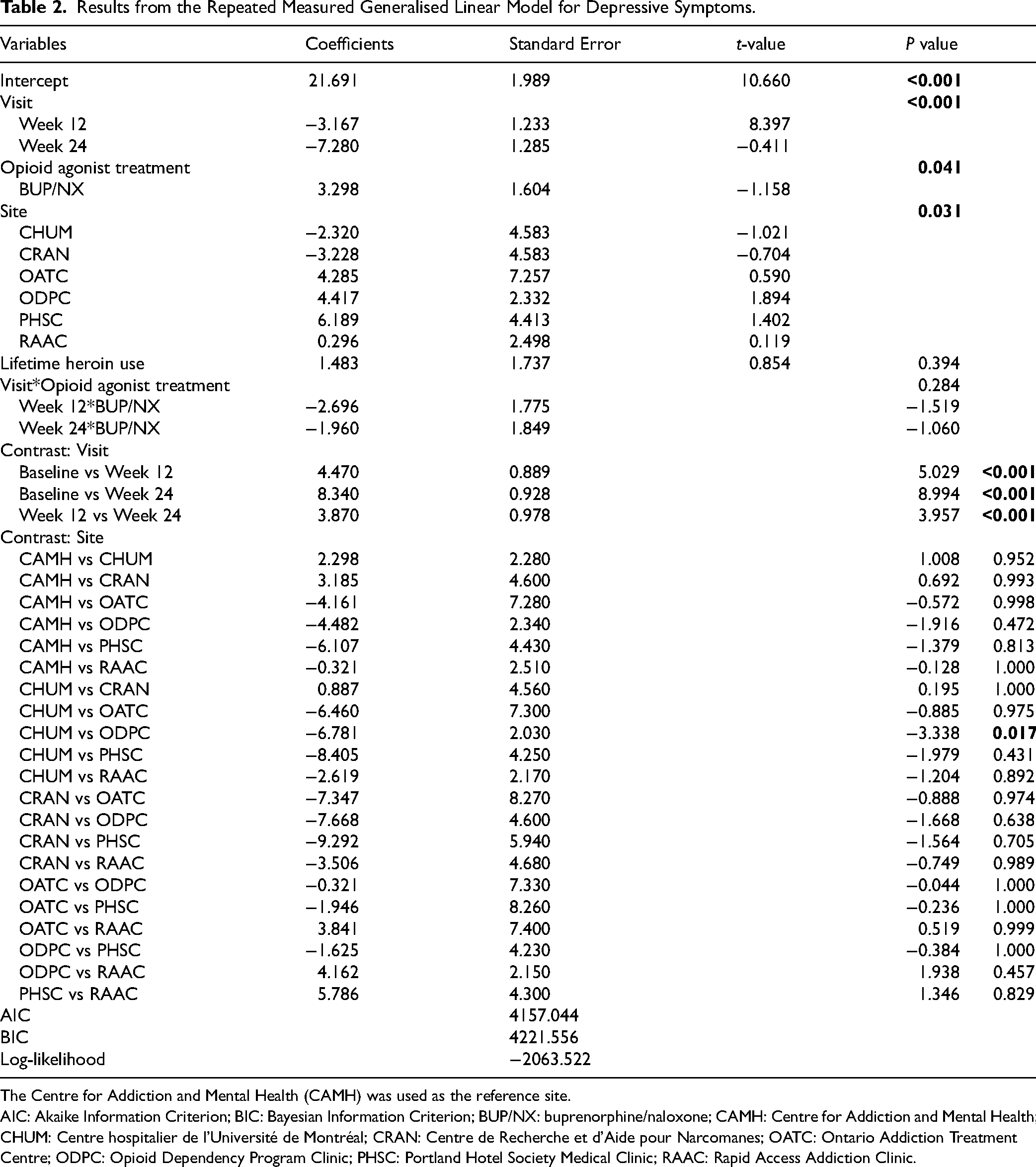
The Centre for Addiction and Mental Health (CAMH) was used as the reference site.
AIC: Akaike Information Criterion; BIC: Bayesian Information Criterion; BUP/NX: buprenorphine/naloxone; CAMH: Centre for Addiction and Mental Health; CHUM: Centre hospitalier de l'Université de Montréal; CRAN: Centre de Recherche et d'Aide pour Narcomanes; OATC: Ontario Addiction Treatment Centre; ODPC: Opioid Dependency Program Clinic; PHSC: Portland Hotel Society Medical Clinic; RAAC: Rapid Access Addiction Clinic.
Mediation Analyses
Table S1 reports the models estimated for the mediation analyses. Table S2 and Figure 3 illustrate the mediation analyses using imputed data. Being on OAT at week 12 or week 24 significantly reduced days of opioid use (
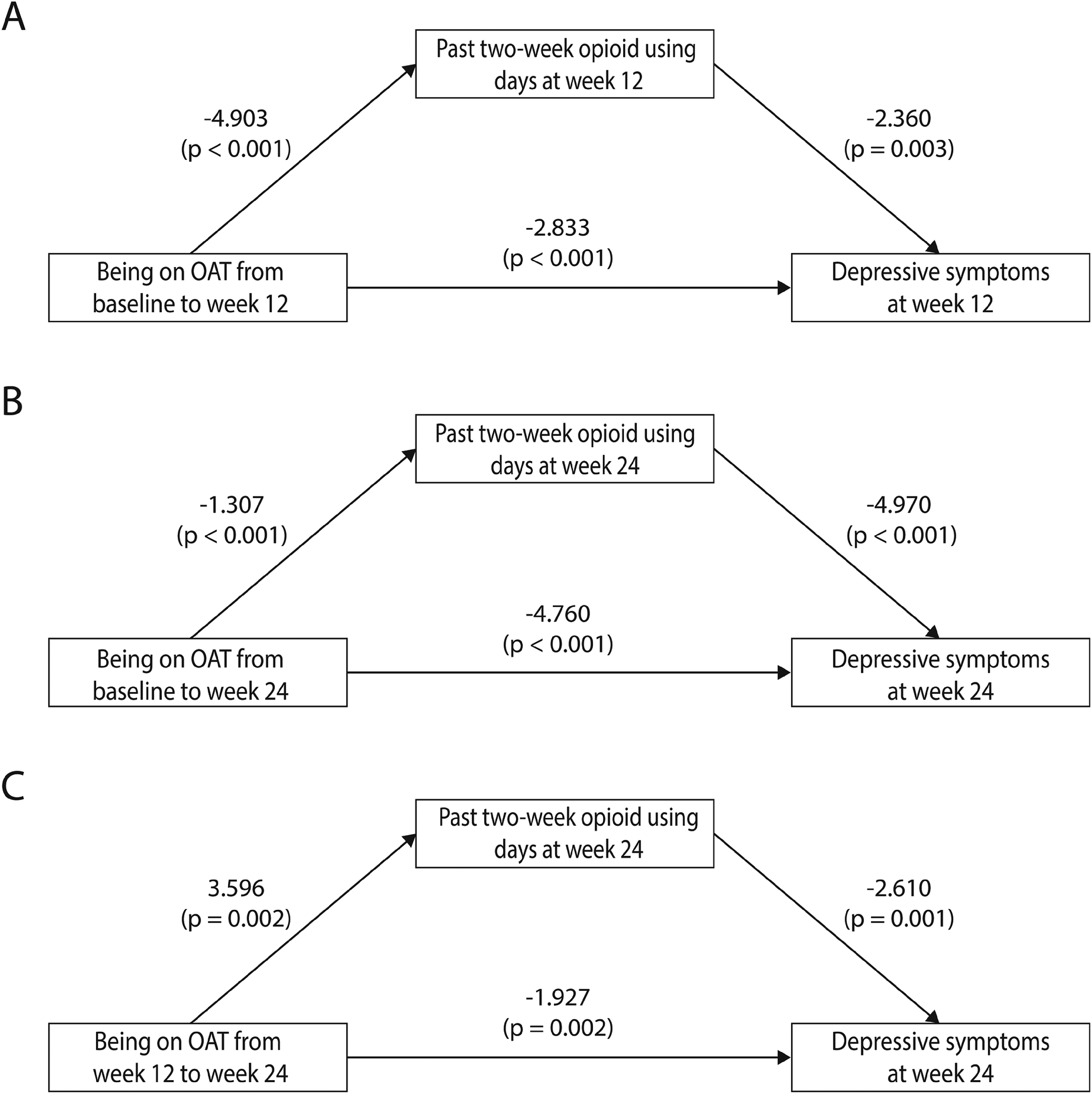
Results from the mediation analyses using imputed data. (A) Being on opioid agonist treatment from baseline to week 12 significantly reduced days of opioid use (
From baseline to week 12, the direct (
Subgroup Mediation Analyses
Figure S1 and Table S2 report the subgroup mediation analyses. For methadone, the direct and total effects were significant from baseline to week 12 (
Sensitivity Analyses
Per-protocol and COVID-19 sensitivity analyses confirmed our results (see Table S3). However, as opposed to our main analysis, BUP/NX did not report overall higher depressive symptoms (marginal mean ± SE for methadone = 21.1 ± 2.1; marginal mean ± SE for BUP/NX = 18.0 ± SE 2.2; mean difference ± SE = 2.047 ± 2.995;
Discussion
To our knowledge, this is the first study to compare the impact of methadone and BUP/NX in decreasing depressive symptoms in people with POUD. Both treatments improved depressive symptoms at a similar rate. This effect was partially mediated by days of opioid use, especially for methadone and during the last 12 weeks of treatment.
Many factors could explain these results. For one, chronic opioid use is known to induce depressive symptoms.32,34 Therefore, by reducing opioid use in the first 12 weeks of treatment, OATs decreased opioid-induced depressive symptoms in the subsequent weeks. The quantity of opioids used during each day might also be lower. However, reductions in opioid use only account for a small proportion of improvements, therefore, suggesting the involvement of other pathways leading to an overall antidepressant effect.
First, BUP/NX can help with depressive symptomatology in people with treatment-resistant depression, possibly through the opioid, endocrinological, and serotoninergic systems.10,19,24 Although long-term methadone treatment can lead to induced dysphoria through hormonal dysregulations,35,36 it has similarly been reported to decrease depressive symptoms, alone or in conjunction with antidepressants.20,22,24 Many studies have reported that both BUP/NX and methadone can help with depressive symptoms independently of opioid use.10,17,20 Therefore, the current evidence underlies that OATs potentially have intrinsic antidepressant properties.
However, beyond the reduction of opioid use and OAT's pharmacological actions, many other OAT-related factors might have lowered depressive symptoms. For example, a previous report of the current study showed that both methadone and BUP/NX increased overall quality of life, 29 which may indicate reduced hopelessness, better hygiene/self-care, and increased involvement in activities. Another report showed that both treatment arms decreased craving frequency, length, and intensity at a similar rate. 37 Both methadone and BUP/NX can also lower withdrawal symptoms, which are associated with increased anxiety and dysphoric moods.38,39 Therefore, by reducing cravings and withdrawal symptoms, both OATs could have reduced anxiety and depressive states, leading to improvements in overall well-being. Also, access to mental health interventions might have been facilitated by the fact that participants were followed at OAT clinics. These interventions could have directly or indirectly reduced primary depressive symptoms. Due to the pragmatic design, access to certain types of mental health interventions (including antidepressants and psychotherapy) may have varied based on geographical location, which could explain differences across study sites. Finally, in the absence of a placebo arm, we cannot exclude a possible Hawthorne effect (i.e., changes in behaviours that could improve physical, psychological, social, and environmental functioning) leading to reduced depressive symptoms. 40 However, many studies looking at methadone or BUP/NX found that these OATs lead to reductions in depressive symptoms comparable to antidepressants and psychotherapy.10,20,22,24,41 Therefore, even if a placebo effect is involved in the improvements seen in depressive symptoms, OATs seem to have an efficacy similar to traditional depression treatments.
Although we found no interaction of treatment and time, depressive symptoms in BUP/NX were on average 3 points higher than methadone. Sensitivity analyses with retained participants did not lead to the same overall difference. This discrepancy seemed to be explained by differences in characteristics between retained and nonretained participants, and BUP/NX participants being less likely to be retained in treatment. Overall opioid use was higher in participants who were not considered as retained. Nonretained participants had also a higher prevalence of some risk factors, such as housing instability, 42 female sex, 43 lower income,42,44 having no drug coverage, 45 and physical/psychiatric comorbidities,46,47 notably more severe depressive symptoms. Previous studies have shown that BUP/NX might lead to lower retention in people with more severe POUD.48,50 As a partial agonist of opioid receptors, BUP/NX leads to a submaximal activation, even at maximum occupancy of µ-opioid receptors. 51 Methadone allows an increased activation of the opioid receptors at higher doses. 52 Consequently, its total agonist effect may make it more suitable for some people who use heavily,53,54 or, such as it is the case in our sample, use highly potent opioids. We might expect that more people with severe POUD switched to another OAT or discontinued treatment and were, therefore, excluded from the retained analyses. Altogether, this may indicate that while participants retained on BUP/NX may report improved depressive symptoms, this OAT is more likely to lead to a treatment switch. 29 Any specific positive effect of BUP/NX may need to be balanced with this lower likelihood of retention, even more so in people with other risk factors for treatment non-adherence.
Since methadone and BUP/NX showed similar trends and pathways in decreasing depressive symptoms, clinicians are encouraged to tailor the selection of OAT to the full array of patients’ needs and characteristics. 54 Assessing patient preferences regarding OAT is also highly relevant in the treatment of POUD and comorbid depression. A recent study on patient preferences for OATs identified that people with OUD attributed many advantages to BUP/NX over methadone, such as the easier switch from BUP/NX to methadone, increased flexibility with take-home doses, less sedation effects, and increased perceived efficacy. 55 Tailoring treatment to patient preferences and needs, including but not only based on the presence of depressive symptoms, might lead to increased treatment adherence and better outcomes. 56
Limitations
This study is not without limitations. Firstly, we had a 44% attrition rate that could have impacted our results. Indeed, most participants not considered as retained showed more psychosocial risk factors. Therefore, we might expect that participants that were more likely to have a beneficial impact on the interventions were retained in treatment. Future studies should explore if our results can be extrapolated to people with more severe psychosocial functioning. Secondly, without a placebo arm, it is impossible to make conclusions about the independent effectiveness of OATs in decreasing depressive symptoms. Our data have shown, however, that having access to methadone or BUP/NX can improve depressive symptoms through a variety of possible mechanisms. Thirdly, having a pragmatic nonblinded design might have biased our data and included more confounders. For example, access to and delivery of other interventions were not standardised across study sites. Fourthly, this randomised controlled trial was not primarily designed to investigate the impact of OATs on depressive symptoms. This secondary analysis is, however, still informative of the possible mechanisms involved in the association between OATs and depressive symptoms. Lastly, we only measured depressive symptoms at week 12 and week 24. Since we did not assess a diagnosis of depression, readers should keep in mind that this study focuses on nonspecified depressive symptoms. Also, we might have found different patterns during the first few weeks of OAT or after 24 weeks. Further trials should be conducted to evaluate whether OATs differ in the very short or long term.
Conclusion Statements
Both the methadone standard model of care and flexible take-home dosing of BUP/NX similarly decreased depressive symptoms in people with POUD. Improvements in depressive symptoms were partially explained by a reduction in days of opioid use. As both treatments are equally effective, clinicians are encouraged to discuss both benefits and disadvantages of OATs and make a shared decision with the patient on which treatment to choose. Future research should investigate how patient preferences and characteristics, such as specific patterns of opioid use or other mental health markers, are likely to influence OAT outcomes.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437221145013 - Supplemental material for Effects of Buprenorphine/Naloxone and Methadone on Depressive Symptoms in People with Prescription Opioid Use Disorder: A Pragmatic Randomised Controlled Trial
Supplemental material, sj-docx-1-cpa-10.1177_07067437221145013 for Effects of Buprenorphine/Naloxone and Methadone on Depressive Symptoms in People with Prescription Opioid Use Disorder: A Pragmatic Randomised Controlled Trial by Gabriel Bastien, Christina McAnulty, Omar Ledjiar, M. Eugenia Socias, Bernard Le Foll, Ron Lim, Ahmed N. Hassan, Suzanne Brissette, Stéphanie Marsan, Annie Talbot, Didier Jutras-Aswad and in The Canadian Journal of Psychiatry
Footnotes
Acknowledgments
The authors would like to acknowledge the work of Denise Adams, Oluwadamilola Akinyemi, Benoit Masse, Jill Fikowski, Aïssata Sako, Katrina Blommaert, Emma Garrod, José Trigo, Amel Zertal, Nirupa Goel, Farihah Ali, Wendy Mauro-Allard, Kristen Morin, Benita Okacha, Eve Poirier, Geneviève St-Onge, and Angela Wallace for assisting in the conduction and administration of the trial. Our team expresses gratitude to Kim Brochu for her clinical input. The authors also thank Léa Gagnon and Helen Kang for reviewing and editing the manuscript. Finally, our team would like to thank all participants and clinical staff who took part in this research.
Data Access
Authors’ Note
GB received scholarships from the Institut universitaire sur les dépendances, the Quebec Network on Suicide, Mood Disorders and Related Disorders, and the Centre hospitalier de l’Université de Montréal Research Centre. CM received funding from the Institut universitaire sur les dépendances, the Gaulin Foundation and the Centre hospitalier de l’Université de Montréal Research Centre. MES is supported by a Michael Smith Foundation for Health Research and St. Paul's Foundation Scholar Award. BLF is supported by a clinician scientist award from the Department of Family and Community Medicine and by the Addiction Psychiatry Chair of the Department of Psychiatry, University of Toronto. BLF receives or has received support from Pfizer Global Research Awards in Nicotine Dependence (GRAND) Program, Brainsway, Bioprojet, Alkermes, Canopy, ACS, and non-financial support from Aurora for work outside this study. KA was supported by an Embedded Clinician Researcher Salary Award from the Canadian Institutes of Health Research. DJA holds a research scholar award from the Fonds de Recherche du Québec en Santé. OPTIMA Research Group:
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funds received from Health Canada and the Canadian Institutes of Health Research (CIHR) through the Canadian Research Initiative in Substance Misuse (CRISM; grant numbers CIS-144301, CIS-144302, CIS-144303, CIS-144304). The four nodes of CRISM received independent funding through a CIHR priority-driven initiative (grant numbers SMN-139148, SMN-139149, SMN-139150, SMN-139151).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
