Abstract
Objectives
With increasing evidence for the clinical utility of pharmacogenomic (PGx) testing for depression, there is a growing need to consider issues related to the clinical implementation of this testing. The perspectives of key stakeholders (both people with lived experience [PWLE] and providers) are critical, but not frequently explored. The purpose of this study was to understand how PWLE and healthcare providers/policy experts (P/HCPs) perceive PGx testing for depression, to inform the consideration of clinical implementation within the healthcare system in British Columbia (BC), Canada.
Methods
We recruited two cohorts of participants to complete individual 1-h, semi-structured interviews: (a) PWLE, recruited from patient and research engagement networks and organizations and (b) P/HCPs, recruited via targeted invitation. Interviews were audiotaped, transcribed verbatim, de-identified, and analysed using interpretive description.
Results
Seventeen interviews were completed with PWLE (7 with experience of PGx testing for depression; 10 without); 15 interviews were completed with P/HCPs (family physicians, psychiatrists, nurses, pharmacists, genetic counsellors, medical geneticists, lab technologists, program directors, and insurers). Visual models of PWLE's and P/HCP's perceptions of and attitudes towards PGx testing were developed separately, but both were heavily influenced by participants’ prior professional and/or personal experiences with depression and/or PGx testing. Both groups expressed a need for evidence and numerous considerations for the implementation of PGx testing in BC, including the requirement for conclusive economic analyses, patient and provider education, technological and clinical support, local testing facilities, and measures to ensure equitable access to testing.
Conclusions
While hopeful about the potential for therapeutic benefit from PGx testing, PWLE and P/HCPs see the need for robust evidence of utility, and BC-wide infrastructure and policies to ensure equitable and effective access to PGx testing. Further research into the accessibility, effectiveness, and cost-effectiveness of various implementation strategies is needed to inform PGx testing use in BC.
Introduction
For people with depression and their healthcare providers, the process of finding a medication that works in relieving symptoms, while minimizing adverse drug reactions, can be a difficult process of trial-and-error.1,2 This may contribute to medication nonconcordance and, subsequently, to poorer health and long-term prognosis 3 and increased costs to the healthcare system.3,4 Pharmacogenomic (PGx) testing is therefore of great relevance in psychiatry, as it aims to use genetic information (about variations that influence the pharmacokinetics and pharmacodynamics of a medication) to aid in finding an antidepressant that works best for each individual patient. PGx tests typically assess multiple genomic variants across multiple genes (“combinatorial PGx testing”) in order to predict gene–drug interactions that can be used to adjust medication choice or dosage.
People with a history of depression have expressed interest in PGx testing, 5 and in Canada, patients who wish/are able to self-pay for PGx tests can access them independently, 6 or with the help of an ordering clinician. There are, however, barriers to the clinical implementation of PGx testing,7,8 and it is not yet clinically routine 9 or – to our knowledge – funded in the vast majority of publicly funded healthcare systems, largely due to the current lack of robust evidence for cost-effectiveness. 10 Evidence for the clinical utility of PGx testing for depression is accumulating,2,7,11–14 including preliminary evidence for improved response and remission rates with PGx-guided antidepressant treatment compared to treatment-as-usual.11–13 And, as evidence for the clinical utility and cost-effectiveness of PGx testing accumulates, it is critical that stakeholders’ interests and concerns – including those of clinicians, administrators, and patients – are also explored and considered in the adoption of PGx testing 15 to help ensure its acceptability and effectiveness within the healthcare system.
Perspectives of people with lived experience (PWLE) and healthcare providers may differ in important ways, and so both are important to explore. With regard to providers’ perspectives, studies to date on PGx testing in psychiatry have largely been based on quantitative survey-based studies and are not specific to depression.16–19 We are aware of only two qualitative studies about PGx for depression specifically.20,21 These data indicate general optimism for the future use of PGx testing and highlight a need for educational and logistical support for prescribing providers, but broader, more conceptual issues related to the implementation of PGx testing in a psychiatric context were lacking.
With regard to the perspectives of PWLE on PGx testing for depression, there have been several qualitative studies exploring why people got testing and their reactions to results, 22 and attitudes towards PGx testing for depression23–25 (among other indications24,25). These studies describe both hopes and concerns for PGx testing. Again, there is little published research concerning perspectives of PWLE on broader, more conceptual issues related to the implementation of PGx testing in a psychiatric context.
Furthermore, no qualitative studies of either PWLE or provider views about PGx testing have been conducted in a Canadian setting. Thus, the particularities of providing PGx testing within the context of the Canadian public healthcare system have not been explored. In order to ensure the effective, equitable, and meaningful provision of PGx testing, we sought to address these gaps. We specifically aimed to explore the perceptions of PGx testing among PWLE and healthcare providers/other professional stakeholders, to inform the process of considering the clinical implementation of PGx testing for depression within the healthcare system in British Columbia (BC), Canada.
Methods
Procedures
As a part of a large, multi-faceted study evaluating the potential effectiveness and cost-effectiveness of introducing PGx testing for depression in BC as a routine part of clinical practice, we conducted a qualitative (interpretive description) semi-structured interview-based study with two groups of stakeholders: (a) PWLE and (b) professional stakeholders (P/HCPs; healthcare providers, policy makers, health insurers, laboratory experts). The Research Ethics Board at the University of British Columbia approved this study (H20-01648).
Data Collection
A semi-structured interview guide was developed for each group with the input of authors CS, EM, LE, AMH, GL, LR, LR, and JA based on their experience with genetics/genetic counselling, PGx research, qualitative research, and/or lived experience with depression (see supplemental information).
Participants for the PWLE group were recruited using a combination of convenience and purposive approaches. Specifically, study advertisements were shared via social media and amplified by mental health networks in BC. A study page was also created on REACH BC (www.reachbc.ca), a provincial initiative connecting BC residents to health researchers. As data collection progressed, in order to target recruitment of people representing groups whom we had not yet captured, we (a) connected with a local men's mental health group who shared our study advertisement with their members/social media followers and (b) connected with the authors of the IMPACT study, 26 who shared our study information with their research participants who had formerly received PGx testing for depression. Interested individuals contacted the study team, were screened for eligibility (i.e., were a resident of BC, fluent in English, and had a self-reported history of depression and antidepressant use), and provided written informed consent prior to the study interview.
For interviews with P/HCPs, targeted recruitment was used to ensure a range of perspectives and professional backgrounds were captured (family physicians, psychiatrists, nurses, pharmacists, genetic counsellors, medical geneticists, lab technologists, private insurers, BC Ministry of Health personnel, and program directors within BC health authorities). Relevant positions and/or specific individuals were identified by the study team and potential participants were contacted individually via email or telephone and invited to participate. Participants provided written informed consent prior to the study interview.
Participants completed one-on-one semi-structured interviews from their workplaces or homes via BlueJeans videoconference software, with a research genetic counsellor; no other observers were present. PWLE were interviewed by CS; P/HCPs were interviewed by EM (both MSc, female; trained in qualitative and quantitative methods with 5 + years of professional research experience). CS had no prior relationships with PWLE participants; EM was a colleague of one P/HCP participant and had interacted in a professional capacity with two others (clinicians and program directors) through her experience working as a genetic counsellor in BC. Participants were informed of the purpose of the research and of the interviewers’ roles as research genetic counsellors. Interviews comprised open-ended questions exploring participants’ perceptions of, experiences with, and opinions and attitudes around PGx testing for depression and its potential use in BC. As data collection and analysis progressed, interview guides were refined (with the input of CS, EM, LE, AMH, GL, LR, LR, and JA) to ensure emerging areas were captured in subsequent interviews. Interviews were audio-recorded, transcribed verbatim, and checked for accuracy before being analysed. NVivo 12 was used to store, organize, and manage data. 27
Analysis
Interview data were analysed concurrently with data collection using interpretive description,28,29 an inductive qualitative approach that aims to understand the range of subjective human experiences to develop clinical understanding in an applied healthcare setting. The two groups of interviews were largely analysed separately by CS and EM (as described below), but salient themes and similarities/contrasts between the two groups were discussed by CS, EM, and JA on a biweekly basis. The frame of reference for this study was the Sustainability of Innovation model, which considers five domains that influence health innovation sustainability (political, organizational, financial, workforce, and innovation-specific factors). 30 This framework was used to “orient [our] inquiry” 28 by informing the development of our interview guide (i.e., raising factors to explore with participants regarding the implementation of PGx), but was not used as a strict analytical structure (as is typical in interpretive description 28 ).
For each group of interviews, analysis began with inductive line by line coding of basic conceptual units and to delineate the properties that characterize them. 29 CS and EM each independently developed and applied a coding framework for each group of interviews. These were iteratively revised based on findings from new transcripts and applied to earlier interviews when relevant. Axial coding was then used to identify the main concepts from the coding framework, the conditions that give rise to them and the relationships between them. These concepts were used to inductively develop visual models of how PWLE and P/HCPs think about PGx testing and its possible use in BC – CS, EM, and JA met twice a month for 6 months to modify and verify the main concepts and to discuss the theoretical linkages between concepts. The authors – including patient partners – met on several occasions to review linkages and discuss the connections and relationships between relevant concepts for each group of interviews until the major concepts formed a cohesive theoretical model. Throughout the analytical process, written memos were used to capture decisions regarding the data and to record salient themes. Transcripts, codes and memos were iteratively reviewed to discuss and resolve discrepancies. Rather than aiming for “saturation” as a recruitment end point (which has been criticized as a concept in qualitative research for a variety of reasons), 31 we employed the concept of theoretical sufficiency, which asks whether the model constructed is adequate in terms of the use for which it was envisioned. 32 Member checking was conducted with two participants from each cohort. Small adjustments were made to the PWLE model; no changes were made to the P/HCP model after member checking.
Results
Visual models representing how PWLE and P/HCPs perceive PGx testing for depression were developed; see Figure 1 and descriptions below.
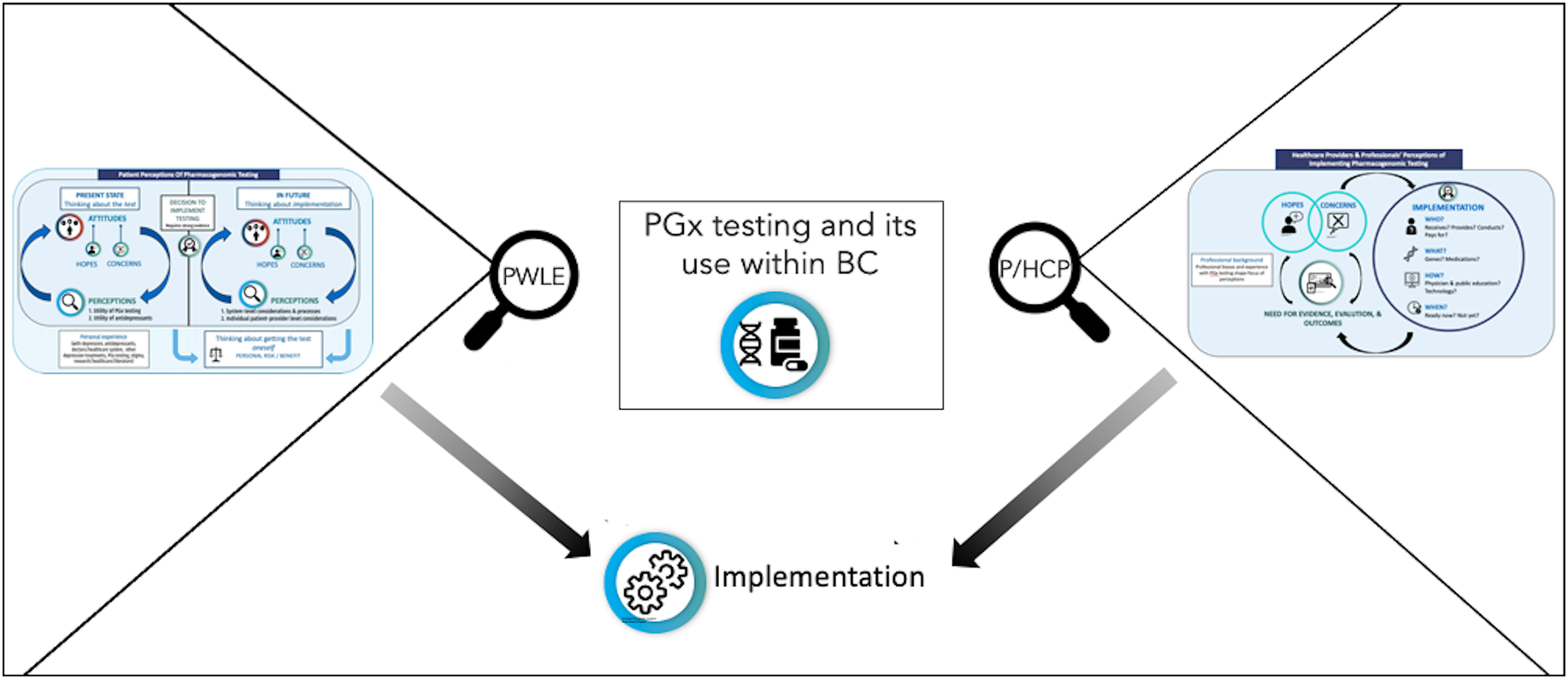
Depiction of study purpose: to describe how the attitudes and perspectives of PWLE and P/HCPs shape the move towards the clinical implementation of PGx to maximize benefits/mitigate harms within the realities of a publicly funded healthcare system.
PWLE
Participants
Seventeen interviews were conducted (Feb–Jun 2021) – with 7 individuals who had previous PGx testing for antidepressant response and 10 without PGx testing experience (see Table 1). Participants were between 21 and 77 years of age, and most identified as women (N = 11) and as White (N = 12). Four of the five regional health authorities in BC were represented across participants. Interviews averaged 62 min in length (range: 38–103 min).
Demographic Information for PWLE and P/HCP Participants.
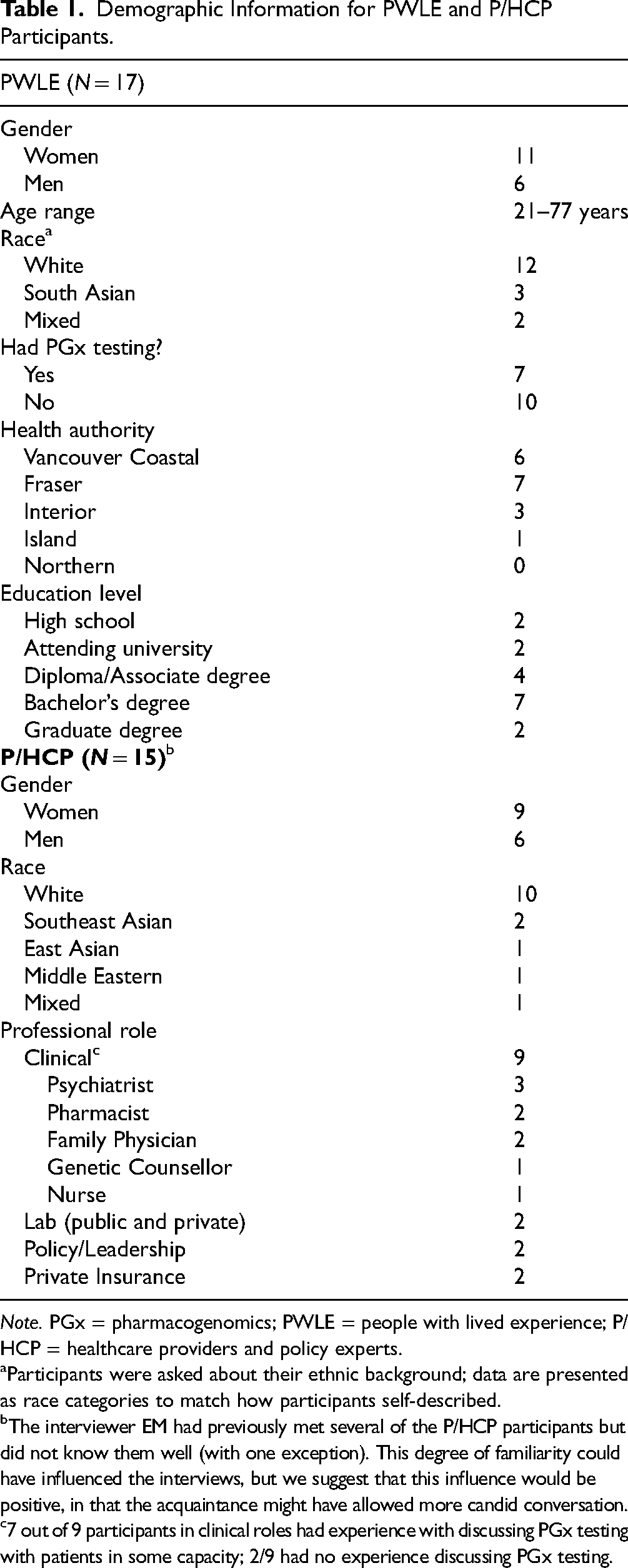
Note. PGx = pharmacogenomics; PWLE = people with lived experience; P/HCP = healthcare providers and policy experts.
Participants were asked about their ethnic background; data are presented as race categories to match how participants self-described.
The interviewer EM had previously met several of the P/HCP participants but did not know them well (with one exception). This degree of familiarity could have influenced the interviews, but we suggest that this influence would be positive, in that the acquaintance might have allowed more candid conversation.
7 out of 9 participants in clinical roles had experience with discussing PGx testing with patients in some capacity; 2/9 had no experience discussing PGx testing.
PWLE model
Participants discussed the present state of PGx testing, its potential use in BC, and their personal process of evaluating PGx testing for themselves (see Figure 2(a)). Illustrative quotes are included within Table 2. Though PWLE's perspectives were influenced by their past experiences, the themes presented were expressed by both those with and without PGx testing experience. Hopes, concerns, and perspectives that were expressed by people without testing experience were also expressed (based on experience, or theoretically) by people with testing experience. Participants in each group had a range of attitudes and perspectives regarding the use of this testing.
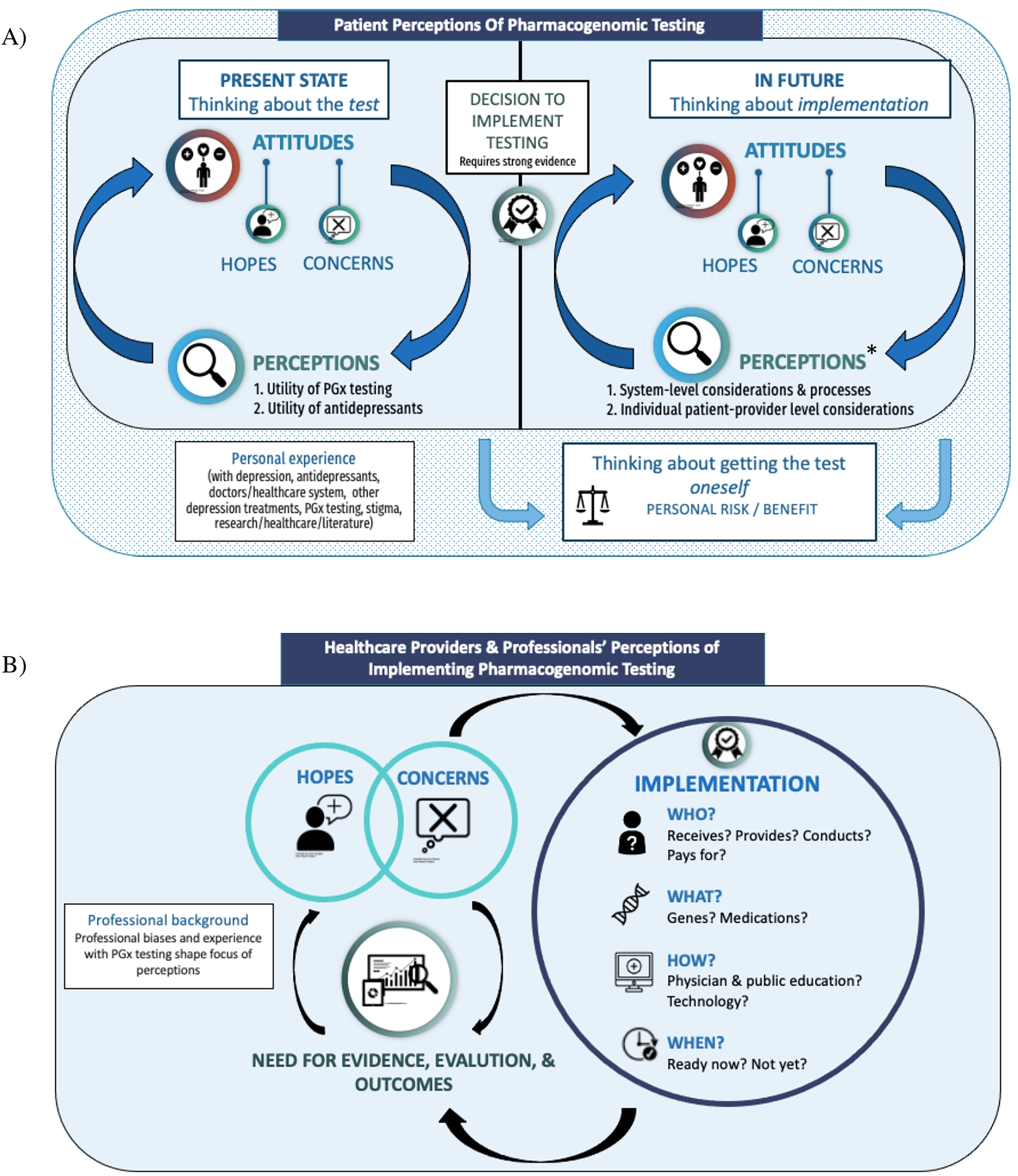
Visual models of participants’ perceptions of PGx testing for depression and its implementation in the BC healthcare system. (a) PWLE model. Participants expressed numerous hopes and concerns related to PGx testing for depression, with a distinction between participants’ thoughts about the present state of PGx testing (and whether or not the test itself was useful), and thoughts about the possible future implementation of PGx testing in BC – at which point significantly more hopes, concerns, and needs for effective implementation arose. In each stage (present and future), there was a cyclical relationship between participants’ perceptions (of the utility of PGx testing and/or antidepressants [present] or of the considerations needed for effective implementation [future]) and their attitudes towards testing. Participants also discussed considering having PGx testing themselves, which was distinct from their discussion about PGx testing in general and was not necessarily tied to whether or not PGx testing was implemented in BC. All aspects of the model were heavily influenced by participants’ past experiences with depression, antidepressants and other depression treatments, the healthcare system, PGx testing, and/or experience with research/academic literature or provision of mental healthcare themselves. See Table 2. (b) P/HCP model. Participants described a number of hopes and concerns for PGx testing, which drove the need for robust evidence of clinical utility of PGx testing and further evaluation and outcomes studies. P/HCP participants did not view implementation as a certain point in time (or an endpoint); rather, the need for evaluation was perceived to be ongoing throughout and beyond the implementation process to monitor its effectiveness. The way that PGx testing is implemented – for whom, by whom, which genes/medications, when and how – and the outcomes that result from that implementation would continue to influence P/HCPs’ hopes and concerns around its use. Participant discussions were strongly influenced by their unique perspectives/professional biases; though most participants discussed each aspect of the model, some components were viewed as more important than others depending on participant background (e.g., psychiatrists focused heavily on the clinical criteria that would warrant PGx testing; insurers and policy makers focused heavily on the need for evidence and cost–benefit analyses). See Table 3.
Elements, Descriptions, and Illustrative Quotes of the Model of How PWLE Perceive PGx Testing and its Implementation Within the BC Healthcare System.

Note. MH = mental health; MoH = Ministry of Health; PGx = pharmacogenomic; PWLE = people with lived experience.
Genetic discrimination was not a large source of concern for the majority of participants, with some expressing that they were already discriminated against because of their depression diagnosis. Concern about genetic discrimination was sometimes based on a perception that PGx testing might reveal risk factors for other disorders (e.g., “one of the breast cancer genes”).
Participants discussed existing challenges accessing appropriate mental health care in BC and how those challenges may present during, or be exacerbated by, the pursuit of PGx testing. While some participants described feeling sufficiently supported to access and make decisions about PGx testing, they acknowledged that many other BC residents did not have those same resources; other participants discussed their own personal challenges accessing care and how they imagined that might impact PGx testing access or the application of PGx test results.
Concerns about genetic discrimination and/or secondary uses of genetic information would be somewhat mitigated if data were to remain in BC/Canada.
The majority of participants – even if they were willing or able to pay for PGx testing out-of-pocket – wanted the BC Medical Services Plan (MSP) to cover the cost of testing for eligible patients in order to ensure equal access to testing and appropriate follow-up care.
Participants varied in the type and source of desired support for PGx testing: many participants wanted to access testing through their GP, either due to ease of access or because of existing trust and familiarity. Others acknowledged that accessing testing through a GP would be difficult because “there are so many people in BC that don’t have GPs,” or thought that PGx testing required more psychopharmacological expertise and should be facilitated by a psychiatrist. Still others wanted every member of their care team (e.g., clinical counsellors or other medical specialists) kept informed of their PGx test results; or wanted minimal support from healthcare providers, with an option to consult one's GP or an “expert” (like a pharmacist, genetic counsellor) if needed.
Elements, Descriptions, and Illustrative Quotes of the Model of How P/HCPs Perceive PGx Testing and its Implementation Within the BC Healthcare System.

Note. PGx = pharmacogenomic; P/HCP = healthcare providers and policy experts; TAT = turnaround time.
Factors that were perceived as both a hope and concern by different participants, or were countered by other participants’ statements. For example, while there was a general hope that PGx testing would result in reduced time to medication response/therapeutic benefit, participants also acknowledged that the turnaround time for PGx testing might actually delay treatment.
Several participants expressed a belief that PGx testing would turn out to be cost effective in terms of reducing the strain on the healthcare system, despite the current lack of evidence. One participant also discussed the societal burden of depression far outweighing the cost of any medication, and the need to include societal costs in future cost–benefit analyses.
A number of participants felt that ideally, PGx testing should be offered to any patient beginning their first antidepressant trial to potentially avoid multiple failed medication trials. From a laboratory perspective, this could also help reduce the cost of testing per patient with batching large numbers of samples for genetic analysis. Others felt strongly that PGx testing should be limited to certain subsets of patients – for example, people with treatment-resistant depression – because clinical utility or cost effectiveness may be limited to certain groups, or because population-wide testing is not feasible.
There was no consensus on which HCP would be the best provider of PGx testing. Tension arose between the need for an established relationship with the patient and knowledge of the full clinical picture in order to make “holistic” treatment decisions (such as the relationship between a patient and a GP or psychiatrist), versus having sufficient time, knowledge or accessibility to have fulsome discussions with patients. Several participants suggested that other practitioners would offer specific advantages: a genetic counsellor (genetic expertise, time), a pharmacist (pharmacological expertise, time, accessibility), or a community nurse or nurse practitioner (existing relationship with patients, time, and accessibility); however, each of these clinicians were perceived to have areas of weakness as well. Other suggestions included a small-scale introduction of PGx testing which could then be expanded with time; integration of genetic counsellors into primary care/psychiatry; or having a specialist multidisciplinary PGx clinic.
Patient access was a major concern for several participants, who discussed the need to have anti-racist structures in place to provide equitable care for Indigenous people, Black people, and people of colour – such as ensuring equal access for patients in rural/Northern BC, or cost coverage in order to avoid a “two-tiered” healthcare system.
Some participants thought testing should not proceed unless there was sufficient evidence of utility; others were confident that this was a question of when (not if) sufficient evidence would be amassed and implementation could proceed – though this notion seemed to be driven by a general sense of optimism rather than any concrete evidence. Finally, some participants felt that since PGx testing was already occurring with or without the BC healthcare system's involvement, there was an imperative to provide the testing in a safe and regulated manner within BC.
Present: thinking about the test
Participants described a cyclical relationship between their hopes and concerns about PGx testing, and their perceptions of utility of both PGx testing and antidepressants. Perceived lack of utility of PGx testing, and/or the perception that antidepressants should not be routinely used seemed to reduce hopes or increase concerns about the testing. Perceived utility of PGx testing and/or antidepressants bolstered hopes and reduced – but often did not eliminate – concerns. Many participants had mixed views.
Decision to implement testing in BC
Participants expressed the need for an authority – such as the BC Ministry of Health, doctors, and/or scientists – to evaluate the literature and decide that there was enough evidence to support the implementation of PGx testing. Despite a general trust and reliance on professionals to make this decision, participants wanted the research evidence to meet standards of rigor and to be available to the public, to potentially aid in personal decision-making.
Thinking about future implementation
As compared to thinking about the test itself, thinking about implementation evoked a greater number of hopes and concerns for participants (see Table 2), which were described with greater emphasis. There was a general feeling that “the testing itself is one thing, and the use of results is entirely different.” Participants discussed both individual-level benefits and risks, such as a person experiencing therapeutic benefit or emotional distress (e.g., from a disappointing result), as well as broader considerations like benefits to the healthcare system and concerns about unequal access to testing (see Table 2).
Thinking about getting the test oneself
Participants also discussed their own decision-making around PGx testing as a risk/benefit assessment of the relative importance of their concerns, hopes, and the current state of their mental health (Table 2).
P/HCPs
Participants
Fifteen interviews were conducted (Oct 2020–Jul 2021) with professional stakeholders in various clinical (N = 9), laboratory (N = 2), policy (N = 2), or health insurance (N = 2) roles (see Table 1). Most participants were White (N = 10); 9 participants identified as women, 6 as men. Interviews averaged 43 min in length (range: 24–59 min).
P/HCP model
Participants discussed their hopes and concerns for PGx testing, the need for outcomes data and evaluation, and considerations for implementation (see Figure 2(b)). Illustrative quotes are included within Table 3. Details about participants’ professional roles are omitted from quotes to protect participant anonymity.
Hopes and concerns
Participants discussed their hopes and concerns for the use of PGx testing (see Table 3), the most prominent of which were hope for therapeutic benefit, concern about the cost of testing, and whether a favourable cost/benefit ratio could be achieved. Participants’ hopes and concerns were sometimes in conflict with each other. For example, some participants thought PGx testing could help, while others thought it may hinder medication concordance.
Need for evidence, evaluation, and outcomes
The need for evidence and evaluation of PGx testing was raised by most participants and was a recurring point of discussion for several participants. This included the need for evidence to support participants’ hopes and the clinical utility of PGx testing (improved patient outcomes) and validity of concerns, as well as evidence to disentangle conflicting hopes and concerns (e.g., whether PGx testing could help or hinder medication concordance). Of particular importance was the need for a conclusive economic analysis, which participants perceived to be currently lacking in the literature.
Implementation
Participants expressed numerous considerations around Who, What, How, and When PGx testing should be implemented (see Table 3), with a focus on mitigating potential harms. For example, participants expressed a need for pretest counselling due to the perception that patients with depression were a vulnerable population that needed to be protected from false hope or unrealistic expectations of PGx testing. However, the ways in which participants thought harms should be mitigated were varied and influenced heavily by professional background.
Discussion
This is the first study to explore the perspectives of both PWLE and professional stakeholders on PGx testing for depression and its potential implementation in a Canadian context. There was both overlap and differences in perspective between PWLE and P/HCPs, as represented in the models. The visual models describing how PWLE and P/HCPs think about the potential introduction of PGx testing in BC can be used to inform the development of implementation strategies that have the best chance of being acceptable and effective within the realities of a public healthcare system.
A major, recurring concern for PWLE and several P/HCP participants was that (assuming appropriate evidence for effectiveness) PGx testing should be paid for through the publicly funded healthcare system. As in a previous Canadian study about PGx that was not specific to depression, 33 many participants were concerned about the potential for inequitable access to testing. There was worry that if PGx were to be available only to those who could private pay, and/or those in urban centres, this could further contribute to inequitable healthcare access and outcomes. The cost of PGx testing – and patients’ means to pay for it – has been cited as a top barrier to implementing PGx. 14 This was also a concern for patients who had previously undergone PGx testing for depression, several of whom thought the test would only be worth taking if it was less expensive. 22
Consistent with previous reports of patient22,34 and provider20,21 perspectives, both PWLE and P/HCP participants were hopeful about the potential therapeutic benefits of PGx testing: faster recovery/symptom reduction, fewer side effects, and reduction in the current practice of trial-and-error prescribing. Both cohorts were also hopeful that PGx testing could result in cost savings for the healthcare system through reductions in mental health crises and hospitalizations. However, we also heard that there would be potential meaningful value of PGx testing even without treatment changes. For example, PWLE said PGx results could validate past experiences with antidepressants and/or current treatment approach (or already had provided validation for several participants with PGx testing experience), while P/HCPs hoped that PGx testing might increase patients’ comfort with taking a medication or their trust in the prescribing clinician.20,21 Both PWLE and P/HCPs also expressed that PGx testing could help validate depression as a legitimate medical condition and thus provide value on a societal level (in terms of decreasing stigma) regardless of its impact on individual treatment decisions.
The development of clinical practice guidelines or resources for P/HCPs to enable them to utilize PGx testing effectively for their patients was identified by both groups in our study, and in previous work14,33 as crucial. Some clinicians expressed that they/their colleagues have limited knowledge of PGx testing (consistent with previous reports16,17,21) or limited time with which to invest in significant interpretation efforts. Concern among patients about the lack of clinical guidelines is a novel finding. Our PWLE (and some P/HCP) participants were concerned that without guidelines, prescribing clinicians might over-utilize PGx testing – and therefore, antidepressants (a concern that has previously been reported 23 ). Participants wanted clinicians to work with each individual in a “holistic” manner, with consideration of patient preferences or lived expertise in their own care and other avenues for mental health management (e.g., therapy). 35
In previous Canadian research in which patients, providers, and policy-makers discussed PGx testing in primary care (not specific to depression), concerns were expressed about genetic discrimination. 33 In contrast, many PWLE participants expressed that the potential for genetic discrimination could not be worse than the discrimination they had already experienced as a result of having depression. If discrimination was raised, it was sometimes connected to misconceptions about what PGx testing would uncover – for example, risk factors for other conditions – suggesting that patients may be confusing PGx testing with predictive testing for susceptibility to illness.23,25 In both PWLE and P/HCP participants, this misconception was also sometimes perceived in a positive manner, in the hope that PGx testing will “explain” their depression. This highlights the need for provider education and pretest counselling to manage expectations and/or concerns about testing.
A number of specific requirements for the effective implementation of PGx testing were expressed by both PWLE and P/HCPs. Both groups emphasized the need for physician education (to enable appropriate ordering and interpretation of PGx testing), patient education (to manage expectations and ensure informed consent/autonomy), testing facilities and infrastructure within BC or Canada (to ensure data privacy and security), and accessible test reports (for patient and clinician understanding of results). P/HCP participants also discussed the need for technological support and clinical protocols. PWLE discussed the need for emotional support for patients through the testing process (if desired), and the need for appropriate and available psychiatric care – a much larger issue in the BC healthcare system that would inevitably affect patient access to PGx testing and subsequent treatment. While PWLE generally trusted that the decision to implement PGx testing would be informed by strong evidence, P/HCP participants discussed the need for ongoing systemic evaluation and outcomes monitoring to continuously support implementation within BC and mitigate concerns about the use of PGx testing.
Opinions varied on exactly how, or through whom, patients should access PGx testing. While family physicians were discussed as a desirable option due to accessibility and an established patient–provider relationship, a number of participants (both PWLE and P/HCPs) had concerns about family physicians’ time and ability to fully educate and support patients throughout PGx testing. Some thought testing should be provided through psychiatrists, but many others thought this would create unnecessary barriers to access. Still others wanted clinical experts – such as genetic counsellors, pharmacists or psychiatrists – available as needed for consultation. Regardless of which provider PWLE wanted to facilitate testing, this was often based on the existence of a trusting relationship, echoing previous reports in the PGx for depression 24 and general PGx literature. 36
P/HCP participants repeatedly discussed the need for conclusive economic analyses – which has been called for numerous times both in the context of PGx for depression9,20 and PGx testing more generally.33,36 We agree that further investigation into the accessibility, effectiveness, and cost-effectiveness (including societal costs) of different implementation strategies for PGx testing is needed. Indeed, our findings are being used to inform the development of a simulation model of the cost-effectiveness of PGx testing for major depression in BC.
Limitations
Our cohort is not fully representative of the BC population and is limited to people who had computer access and were engaged in social media or research networks. Both cohorts were largely White, and the perspectives of Indigenous people, Black people, and people of colour are absent. Future areas of study should prioritize engagement with Indigenous communities of BC and the co-creation of PGx research priorities, protocols, and knowledge.
Our models of how PWLE and P/HCPs perceive PGx testing for depression were developed from the experiences and perspectives of residents of BC, and are thus specific to our healthcare context. However, they provide useful starting points for the consideration of clinical implementation of PGx testing for depression in public healthcare settings.
Conclusion
PWLE and P/HCPs have numerous hopes, concerns, and opinions on how PGx testing should be implemented in BC. PWLE generally trust that the decision to implement testing will be based on sound evidence, but want the evidence to be robust and made available to them for personal decision-making. PWLE also want the plan for PGx implementation to ensure low-barrier, equitable, and effective access to testing for eligible patients. P/HCPs were also hopeful about the potential for therapeutic benefit from PGx testing, but see the need for ongoing outcomes evaluation and monitoring to ensure that PGx testing is introduced in a balanced and cost-effective manner.
Supplemental Material
sj-pdf-1-cpa-10.1177_07067437221140383 - Supplemental material for Pharmacogenomic Testing for Major Depression: A Qualitative Study of the Perceptions of People with Lived Experience and Professional Stakeholders
Supplemental material, sj-pdf-1-cpa-10.1177_07067437221140383 for Pharmacogenomic Testing for Major Depression: A Qualitative Study of the Perceptions of People with Lived Experience and Professional Stakeholders by Caitlin Slomp, Emily Morris, Louisa Edwards, Alison M. Hoens, Ginny Landry, Linda Riches, Lisa Ridgway, Stirling Bryan and Jehannine Austin in The Canadian Journal of Psychiatry
Supplemental Material
sj-docx-2-cpa-10.1177_07067437221140383 - Supplemental material for Pharmacogenomic Testing for Major Depression: A Qualitative Study of the Perceptions of People with Lived Experience and Professional Stakeholders
Supplemental material, sj-docx-2-cpa-10.1177_07067437221140383 for Pharmacogenomic Testing for Major Depression: A Qualitative Study of the Perceptions of People with Lived Experience and Professional Stakeholders by Caitlin Slomp, Emily Morris, Louisa Edwards, Alison M. Hoens, Ginny Landry, Linda Riches, Lisa Ridgway, Stirling Bryan and Jehannine Austin in The Canadian Journal of Psychiatry
Footnotes
Data Access
Data are available from the corresponding author on reasonable request.
Acknowledgments
The authors thank the study participants and offer gratitude to the Coast Salish Peoples, including the xʷməθkʷəy̓əm (Musqueam), Skwxwú7mesh (Squamish), and Səl̓ílwətaʔ/Selilwitulh (Tsleil-Waututh) Nations, on whose traditional, unceded, and ancestral territory we have the privilege of working.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Genome BC and Genome Canada (project #B26PMH) and Michael Smith Health Research BC (award #18932). JA was supported by BC Mental Health and Substance Use Services.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
