Abstract
Objective
Serotonergic psychedelics are re-emerging as potential novel treatments for several psychiatric disorders including major depressive disorder. The Canadian Network for Mood and Anxiety Treatments (CANMAT) convened a task force to review the evidence and provide a consensus recommendation for the clinical use of psychedelic treatments for major depressive disorder.
Methods
A systematic review was conducted to identify contemporary clinical trials of serotonergic psychedelics for the treatment of major depressive disorder and cancer-related depression. Studies published between January 1990 and July 2021 were identified using combinations of search terms, inspection of bibliographies and review of other psychedelic reviews and consensus statements. The levels of evidence for efficacy were graded according to the Canadian Network for Mood and Anxiety Treatments criteria.
Results
Only psilocybin and ayahuasca have contemporary clinical trials evaluating antidepressant effects. Two pilot studies showed preliminary positive effects of single-dose ayahuasca for treatment-resistant depression (Level 3 evidence). Small randomized controlled trials of psilocybin combined with psychotherapy showed superiority to waitlist controls and comparable efficacy and safety to an active comparator (escitalopram with supportive psychotherapy) in major depressive disorder, with additional randomized controlled trials showing efficacy specifically in cancer-related depression (Level 3 evidence). There was only one open-label trial of psilocybin in treatment-resistant unipolar depression (Level 4 evidence). Small sample sizes and functional unblinding were major limitations in all studies. Adverse events associated with psychedelics, including psychological (e.g., psychotomimetic effects) and physical (e.g., nausea, emesis and headaches) effects, were generally transient.
Conclusions
There is currently only low-level evidence to support the efficacy and safety of psychedelics for major depressive disorder. In Canada, as of 2022, psilocybin remains an experimental option that is only available through clinical trials or the special access program. As such, Canadian Network for Mood and Anxiety Treatments considers psilocybin an experimental treatment and recommends its use primarily within clinical trials, or, less commonly, through the special access program in rare, special circumstances.
Keywords
Introduction
Major depressive disorder (MDD) is a common and often disabling condition affecting more than 300 million people globally.1,2 Currently available treatments are sometimes ineffective with over one-third of patients failing to respond adequately to multiple first-line treatments. 3 Evidence-based MDD treatment recommendations are provided by the Canadian Network for Mood and Anxiety Treatments (CANMAT) and acknowledge the urgent need for novel treatments.4–6 Over the past two decades, there has been a resurgence of interest in psychedelics combined with psychological support for numerous psychiatric disorders, creating a “second wave” of psychedelic research. 7 Promising preliminary results from the recent pilot clinical trials have fuelled interest from clinicians, patients, regulatory bodies and industry partners alike.8,9 Accordingly, the Food and Drug Administration (FDA) has granted breakthrough therapy designation for psilocybin for MDD.10,11
Classic serotonergic psychedelics (to be referred to as “psychedelics” moving forward) have a high affinity for the serotonin 2A receptor (5HT2A) with potent agonist or partial agonist effects inducing an acute altered state of consciousness (e.g., acute cognitive, perceptual, psychological and emotional effects).9,12 While the debate continues regarding the inclusion of other mind-altering substances (e.g. cannabis, ketamine or 3,4-methyl enedioxymethamphetamine (MDMA)) within the psychedelic class, 13 for the purpose of the current review, we have restricted the analysis based on the pharmacological definition, exclusively 5HT2A agonists and partial agonists. This definition includes the following agents (and their psychoactive derivatives or metabolites): psilocybin, lysergic acid diethylamide (LSD), N,N-dimethyltryptamine (DMT; ayahuasca; 5-methoxy-N,N-dimethyltryptamine (5-MeO-DMT)) and mescaline. As such, the evidence for ketamine, cannabis and MDMA are not reviewed herein; however, CANMAT has recently reviewed and provided recommendations for ketamine 14 and cannabis. 15
The objective of the present CANMAT Task Force review is to summarize and synthesize evidence for the use of psychedelics in depression. We follow the previous CANMAT question and answer format, focusing on areas that are of greatest importance to clinicians, researchers and individuals with lived experience. We focus exclusively on antidepressant efficacy in clinical samples, although we recognize research activity in other areas including substance use disorders, anxiety, existential distress, post-traumatic stress disorder (PTSD) and obsessive-compulsive disorder (OCD).
Methods
We followed similar methods as described in previous CANMAT guidelines to identify clinical trials evaluating serotonergic psychedelics.4,14,16,17 As described above, the term “psychedelic” refers to 5HT2A agonists or partial agonists throughout this manuscript, primarily including psilocybin, LSD, DMT, mescaline and their derivatives. Given that DMT is the primary active psychedelic ingredient in ayahuasca, clinical trials evaluating ayahuasca were also included. Relevant prospective clinical trials published in English or French were identified using searches of electronic databases (PubMed, PsychInfo and Cochrane Registry of Clinical Trials) from 1 January 1990 to 31 July 2021, an inspection of bibliographies, and a review of other guidelines and major reports. Combinations of the following search terms were used (including relevant variations in spelling and abbreviations): depression, MDD, mood disorder, psychedelic, 5HT2A, psilocybin, LSD, DMT, ayahuasca, mescaline and 5-MeO-DMT. Given known ethical and scientific concerns with the first wave of psychedelic research in the mid-1900s, only contemporary trials completed after 1990 were included. We also reviewed recent consensus statements and guidelines on psychedelic use in psychiatry.
While human research with psychedelics has been completed in numerous populations outside of mood disorders (e.g., healthy volunteers, substance use disorders, PTSD, anxiety, etc.), the current review focuses on studies evaluating clinical samples with a primary diagnosis of MDD, as well as studies evaluating cancer-related depression. However, we excluded studies that exclusively evaluated effects on cancer-related anxiety or existential distress, where depression was not an inclusion criterion or primary outcomes of interest.
Level of evidence for antidepressant efficacy was evaluated using CANMAT's standard grading system (Table 1). Levels of evidence range from highest at 1 (meta-analysis with narrow confidence intervals or replicated double-blind randomized controlled trials [RCTs] with n ≥ 30 in each arm) to lowest at 4 (pilot RCTs with n < 10 in each arm, uncontrolled trials, anecdotal reports or expert opinion). Level of evidence was also supplemented by expert consensus among the task force assessing tolerability, safety, feasibility and risk of bias.
Canadian Network for Mood and Anxiety Treatments (CANMAT) Evidence Grading Criteria.

Results
The task force developed eight clinically oriented questions with responses presented below. Table 2 summarizes findings and recommendations relating to these eight questions.
Summary of the Canadian Network for Mood and Anxiety Treatments (CANMAT) Psychedelic Task Force Findings.

Note. BDNF = brain-derived neurotrophic factor; CANMAT = Canadian Network for Mood and Anxiety Treatments; LSD = lysergic acid diethylamide; MDD = major depressive disorder; MAOI = monoamine oxidase inhibitor; TRD = treatment-resistant unipolar depression; 5-MeO-DMT = 5-methoxy-N,N-dimethyltryptamine.
What is the Mechanism of Action of Psychedelics?
Serotonergic psychedelics include psilocybin, LSD, DMT and mescaline, all with tryptamine structures similar to serotonin.18–20 5HT2A receptor agonist effects have been demonstrated to induce the hallucinogenic experiences that distinguish psychedelics. 21 There is also evidence that pre-medicating with 5HT2A receptor antagonists, such as ketanserin, prevents the psychoactive effects of psychedelics.21,22 5HT2A receptors are highly expressed in brain regions involved in perception, emotional processing, memory, executive function and self-awareness, including the prefrontal cortex, anterior cingulate and amygdala.21,23 There is evidence that effective treatments for depression such as antidepressant medications and electroconvulsive therapy down-regulate brain 5HT2A receptors.24–26 However, the potential role of 5HT2A receptor downregulation following psychedelic treatment has yet to be determined. Furthermore, these burst 5HT2A effects stimulate a downstream cascade that triggers increased production and release of brain-derived neurotrophic factor (BDNF), ultimately increasing neuroplasticity, a common pathway for mechanistically distinct antidepressant treatments.27,28 Immunomodulatory and anti-oxidant effects of psychedelics have also been implicated in their potential antidepressant effects.18,28,29
On a network level, preliminary imaging and electrophysiological studies have implicated the modulation of several key brain regions. Modulation of connectivity patterns and the default mode network (DMN) has been a major focus of neuroimaging studies to date. 30 Activity and connectivity of brain regions crucial to self-referential thinking in the DMN are most strongly impacted by psychedelics.31–33 Notably, however, the DMN theory of psychedelics has been criticized, with some studies suggesting that the observed changes in the DMN are not specific to classic psychedelics, but may be a non-specific change observed with other mind-altering substances. 34
Beyond material changes observable through molecular, electrophysiological and neuroimaging studies, significant theories have been advanced to determine the psychological mechanisms underlying the antidepressant effects of psychedelic-assisted psychotherapy.7,9 The belief that psychedelics may act as a catalyst to achieve greater insight and have emotional breakthroughs in therapy has been a central theory for the mechanism of action of psychedelics.23,35,36 Another theme is the possibility that psychedelics address rigid, inflexible and maladaptive thoughts and ruminations patterns, providing the opportunity for accessing and shifting these negative core beliefs. This theory is supplemented by the entropy hypothesis that posits that concrete, rigid negative thought patterns underlie depression. A robust acute increase in entropic brain activity, or randomness, during a psychedelic experience, may allow these ruminative and rigid thought patterns to be released and potentially replaced with more adaptive core beliefs and psychological flexibility.31,37 Additionally, spiritual or mystical experiences have been shown to predict sustained psychological benefits, suggesting that they may play a key psychological role in facilitating the antidepressant effects of psychedelics.38,39
Lastly, it has also been suggested that the large antidepressant effect sizes associated with psychedelics may be primarily driven by placebo or expectancy effects.40–43 The positive effects of placebo have been demonstrated for both mental and physical diagnoses.44–47 Given the dramatic experiential effects and propensity for functional unblinding with psychedelics, the neurobiological mechanisms of the placebo effect (as summarized in recent reviews44,48,49) may play an important role.
Notably, the negative connotations of placebo effects have been questioned in recent years, and are considered legitimate neurobiological effects that should not be avoided, but robustly evaluated and optimized to improve patient outcomes. 44 Moreover, patient improvements in clinical practice secondary to placebo effects are still beneficial, regardless of the underlying neurobiological mechanism at play (e.g., placebo vs. non-placebo mechanisms). As such, an agnostic approach may be advantageous; considering placebo and expectancy effects as a single contributing factor to psychedelics’ central mechanism of action, rather than just a troublesome and unavoidable confounder. Nevertheless, placebo effects may still be problematic for researchers attempting to design rigorous, blinded, clinical trials evaluating psychedelics with comparable scientific standards as clinical trials evaluating standard antidepressant medications. Notably, however, challenges with blinding are not unique to psychedelics as functional unblinding frequently occurs with disparate pharmacological and non-pharmacological interventions. For example, psychotherapy trials comparing cognitive behavioural therapy to non-directive therapy or wait-list controls would similarly have high rates of functional unblinding given how dramatically different these interventions are subjectively experienced by trial participants.
What is the Pharmacokinetic Profile of Psychedelics?
Psychedelics show significant variability in their pharmacokinetics, specifically in terms of the overall duration of their hallucinogenic effects. While there is some variability and dose dependence, the overall duration of effects with moderate to high doses following oral administration is shortest for ayahuasca (∼4 h), moderate with psilocybin (∼6 h) and longest with mescaline and LSD (8–12 h).9,50,51 Notably, oral DMT is rapidly metabolized by monoamine oxidases in the gastrointestinal tract and liver and thus has a negligible bioavailability in the absence of a monoamine oxidase inhibitor (MAOI, such as harmine, which is present in ayahuasca).9,50,52 Conversely, intravenous and inhaled DMT may have brief bioavailability with strong psychoactive effects over 5–15 min. 53 Details of onset of clinical effects, half-lives, timing of peak psychedelic effects and time to complete resolution of the psychedelic experience are summarized in Table 3.
Pharmacokinetic Profile of Serotonergic Psychedelics With Time to Onset, Timing of Peak Effects and Total Duration of Psychoactive Effects.
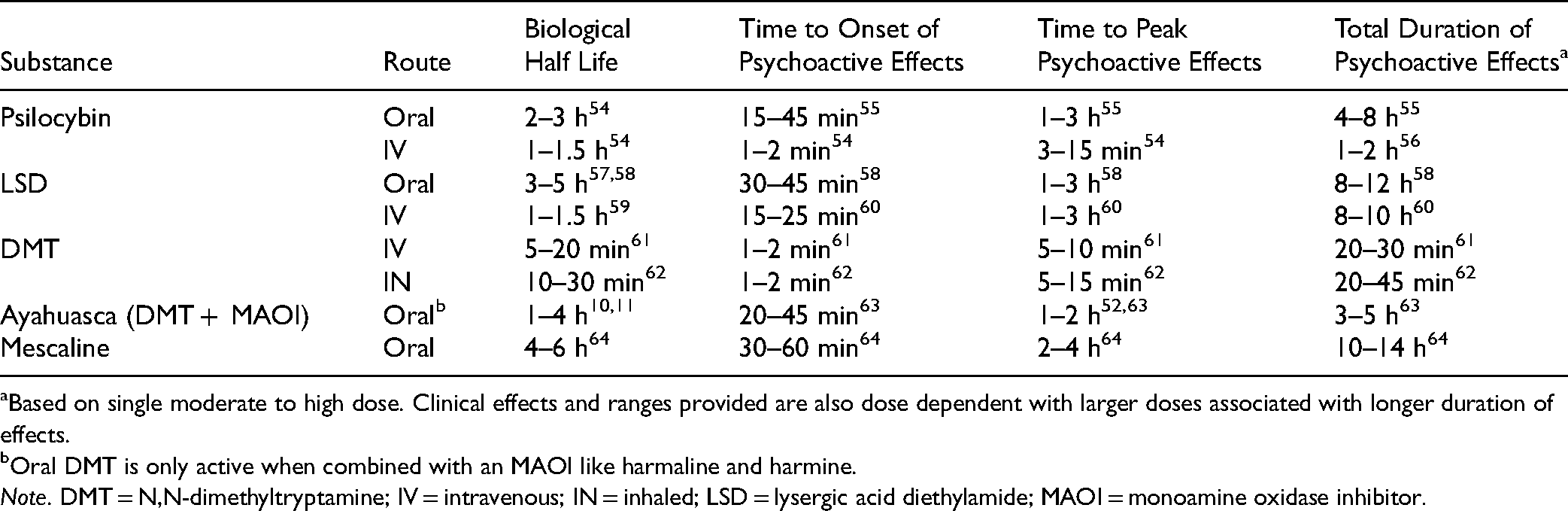
Based on single moderate to high dose. Clinical effects and ranges provided are also dose dependent with larger doses associated with longer duration of effects.
Oral DMT is only active when combined with an MAOI like harmaline and harmine.
Note. DMT = N,N-dimethyltryptamine; IV = intravenous; IN = inhaled; LSD = lysergic acid diethylamide; MAOI = monoamine oxidase inhibitor.
Do Psychedelics Have Antidepressant Efficacy?
Our systematic literature search of studies published from 1 January 1990 to 31 July 2021 identified eight (k = 8) prospective clinical trials evaluating antidepressant effects of psychedelics in depressed patients (summarized in Table 4). Three trials evaluated psilocybin for MDD (two RCTs and one open-label trial) with three additional RCTs evaluating psilocybin in cancer-related depression and anxiety. Two trials (one open label and one RCT) evaluated ayahuasca for treatment-resistant unipolar depression (TRD). We identified no contemporary trials evaluating LSD, mescaline, pure DMT or 5-MeO-DMT for depression. Of note, trials with LSD and DMT for depression have been registered, however, did not have published results at the time of our literature search. Registered trials (including ongoing and unpublished studies) have been recently reviewed elsewhere. 65
Prospective Clinical Trials Evaluating Antidepressant Effects of Serotonergic Psychedelics.
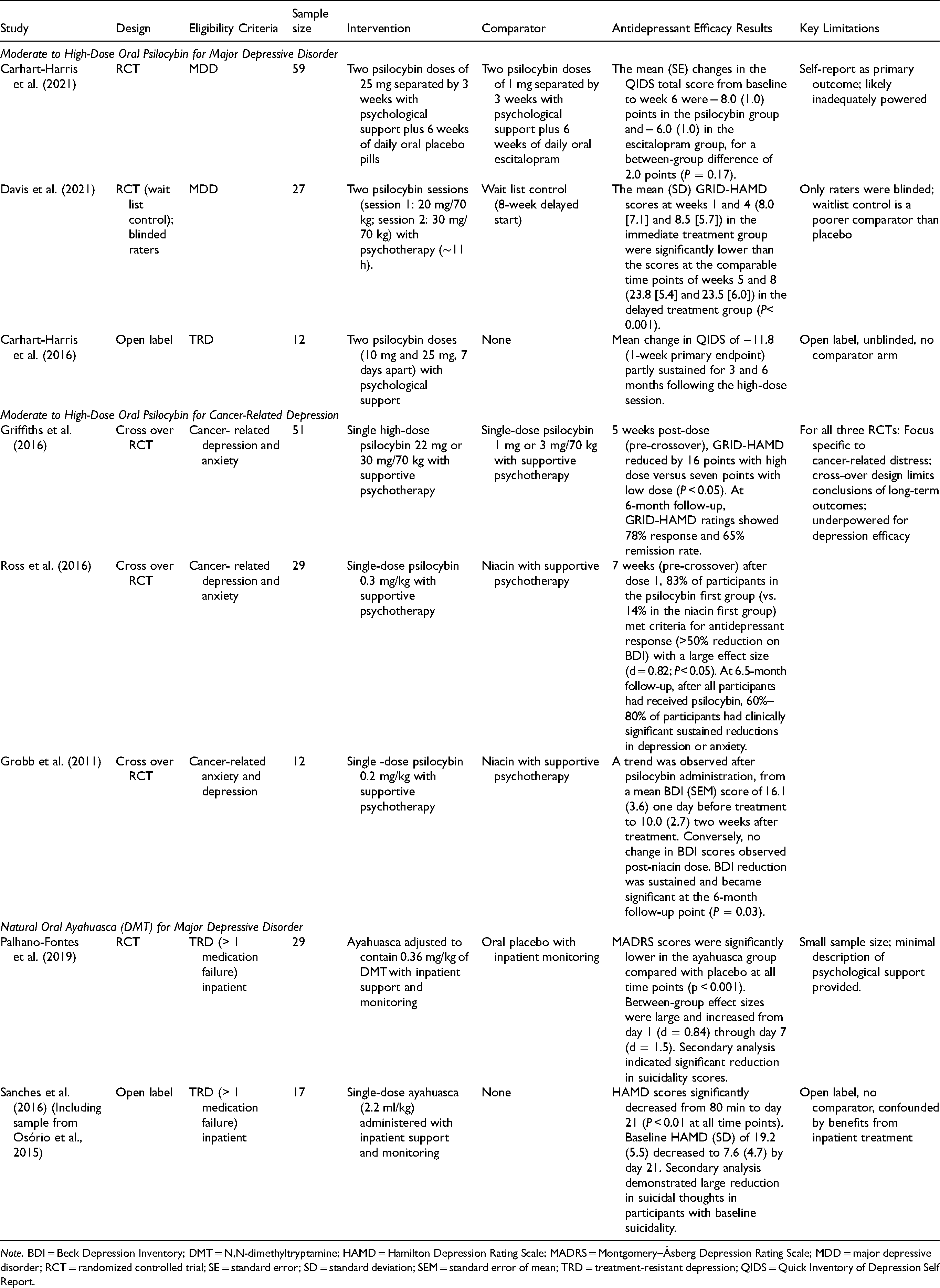
Note. BDI = Beck Depression Inventory; DMT = N,N-dimethyltryptamine; HAMD = Hamilton Depression Rating Scale; MADRS = Montgomery–Åsberg Depression Rating Scale; MDD = major depressive disorder; RCT = randomized controlled trial; SE = standard error; SD = standard deviation; SEM = standard error of mean; TRD = treatment-resistant depression; QIDS = Quick Inventory of Depression Self Report.
Psilocybin for MDD
All three trials evaluated the effects of moderate to high dose (>0.2 mg/kg; >20 mg) oral psilocybin combined with psychotherapy.66–68 The psychotherapy component shared similar elements across all three trials, that is, supportive preparatory sessions before dosing; monitoring and non-directive support during dosing; supportive, non-structured, transtheoretical integration sessions after dosing. None of these studies identified used psilocybin without psychotherapy and none evaluated micro-dosing (i.e., repeated low doses of psilocybin); however, one observational study suggested benefits of micro-dosing were equivalent to placebo. 69
The first study was an open-label, single-armed, feasibility trial involving two sessions (first dose 10 mg, second dose 25 mg oral psilocybin) of psilocybin-assisted psychotherapy spaced 7 days apart in 12 participants with TRD. There was a significant reduction in depressive symptoms (as measured by the Quick Inventory of Depressive Symptomatology Self-Report [QIDS-SR]) after 7 days (hedges’ g = 3.1; P = 0.002) and 3 months (g = 2.0; P = 0.003) after the second (high dose; 25 mg) session. 66 At follow-up, QIDS-SR scores remained low at 3 and 6 months (Cohen's d = 1.5 and 1.4, respectively; both P < 0.001) with no participants seeking antidepressant treatment within 5 weeks of psilocybin sessions. 70
The same research group completed a phase 2, double-blind RCT involving patients with long-standing, moderate-to-severe MDD, comparing psilocybin-assisted psychotherapy with escitalopram, over a 6-week period (n = 59). 67 Participants were randomized to receive two separate doses of 25 mg of psilocybin with psychotherapy, 3 weeks apart in addition to 6 weeks of daily oral placebo (psilocybin group) or two separate doses of 1 mg of psilocybin with psychotherapy, 3 weeks apart as well as 6 weeks of daily oral escitalopram (escitalopram group). All participants received the same psychological support. The mean changes in score from baseline to week 6 on the primary outcome measure (QIDS-SR) was a between-group difference of 2.0 points that failed to reach statistical significance (P = 0.17). There was no statistically significant difference in response rate (proportion with ≥50% QIDS-SR reduction; 70% in the psilocybin group vs. 48% in the escitalopram group) but rate of remission (endpoint QIDS-SR score ≤5) was significantly superior in the psilocybin group (57% vs. 28%, respectively). However, these significant differences in remission were not observed after adjusting for multiple comparisons. Other secondary outcomes (such as clinician-rated depression scales) significantly favoured psilocybin over escitalopram, but were not adjusted for multiple comparisons. The incidence of adverse events was similar in both groups.
In an 8-week RCT, Davis et al. (2021) 71 utilized a waitlist control design. The intervention consisted of preparation sessions for 3 weeks followed by two full-day psilocybin sessions (session 1: 20 mg/70 kg; session 2: 30 mg/70 kg) separated by 1–2 weeks. Participants were randomized to begin treatment immediately (n = 12) or after an 8-week delay (n = 15), with all participants eventually receiving active treatment. The mean (SD) GRID-Hamilton Depression Rating Scale (GRID-HAMD) scores at weeks 5 (8.0 [7.1]) and 8 (8.5 [5.7]) (representing 1 week and 4 weeks following the second psilocybin session) in the immediate treatment group were significantly lower than weeks 5 and 8 scores (23.8 [5.4] and 23.5 [6.0]. respectively) in the delayed treatment group. The effect sizes were large at week 5 (d = 2.5; P < 0.001) and week 8 (d = 2.6; P < 0.001). Following intervention periods in both the immediate and delayed treatment groups, 71% of participants at week 1 and 71% at week 4 had a response with psilocybin (>50% reduction in GRID-HAMD score). The remission rate (<7 GRID-HAMD score) was also high: 58% at week 1 and 54% at week 4.
Analysis of long-term outcomes with prospective 12-month follow-up was also completed for the above study. 72 Large decreases from baseline in GRID-HAMD scores were observed at 6-month (d = 2.6) and 12-month endpoints (d = 2.4). At 12 months, 75% of participants met the criteria for response and 58% for remission. There were no serious adverse events judged to be related to psilocybin in the long-term follow-up period. No participants reported psilocybin use outside of the context of the study.
These three clinical trials provide preliminary evidence of antidepressant efficacy following one or two moderate to high doses of oral psilocybin combined with supportive psychotherapy. 73 All three trials are limited by small sample sizes, inadequate control arms and weak blinding. These results represent Level 3 evidence for acute antidepressant efficacy for psilocybin-assisted therapy. While all three trials included participants with TRD, only one open-label trial specifically evaluated psilocybin-assisted psychotherapy in TRD exclusively (e.g., TRD required as part of eligibility criteria), supportive of Level 4 evidence in TRD populations. Superiority of psilocybin over standard treatments has not been established.
Psilocybin for Cancer-Related Depression and Anxiety
We identified three crossover RCTs evaluating the antidepressant and anxiolytic effects of psilocybin-assisted psychotherapy in cancer populations with depression and anxiety. Grob et al. (2011) completed the first double-blind, placebo-controlled, crossover pilot trial involving twelve participants with advanced-stage cancer-related depression and anxiety, using a moderate dose (0.2 mg/kg) of psilocybin compared with niacin as an active placebo, with both groups receiving supportive psychotherapy. While safety and feasibility were the primary outcomes of this study, depressive symptoms were measured using the Beck Depression Inventory (BDI). For the BDI, there was an overall interaction of psilocybin by day that did not attain statistical significance at the 5% level (F[1,11] = 3.75, P = 0.08). BDI scores decreased by almost 30% 1 month after the second treatment session (P = 0.05), a difference that was sustained and became statistically significant at 6-month follow-up (P = 0.03).
Ross et al. (2016) completed a similar trial in 29 patients with cancer-related anxiety and depression, comparing single-dose psilocybin (0.3 mg/kg) to niacin, both in conjunction with psychotherapy. Prior to the crossover (e.g., 7-week primary outcome), psilocybin was associated with immediate, substantial and sustained improvements in anxiety and depression, along with decreases in cancer-related demoralization. On the BDI at 7 weeks after dose 1, 83% of participants in the psilocybin first group (vs. 14% in the niacin first group) met the criteria for antidepressant response (>50% reduction of BDI scores). At the 6.5-month follow-up, psilocybin was associated with enduring anxiolytic and antidepressant effects (approximately 60%–80% of participants continued with clinically significant reductions in depression or anxiety). An additional long-term follow-up study found that antidepressant effects were still observed in 60%–80% of participants years after the trial was completed (average follow-up of 4.5 years). 74 The degree of psilocybin-induced mystical experience correlated with the therapeutic effect of psilocybin on anxiety and depression.75,76
Griffiths et al. (2016) conducted a similar study in a larger sample using low-dose psilocybin as the control. 39 The study population was comprised of 51 people with life-threatening cancer diagnoses and symptoms of depression and anxiety. This double-blind, crossover RCT compared psychotherapy assisted by very low psilocybin dose (1 or 3 mg/70 kg) to psychotherapy with -ose psilocybin (22 or 30 mg/70 kg) administered in counterbalanced sequence with 5 weeks between sessions and a 6-month follow-up. Compared to the very low-dose group, the high-dose psilocybin group had large decreases in clinician- and self-rated measures of depressed mood and anxiety, along with increases in quality of life (prior to the respective crossover). At 6-month follow-up, these changes were sustained, with 80% of participants continuing to show clinically significant decreases in symptoms of anxiety and depression. Similar to earlier trials, mystical-type psilocybin experience on the session day was associated with positive outcomes. The only serious medical or psychiatric adverse event reported in any of the three trials was one patient dying by suicide 11 days after receiving a very low dose of psilocybin in the Griffith's trial. However, the authors reported that the death was not deemed to be related to the research procedure or psilocybin. 39
Ayahuasca for MDD
While no clinical trials were identified evaluating pure DMT or 5-MeO-DMT, two trials evaluated natural ayahuasca (containing DMT plus the MAOI harmine) administered orally in TRD. The first pilot study evaluated the effects of a single dose of ayahuasca in six participants in a current major depressive episode who had failed at least one antidepressant trial. 77 This trial was then extended to include a total of 17 participants. 33 In this open-label trial conducted in an inpatient psychiatric unit, each participant drank 120–200 ml of ayahuasca (2.2 ml/kg body weight). No specific psychotherapy sessions were described in the published report. Ayahuasca administration was associated with significant decreases in depression severity (i.e. HAMD and Montgomery–Åsberg Depression Rating Scale [MADRS] total scores) from 80 min through day 21. Ayahuasca was described as well tolerated, but emesis was reported in 47% of participants. However, the authors note that acute emesis is believed to be part of the therapeutic effect, as seen in ayahuasca ceremonial rituals, and therefore should not be necessarily interpreted as a problematic adverse effect. In a secondary analysis, among individuals with suicidality at baseline (n = 15), post-acute effect sizes for decreases in suicidality were large (g = 1.31–1.75), with the largest effect size occurring 21 days after the intervention (g = 1.75). 78
The same investigators led a parallel-arm, randomized, double-blind, placebo-controlled trial in 29 people with TRD (defined as >1 antidepressant failure), who received a single dose of either ayahuasca or placebo. In contrast to the open-label studies, supportive care was provided before, during and after the dosing session, utilizing common elements of psychedelic-assisted psychotherapy, such as music. 79 Participants received a single dose of 1 ml/kg of liquid placebo or ayahuasca adjusted to contain 0.36 mg/kg of N,N-DMT with support throughout the session from at least two investigators. When the acute psychedelic effects were resolved, each participant received a final psychiatric evaluation and debrief. Although no additional therapy was described, four participants remained as inpatients for an entire week. Significant antidepressant benefits of ayahuasca were observed compared with placebo at all-time points (P < 0.05). MADRS scores were significantly lower in the ayahuasca group compared with placebo at days 1 and 2 (P = 0.04), and at day 7 (P < 0.0001). Between-group effect sizes increased from day 1 (d = 0.84) to day 7 (d = 1.49). Response rates were higher in the ayahuasca group compared to placebo at day 7 (64% vs. 27%; P = 0.04), although the remission rate failed to reach statistical significance (36% vs. 7%; P = 0.054). An additional secondary analysis evaluated reduction in suicidal thoughts with medium between-group effect sizes favouring ayahuasca at all time points (ayahuasca vs. placebo; day 1: d = 0.58; day 2: d = 0.56; day 7: d = 0.67). 80
Overall, evidence from these two small trials supports acute antidepressant efficacy following a single dose of oral ayahuasca. Given the small sample sizes, methodological limitations and lack of replication from independent centres, there is Level 3 evidence for acute antidepressant efficacy of single-dose oral ayahuasca for TRD. Preliminary evidence also supports potential benefits for the reduction in suicidality.78,80
Is Psychotherapy Necessary for Psychedelic Treatments?
All published trials of psilocybin include psychotherapy as an integral part of the intervention. Psychotherapeutic support has been associated with a greater likelihood of occasioning mystical-like experience and has been generally accepted as an essential part of psychedelic treatments.50,81 The therapy includes preparatory session(s), non-directive dosing session monitoring by two individuals (referred to as guides, monitors or therapists) followed by integration sessions to support individuals in processing their psychedelic experience. However, only brief descriptions of the psychotherapy itself are provided in published studies. Nevertheless, a systematic review of common components of psychedelic therapy, identified the following elements. 82 In preparation sessions, there is a focus on building therapeutic alliance and trust, understanding the patient narrative, setting intentions and providing psychoeducation on what to expect from the treatment session. Dosing sessions include two providers who are non-directive, with an emphasis on the patient focusing inwards, rather than actively engaging with external therapists during the actual dosing session. This role is primarily described as safety and support, rather than playing an active or directive role in the patient's psychedelic experience. 12 A pre-selected music playlist was also included in all trials, most commonly consisting of classical music with minimal lyrics. The use of music is described as an essential aspect of dosing sessions; however, superiority with specific types of music has not been demonstrated.83,84 Integration sessions are described as helping the patient retell their experience and connect the experience with the initial intentions set out in preparatory sessions. 82
In contrast to psilocybin trials, the three ayahuasca studies had fewer psychotherapeutic components described. However, based on the inpatient milieu used in all three trials, a certain degree of preparation and support may be assumed.33,77,79 Although some form of psychological support may be necessary for the safety and antidepressant efficacy of psychedelic treatments, it is undetermined whether psychedelics would be as safe and effective in the absence of psychotherapy.
Despite the generally accepted importance of psychotherapy, there has been minimal research to determine optimal therapy protocols or approaches. Therapy protocols and manuals have been largely developed based on expert opinion rather than empirical evidence supporting any specific component. Even basic parameters, such as the minimum amount of psychotherapy (e.g., hours or sessions) needed to adequately prepare for and integrate a psychedelic experience have yet to be evaluated, relying on expert opinion exclusively. Optimal setting (e.g., physical space, music selection, etc.) is also of great interest with potential impacts on safety and efficacy. The role of manualized therapies known to be effective for MDD, such as cognitive behavioural therapy, remains unknown with less directive, supportive approaches predominating in completed trials to date. 82
Are There Drug Interactions With Psychedelics?
Studies to date have focused on using psychedelics as monotherapy requiring a full washout of antidepressants, antipsychotics and mood stabilizers. From a safety perspective, this is based on the theoretical risk of inducing serotonin syndrome or serotonin toxicity; however, the actual clinical relevance and risk remain unknown, with only case reports and one small pilot study in healthy controls providing guidance.12,22,85,86 Downregulation of 5HT2A receptors may also occur with chronic use of serotonin reuptake inhibitors that may theoretically reduce acute psychedelic effects. 21 Nevertheless, concerns regarding combining a monoaminergic antidepressant with a potent 5HT2A receptor agonist are appreciable. Specifically, combining antidepressants with ayahuasca may pose a high risk of serotonin syndrome and hypertensive crisis because in addition to DMT, ayahuasca contains MAOIs (harmaline and harmine).53,85,87 There have also been concerns regarding seizures if psychedelics are combined with lithium, based on naturalistic data from online reports. 88 Of note, this naturalistic study did not find safety concerns with other mood stabilizers, namely lamotrigine, that was not associated with seizures or other adverse effects. Drug–drug interactions may also negatively impact potential antidepressant efficacy as any psychotropic that has significant antagonist effects on the 5HT2A receptor (e.g. atypical second antipsychotics and some antidepressants), would block the putative pharmacodynamic target of psychedelics.21,22,89
What are the Acute Side Effects, Safety Concerns and Contraindications for Psychedelics?
When using moderate to high psychedelic doses, acute psychotomimetic effects would be anticipated in close to 100% of individuals. 9 Acute psychological effects broadly include changes in six domains: cognition, perception, negative emotion, positive emotion, mystical experience and social connectedness. 9 All domains may vary greatly among individuals with a reliable dose–response effect and increased intensity at high doses. Drawing the line between therapeutic effect and adverse effect is challenging, as acute psychological effects (both positive and negative) may be a key component in mediating the psychedelic-induced antidepressant effects. The subjective characterization of therapeutic effect versus adverse effect may also impact how adverse effects are reported by investigators with differing views on this categorization with inconsistencies observed between published trials reviewed herein. Additionally, given the challenges of using safety data from homogenous clinical trial samples (e.g., without comorbidities, history of psychosis, substance use disorders, personality disorders, lack of demographic diversity, etc.), adverse effects and safety concerns might be more common in real-world clinical settings including more complex patients. 90
Cognitive changes associated with psychedelics include increased flexibility and creativity, but may also include disorganized and distractible thought patterns. Perceptual disturbances may be hallucinatory (primarily visual and auditory) or dissociative (derealization, depersonalization and time distortion). Negative emotions include anxiety, irritability, agitation, paranoia and mood lability. These negative emotions may be markedly distressing and cause patients to want to leave the dosing session, which is why close monitoring is thought to be required. Conversely, acute positive emotions are also observed with elevated mood, euphoria and general feelings of improved wellbeing. Spiritual and mystical experiences are also common and unique to psychedelics, and have been associated with sustained psychological benefits.39,91 Lastly, social connectedness with enhanced empathy and general openness to others are also commonly observed. 91 The relative importance of these acute effects to longer-term benefits is an active area of research. Of interest, some preclinical models have even called into question if acute psychedelic, mind-altering effects are necessary for antidepressant benefits.21,92
A major concern with psychedelic use is the risk of harm to self and others while in an altered state of consciousness. Case reports and media coverage have highlighted tragic adverse outcomes of physical harm to others and self (including lethal harm). 12 Clinical trials to date suggest that these are rare events that are preventable with adequate participant screening, monitoring and safety protocols. Identified case reports of treatment-emergent suicidality have occurred outside of clinical trial settings, with the exception of one reported suicide in a participant receiving a very low dose of psilocybin.39,93 Nevertheless, more trials are needed to better determine the frequency of these likely rare but serious adverse events, and to delineate safety protocols required to prevent them.
Given the concerns for potentially serious adverse effects, contraindications for clinical use of psychedelics relate to risk factors for greater psychological adverse effects and exacerbating pre-existing disorders. Specifically, personal history of psychosis, mania, hypomania or violence were exclusion criteria in all clinical trials identified. Further, first or second-degree relatives with psychosis or bipolarity are a common exclusion criterion in most studies. However, these exclusion criteria are based on a theoretical risk of vulnerability to adverse reactions. Given the high prevalence of treatment resistance with bipolar depression, it is important to determine whether psychedelics may be safely and effectively used in this population.
In addition to acute psychological effects, psychedelics may also transiently cause increases in blood pressure, acute gastrointestinal side effects and headaches that may persist a few days after a dosing session. Studies evaluated blood pressure regularly during dosing sessions and found relatively mild and transient increases with no hypertensive crises observed. Ayahuasca is specifically associated with significant nausea and emesis in approximately half of the study participants. However, investigators described this as potentially an essential part of the ayahuasca experience (in line with traditional practices 94 ) and report the subjective distress related to emesis as mild. A theoretical concern of possible drug-induced valvular disease due to 5HT2B effects of LSD and psilocin has also been recently suggested. 95
What are the Potential Long-Term Risks Associated With Psychedelics?
In a systematic review evaluating the long-term effects of psychedelics, data from time points greater than 2 weeks after a dosing session were synthesized from 34 contemporary clinical trials. 96 While long-term clinical benefits were observed, limited aversive side effects were noted by study participants past the 2-week point. While there are no documented cases within the short-term clinical trials reviewed herein, the concern of developing a hallucinogen use disorder is of theoretical concern.8,12 Expert opinion suggests a low risk of developing physical or psychological dependence with prescribed clinical use.8,12,97 Another potential long-term risk of concern is hallucinogen-persisting perception disorder (HPPD), an uncommon adverse effect seen in individuals who regularly use psychedelics, as in hallucinogen use disorders.98–100 However, no cases of HPPD were identified in the included trials.101,102 HPPD is likely very rare with the controlled protocols used in clinical trials, especially when personal and family histories of psychosis are exclusion criteria. Concerns remain for patients who might require repeated doses to maintain therapeutic benefits. For example, if a patient needs multiple doses per year for relapse prevention, or for treatment or relapse/recurrence, the threshold for use that may be associated with HPPD risk remains unclear. 99
Research in the 1960s also raised concerns about chromosomal damage and risk of cancer with LSD, but these findings have never been replicated in human clinical trials.103,104 As with any potentially carcinogenic substance, the effects are likely more relevant with repeated doses and prolonged exposure. Hence the unknown risk of repeated doses to treat mood episode relapse over time warrants further investigation.
What are the Ethical and Medico-Legal Considerations for Psychedelics?
Currently, no psychedelics are available by routine prescription in Canada. The only way to access psilocybin at this time is through a Subsection 56(1) Exemption issued by the federal Minister of Health under the Controlled Drugs and Substance Act (CDSA) or through the special access program (SAP).105,106 A Subsection 56(1) exemption is typically only granted for human use as part of Health Canada approved clinical trials. Recently, a small number of exemptions were granted to allow patients with refractory distress associated with life-threatening illness, especially cancer, to consume psilocybin mushrooms with the support of a therapist. However, the Exemption Office has provided clear and direct guidance to the public and providers that these exemptions are exceptionally rare and that the proper route for access is through clinical trials. Further, exemptions have been typically only granted for medically ill patients, usually near the end of life, along with a small number of therapists for experiential training.
In 2022, following substantial advocacy to reclassify psilocybin and other psychedelics under the SAP, the federal government reclassified psychedelics to permit applications for access through the SAP outside of clinical trials for serious or life-threatening illnesses. 106 Given the recency of these changes to the SAP, it remains unclear the criteria for approving SAP applications for patients with MDD in the absence of terminal medical illness. It is also unclear if this change to the SAP will be followed by additional policy changes that may allow for easier access to psychedelics as part of off-label clinical care. Notably, a limited number of districts, such as Oregon and California, have recently decriminalized specific psychedelics, creating avenues for legal use as part of off-label clinical care outside of Canada.107,108
While clinical trials may be an optimal avenue to provide access to psychedelics safely, Health Canada requires rigorous toxicology and animal testing of any substance prior to allowing its use in clinical trials. 109 This cost for testing is typically prohibitive and is not routinely completed for natural psilocybin. Sourcing psilocybin (with adequate pharmacological and toxicology testing) for clinical trials has proven to be a significant barrier to research locally. Given these barriers, and despite the tremendous interest and investment in psilocybin from physicians, researchers, industry and patients, only two registered psilocybin clinical trials have received Health Canada approval in the past 40 years.110,111
Considering the challenges in accessing psychedelics, clinicians are often confronted with the ethical dilemma of a harm reduction approach, when patients request monitoring and support for self-sourced, illegal psychedelics. Importantly, current federal laws strictly prohibit any healthcare provider involvement, including but not limited to, monitoring dosing sessions, facilitating access or encouraging use in the absence of a Subsection 56(1) exemption or SAP approval. 105 Additionally, self-sourced psychedelics may contain potentially dangerous impurities and an unknown dosage of active ingredients leading to unpredictable outcomes. Accordingly, CANMAT strongly recommends against any physician involvement in such practices, outside of approved clinical trials or the SAP, given the significant medico-legal and ethical concerns.
Lastly, a significant ethical concern, which is not unique to the Canadian context, is the self-administration of psychedelics by therapists, as part of a therapist training program, also known as “experiential training.” A small number of individual therapists in Canada have received exemptions for psilocybin and MDMA therapy training programs, under the assumption that first-hand psychedelic experience is essential to be a competent therapist. Proponents of experiential training believe that it would be beneficial, or should even be required, for psychedelic therapists.7,38,112,113 However, this view is formed without empirical evidence to support the relevance or utility of experiential training. On the contrary, many concerns have been raised about such practices given the allegiance effect that may bias clinical trial results, and human rights concerns restricting therapists who are prescribed psychotropics from participating in the training.112–114 Additionally, there are concerns that history may repeat itself, given the catastrophic effects of investigator personal psychedelic use contributing to the demise of psychedelic research in the 1960s, as most typified by Timothy Leary's personal use.7,9,112,115,116
In summary, several ethical, medico-legal and regulatory challenges continue to influence the potential translation of preliminary findings from psychedelic clinical trials to clinical practice. Moving forward, balancing patient, provider and societal safety with ethical access will be paramount in Canada and globally.
Conclusions
Given the current legislation and lack of high-quality evidence to support efficacy and safety, the CANMAT Psychedelic Task Force reached a consensus that psychedelics can only be considered an experimental treatment of MDD. There is a great need to conduct clinical trials locally to add to the science while simultaneously developing a route for legal access for depressed patients in Canada. A search of ongoing clinical trials evaluating psychedelics identified 70 registered clinical studies, providing hope that our understanding of clinical applications of psychedelics will dramatically improve in the coming years. 65 Optimizing treatment protocols (e.g., medication dosage, number of psychedelic sessions, role of repeated doses, type of psychotherapy, music selection, follow-up care, monitoring requirements, etc.) to improve efficacy and safety is a priority area of research as current protocols are largely driven by expert opinion rather than empirical evidence. In the interim, cautious optimism is warranted that psychedelic-assisted psychotherapy may provide a novel class of interventions distinct from currently available treatments, with hope of potentially improving patient outcomes in the near future.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest:
Joshua D. Rosenblat has received research grant support from the Canadian Institute of Health Research (CIHR), Physician Services Inc. (PSI) Foundation, Labatt Brain Health Network, Brain and Cognition Discovery Foundation (BCDF), Canadian Cancer Society, Canadian Psychiatric Association, Academic Scholars Award, American Psychiatric Association, American Society of Psychopharmacology, University of Toronto, University Health Network Centre for Mental Health, Joseph M. West Family Memorial Fund and Timeposters Fellowship and industry funding for speaker/consultation/research fees from Janssen, Allergan, Lundbeck, Sunovion and COMPASS. He is the Chief Medical and Scientific Officer of Braxia Scientific Corp and the Medical Director of Braxia Health.
M. Ishrat Husain reports grants from the Brain and Behavior Research Foundation, CAMH Foundation, Canadian Institutes of Health Research, Physicians Services Incorporated Foundation and University of Toronto. Dr Husain has been a PI for a trial sponsored by COMPASS Pathways Limited and is on the advisory boards of MindSet Pharma Inc., Wake Network Inc. and PsychEd Therapeutics. He owns shares of MindSet Pharma Inc.
Yena Lee was previously the Chief Research Officer of Braxia Scientific Corp.
Roger McIntyre has received research grant support from Global Alliance for Chronic Diseases/Canadian Institutes of Health Research (CIHR)/National Natural Science Foundation of China's Mental Health Team Grant; speaker/consultation fees from Lundbeck, Janssen, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Sunovion, Bausch Health, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Abbvie. He is the CEO of Braxia Scientific Corp.
Rodrigo B. Mansur has received research grant support from the Canadian Institute of Health Research (CIHR), Physician Services Inc. (PSI) Foundation, Baszucki Brain Research Fund, and an Academic Scholars Award from the University of Toronto, Department of Psychiatry.
David Castle has received grant monies for research from Eli Lilly, Janssen Cilag, Roche, Allergen, Bristol-Myers Squibb, Pfizer, Lundbeck, Astra Zeneca, Hospira; Travel Support and Honoraria for Talks and Consultancy from Eli Lilly, Bristol-Myers Squibb, Astra Zeneca, Lundbeck, Janssen Cilag, Pfizer, Organon, Sanofi-Aventis, Wyeth, Hospira, Servier, Seqirus; and is a current or past Advisory Board Member for Lu AA21004: Lundbeck; Varenicline: Pfizer; Asenapine: Lundbeck; Aripiprazole LAI: Lundbeck; Lisdexamfetamine: Shire; Lurasidone: Servier; Brexpiprazole: Lundbeck; Treatment-Resistant Depression: LivaNova; Cariprazine: Seqirus. He is a founder of the Optimal Health Program (OHP), and holds 50% of the IP for OHP; and is part owner of Clarity Healthcare. He is on the scientific advisory of The Mental Health Foundation of Australia. He does not knowingly have stocks or shares in any pharmaceutical company.
Hilary Offman has received consulting fees from Braxia Scientific Corp.
Sagar V. Parikh has received honoraria or research funds from Assurex, Takeda, Janssen, Mensante, Aifred, Sage.
Benicio N. Frey has no disclosures.
Ayal Schaffer has received funding (past 3 years) from AbbVie, GlaxoSmithKline, Janssen, Lundbeck, Otsuka, Sunovion.
Kyle T. Greenway has served as an instructor for psychedelic support and an uncompensated scientific advisor for Wake Network, Inc. and grant support from the Quebec Network on Suicide, Mood Disorders and Related Disorders; the Northern Ontario Academic Medicine Association; and the Foundation for Psychocultural Research
Nicolas Garel has no disclosures.
Serge Beaulieu has received honoraria or research funds from CIHR, Pfizer, Otsuka, Lundbeck, Sunovion, Allergan, Abbvie, Janssen-Ortho and Takeda.
Sidney H. Kennedy has received grants from Abbott, Allergan, Brain Canada, Canadian Institutes for Health Research (CIHR), Janssen, Lundbeck, Ontario Brain Institute, Ontario Research Fund(ORF), Otsuka, Pfizer, Servier, Sunovion and Xian-Janssen and consulting fees or honorarium from Abbott, Alkermes, Allergan, Boehringer Ingelheim, Brain Canada, Canadian Institutes for Health Research (CIHR), Janssen, Lundbeck, Lundbeck Institute, Ontario Brain Institute, Ontario Research and has Stock/Stock Options from Field Trip Health
Raymond W. Lam has received honoraria for ad hoc speaking or advising/consulting, or received research funds, from Allergan, Asia-Pacific Economic Cooperation, BC Leading Edge Foundation, Canadian Institutes of Health Research, Canadian Network for Mood and Anxiety Treatments, Healthy Minds Canada, Janssen, Lundbeck, Lundbeck Institute, Michael Smith Foundation for Health Research, MITACS, Myriad Neuroscience, Ontario Brain Institute, Otsuka, Unity Health, Vancouver Coastal Health Research Institute, Viatris and VGH-UBCH Foundation.
Roumen V. Milev has received honoraria or research funds from Ontario Brain Institute, Allergan, Janssen, Lallemand, Kye, Lundbeck, Nubiyota, Otsuka, Pfizer and Sunovion.
Arun V. Ravindran has received grants from Janssen.
Valerie Tourjman has no disclosures.
Michael van Ameringen has received honoraria or research funds from Allergan, Almatica, Brainsway, Lundbeck, Myriad Neuroscience, Otsuka, Purdue, Janssen, Canadian Foundation for Innovation, Hamilton Academic Health Sciences Organization and Pfizer.
Lakshmi N. Yatham has received honoraria or research grants from Allergan, CANMAT, Lundbeck Institute, Otsuka, DSP and Sanofi.
Valerie H. Taylor has received honoraria or research funds from Optmi, DiaMentis, Janssen, Lundbeck, Otsuka, Purdue, Shire and Takeda.
