Abstract

Introduction
Treatment-resistant schizophrenia (TRS), typically defined as a failure to respond to multiple antipsychotic drugs from at least two different classes prescribed in adequate dosages and durations, 1 is one of the most disabling psychiatric conditions. 2 Clozapine has shown superiority among antipsychotic medications and is the only approved drug for the management of TRS. 1 However, it can cause rare, severe adverse effects such as myocarditis and agranulocytosis. 3 Consequently, adherence to rigorous hematological monitoring is demanding and contributes to clozapine underutilization. 4 Adopting a patient-centred approach is especially relevant in TRS when traditional monitoring schedules for clozapine is unfeasible. Herein we outline a patient-centred approach to clozapine therapeutic monitoring.
Case
The patient is a 60-year-old Caucasian man diagnosed with TRS, tobacco use disorder, venous insufficiency, deep vein thrombosis, epilepsy, type 2 diabetes, chronic kidney disease, hypothyroidism, benign prostatic hypertrophy and medication-induced parkinsonism. The patient has limited community supports, and his substitute decision-maker is the public guardian. He previously benefitted from clozapine and had a period of stability between 2006 and 2018. However, clozapine treatment ended because of poor compliance with the required CBC monitoring. He was admitted to hospital in February 2020 owing to the risk of harming others and poor self-care, and he continued to pose a significant threat of violence during hospitalization. We considered a retrial of clozapine to help address his psychosis and risk of violence. We convened departmental rounds to review this patient's case, sought consultation with the hospital ethicist and consulted the Canadian clozapine monitoring network (Clozaril Support and Assistance Network) which supported a reduced blood work frequency to once a month instead of once a week. We reviewed the proposed treatment, ethical considerations, risks and benefits with the public guardian, the patient's substitute decision-maker, which consented. We gradually started clozapine at 12.5 mg and then increased it by 25 mg increments every 3 days. The patient tolerated clozapine well and had reassuring blood work and vital signs. The patient improved significantly: his physical aggression and hostility diminished considerably, and his quality of life improved dramatically.
Discussion
Patient-centred care opens the way for patients who would benefit from clozapine yet are ineligible for treatment due to factors such as non-adherence to bloodwork. A potential approach to patient-centred clozapine care involves: (a) assessing for potential suitability of reduced monitoring, (b) obtaining the second opinion and integrating shared decision making (SDM), (c) obtaining informed consent and (d) proceeding with reduced monitoring schedule and ongoing reassessment (Figure 1).
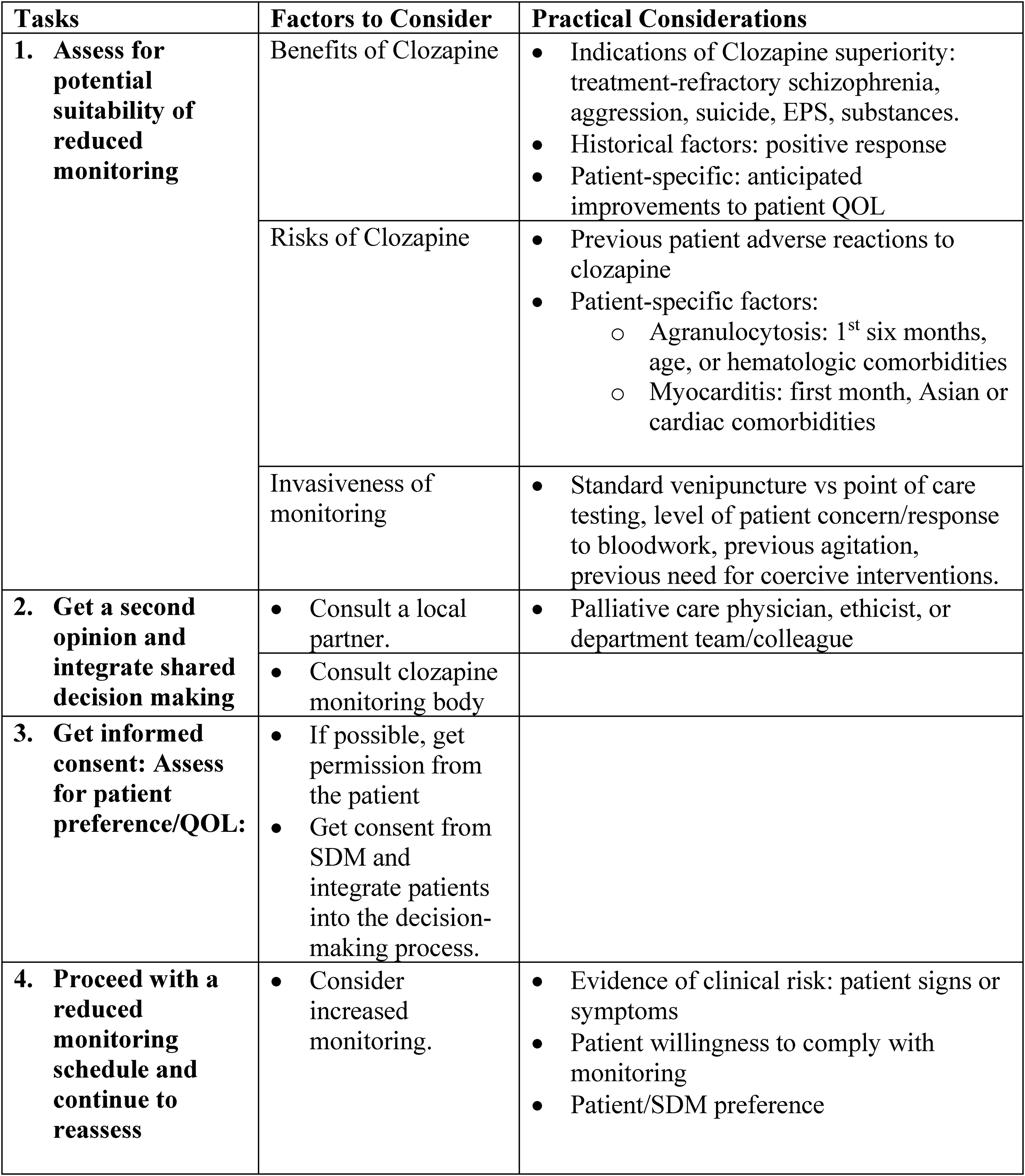
Schema for a patient-centred approach to clozapine monitoring.
Assess for Potential Suitability of Reduced Monitoring
The primary step in approaching reduced clozapine monitoring involves contextualizing the potential benefits and risks of clozapine for the particular patient, in the context of the feasibility and risks of clozapine monitoring. Clozapine boasts superiority among other antipsychotics and is the only first-line treatment for TRS, which underscores the importance of minimizing barriers to clozapine utilization for patients with severe illness. 3 Patients who previously improved and tolerated clozapine but discontinued due to non-adherence with laboratory monitoring are prime candidates for reduced monitoring. At the same time, due to clozapine's severe adverse reactions, it is essential to consider the patient's associated risks before adopting mandatory blood monitoring variations. Patients with a history of severe hematologic reactions or possible myocarditis while on clozapine are not recommended for reduced monitoring as they are likely to develop side effects again. A review of the risks and benefits of clozapine needs to be considered concerning the patient's attitude toward blood monitoring, which may range from mildly resistant—which may be simply addressed through psychoeducation, to profound resistance with extreme hostility and aggression—which may otherwise negate the possibility of community clozapine trial.
Get a Second Opinion and Integrate SDM
Consulting a local partner, such as a palliative care physician, ethicist, or even department team/colleague, can provide a shared clinical decision for the patient's best interest. Consulting a clozapine monitoring body can further guide care plans to use revised monitoring protocols.
Get Informed Consent: Assess for Patient Preference/Quality of Life
Patient-centred care requires core skills of having an open and honest conversation between the patient and/or their caregiver regarding the patient's values, illness, prognosis, and management. Patients should receive education on the risks, benefits, and alternative treatments. Clinicians should get informed consent from the patient or their SDM for a care plan reflecting the patient's preferences to improve their quality of life.
Proceed With a Reduced Monitoring Schedule and Continue to Reassess
Finally, clinicians can implement an action plan involving reduced monitoring with continued clinical reassessment. Clinicians should increase blood monitoring if there is evidence of clinical risk, if the patient agrees to increased monitoring, or based on a change in the patient/SDM treatment preference.
Conclusion
Implementing a patient-centred approach to hematologic monitoring may facilitate more patients’ acceptance of clozapine and hence achieve greater symptomatic relief and improved quality of life for patients with schizophrenia.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MD reports personal fees from Eisai Ltd and Winterlight Labs. AB is a recipient of the 2020 Friends of Matt Newell Endowment. RFW has received honoraria from HLS Therapeutics. There are no other relevant conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
