Abstract
Objectives
Despite increasing rates of legalization of medical cannabis worldwide, the current evidence available on its effect on mental health outcomes including anxiety is of mixed results. This study assesses the effect of medical cannabis on generalized anxiety disorder 7-item (GAD-7) scores in adult patients between 2014 and 2019 in Ontario and Alberta, Canada.
Methods
An observational cohort study of adults authorized to use medical cannabis. The GAD-7 was administered at the time of the first visit to the clinic and subsequently over the follow-up time period of up to 3.2 years. Overall changes in GAD-7 scores were computed (mean change) and categorized as: no change (<1 point); improvement; or worsening—over time.
Results
A total of 37,303 patients had initial GAD-7 scores recorded and 5,075 (13.6%) patients had subsequent GAD-7 follow-up scores. The average age was 54.2 years (SD 15.7 years), 46.0% were male, and 45.6% noted anxiety symptoms at the baseline. Average GAD-7 scores were 9.11 (SD 6.6) at the baseline and after an average of 282 days of follow-up (SD 264) the average final GAD-7 score recorded was 9.04 (SD 6.6): mean change −0.23 (95% CI, −0.28 to −0.17,
Conclusions
Overall, there was a statistically significant decrease in GAD-7 scores over time (in particular, in the 6–12-month period). However, this change did not meet the threshold to be considered clinically significant. Thus, we did not detect clinical improvements or detriment in GAD-7 scores in medically authorized cannabis patients. However, future well-controlled clinical trials are needed to fully examine risks or benefits associated with using medical cannabis to treat anxiety conditions.
Objectifs
Malgré les taux croissants de la légalisation du cannabis médical dans le monde entier, les données probantes actuellement disponibles concernant son effet sur les résultats de santé mentale, notamment l’anxiété, sont des résultats mixtes. La présente étude évalue l’effet du cannabis médical sur les scores au trouble d’anxiété généralisée en 7 items (TAG-7) chez les patients adultes entre 2014 et 2019 en Ontario et en Alberta, Canada.
Méthodes
Une étude de cohorte observationnelle d’adultes autorisés à consommer du cannabis médical. Le TAG-7 a été administré lors de la première visite à la clinique et subséquemment, durant la période de suivi de jusqu’à 3,2 ans. Les changements généraux des scores au TAG-7 ont été calculés (changement moyen) et catégorisés comme étant : aucun changement (< 1 point); amélioration ou dégradation – au fil du temps.
Résultats
Les premiers scores au TAG-7 de 37 303 patients ont été enregistrés et 5 075 (13,6 %) patients ont eu des scores subséquents au suivi du TAG-7. L’âge moyen était de 54,2 ans (ET 15,7), 46,0 % étaient de sexe masculin, et 45,6 % ont mentionné des symptômes anxieux au départ. Les scores moyens au TAG-7 étaient de 9,11 (ET 6,6) au départ et après une moyenne de 282 jours de suivi (ET 264) le score moyen final au TAG-7 s’inscrivait à 9,04 (ET 6,6) : changement moyen -0,23 (IC à 95 % -0.28 à -0.17;
Conclusion
En général, il y avait une diminution statistiquement significative des scores au TAG-7 avec le temps (en particulier, dans la période de 6 à 12 mois). Toutefois, ce changement n’atteignait pas le seuil pour être considéré cliniquement significatif. Nous n’avons donc pas détecté d’améliorations cliniques ni de détriment aux scores du TAG-7 chez les patients autorisés au cannabis médical. Cependant, il faut de futurs essais cliniques bien contrôlés afin d’examiner pleinement les risques ou les avantages associés à la consommation de cannabis médical pour traiter les troubles anxieux.
Introduction
A growing number of jurisdictions are enabling legal access to cannabis for medical purposes. Despite this, cannabis’ therapeutic benefits or potential health harms 1 are still unclear. Indeed, clinical outcomes in individuals who use medical cannabis are understudied—the majority of studies do not assess the effect of medical cannabis as a medical therapy.2,3 Since its legalization, medical cannabis has been utilized for various therapeutic interventions (such as pain, sleep disorders, and others) 4 —one major area of utilization being for the treatment of anxiety disorders and anxiety-related symptoms. 5 However, strong evidence regarding the safety and efficacy of medical cannabis use for anxiety is lacking.6,7 In Canada, the recent legalization of cannabis for non-medical use has corresponded with increasing interest in the therapeutic benefits of cannabis, and with more people considering cannabis use as one option for managing health conditions, including anxiety. Thus, there is a need to further clarify evidence on medical cannabis’ effect on anxiety—to assist clinicians and patients in evaluating the potential risks and benefits of cannabis use on anxiety disorders.
Past systematic reviews on medical cannabis’ effects on anxiety and anxiety-related symptoms have shown mixed results8–11—with significant research evidence linking cannabis use with increased anxiety symptoms.12–14 One meta-analysis on cannabis use and anxiety by Twomey, 15 reported that cannabis use was only a minor risk factor and showed a very small association with increasing anxiety symptoms. Conversely, other studies, such as Gobbi et al., 16 report that cannabis use is significantly associated with a higher risk for depression, anxiety, and suicide in young adults. However, the majority of former studies are limited due to the following: studies focus on how cannabis increases anxiety levels12,14,17 rather than how cannabis could potentially improve anxiety18,19; few accurately differentiate cannabis prescribed medically versus non-medical use13,20; studies are typically age-specific with small cohort sample sizes3,16,21–24; and the studies primarily measure anxiety and concomitant use of cannabis in the context of other illegal substance use (not medical cannabis use).25,26
Despite the evidence on its well-validated measure for anxiety, few studies27–29 have directly utilized the generalized anxiety disorder 7-item (GAD-7) 30 questionnaire as a method of measuring anxiety levels in association with cannabis use. To date, the most recent clinical recommendations from both Canada and the US (based on the best available evidence)31,32 concur that there is limited evidence on cannabis’ effectiveness in improving anxiety symptoms. Concerns also exist on whether medical cannabis use could iatrogenically increase anxiety and anxiety-related symptoms. Thus, clarity of anxiety-related health outcomes among patients accessing medical cannabis is needed to understand its downstream influence on the overall healthcare system. 33 To address this gap, the present longitudinal cohort observational study assesses changes in GAD-7 scores from adult patients medically authorized to access cannabis from 2014 to 2019.
Materials (or Patients) & Methods
Study Design
We conducted a cohort study of adult patients authorized for medical cannabis use from specialized cannabis clinics in Alberta and Ontario, Canada between 2014 and 2019.
Study Population
Inclusion Criteria
The study population consisted of all adult patients authorized medical cannabis (inhaled [smoked or vaporized] or orally consumed [oils] cannabis) attending a chain of specialized clinics in the province of Alberta and Ontario (Canada) between June 2014 and January 2019. Our study includes individuals at least 18 years of age, of any sex and ethnicity, who received medical cannabis authorization for a wide range (over 1,000 reasons) of health conditions (acute and chronic). Patients may choose to seek assessment for medical cannabis through the clinic via a self-referral or by a physician referral. The index date for each patient was the first recorded initial GAD-7 score following medical cannabis authorization at the clinics.
Exclusion Criteria
We excluded patients whose records were incomplete (i.e. they had no recorded GAD-7 score at the baseline and/or follow-up appointments). Patients with GAD-7 scores that were recorded as a number outside (lower or higher) of the range of scores 0–21 due to input error, were also excluded from the study.
Data Source
Between June 2014 and January 2019, all patients seeking assessment at included cannabis clinics in Alberta and Ontario, Canada were eligible. Informed consent was provided by the patient at the time of first referral which allowed data to be collected and used for clinical and research purposes. As part of the intake process, each patient seeking medical cannabis meets with a trained counselor who performs an initial assessment and collects relevant data. All patients must provide sociodemographic information and disclose their primary medical complaints that constitute their rationale for requesting a medical cannabis authorization. In addition, the majority of patients complete several validated questionnaires at the baseline, which includes the GAD-7 questionnaire. Following their initial intake interview, the patient is referred to a physician who makes their assessment based on the self-reported information, the patient's health record, and any additional assessments conducted by the physician. All data was released as de-identified data.
Patient Consent
Informed patient consent was provided by the patient at the time of the first intake, which allows data to be collected and used for clinical and research purposes.
Patient and Public Involvement
Patients and the public were not involved in the design, conduct, and reporting of this research project.
Ethics Approval
This study was approved by the University of Alberta Health Research Ethics Board (PRO 00068887) and the Veritas Research Ethics Board in Ontario (16,111-13:21:103-01-2017). Informed signed consent is provided by the patient at the time of first referral, which allows data to be collected and used.
Outcomes
To assess the association between medical cannabis and anxiety and anxiety-related health outcomes, our study utilized the GAD-7 questionnaire. 30 The GAD-7 questionnaire is a self-administered tool for assessing generalized anxiety disorder and anxiety-related symptoms. The GAD-7 was selected for this study because it is currently one of the assessment instruments routinely administered by physicians during clinical encounters within the clinics. Using the threshold score of 10, the GAD-7 has a sensitivity of 89% and a specificity of 82% for GAD. It is moderately good at screening three other common anxiety disorders—panic disorder (sensitivity 74%, specificity 81%), social anxiety disorder (sensitivity 72%, specificity 80%), and post-traumatic stress disorder (sensitivity 66%, specificity 81%). 30 At every clinical encounter, medically authorized cannabis patients reported on the frequency (from not at all to nearly every day) via the completion of seven question items, to determine the severity of anxiety symptoms experienced. The outcome of the questionnaire is reported as a score between 0 and 21 with 0 indicating no anxiety and 21 indicating the highest level of anxiety.
Other Variables
Covariates including sociodemographic characteristics: age at authorization, sex, neighbourhood average income quintile, length of follow-up, and reason for cannabis use were considered. Neighbourhood average income was determined by matching census data to the current area of residence for the patient. Length of follow-up was recorded as the number of days between the patients’ first appointment when the baseline GAD-7 questionnaire was administered and the appointment when the follow-up GAD-7 questionnaire was conducted. The reason for seeking medical cannabis was coded into grouped categories (Table 1). Due to the scope of the study (anxiety outcomes)—mental health was further divided into subcategories: anxiety, depression, post-traumatic stress disorder, bipolar, panic disorder, and mood disorder (Supplemental Table 1). It is important to note that some patients’ records did not identify their reasons for seeking medical cannabis. Hence, for this subset of patients, reasons for cannabis authorization were coded as “unknown.”
Characteristics of Patients Authorized Medical Cannabis and Screened Using the GAD-7 Questionnaire (
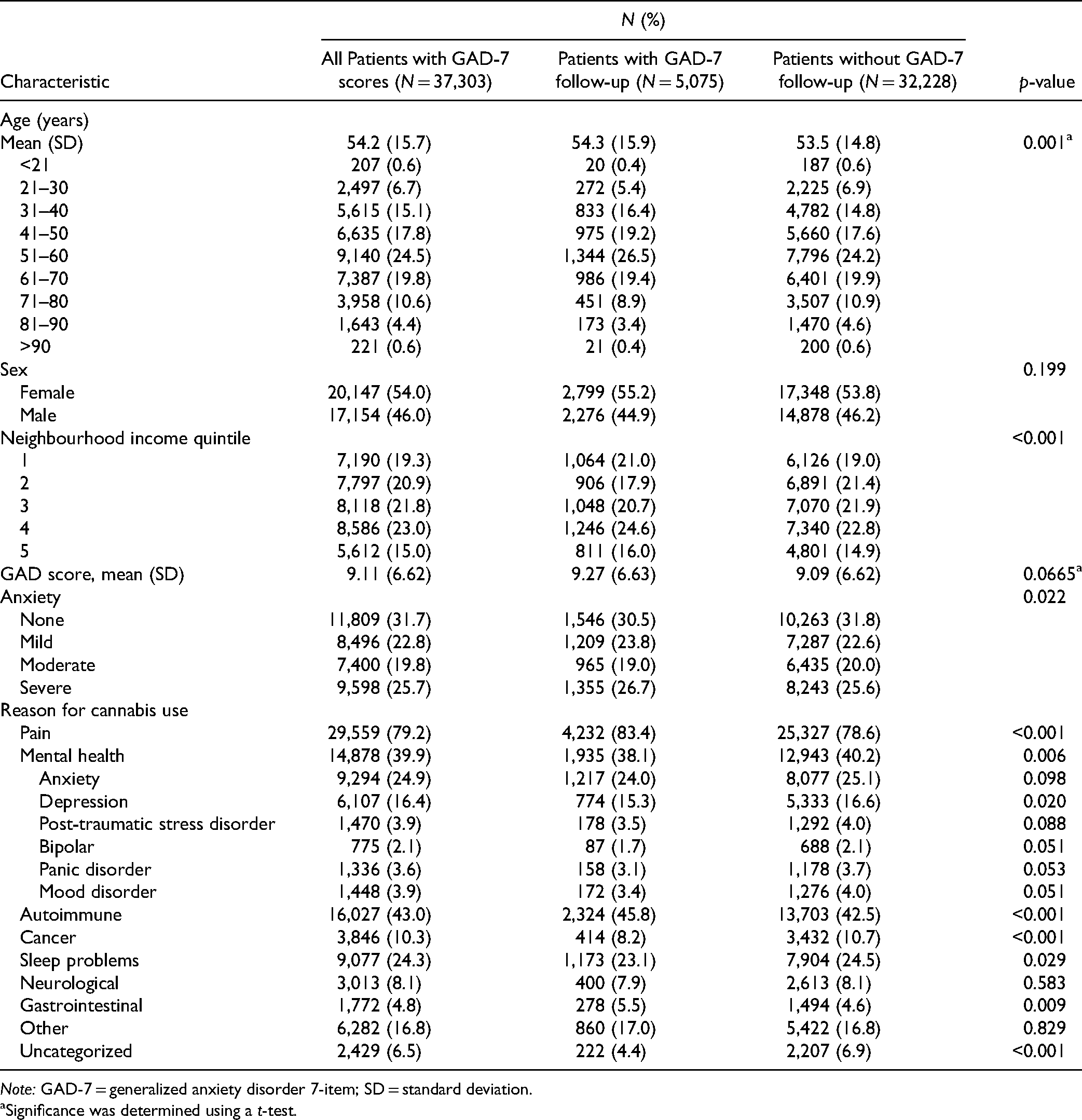
Significance was determined using a
Statistical Analysis
The primary analysis evaluated the mean change in GAD-7 scores by subtracting the baseline GAD-7 scores from the final GAD-7 score. Therefore, a mean change in GAD-7 score with a negative value represents a decrease in anxiety; and a positive value represents an increase in anxiety from the baseline to follow-up (Figure 1). The GAD-7 scores were also represented with clinical categorizations of anxiety levels as follows: GAD-7 score of 0–4 (none), 5–9 (mild), 10–14 (moderate), and 15–21 (severe). 30 These qualitative measures in anxiety categories ensured clearer observance of any clinically relevant changes, in addition to a numeric change in the GAD-7 score. Finally, we used a minimally clinically important difference in the GAD-7 of at least a 4-point change as potentially clinically meaningful, which is supported by Toussaint et al.'s 34 sensitivity to change analysis of the GAD-7 scale.
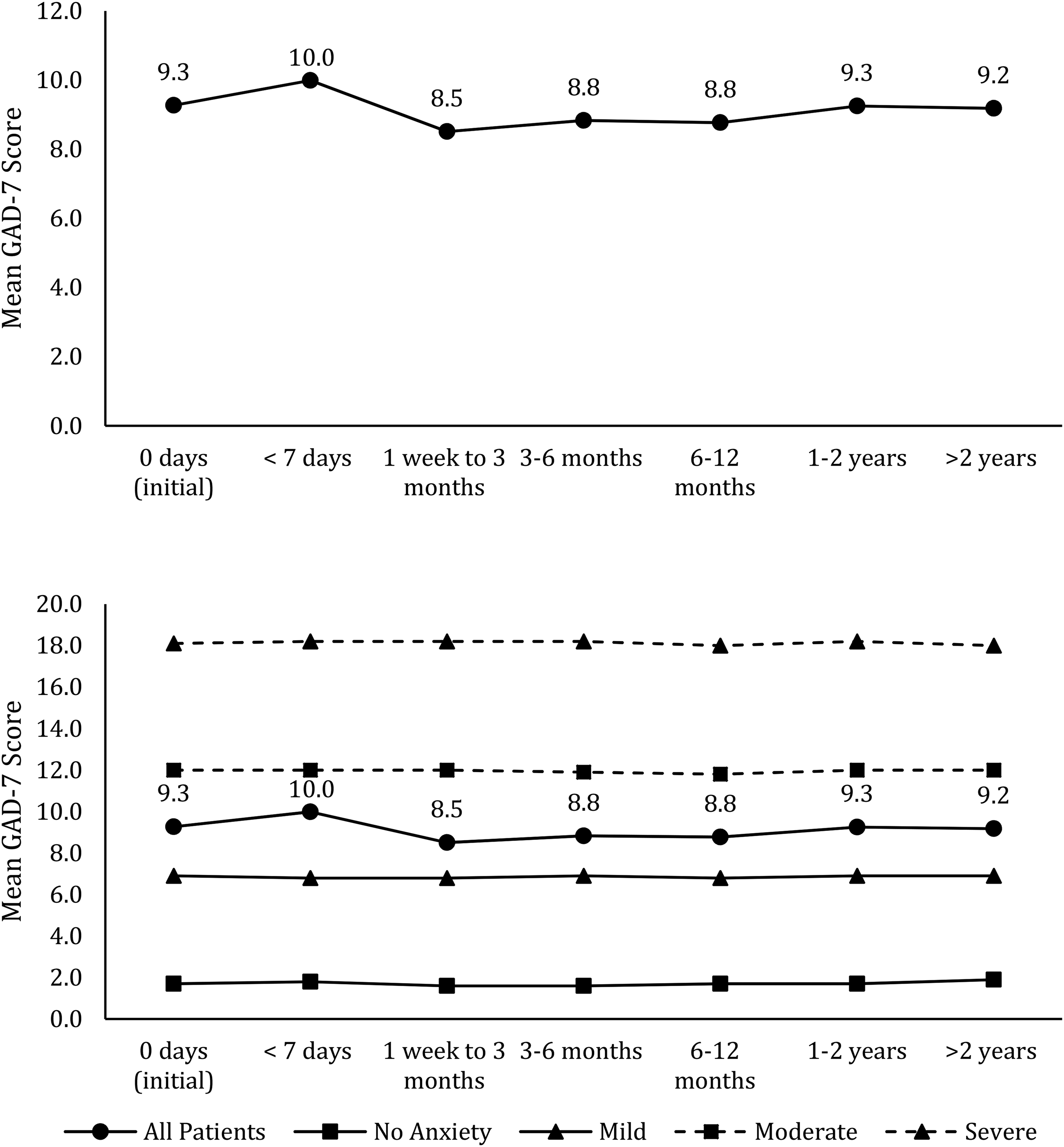
Change in mean generalized anxiety disorder 7-item (GAD-7) scores over time.
Descriptive statistics using counts, percentages, means, and standard deviation were used to describe the patient's demographics. Paired
Sensitivity Analysis
To assess the effect of length of exposure to medical cannabis on GAD-7 scores, follow-up time was divided into six categories: less than seven days; one week to three months; three to six months; six to twelve months; one to two years, and greater than two years as these time frames represented the most common measurement periods of the GAD-7. In a posthoc analysis, we also specifically evaluated the association between authorization for medical cannabis and change in GAD-7 scores among those who reported moderate to severe GAD-7 scores at baseline (i.e., score ≥10). In addition, we restricted the cohort to only patients who had at least 6 months of follow-up data to evaluate the change in GAD-7 scores.
Results
In total, 84,809 patients were authorized for medical cannabis. From this, 37,303 patients completed the initial GAD-7 questionnaire, and 47,506 patients (56%) did not have their GAD-7 score recorded, in which 237 patients had a reported score that was outside of the range of 0–21. However, only 5,075 patients (6%) completed the GAD-7 questionnaire at a consequent follow-up appointment(s) (Figure 2), which were the number of patients included in the analysis. The large loss of follow-up is largely due to the GAD-7 questionnaire not being re-administered at follow-up appointments. Some of the patients had multiple follow-up appointments where the GAD-7 questionnaire was retaken, resulting in 5,732 post-follow-up GAD-7 measurements.
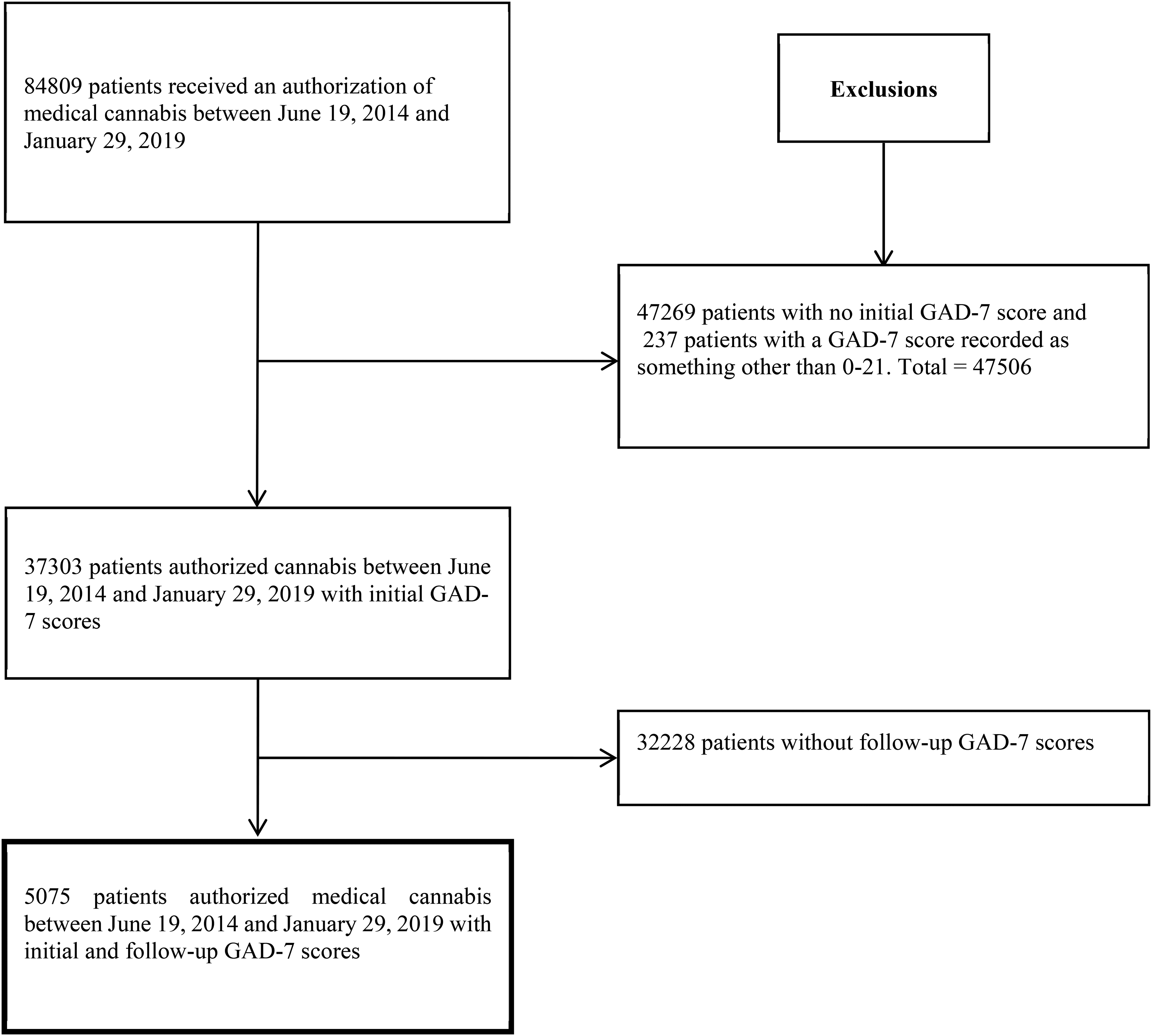
Selection of study population.
The demographics of the patients with GAD-7 retested were similar to those of the patients who did not have their GAD-7 retested; however, there were statistically significant differences in age (
Overall, the mean GAD-7 score at the baseline was 9.11 (SD 6.6). After an average follow-up of 286 days (SD: 268) (median: 195 days, interquartile range 77 to 453), the mean GAD-7 score was 9.04 (SD 6.6). The majority of patients experienced no change in their GAD-7 score (90.8%) or anxiety classification (93.3%) (Table 2; Supplemental Table 3). The mean difference between initial and final GAD-7 scores for all patients with follow-up scores was −0.23 (95% CI, −0.28 to −0.17,
Changes in Anxiety Classification From Initial to Final Generalized Anxiety Disorder 7-Item (GAD-7) Assessment Based on Initial Anxiety Level Classification (
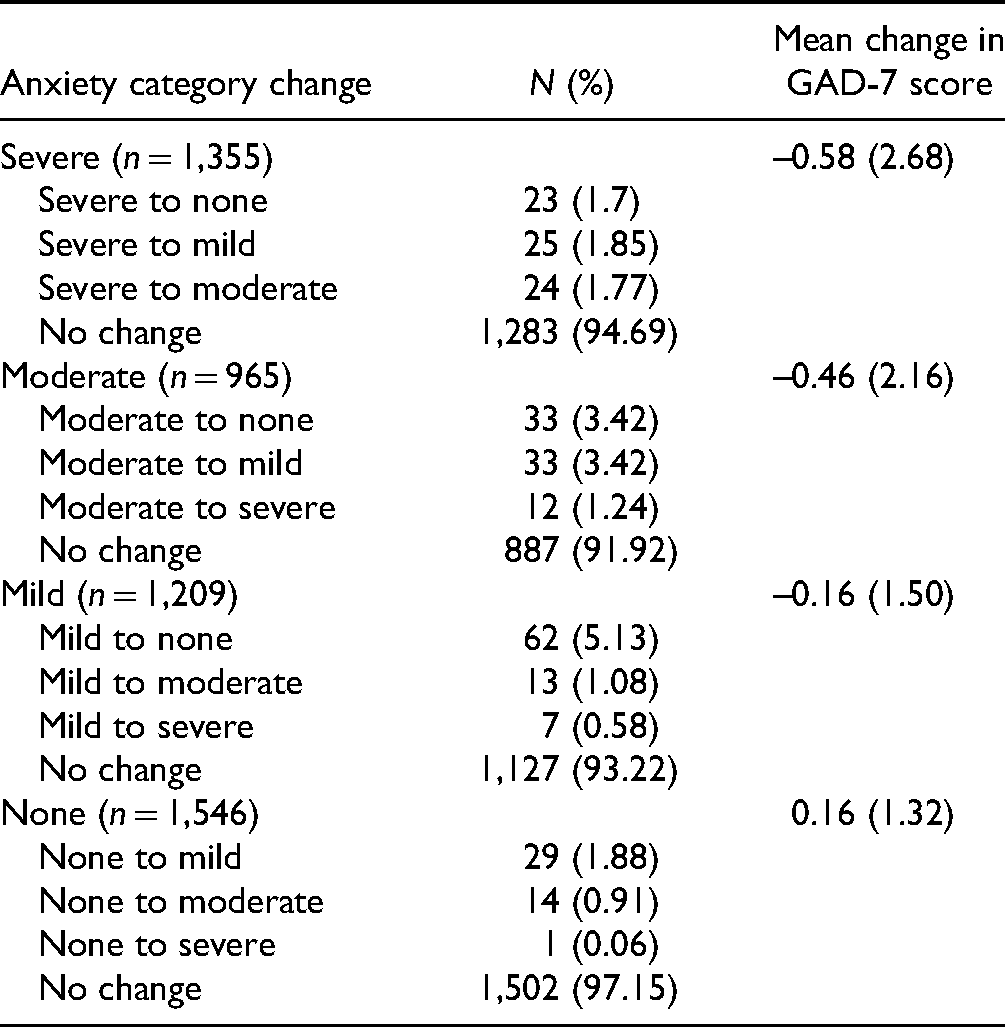
The multiple linear regression analysis showed that the initial GAD-7 score, length of follow-up, pain, anxiety, depression, and sleep disorder were statistically associated with changes in GAD-7 scores (Supplemental Table 4). However, none of the individual coefficients contributed to a clinically important change in the GAD-7 score of at least four points. The other variables (i.e. sex, age, neighbourhood income quintile, and the presence or absence of neurological conditions, gastrointestinal conditions, autoimmune conditions, cancer, general mental health conditions, post-traumatic stress disorder, panic disorders, mood disorders, or bipolar disorder) were determined not to be associated with changes in GAD-7 (Supplemental Table 5).
Sensitivity Analyses Results
The analysis based on the timing of the follow-up appointment revealed that the most frequent change in GAD-7 scores occurred in the 6- to 12-month time period (Table 3). Among the 1,090 (19.0%) patients with a measurement in this time period, 10.6% had a decrease and 3.9% had an increase in their GAD-7 score: mean difference −0.50 (95% CI, −0.67 to −0.34,
Change in Anxiety Categories for Each Patient's Follow-up Generalized Anxiety Disorder 7-Item (GAD-7) Testing Over Each Patient's Entire Follow-up Period (
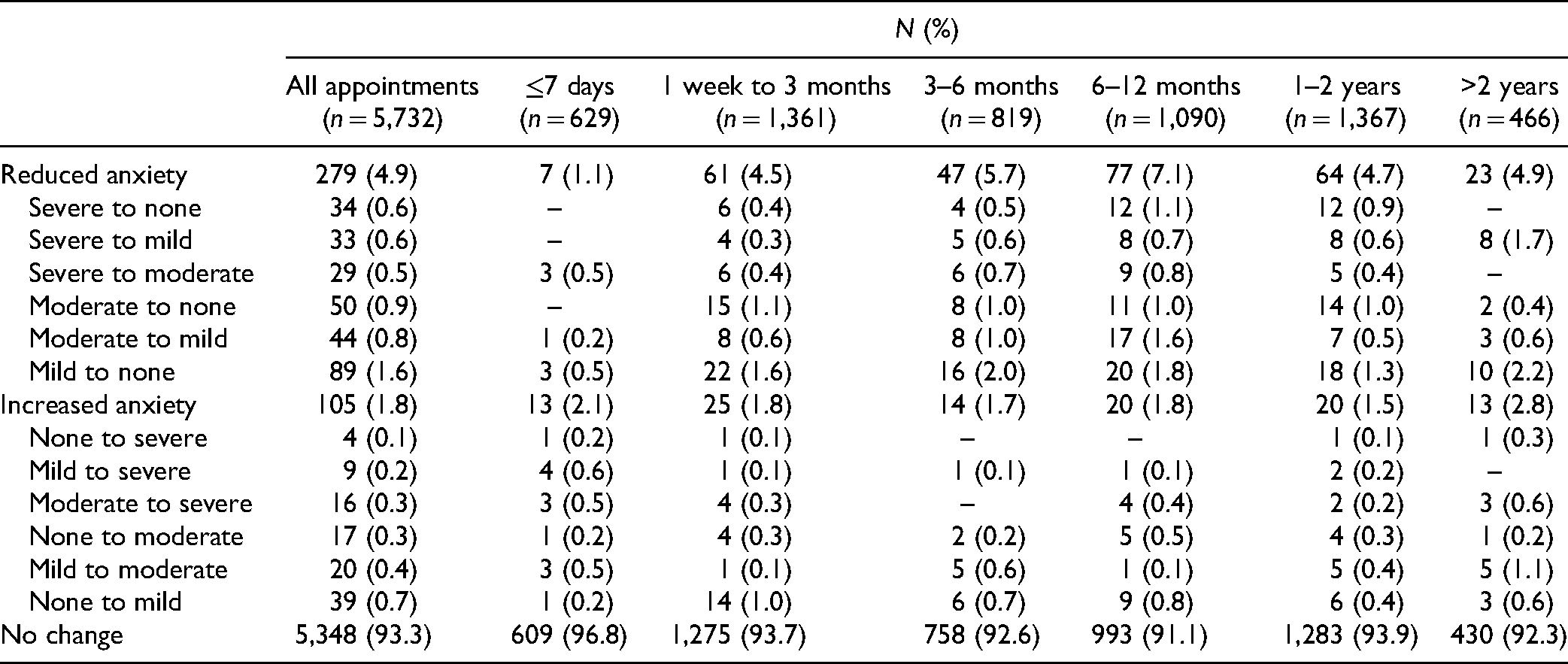
In our posthoc analyses, authorization for medical cannabis was not associated with a clinically important change in GAD-7 scores among those within initially moderate to severe GAD-7 scores. Overall, among these patients (
Discussion
This population-based study showed no clinically important improvements associated with authorization for medical cannabis use and changes in GAD-7 scores (90.8% observed no change) and anxiety classification (93.3%) over time for the majority of patients. Despite a statistically significant decrease in GAD-7 scores over time (in particular, in the 6–12-month period), this change did not meet the threshold to be considered clinically significant. Although most patients demonstrated no benefit, there was no evidence of any worsening in GAD-7 scores or in anxiety classification either.
In comparison with previous studies, there are only three studies that specifically utilized the GAD-7 to measure anxiety levels and cannabis use. Turna et al.
29
reported that in 92% of their participants (
Conversely, the majority of studies on medical cannabis and anxiety-related symptoms reported that anxiety symptoms were higher post-medical cannabis use; however, our data did not support this premise. For GAD-7 scores in the study by Bahorik et al., 35 the results showed that cannabis use worsened anxiety levels. The systematic review by Moore et al. 17 suggested that anxiety outcomes were consistently higher for those who used cannabis more frequently than individuals who did not. Mammen et al. 14 and Hill 12 echoed this result in that cannabis use was often linked with individuals with substance use disorders, which in turn frequently results in higher anxiety levels. For the remaining studies identified, the level of anxiety level reduction/increase was difficult to quantify as every study used a different standard of measuring anxiety (not the GAD-7)9,18,36 or utilized primarily surveys/databases to measure anxiety.13,20,25,37,38 Collectively, these data are in contrast to our findings where the vast majority of patients did not show any worsening in their GAD-7 scores or anxiety classification over time.
The strength of our study is that it is currently, to our knowledge, the largest population-based study on medical cannabis users and changes in anxiety levels, and one of few studies that assess cannabis with validated anxiety-related outcomes using the GAD-7 questionnaire.
However, our study is not without limitations. First, attrition bias was a limitation as a large proportion of baseline medical cannabis patients did not complete their follow-up. From this, it may be possible that those who did not follow-up may have had higher increases in anxiety symptoms. Second, although initial and follow-up GAD-7 scoring is a clinical practice recommendation for medical cannabis authorizing physicians in Canada, the GAD-7 was not consistently administered during the initial/follow-up assessments. Consequently, patients who completed follow-up assessments may represent a specific subset of patients, although baseline characteristics were similar between those completing and not completing follow-up GAD-7 scores. Third, although patients were authorized to use medical cannabis, we have no way of ensuring the products were consumed as prescribed or if patients elected to use alternative agents than what was prescribed for anxiety. Fourth, some patients who are seeking medical cannabis may have had previous experience with cannabis use. Therefore, the GAD-7 baseline score may not represent a true “baseline.” Another limitation of our study is the lack of a control group. We do not know whether GAD-7 scores would change over the same period of time in individuals who did not seek medical cannabis for treating their anxiety symptoms. Lastly, our study is limited by the lack of clinical details: exposure, the amount, route, strain, onset/age of anxiety symptoms, and frequency of consumption of medical cannabis. We also did not have information on any concomitant use of other medications or treatments, such as the use of anti-depressants, mood stabilizers, or benzodiazepines.
Conclusions
Overall, we did detect a statistically significant numerical decrease in anxiety over time, particularly in the 6- to 12-month time period. However, we found no evidence of a clinical or therapeutic benefit associated with authorizing medical cannabis for patients seeking help with anxiety conditions, disorders, and related symptoms. With the increasing availability of cannabis for medical purposes in many areas of the world, we believe our findings contribute important information to the field of mental health—in specific, for clinicians who currently counsel medical cannabis for patients seeking anxiety-related symptom treatment. Although a substantial number of studies have been conducted on acute cannabis use and anxiety, clarity is needed on the exact clinical outcomes of medical cannabis use on anxiety levels, particularly for long-term use. The present study suggests medical cannabis may not impact anxiety levels negatively or positively, however, future well-controlled clinical trials are needed to fully examine risks or benefits associated with using medical cannabis to treat anxiety conditions.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437211043393 - Supplemental material for Generalized Anxiety Disorder 7-Item (GAD-7) Scores in Medically Authorized Cannabis Patients—Ontario and Alberta, Canada
Supplemental material, sj-docx-1-cpa-10.1177_07067437211043393 for Generalized Anxiety Disorder 7-Item (GAD-7) Scores in Medically Authorized Cannabis Patients—Ontario and Alberta, Canada by Cerina Lee, Jessica M. Round, John G. Hanlon, Elaine Hyshka, Jason R.B. Dyck and Dean T. Eurich in The Canadian Journal of Psychiatry
Footnotes
Authors’ Note
Informed written signed consent was obtained from the patient at the time of first referral which allowed data to be collected and used for clinical and research purposes. All data was released as de-identified data to the researchers. Patients and the public were not involved in the design, conduct, and reporting of this research project as.
Acknowledgments
The views expressed herein do not necessarily represent the views of CCSA or its funders. The funders did not participate in the design of the study, collection, analysis, interpretation of the data, and in writing the manuscript.
Authors’ Contributions
DTE, JRBD, JGH, and EH designed the study, and DTE and JRBD acquired the data. DTE and JMR analyzed the data. CL, JMR, and DTE drafted the manuscript. All other authors revised it critically for important intellectual content and approved the final version to be published. All authors are accountable for the work and integrity of the work. The corresponding author and guarantor accepts full responsibility for the work and/or conduct of the study, had access to the data, and controlled the decision to publish. DTE attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted. All authors have read and approved the manuscript.
Data Access
The dissemination of data results to study participants and/or patient organizations in this research project is not possible/applicable. The data from the study will not be shared as only the researchers can have access to the data as per the data sharing agreements.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JRBD is a former member of the board of directors of Aurora Cannabis Inc., which is a for-profit, company licensed for the cultivation and sale of medical cannabis. In the past, JGH has worked as a paid advisor and speaker for Canadian Cannabis Clinics, however, does not have any current ties. JRBD has a financial interest in Aurora Cannabis Inc. DTE and JRBD hold a Mitacs Grant with Aurora as a partner. Mitacs is a national, not-for-profit organization that works with universities, private companies, and both federal and provincial governments, to build partnerships and administer research funding that supports industrial and social innovation in Canada. DTE does not have any past or present financial interest in the companies involved. CL, JMR, and EH have no conflicts of interest to declare. Moreover, the research funders and companies listed were not involved in any aspect of the design or write-up of the study, and all analyses were performed independently from the funders and companies.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Production of this study has been made possible through a CIHR Catalyst Grant for Cannabis Research in Urgent Priority Areas, funded by the Canadian Centre on Substance Use and Addiction using Health Canada Cannabis Research Initiative funds. The views expressed herein do not necessarily represent the views of CCSA or its funders. (grant number CCSA 163022).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
