Abstract

Introduction
Preventative treatment before the diagnosis of a serious mental illness (SMI) is an important area for further research and development. 1 More specifically, treatment of putatively prodromal symptoms may reduce the risk of transitioning to a full-blown SMI. One way to examine the risk of developing an SMI is by using the clinical transdiagnostic staging model proposed by McGorry and colleagues, which outlines stages of the expression of an SMI. 2 The first 3 stages are indicative of individuals at-risk for SMI: stage 0 (no symptoms but having factors that are considered risk for SMI, such as a history of an SMI in a first degree relative, evidence of obstetric complications or a history of severe trauma), stage 1a (experiencing low mood or distress), or stage 1b (experiencing subthreshold or attenuated symptoms for an SMI). The later stages (2 to 4) of the model describe individuals meeting full diagnostic criteria for SMI (Supplementary Table 1).
We previously reported the lifetime history of and current medication treatment for individuals in various stages of risk for an SMI in the Canadian Psychiatric Risk and Outcome Study (PROCAN). 3 We found that not only did treatment history vary by at-risk groups, but that many young people with no formal diagnoses were taking medications, and that some of those young individuals had been taking medications for at least several years. 3 Our aim for this report was to determine whether the same medication treatment patterns persisted through 2 years of the PROCAN follow-up.
Method
PROCAN was a naturalistic longitudinal study conducted at 2 Canadian sites (Calgary and Toronto) following 201 participants (43.3% males), aged 12 to 25 years (17.8 ± 3.7 years) categorized into various stages of risk for an SMI, which is defined as schizophrenia and other psychotic disorders, bipolar disorder or recurrent major depression. 4 At baseline, there were 41 stage 0, 53 stage 1a, and 108 stage 1b participants. PROCAN participants and study design are described in detail elsewhere. 4 Details of demographics and Diagnostic and Statistical Manual of Mental Disorders diagnoses are presented in Supplementary Tables 2 and 3.
Participants were asked at baseline, 6-, 12-, and 24-month assessments what psychotropics they were receiving and had been receiving since their last follow-up visit. This allowed for a continuous record of medication taken during the PROCAN follow-up period. Medications were categorized into 6′ groups: antidepressants, mood stabilizers, anxiolytics, antipsychotics, simulants (attention deficit hyperactivity disorder [ADHD]), and nonstimulants (ADHD). Participants who transitioned to an SMI during the PROCAN follow-up period were excluded from follow-up analyses after transition. Of note, the 43 participants from the Toronto site were only followed up for 12 months and accounted for 36.5% of the 52 dropouts at 24 months.
Results
Overall, the proportion of PROCAN participants receiving medications remained relatively stable across the follow-up timepoints (Table 1), although there were slight increases for all stages at 12 months. Stage 1a participants’ current medication use ranged from 32.7% to 40.5% and stage 1b participants, 31.8% to 46.4%. The exception was that stage 0 participants’ current medication use increased from 14.6% at baseline to 26.5% at 12 months. Stimulants were the most reported medications for stage 0 participants. Among stage 1a and 1b participants, antidepressants were the most reported medications, followed by stimulants. Stimulants (ADHD) increased in frequency in stage 1a and 1b participants from baseline, whereas antipsychotics decreased in frequency after baseline. Finally, mood stabilizers were the least commonly reported medication among all stages and were only reported at baseline.
Medication Use Over Time for PROCAN Participants Stratified by the Baseline Clinical Stage of Risk for SMI.
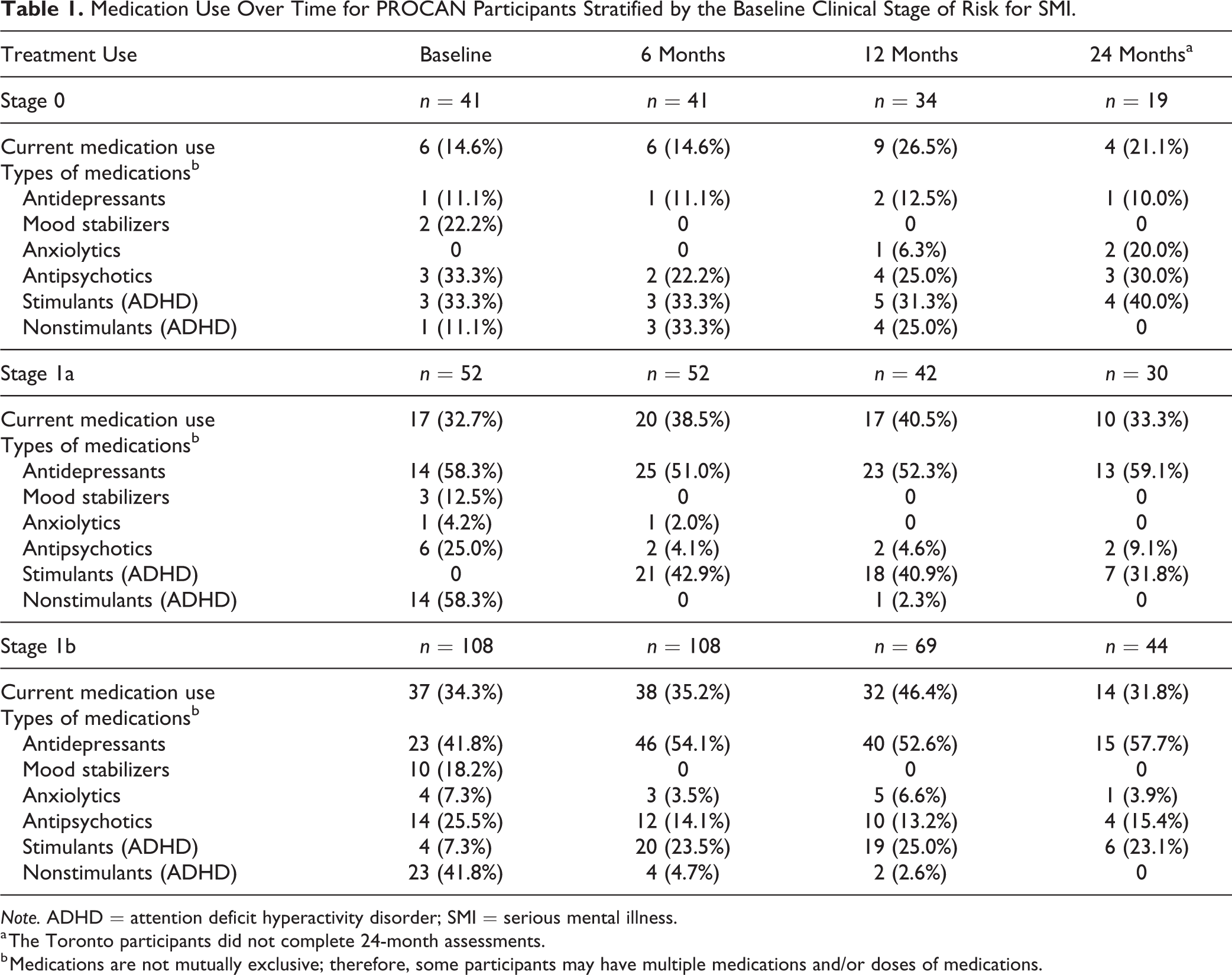
Note. ADHD = attention deficit hyperactivity disorder; SMI = serious mental illness.
a The Toronto participants did not complete 24-month assessments.
b Medications are not mutually exclusive; therefore, some participants may have multiple medications and/or doses of medications.
Discussion
Similar to our previous publication, 3 there were differences between the at-risk clinical stage groups, and by having 2 years of follow-up assessments, we observed that these same treatment patterns persisted over time. The stage 1a and 1b participants reported the highest proportions of medication use, whereas stage 0 participants, although much lower, still reported medication use across the follow-up timepoints. Specifically, antidepressant use increased over time in the symptomatic (stage 1a and 1b) participants.
PROCAN was a naturalistic study. Even though those who had developed an SMI were not included in our analyses, a significant proportion of the participants continued to be in prescribed medication over a 2-year period. Many presented initially with mild symptoms, and the most symptomatic could only be described as having attenuated syndromes. This suggests that there may be gaps in the identification and treatment for the mental health concerns of these young people. The mean age of the sample was 17 years, and so, most participants could be described as emerging adults, for which it is known that there is a lack of developmentally sensitive treatments to meet the needs of these young people. Results presented here offer added support to address the specific needs of youth with mental health concerns.
Supplemental Material
Supplemental Material, sj-docx-1-cpa-10.1177_0706743720988450 - Longitudinal Trends in Medication Treatment for Youth At-Risk for Serious Mental Illness
Supplemental Material, sj-docx-1-cpa-10.1177_0706743720988450 for Longitudinal Trends in Medication Treatment for Youth At-Risk for Serious Mental Illness by Megan S. Farris, Amy Braun, Glenda MacQueen, Benjamin I. Goldstein, JianLi Wang, Sidney H. Kennedy and Jean Addington in The Canadian Journal of Psychiatry
Footnotes
Authors’ Note
Data are not available at this time, due to ongoing data collection. The opinions, results, and conclusions are those of the authors and no endorsement by either funding source is intended or should be inferred.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.A. has received research support from National Institute of Mental Health, Brain Canada, and the Mathison Centre at the University of Calgary. S.H.K. has received funding or honoraria from the following sources: Abbott, Allergan, AstraZeneca, BMS, Brain Cells Inc., Brain Canada, Clera, CIHR, Eli Lilly, Janssen, Lundbeck, Lundbeck Institute, OMHF, Ontario Brain Institute, Otsuka, Pfizer, Servier, St. Jude Medical, Sunovion, and Xian-Janssen. G.M. has been on advisory board or speaker for Allergen, Lundbeck, Lilly, Pfizer, Janssen. B.G., J.W., M.F., and A.B. list no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Brain Canada Foundation and the Mathison Centre for Research & Education at the University of Calgary.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
