Abstract
Objective:
Patients with major depressive disorder often have limited response to first-line and second-line medications; hence, novel pharmacological treatments are needed for treatment-resistant depression (TRD). Ketamine, an N-methyl-
Methods:
A systematic review was conducted with computerized search of electronic databases up to January 31, 2020 using combinations of search terms, inspection of bibliographies, and review of other ketamine guidelines and consensus statements. The level of evidence and lines of treatment were assigned according to CANMAT criteria. Recommendations were given in question–answer format.
Results:
Intravenous (IV) racemic ketamine given as a single infusion has Level 1 evidence for efficacy in adults with TRD. The evidence for multiple infusions, given as an acute series or as ongoing maintenance treatment, is limited to Level 3. Adverse events associated with ketamine infusions include behavioral (e.g., dissociative symptoms) and physiological (e.g., hypertension) events. There is only Level 3 or 4 evidence for non-IV formulations of racemic ketamine. Consensus recommendations are given for clinical administration of IV ketamine including patient selection, facility and personnel issues, monitoring, and maintaining response.
Conclusions:
Single-dose IV racemic ketamine is a third-line recommendation for adults with TRD. The need for repeated and maintenance ketamine infusions should be carefully assessed on a case-by-case basis with consideration of potential risks and benefits. Because of limited evidence for efficacy and risk for misuse and diversion, the use of oral and other formulations of racemic ketamine should be limited to specialists with ketamine-prescribing expertise and affiliations with tertiary or specialized centers.
Introduction
Major depressive disorder (MDD) is a significant public health problem and a leading cause of disability worldwide. Although there are many effective treatments for MDD, including antidepressant medications and psychological treatments, 1 more than 30% of patients are unable to achieve full symptom remission even after several treatment steps. 2 Clinical response with antidepressants, most of which affect monoamine neurotransmitters, often takes 2 weeks or more. Hence, there is still an unmet need for new treatments with greater efficacy and rapid onset of benefit.
One promising nonmonoaminergic neuronal target is the glutamatergic system.
3,4
Ketamine, which has long been used as a general anesthetic, is an antagonist of a specific class of glutamate receptors referred to as N-methyl-
The 2016 Canadian Network for Mood and Anxiety Treatments (CANMAT) guidelines for pharmacological management of MDD listed IV ketamine as an experimental treatment because the available evidence was limited to single-dose studies with only brief follow-up. 7 Since then, the evidence base for ketamine and one of its enantiomers, esketamine, has substantially expanded. There is still a pressing need among patients with TRD for faster-acting and more effective medications. Racemic ketamine is being used off-label in the community and evidence to date suggests noninferiority to IV esketamine. 8 For these reasons, CANMAT organized a task force to review the evidence and provide updated recommendations for the clinical use of ketamine as a pharmacological treatment in adults with MDD. We limited the scope of the task force review to racemic ketamine because intranasal esketamine (Spravato®) has been approved by Health Canada, the United States Food and Drug Administration (FDA), and the European Medicines Agency for patients with TRD, and other reviews are available for its use. 9,10 The use of adjunctive ketamine for psychological or somatic treatments was also outside the scope of this review.
Methods
We followed similar methods as described in previous CANMAT guidelines. 1,11 In summary, relevant studies published in English and French were identified using computerized searches of electronic databases (PubMed, PsychInfo, and Cochrane Register of Clinical Trials) from inception to January 31, 2020, inspection of bibliographies, and review of other guidelines and major reports. We also reviewed recent consensus statements and guidelines on ketamine. 5,12
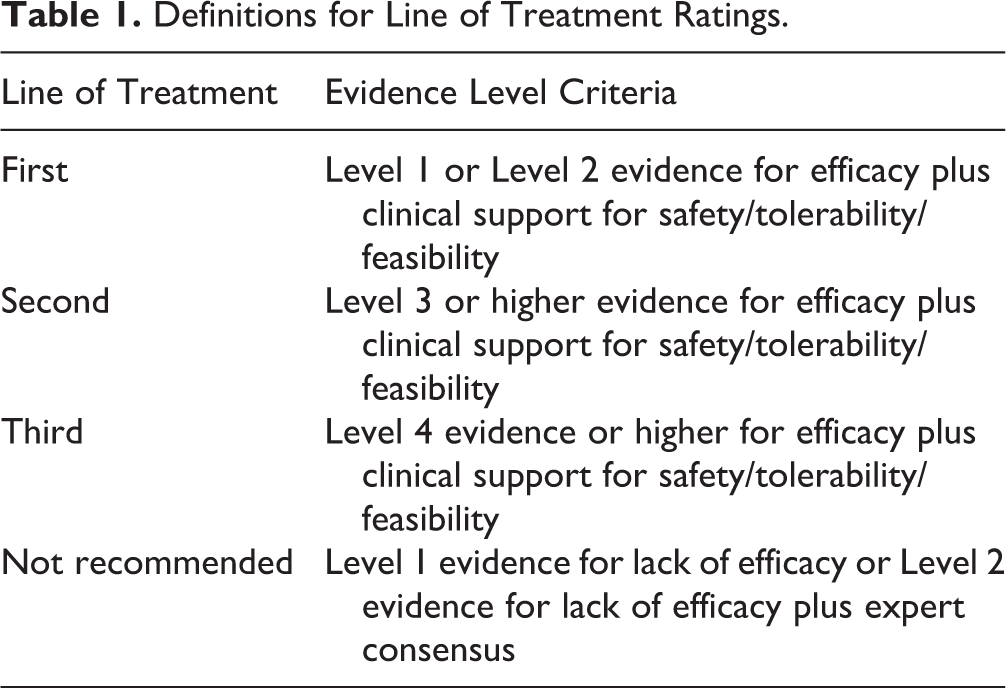
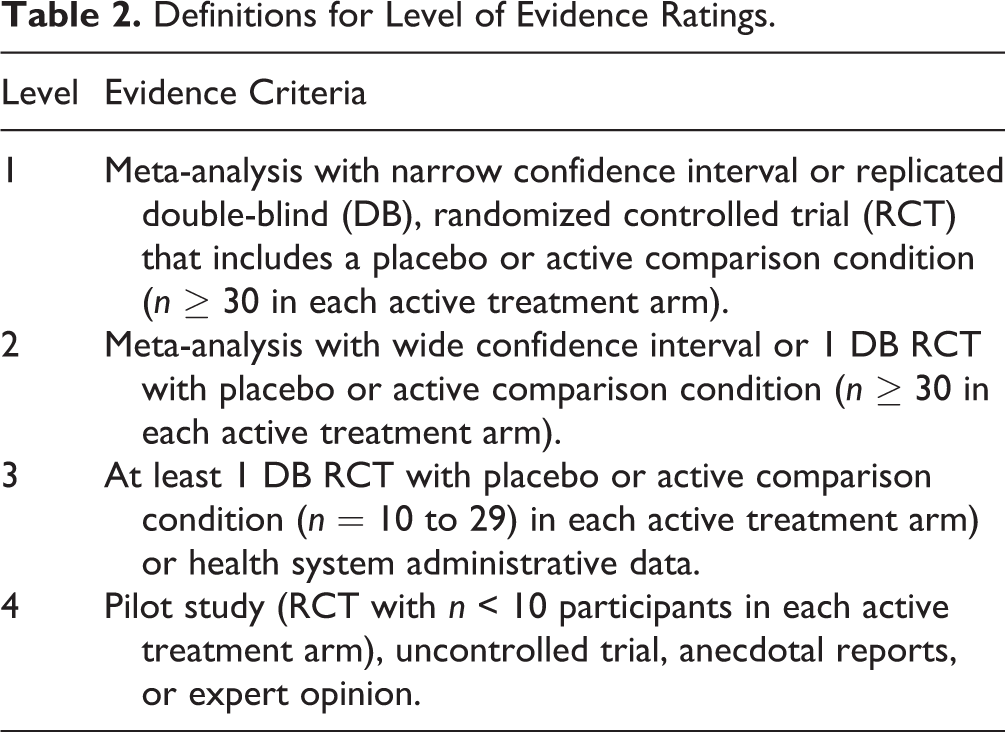
Clinical recommendations are graded by line of treatment (Table 1) with level of evidence (Table 2) supporting each recommendation. Levels of evidence range from highest at 1 (meta-analysis with narrow confidence intervals or replicated double-blind randomized controlled trials [RCTs] with n ≥ 30 in each arm) to lowest at 4 (pilot RCTs with n < 10 in each arm, uncontrolled trials, anecdotal reports, or expert opinion). The Cochrane risk of bias tool was used to assess the study quality. Line-of-treatment recommendations were also informed by clinical support, defined as expert consensus of the task force assessing tolerability, safety, and feasibility. Higher order recommendations (e.g., principles of care) reflect higher level judgment of the strength of evidence from various data sources and therefore are primarily Level 4 evidence. Based on clinician feedback, we retained the question–answer format used in previous CANMAT depression guidelines. The term “ketamine” refers to racemic ketamine unless otherwise indicated. For clarity and to differentiate from compounded formulations of esketamine, we use the brand name “Spravato®” when referring to the intranasal esketamine approved by regulatory agencies.
Definitions for Line of Treatment Ratings.
Definitions for Level of Evidence Ratings.
Results
The task force developed 14 clinical questions with responses presented below. Box 1 summarizes recommendations for the use of racemic ketamine. These recommendations are presented as guidance for clinicians who should consider them in the context of individual patients and not as standards of care.
Summary of Recommendations for the Use of Racemic Ketamine in Adults with Treatment-resistant Depression (TRD) (Level of Evidence in Brackets).

What Is the Mechanism of Action of Ketamine?
The antidepressant mechanism of subanesthetic ketamine remains unclear, but antidepressant doses of ketamine produce a burst in glutamate release 13 and sustained synaptogenesis in depression-related neural circuits, 14 dependent on an increase in brain-derived neurotrophic factor 15 and protein synthesis. 16 A leading hypothesis is that antagonism of NMDA receptors on gamma-aminobutyric acid (GABA)-ergic interneurons in the prefrontal cortex leads to disinhibition and initiation of a cellular and molecular cascade resulting in antidepressant effects. 17 This potential mechanism of action is relevant to esketamine, as (S)-ketamine is approximately 4 times more potent at inhibiting NMDA receptors than (R)-ketamine. 16 However, several adequately powered RCTs of other NMDA receptor antagonists have failed to demonstrate antidepressant efficacy. 18,19 Alternatively, metabolites of ketamine may be involved in the antidepressant mechanism, particularly hydroxynorketamine, a metabolite of (R)-ketamine that affects α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors. 16
Ketamine acts on other neurotransmitter systems, including dopaminergic, serotonergic, and cholinergic systems, in addition to acting on opioid receptors. However, apart from dopamine-2 receptors, ketamine has low affinity within these systems and likely does not have functional agonist or antagonist properties at clinically relevant concentrations. 16 Nevertheless, a small study (n = 14 participants with TRD) recently tested the hypothesis that ketamine may require the opioid system for antidepressant effects. 20 This study randomized individuals to pretreatment with either naltrexone (a nonselective opioid antagonist) or placebo prior to an infusion of ketamine. There was a marked reduction in antidepressant efficacy in the naltrexone-treated group in the 7 ketamine responders, and therefore, the study authors raised caution about the potential for abuse and misuse of ketamine. 21
What Are the Pharmacokinetics of Ketamine?
Ketamine can be administered through multiple routes including IV, intramuscular, intranasal, sublingual, and oral. IV administration achieves maximum plasma concentrations within 1 min of infusion if administered as a bolus, 22 and when infused over 40 min at an antidepressant dose, its concentration progressively decreases after the end of the infusion. 23 Intramuscular administration peaks within 5 to 30 min, 24 sublingual within 45 min, and oral within 20 to 120 min. 24 Bioavailability of ketamine varies widely with non-IV formulations, ranging from approximately 90% for intramuscular injection 24 to 50% for intranasal, 25 29% for sublingual, 26 and 17% for oral ketamine. 24
Ketamine disperses rapidly into perfused tissues including the brain. Racemic (R, S)-ketamine results in an (S) to (R) ratio of 0.84 at antidepressant dosing. 27 Ketamine undergoes extensive metabolism to norketamine by cytochrome P450 (CYP) enzymes CYP3A4, CYP2C19, CYP2B6, CYP2A6, and CYP2D6, and demethylation to hydroxynorketamine by CYP2B6 and CYP3A4. 28 Moreover, there appears to be isomeric specificity, with CYP3A4 metabolizing (S)-ketamine more rapidly than (R)-ketamine. 28 Accordingly, oral (S)-ketamine has bioavailability less than 10% due to extensive first-pass hepatic metabolism. 29 Both (S)- and (R)-ketamine have plasma half-lives of approximately 15 min and elimination half-lives of 155 min. 30 Ketamine and its metabolites are predominantly eliminated in urine and bile. 31 Following single infusions, plasma levels of ketamine are undetectable within 24 hr of administration, but very low levels of ketamine metabolites are found up to 3 days postadministration. 27 Repeated infusions may decrease clearance of ketamine but not norketamine. 32
Is IV Ketamine Effective in Acute Treatment of MDD?
At least 7 meta-analyses synthesizing the results of placebo- and active-controlled RCTs have been published in the last 5 years. These studies consistently conclude that single infusions of IV ketamine have rapid antidepressant effects that peak within 24 hr and last 3 to 7 days, comprising Level 1 evidence for efficacy. 33 –39 A limitation of this evidence is the small set of included RCTs with small sample sizes in individual trials (i.e., 6 to 9 RCTs with a total of 103 to 368 participants), in addition to methodological issues such as crossover study designs and limited follow-up beyond 1 week. Most of the RCTs examined ketamine at a subanesthetic dose of 0.5 mg/kg infused over 40 min. Lower doses were ineffective in 2 RCTs 40,41 and in a meta-analysis. 42 In one of the RCTs, a higher dose (1 mg/kg; n = 20 patients) was similar in efficacy to the standard dose and was well tolerated. 41
Relapse after a single infusion usually occurs within 10 days, 37,39 but sustained response can be experienced in some patients for weeks. Two RCTs followed patients for up for 35 days after a single infusion. The first study (using a ketamine dose of 0.5 mg/kg) reported sustained clinical response in 21 of 47 (45%) patients at 7 days and 9 of 47 (19%) patients at 35 days postinfusion. 43 The second study (using doses of 0.5 mg/kg and 1.0 mg/kg) found similar rates of response in both dose groups, totaling 18 of 41 (44%) patients at 7 days and 13 of 41 (34%) patients at 30 days postinfusion. 44
Several open-label studies have examined repeat infusions, typically given twice or thrice weekly to a total of 4 to 6 sessions. In a large case series conducted in China, patients with TRD (n = 97; 77 with MDD and 20 with bipolar disorder) showed higher response and remission rates after 6 infusions (68% and 51%, respectively) than after the first infusion (14% and 9%, respectively). 45 This pattern was also seen in open-label studies of repeat infusions, 46 suggesting that some patients require several infusions for initial and/or full benefit. Repeated infusions may also extend time to relapse to an average of 18 to 19 days after infusions are stopped. 47,48
The RCT evidence is mixed. A placebo-controlled RCT randomized 67 patients with TRD to 1 of 4 conditions: twice-weekly IV ketamine infusions or placebo and thrice-weekly IV ketamine or placebo. 49 At day 15, both active ketamine conditions were significantly superior to placebo, with no differences between schedules; summing the conditions, IV ketamine compared to placebo had higher response rates (51% vs 9%, respectively) and remission rates (26% vs 3%). However, another RCT (n = 26) involving repeated infusions (2 infusions given twice a week for 3 weeks) found no difference in efficacy between the ketamine and placebo infusions; of note, this was in a sample of patients with chronic suicidal ideation and highly resistant TRD, with over 50% having failed electroconvulsive therapy (ECT). 50 In summary, there is only Level 3 evidence for efficacy of repeated ketamine infusions.
Are Other Formulations of Ketamine Effective in Acute Treatment of MDD?
The wide inter- and intraindividual variability in bioavailability for non-IV formulations of ketamine is problematic for dose comparisons. There are few RCTs of oral and non-IV formulations, and fewer have assessed bioavailability with plasma ketamine levels.
A systematic review of oral ketamine studies highlighted the limited evidence for safety and efficacy. 51 Only 3 small RCTs are published; all involved very different doses and populations, and 2 RCTs were from the same center. One study involved medication-free patients with MDD (n = 81) who were started concurrently on sertraline titrated to 150 mg/d while randomized to oral ketamine 50 mg/d (25 mg bid) or placebo for 6 weeks; the adjunctive ketamine group had greater improvement in depressive symptoms and response but not remission. 52 A second study assessed medication-free patients with MDD and comorbid chronic pain with mild depressive severity (n = 41) and compared oral ketamine 150 mg/d (50 mg tid) or diclofenac 150 mg/d (50 mg tid) for 6 weeks; the ketamine group showed greater improvement in depressive symptoms, response, and remission. 53 The third RCT studied medicated patients with TRD (n = 41) and randomized participants to add-on treatment with liquid ketamine 1 mg/kg or saline placebo given thrice weekly for 3 weeks; the add-on ketamine showed greater improvement in depressive symptoms, response, and remission. 54 In all the studies, oral ketamine was well tolerated.
Two RCTs investigated intranasal racemic ketamine. One placebo-controlled crossover RCT (n = 20) reported a significant antidepressant effect with a single dose of 50 mg. 55 However, an RCT using a dose of 100 mg in 10 repeated doses over 6 weeks was terminated because of poor tolerability experienced by the first 3 patients. 56 In contrast, a retrospective study of 17 patients treated clinically with up to 100 mg once or twice a week as maintenance treatment after 6 to 8 IV ketamine infusions found good tolerability and benefit in 13 patients (76%). 57 Differences in these reports likely reflect variability in treatment devices, intranasal absorption, and bioavailability of racemic ketamine. The only RCT with subcutaneous ketamine was in a small sample of geriatric patients. 58 One small crossover RCT (n = 15) compared IV, intramuscular, and subcutaneous routes of administration; there were no differences in antidepressant effect across the 3 conditions. 59
In summary, there are still very few RCTs of oral and other non-IV formulations of ketamine in patients with MDD. Given the small sample sizes, diverse populations, and different doses studied, the evidence for efficacy of oral ketamine was graded as Level 3 and other formulations as Level 4. There is also potential risk of misuse and diversion with these formulations. For these reasons, the use of non-IV formulations of ketamine should be limited to mood disorders specialists affiliated with tertiary or specialized centers, with patients informed about the risks and lack of evidence.
Is Ketamine Effective in Resolving Suicidal Ideation?
There is evidence that ketamine may have specific antisuicidal effects. Rapid reductions in suicidal ideation that appeared independent of overall depressive symptom response were described in pooled analyses of single-infusion ketamine trials 60 and in meta-analyses, 61,62 although there was significant heterogeneity of results. A randomized, midazolam-controlled crossover study found that suicidal ideation was significantly reduced after a single ketamine infusion and continued to decrease with 6 open-label infusions repeated thrice weekly over 2 weeks; suicidal ideation remained low after 4 additional open-label weekly maintenance infusions. 63 In these studies, suicidal ideation was measured using a single suicidality item on various clinician- and self-rated depression scales. Other studies have examined suicidal ideation as a primary outcome using validated scales such as the Scale for Suicide Ideation, with midazolam-controlled RCTs showing significant reductions in suicidal ideation on these measures. 64 –66 While encouraging, these results are considered preliminary because they are based on mostly small, single-infusion studies, and, as yet, there are no data on longer term effects of ketamine on suicidal ideation or behaviors.
How Effective Is Ketamine Compared to Electroconvulsive Therapy?
There are no published RCTs comparing the antidepressant effect of ketamine infusions to a standard course of ECT, but several trials are in progress. 67,68 In contrast, many RCTs have addressed the question of whether general anesthesia with ketamine, given alone or in combination with other anesthetics, can improve the response of patients with MDD during ECT. Although an initial meta-analysis suggested some positive effects of ketamine anesthesia, 69 4 subsequent meta-analyses (involving 10 to 18 RCTs, 612 to 1,035 participants) have not shown any significant effects either early or later in a course of ECT. 70 –73
Who Should Receive Ketamine?
To date, most studies of ketamine have involved adult patients with TRD, many of whom had failed numerous medication, neurostimulation, and other treatments. Given the variable definitions of TRD used in studies and the lack of a specific “patient profile” for response to IV ketamine, candidate patients should undergo a comprehensive assessment, 1 with special attention to comorbidity for psychiatric and other medical conditions, history of substance abuse/dependence, and detailed evaluation of adequacy of previous treatments. Generally, patients under consideration for ketamine should have exceeded minimum criteria for treatment resistance such as trials of at least 2 first-line antidepressants from different classes and at least 1 adjunctive medication. 7 Table 3 lists factors to consider during assessment for ketamine treatment. The evidence for ketamine use in pediatric and geriatric age groups is limited to Level 4 quality (small case series); hence, there is insufficient evidence for any recommendation in those age groups.
Factors to Consider for Ketamine Treatment.

Table 4 lists recommended assessments prior to initiation of ketamine treatment. Because of the potential to increase blood pressure (see side effects), ketamine should not be administered if a patient has serious risks for hypertension, such as increased intracranial or intraocular pressure. There are also relative contraindications to the use of ketamine, which include psychotic symptoms, poorly controlled hypertension, unstable medical conditions such as cardiovascular or respiratory disease, substance use/dependence, and pregnancy/breastfeeding.
Recommended Assessments and Monitoring for Ketamine Infusions.

a Do not initiate ketamine infusion if blood pressure > 140/90; stop infusion if blood pressure > 160/100.
Are There Drug Interactions with Ketamine?
Ketamine does not inhibit or induce CYP isoenzymes and is unlikely to cause clinically significant pharmacokinetic interactions when used in subanesthetic doses for the treatment of depression. The plasma levels of ketamine may be increased by moderate-to-strong inhibitors of CYP2B6, CYP2C9, and CYP3A4 inhibitors (e.g., clarithromycin, ketoconazole, among others) and decreased by CYP2C9 inducers. 74
Patients can generally continue antidepressant medications during ketamine treatment. Case reports (see example) 75 have described concurrent use of monoamine oxidase inhibitors and ketamine without incident, but because of limited experience and theoretical risk of hypertensive crisis, the combination should only be used with caution.
Given the putative mechanism of ketamine, medications that act on the glutaminergic or GABAergic system have the potential to affect the antidepressant response. For example, small studies suggest that benzodiazepines, which modulate GABA receptors, may attenuate or delay the antidepressant effect of IV ketamine. 76 Hence, some investigators have recommended holding or discontinuing any GABAergic or glutaminergic agents prior to a ketamine infusion. 76
Ketamine may also increase the effects of other sedatives including barbiturates, opioids, anesthetics, and alcohol. Ketamine should be used with caution when patients are taking other drugs that are NMDA antagonists, for example, ketamine may enhance the adverse/toxic effects of dextromethorphan and memantine. 77
What Settings and Personnel Are Required to Administer Ketamine Infusions?
There are no widely accepted facility standards or staffing credentials for ketamine administration as an antidepressant. Most ketamine studies have been conducted in academic medical center hospitals or outpatient clinics with ketamine administered by medical practitioners who are qualified for advanced cardiac life support (ACLS). In view of these limitations, the following recommendations are based on balancing the potential harms associated with low-dose ketamine infusions and the practicality of allowing reasonable access to this treatment. These recommendations are similar to those of other consensus groups. 5,12
Ketamine infusions should be administered in facilities that have secure storage for controlled medications, physiological monitoring requirements, and available medications to manage adverse events. Clinicians who administer ketamine infusions should have medical training and be licensed to administer controlled substances. They, or a member of the infusion team, should be qualified to manage neurologic/psychiatric adverse events, including agitation and psychotomimetic symptoms, and cardiorespiratory adverse events, including hypertension and airway management. In most settings in Canada, the administering clinician will be a physician or nurse. Generally, an on-site anesthesiologist is not required for subanesthetic ketamine dosing for low-risk patients. Rapid access to ACLS should be available in the event of severe or persistent cardiovascular or respiratory dysfunction. In nonhospital facilities, the administering clinician or member of the infusion team should be qualified for resuscitation and airway support, such as an anesthesiologist or ACLS-qualified physician or nurse.
What Are the Acute Side Effects of Ketamine?
The most clinically relevant side effects of antidepressant doses of IV ketamine include dissociative symptoms, increased blood pressure and heart rate, and drowsiness. 38 Other transient, mild side effects of IV ketamine systematically recorded in an RCT (n = 47) included blurred vision (43%), headache (32%), nausea and vomiting (34%), dry mouth (26%), poor coordination (26%), poor concentration (26%), and restlessness (21%). 43
Mild-to-moderate dissociative symptoms such as dream-like feelings, dizziness, confusion, unusual thoughts, and perceptual illusions are experienced by 50% to 80% of patients, peak within 30 to 60 min, and usually resolve within an hour of infusion; more severe psychotomimetic effects are rare. 78,79 Studies using validated rating scales of dissociative and psychotic symptoms have shown only small, transient increases in symptoms that usually do not require clinical intervention other than occasionally pausing or stopping the infusion.
Increased blood pressure and heart rate are also usually mild and transient, with no serious or persistent cardiovascular events reported. 80 A relatively large case series (n = 66 participants, 684 total infusions) reported that blood pressure peaked (mean systolic increase of 3.3 mmHg, diastolic 3.2 mmHg) at 30 min after the start of infusion; only 9% of infusions showed clinically significant increases in blood pressure (defined as systolic > 30 mmHg, diastolic > 15 mmHg), and none required intervention. 81 In contrast, an earlier report on pooled data from 3 studies (n = 84 participants, 205 total infusions) described larger mean increases in blood pressure (systolic 19.6 mmHg, diastolic 13.4 mmHg), but the mean peak blood pressures were not of clinical concern (systolic 141.9 mmHg, diastolic 86.4 mmHg) and blood pressures returned to baseline at 60 to 70 min after infusion. 78 However, 25 participants (30%) experienced a clinically significant increase in blood pressure, and 12 participants (14%) received antihypertensive medications; 2 participants (2%) did not respond to medication, and the infusions were stopped, after which vital signs returned to baseline. It is unclear what accounts for the discrepant findings in these studies. Clinical experience aligns with the lower rate of hypertensive events. Regardless, blood pressure should be monitored during ketamine infusions because some patients may experience clinically significant hypertension that requires intervention.
What Are Monitoring Requirements for Ketamine Infusions?
Compared to anesthetic doses, the antidepressant doses of ketamine carry lower risk for marked cardiovascular changes or respiratory depression. The physiological changes occurring during infusion also return to baseline within 40 to 80 min postinfusion, consistent with the fall in plasma ketamine levels. Hence, monitoring during IV ketamine administration in clinical studies has focused primarily on the transient cardiovascular and behavioral effects. 80,81
Table 4 lists recommendations for monitoring. At minimum, physiological measures should be assessed at baseline, midway through the infusion, at the end of infusion, and at 1 hr postinfusion. Ketamine infusion should not be initiated if baseline blood pressure is greater than 140/90. If blood pressure rises above 160/100, which is the demarcation between Stage 1 and Stage 2 hypertension, 82 the infusion should be paused and may be resumed when blood pressure returns below that upper threshold. If hypertension cannot be managed conservatively, antihypertensive medications can be used.
Patients should be informed about potential dissociative experiences as they may subjectively be experienced as positive or negative. Severity of symptoms can be assessed using a validated symptom rating scale such as a modified Brief Psychiatric Rating Scale 83 or the Clinician-Administered Dissociative States Scale. 84 Distressing or severe symptoms can usually be managed with clinician support, pausing or stopping the infusion, or judicious use of atypical antipsychotics (instead of benzodiazepines).
Monitoring should continue for at least 1 hr following infusion, and patients may be considered for discharge once vital signs and mental state have returned to baseline. Note that the FDA has required that Spravato® be used within a Risk Evaluation and Mitigation Strategy 85 that includes a monitored distribution system of pharmacies and clinics as well as a recommended postdose monitoring period of 2 hr. 86
IV ketamine may cause significant impairment in driving performance even at low doses and blood levels, but this effect appears to diminish at 2 hr postinfusion. 87 A study of Spravato® found that driving performance was not impaired 8 hr following a dose 88 ; hence, the Health Canada recommendation is not to drive until the following day after a restful sleep. 86 A similar recommendation for IV ketamine seems prudent.
How Do You Maintain Response Following Acute Ketamine Infusions?
There is little information on maintenance strategies following ketamine infusions. Most RCTs show that after a single infusion, many patients lose the antidepressant effect within 1 week, although 45% of patients still have response at 7 days and 19% to 24% at 30 days or longer. 43,44 In repeated infusion studies, relapse may be delayed for longer periods. 47,48 In these ketamine studies, patients were either medication-free or remained on medications that had been ineffective. Although there is no supporting evidence, it seems reasonable to start the patient on a previously untried antidepressant (or adjunctive agent) when initiating an acute course of ketamine infusions, or shortly after, in the hope that response can be maintained on the medication alone. This is an approach used in the Spravato® trials.
There is limited evidence for racemic ketamine infusions for relapse prevention. Only small, open-label studies have examined maintenance schedules after a course of repeated ketamine infusions. Two studies (n = 12, n = 23) treated responders after 6 repeated infusions (thrice weekly for 2 weeks) with weekly infusions for 4 weeks, 46,89 and a third study (n = 23) administered infusions every 2 weeks for 7 weeks. 90 In these open-label studies, most patients continued to have response over the follow-up period. Retrospective case series have reported benefit from ongoing maintenance ketamine given at varying intervals for longer durations. 91,92 Acute dissociative effects have been reported to be more mild or noted to dissipate with repeated infusions during maintenance. 46 Anecdotal reports suggest that some patients may be able to transition to intranasal or sublingual formulations of racemic ketamine for maintenance after ketamine infusions. 57
In summary, because of small sample sizes and short follow-up periods, the evidence for maintenance racemic ketamine is graded as Level 4. Although Spravato® was superior to placebo in a relapse prevention trial, 93 we cannot conclude the same efficacy for racemic ketamine. Until there is higher quality evidence for relapse prevention, maintenance ketamine should be considered only when other evidence-based treatment options have been exhausted and the potential benefit is felt to outweigh potential risks of not offering maintenance ketamine.
What Are the Potential Long-term Risks Associated with Ketamine?
There are no long-term studies of ketamine treatment for depression; hence, many of the long-term risks derive from studies of chronic ketamine misuse and abuse/dependence. While true risk of ketamine abuse when used as an antidepressant is unknown, it has been ranked similarly to alcohol, benzodiazepines, and amphetamines in regard to potential risk of overall harm. 94 Due to potential for ketamine abuse, 95 clinicians prescribing ketamine should carefully consider patient selection for risks of misuse and diversion. If oral or intranasal ketamine is prescribed, it should be dispensed in limited quantities with close monitoring to prevent misuse. 96
Chronic abuse of high-dose ketamine has been linked to cognitive dysfunction 97 and “ketamine bladder,” a syndrome consisting of cystitis (painful bladder, frequency, incontinence, and hematuria) with upper tract obstruction and papillary necrosis. 98 However, single and repeated infusions of ketamine as an antidepressant have not been associated with changes in performance on neurocognitive tasks, 99,100 and there are no reports of excessive urinary symptoms in placebo-controlled trials or maintenance studies of ketamine for TRD. 47,91 Nevertheless, these remain possible long-term side effects of ketamine.
Does Esketamine Differ from Racemic Ketamine in Efficacy or Tolerability?
One study compared IV esketamine (0.25 mg/kg) to IV racemic ketamine (0.5 mg/kg) in patients with TRD (n = 63) using a randomized, single infusion noninferiority design; no placebo condition was included. 8 This study reported noninferiority between conditions, with similar rates of symptom improvement, response, and remission at 24 hr and 3 days postinfusion. The IV esketamine-treated group had a lower response rate compared to the IV ketamine-treated group at 7 days postinfusion (44% vs. 62%), but the difference was not statistically significant. There were no differences in adverse events between conditions.
Spravato® has Level 1 evidence for efficacy based on large-sample RCTs; however, there have been no direct comparisons to racemic ketamine treatment. Nevertheless, a meta-analysis (n = 4 trials, 708 participants) reported significant differences between Spravato® and placebo as early as 2 hr postdose and continuing through 8 to 28 days of treatment. 9 Although early response rates appear lower than IV ketamine trials, major differences in study designs preclude any comparison of results. For example, all the trials with Spravato® involved patients with TRD who initiated a previously untried antidepressant at the same time as study drug, whereas the IV ketamine trials involved patients who were medication-free or had ketamine added on to ineffective treatments. Moreover, the adverse events associated with Spravato® were similar to those reported in IV ketamine studies. 10,86
Conclusions
There is now Level 1 evidence for efficacy of single-infusion IV ketamine as an antidepressant. The antisuicidal effects of ketamine make it a particularly promising novel treatment. Side effects of ketamine treatment are generally mild, short-lasting, and infrequently require clinical intervention. Unfortunately, however, there is still only Level 3 evidence for efficacy of repeated infusions and Level 4 evidence for relapse prevention. There are also feasibility challenges for access to monitored infusions. For these reasons, we consider IV ketamine to be a third-line recommendation for adults with TRD.
Ketamine is not approved by Health Canada or other regulatory agencies for any depression indication. There is significant interest in off-label use of ketamine in psychiatry, and clinicians should weigh potential benefits against unknown long-term side effects and the risk of diversion and misuse. The latter is of particular concern for oral and other non-IV formulations of ketamine, for which there is only Level 3 or 4 evidence of efficacy and unanswered questions about dosing, given the wide variations in bioavailability. With sensible guidance for monitoring and supervising ketamine use, overly restrictive regulations that reduce access for appropriate patients may not be necessary. However, the personnel and facility costs for monitored infusions in the Canadian healthcare system will likely limit ketamine use to tertiary or specialized centers treating patients who have exhausted other more accessible, evidence-based treatment options.
In contrast to racemic ketamine, Spravato® has been approved by Health Canada and other regulatory agencies. While the scope of this task force was limited to racemic ketamine, we note that there are no available data comparing Spravato® with IV ketamine. Hence, in settings where both medications are available, it is unclear where each fit in a treatment algorithm for TRD. Prescribing choices will also be influenced by accessibility and cost. Whereas Spravato® has much higher acquisition cost, IV ketamine has higher facility and personnel costs.
Finally, we recognize that these recommendations for administering and monitoring ketamine are based mainly on clinical research protocols that have yet to be rigorously evaluated in real-world settings and may change with more evidence. We emphasize the need for continued education, shared decision-making with patients, responsible practice, and gathering of systematic, long-term data to inform the use of ketamine in clinical practice.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest: Jennifer Swainson has received honoraria from Bausch, Janssen, Lundbeck, Otsuka, Sunovion, and Eisai. Alexander McGirr has no disclosures. Pierre Blier has received honoraria or research funds from Allergan, Bristol Myers Squibb, Janssen, Lundbeck, Pierre Fabre Médicaments, Pfizer, Sunovion, CIHR, Ontario Brain Institute, and Otsuka. Elisa Brietzke has received honoraria or research funds from Queen’s University School of Medicine, SEAMO, CNPq-Universal Grant, Otsuka, Daiichi-Sankyo, and FAPESP. Nisha Ravindran has received grants from Janssen. Serge Beaulieu has received honoraria or research funds from Pfizer, Otsuka, Lundbeck, Sunovion, Allergan, Janssen-Ortho, and Takeda. Benicio N. Frey has received research funds from Pfizer. Sidney H. Kennedy has received honoraria or research funds from Abbott, Alkermes, Allergan, BMS, Brain Canada, CIHR, Janssen, Lundbeck, Lundbeck Institute, Ontario Brain Institute, Ontario Research Fund, Otsuka, Pfizer, Servier, Sunovion, Xian-Janssen, and Field Trip Health. Roger S. McIntyre has received honoraria or research funds from Lundbeck, Janssen, Shire, Purdue, Pfizer, Otsuka, Allergan, Takeda, Neurocrine, Sunovion, Minerva, Stanley Medical Research Institute, CIHR, GACD, and National Natural Science Foundation of China. Roumen V. Milev has received honoraria or research funds from Ontario Brain Institute, Allergan, Janssen, Lallemand, Kye, Lundbeck, Nubiyota, Otsuka, Pfizer, and Sunovion. Sagar V. Parikh has received honoraria or research funds from Assurex, Takeda, Janssen, Mensante, Aifred, Sage. Ayal Schaffer has received honoraria from Allergan, Janssen, Lundbeck, Otsuka, and Sunovion. Valerie H. Taylor has received honoraria or research funds from DiaMentis, Janssen, Lundbeck, Otsuka, Purdue, Shire, and Takeda. Valérie Tourjman has no disclosures. Michael van Ameringen has received honoraria or research funds from Allergan, Almatica, Brainsway, Lundbeck, Myriad Neuroscience, Otsuka, Purdue, Janssen, Canadian Foundation for Innovation, Hamilton Academic Health Sciences Organization, and Pfizer. Lakshmi N. Yatham has received honoraria or research grants from Allergan, CANMAT, Lundbeck Institute, Otsuka, DSP, and Sanofi. Arun V. Ravindran has received grants from Janssen. Raymond W. Lam has received honoraria or research funds from Allergan, Asia-Pacific Economic Cooperation, BC Leading Edge Foundation, CIHR, CANMAT, Canadian Psychiatric Association, Hansoh, Healthy Minds Canada, Janssen, Lundbeck, Lundbeck Institute, MITACS, Ontario Brain Institute, Otsuka, Pfizer, St. Jude Medical, University Health Network Foundation, and VGH-UBCH Foundation. Other authors have no disclosures.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
