Abstract
Objective:
Bright light therapy is increasingly recommended (alone or in combination with antidepressant medication) to treat symptoms of nonseasonal major depressive disorder (MDD). However, little is known about its impacts on quality of life (QoL), a holistic, patient-valued outcome.
Methods:
This study utilizes secondary outcome data from an 8-week randomized, controlled, double blind trial comparing light monotherapy (n = 32), fluoxetine monotherapy (n = 30), and the combination of these (n = 27) to placebo (n = 30). QoL was assessed using the Quality of Life Enjoyment and Satisfaction Questionnaire Short Form (Q-LES-Q-SF). Treatment-related differences in QoL improvements were assessed using a repeated measures analysis of variance. The influence of potential predictors of QoL (demographic variables and change in depressive symptoms) were investigated via hierarchical linear regression.
Results:
Q-LES-Q-SF scores significantly improved across all treatment conditions; however, no significant differences were observed between treatment arms. QoL remained poor relative to community norms by the end of the trial period: Across conditions, 70.6% of participants had significantly impaired QoL at the 8-week assessment. Reduction in depressive scores was a significant predictor of improved QoL, with the final model accounting for 54% of variance in QoL change scores.
Conclusion:
The findings of this study emphasize that improvement in QoL and reduction in depressive symptoms in MDD, while related, cannot be taken to be synonymous. Adjunctive therapies may be required to address unmet QoL needs in patients with MDD receiving antidepressant or light therapies. Further research is required to explore additional predictors of QoL in order to better refine treatments for MDD.
Introduction
Major depressive disorder (MDD) is a major public health challenge in Canada, with an estimated lifetime prevalence of 11.3%. 1 Depressive disorders have been ranked as the second leading cause of disease burden 2 and are associated with significant impacts to quality of life (QoL). 3
Bright light therapy has support as a first-line treatment for seasonal affective disorder 4,5 and holds promise as a treatment for nonseasonal MDD: It is safe, well-tolerated, and inexpensive. 6 There is emerging evidence to support the safety, tolerability, and efficacy of bright light therapy in treating depressive symptoms in nonseasonal MDD, both alone 4,7 and adjunctive to pharmacological treatment. 8 –11 The Canadian Network for Mood and Anxiety Treatments guidelines support light therapy as second-line monotherapy for mild-to-moderate nonseasonal MDD or as adjunctive treatment for more severe cases. 12
Despite significant impacts of depression on QoL, 13 few clinical trials of light therapy in MDD have reported the impact of this treatment on this outcome. Defined as “an individual’s perception of their position in life in the context of the culture and value systems in which they live and in relation to their goals, expectations, standards and concerns” (p.1405), 14 QoL includes physical, psychological, social, and occupational components. Such diverse outcomes are valued by patients alongside or above symptom reduction 15,16 and are increasingly recommended as primary outcome measures in clinical trials for mood disorders. 17 Although light therapy has been shown to improve depressive symptoms in nonseasonal MDD, the impacts of an intervention on QoL cannot be presumed to be equivalent to reduction in symptom severity. For example, a large-scale prospective randomized controlled trial (RCT) of antidepressant treatment showed that of participants who could be classified as “remitted,” only 68% reported within normal QoL scores, 18 suggesting that improving depressive symptoms alone is not sufficient to improve QoL. A recent meta-analysis showed both pharmacotherapy and psychotherapy improved depressive symptom severity to a greater extent than functioning or QoL. 19 It is not yet known whether similar findings may be observed in light therapy, contributing to a gap in understanding of effective, holistic treatments in MDD.
To address this question, we analyzed QoL outcome data from an RCT investigating the efficacy of bright light therapy, an antidepressant (fluoxetine), and combination therapy for outpatients with MDD. Primary outcome analyses found that bright light therapy (as monotherapy and in combination with fluoxetine) was superior to placebo in reducing depressive symptoms. 10 The aims of this study were (i) to compare the impact of light therapy, fluoxetine, and the combination on QoL among patients with MDD and (ii) to explore predictors of change in QoL under treatment. Based on the primary outcome analysis, we hypothesized that combination and light monotherapy would be superior to placebo in improving QoL. Analyses of potential predictors of change in QoL (demographics and change in depressive symptoms) are exploratory.
Methods
Design
The Light, Ion, and Fluoxetine Efficacy in Depression trial (ClinicalTrials.gov identifier: NCT00958204) was an RCT conducted between October 7, 2009, and March 11, 2014. The majority of participants were recruited from 3 outpatient clinics in Canada (1 in Vancouver, British Columbia, and 2 in Toronto, Ontario). Due to slow recruitment, the study was halted in 2 other sites. This study was approved by the University of British Columbia Clinical Ethics Review Board and the University of Toronto Health Sciences Research Ethics Board.
Participants
Participants were recruited by referral from psychiatrists at the UBC Mood Disorders Centre, as well as a combination of web, newspaper, and radio advertisements. Participants were required to be aged 19 to 60, have a DSM-IV-TR diagnosis of MDD as assessed by a psychiatrist and confirmed via the Mini International Neuropsychiatric Interview, 20 and Hamilton Depression Rating Scale (HAM-D) 21 score >20 at screening and baseline. Exclusion criteria were seasonal pattern of depressive episodes, serious suicide risk as judged by the clinician, substance abuse or dependence in the past year, comorbid psychotic, bipolar, obsessive compulsive, post-traumatic or organic mental disorders, and primary generalized anxiety disorder or panic disorder. Patients were also excluded on the basis of unstable medical illness, retinal disease, pregnancy or breastfeeding, treatment resistance during the current episode, and previous use of light therapy or fluoxetine. Participants were required to not be using other concurrent treatments for depression, including psychotherapy.
Procedure
Individuals who provided informed consent were monitored for 1 week following the screening visit to eliminate spontaneous responders (defined as
Interventions
Light therapy
The active treatment was daily exposure to a fluorescent light box (Carex Day-Light Classic, emitting 4,000-k white light at 10,000 lux at 35.6 cm from screen to cornea, with a UV filter). Device specifications are described in full elsewhere. 10 Participants were instructed to use the light box for 30 minutes per day, within 15 minutes of awakening.
Placebo treatment consisted of daily use of a deactivated negative ion generator (SphereOne Inc.) modified to emit an audible hum. Participants were given the same instructions for use as per the active light box. This sham treatment is considered credible for clinical trials of light therapy, 22 and in this study, expectations for effectiveness of treatment did not differ across conditions. 10
Medication
The active medication was a fixed 20 mg dose of fluoxetine taken daily
Assessment/Measures/Outcomes
QoL was assessed with the Quality of Life Enjoyment and Satisfaction Questionnaire Short Form (Q-LES-Q-SF), 23 a 14-item measure of self-reported satisfaction across various life domains (including physical health, mood, sexual drive and interest, ability to function in everyday life, ability to get around physically, living/housing situation, economic status, social relationships, work performance, leisure activities, household duties, and overall sense of well-being). Ratings are on a 5-point Likert scale, with higher scores representing better QoL. Total Q-LES-Q-SF scores are calculated as a percentage of the maximum possible score of 70. The Q-LES-Q was developed and validated for use with depressed outpatients although no condition-specific content was included to reduce measurement redundancy with symptom scales. The Q-LES-Q long and short forms are some of the most frequently used QoL assessments in psychiatry research, 24 and both have been shown to have appropriate reliability, validity, and responsiveness to change. 23,24 QoL outcome data were collected at baseline, after 4 weeks of treatment and on termination of the 8-week treatment period.
Depression symptoms were assessed using the Montgomery-Asberg Depression Rating Scale (MADRS), 25 evaluated using a structured interview guide 26 by telephone with a rater blinded to treatment condition. Depressive symptoms were assessed at baseline, and Weeks 1, 2, 4, 6, and 8 of treatment.
Statistical Analysis
Randomized patients were included in the analysis based on modified intent to treat: All patients who had baseline Q-LES-Q-SF data were included in the analysis. 27 Postbaseline missing data were imputed using last observation carried forward, which produces conservative estimates of treatment effects in populations which may spontaneously improve, such as depression. 28
Equivalence between intervention groups at baseline was established using univariate tests. Baseline and endpoint Q-LES-Q-SF scores were compared against previously defined cut-offs to evaluate the percentage of participants with normative QoL scores (defined as within 1 standard deviation of the community norm of 78.3%, i.e.,
A repeated-measures analysis of variance (ANOVA) was used to compare the condition arms in terms of their effect on Q-LES-Q scores across baseline, 4-, and 8-week timepoints. Post hoc tests were used to test the differences between timepoints and conditions if significant main or interaction effects were observed. Effect sizes were estimated for overall Q-LES-Q-SF change scores (baseline to endpoint) using Cohen’s d.
To evaluate predictors of change in QoL (baseline to 8-week Q-LES-Q-SF change scores), a series of hierarchical linear regressions were performed. The first model controlled for baseline Q-LES-Q-SF scores. At Step 2, general demographic variables (age, gender) were added. At Step 3, treatment group was added to the model at Step 3, and at Step 4, the impact of depression change scores from baseline to treatment endpoint were tested by (with negative scores indicating amelioration in depressive symptoms).
Significance was set at α = 0.05 for all tests. All analyses were performed using IBM SPSS Statistics for Windows, version 26.0 (IBM).
Results
Sample
A total of 131 eligible participants were recruited, and 122 were randomized. A Consolidated Standards of Reporting Trials (CONSORT) recruitment and randomization flowchart is provided in the primary outcome paper. 10 Three cases were excluded from the present analysis for lacking baseline Q-LES-Q-SF data. The final included cases were as follows: light monotherapy (n = 32), fluoxetine monotherapy (n = 30), combination light therapy and fluoxetine (n = 27), and placebo (n=30). Prior to imputing missing postbaseline data with last observation carried forward, rates of missing data were reviewed. Rates of missing Q-LES-Q-SF data at Weeks 4 and 8 were 16.8% (n = 20) and 16% (n = 19), respectively. Little’s Missing Completely at Random Test of Weeks 4 and 8 Q-LES-Q data were nonsignificant, χ2(2) = 2.46, P = 0.29, indicating there was no evidence to suggest that data were not missing completely at random. Table 1 describes the demographics and baseline scores characteristics of the final sample (n = 119). There were no significant differences between groups at baseline on any demographic or clinical characteristics.
Baseline Demographic Characteristics, Q-LES-Q-SF, and MADRS Scores.
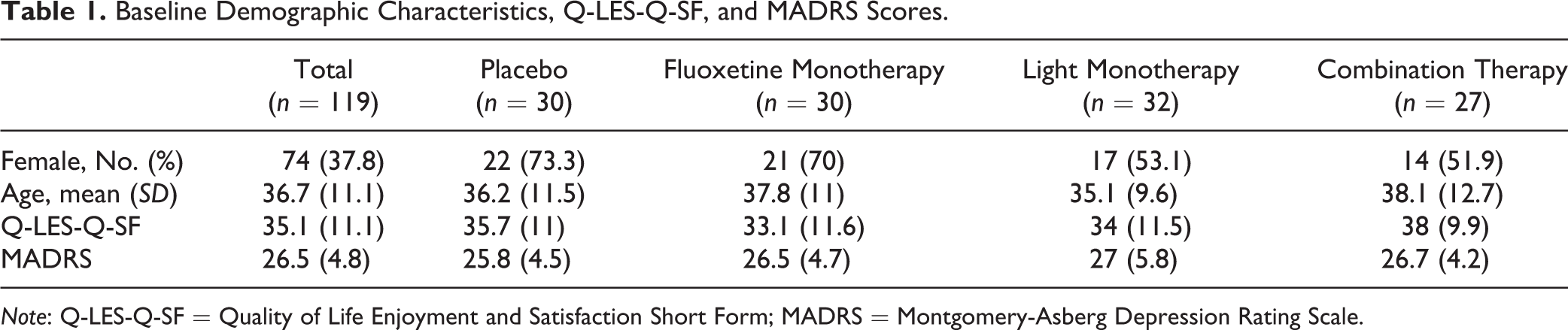
Note: Q-LES-Q-SF = Quality of Life Enjoyment and Satisfaction Short Form; MADRS = Montgomery-Asberg Depression Rating Scale.
The one-way ANOVA for baseline Q-LES-Q-SF scores showed no differences between patients randomized to each condition F(3, 115) = 1.09, P = 0.36. Mean Q-LES-Q-SF scores (collapsed across treatment groups) at baseline were significantly lower than the community norm: t(646) = 37.8, P < 0.001. No participants had QoL within normative standards at baseline, 1.7% had moderately impaired QoL, and 98.3% had severely impaired QoL.
Change in QoL Following Treatment
Mean Q-LES-Q-SF scores collapsed across treatment groups at 8 weeks (M = 49.6, SD = 18.2) remained significantly poor compared to community norms, t(646) = 22, P < 0.001. The proportion of participants across each intervention condition with normative, moderately impaired, and severely impaired QoL at the 8-week endpoint are represented in Figure 1. By the 8-week endpoint, 21% of participants had achieved QoL within a normative range, 8.4% had moderately impaired QoL, and 70.6% remained within the severely impaired range. Fisher’s exact test showed no significant differences in the proportion of normative versus impaired QoL at 8 weeks (P = 0.11). Of the participants who were classified as achieving remission according to depression ratings (MADRS endpoint score
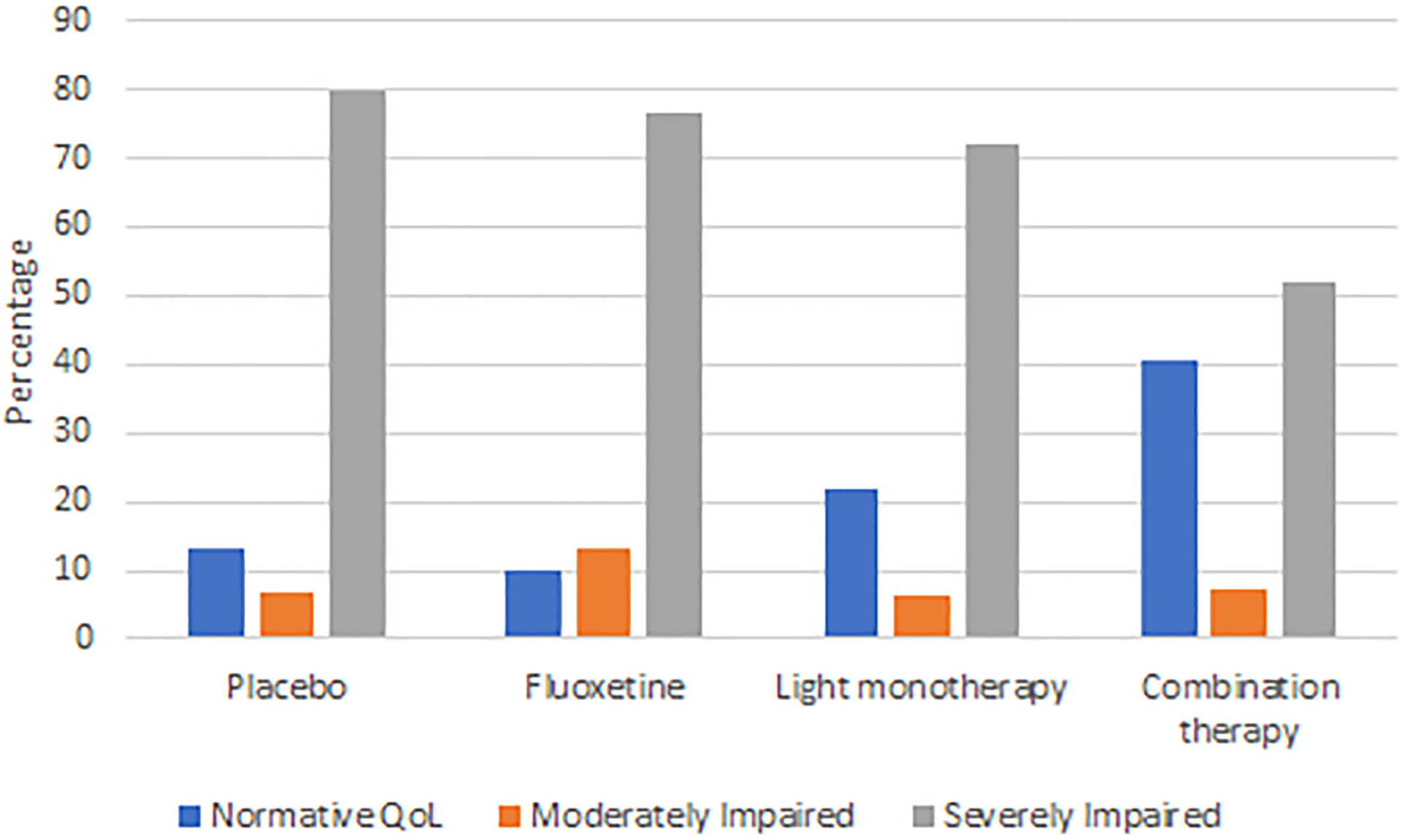
Percentage of participants attaining normative, moderately impaired, or severely impaired QoL at 8 weeks.
A repeated measures ANOVA was performed to evaluate differences in change in Q-LES-Q-SF scores between treatment conditions. Levene’s test for equality of error variances was not significant across all timepoints. The influence of potential outliers was evaluated by comparing the trimmed mean and mean value of Q-LES-Q-SF scores at each timepoint. As these did not substantially differ, these cases were retained for the final analysis.
Results of the repeated measures ANOVA showed that Q-LES-Q-SF scores significantly improved over time, F(1.5, 181.6) = 63, P < 0.001. As Mauchly’s test showed the assumption of sphericity was violated, χ2(2) = 0.69, P < 0.001, Huynh-Feldt corrected values are reported. Bonferroni post hoc tests comparing adjacent timepoints showed Q-LES-Q-SF scores significantly improved across all conditions between baseline and 4 weeks (P < 0.001) and between 4 weeks and 8 weeks (P < 0.001). Partial η2 effect sizes indicated a large effect of time on Q-LES-Q-SF scores (partial η 2 = 0.35).
There was no significant difference in rates of change to Q-LES-Q-SF scores over time between the treatment groups F(4.7, 181.6) = 1.27, P = 0.28, partial η 2 = 0.03. Figure 2 shows the trajectory of change in Q-LES-Q-SF scores across intervention conditions. The mean (SD) change in Q-LES-Q-SF scores from baseline to the 8-week endpoint was 19.2 (SD = 21.6) in those who received combination therapy, 15.8 (SD = 15.4) in those who received light monotherapy, 12.1 (SD = 16.9) in those who received fluoxetine monotherapy, and 11.3 (SD = 16.2) in those who received placebo. Cohen’s d effect sizes for the combination, light, and fluoxetine treatment arms relative to placebo were small to moderate (0.41, 0.28, and 0.05, respectively).
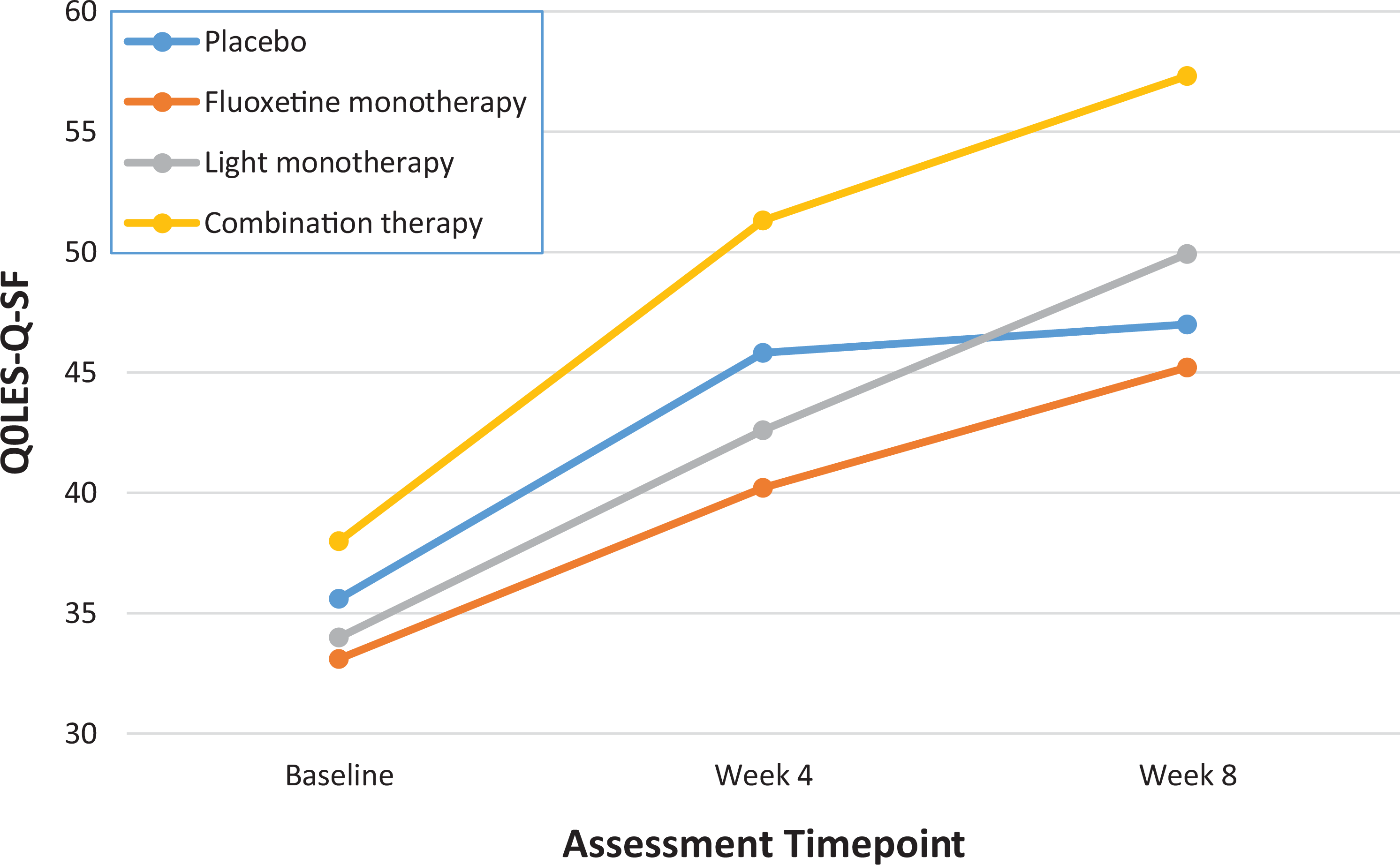
Scores on the Quality of Life Enjoyment and Satisfaction Questionnaire: Short Form (Q-LES-Q-SF) from baseline to 8-week endpoint across intervention conditions.
Predictors of Change in QoL
Potential predictors of change in QoL were investigated (Table 2). After controlling for baseline levels of QoL at Step 1, the addition of demographic predictors (age and gender) did not improve the fit of the model. In line with the results of the repeated measures ANOVA, inclusion of treatment group at Step 3 did not improve model fit. Fit was significantly improved with the addition of change in depressive symptoms (MADRS scores at baseline subtracted from the 8-week endpoint); reduction in depressive symptoms was significantly associated with improvements in QoL. The final model accounted for 54% of the variance of change in QoL.
Results of Hierarchical Linear Regressions Predicting Change in Q-LES-Q-SF Scores with Treatment.
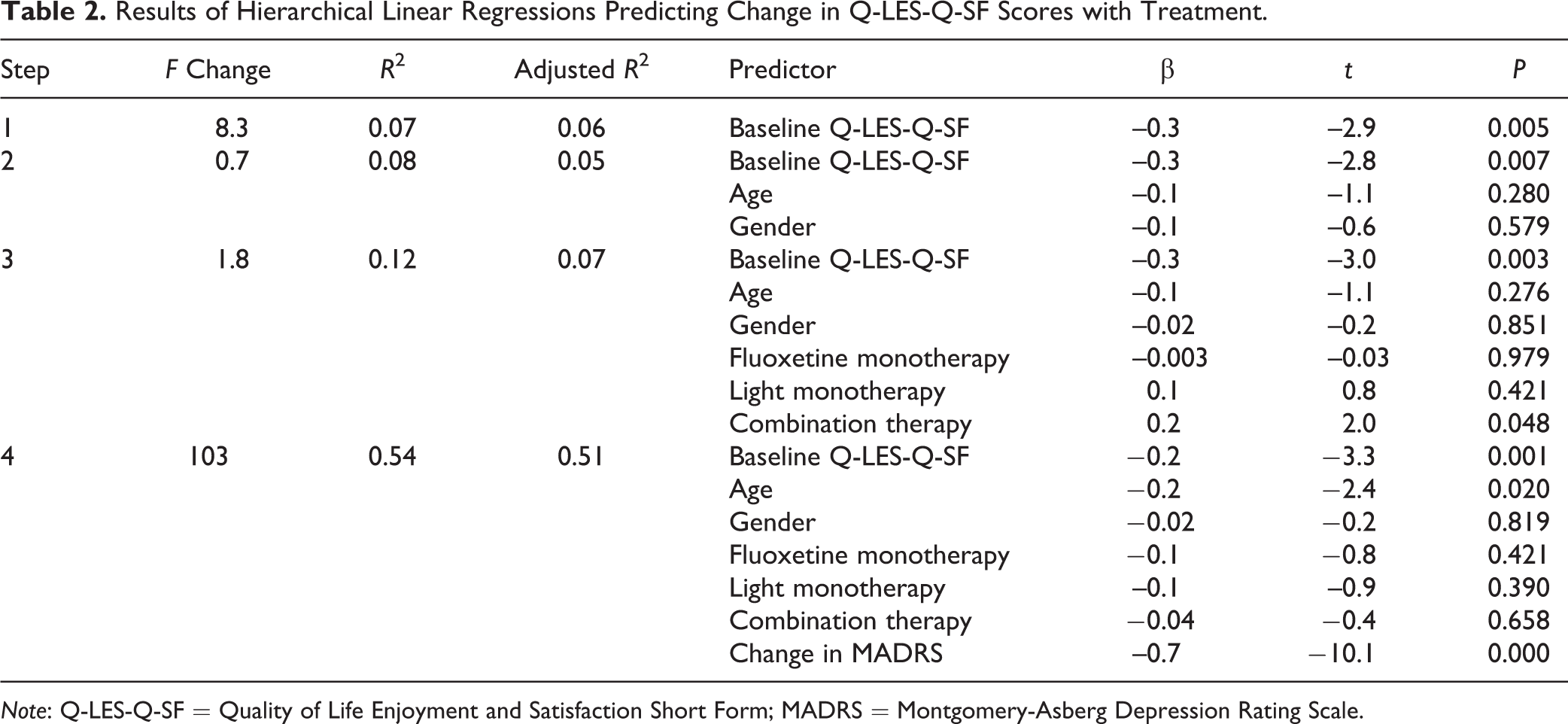
Note: Q-LES-Q-SF = Quality of Life Enjoyment and Satisfaction Short Form; MADRS = Montgomery-Asberg Depression Rating Scale.
Discussion
This study analyzed the impact of light therapy, fluoxetine, and the combination of these on QoL in nonseasonal MDD in an 8-week RCT. While bright light therapy has support for its efficacy in ameliorating depressive symptom, 11 its impact on QoL has not been evaluated. In this study, QoL as measured by the Q-LES-Q-SF significantly improved across all intervention conditions; however, there were no significant differences between treatment conditions regarding their efficacy for improving QoL. This contrasts with the findings of the primary outcome analysis, which found that bright light therapy, both as monotherapy and in combination with fluoxetine, was significantly superior to placebo at improving depression (MADRS scores). Furthermore, only 21% of participants achieved a QoL score within a normative range by the 8-week endpoint. In comparison, the analysis of primary outcomes showed that in the light therapy treatment arms (monotherapy and combination), 50% to 76% demonstrated response on the MADRS (defined as a 50% or greater reduction in depressive symptoms), and 44-59% of participants could be classified as achieving remission (defined as a MADRS score of 10 or less). Even when considering only the subgroup who achieved remission according to MADRS scores, QoL remained impaired for 46.6% of participants. While change in depressive symptoms was found to be a significant predictor of improvement in QoL, the final model accounted for only 54% in the variance in Q-LES-Q-SF change scores. Taken together, this study’s findings add to a body of literature emphasizing that reduction in depressive symptoms and improvement in QoL, while related, cannot be presumed to be synonymous. 13
Three methodological explanations may account for the finding that the intervention arms differed in efficacy for improving depressive symptoms, but not QoL. First, the impact of expectations on subjective outcome measures relative to clinician rated outcomes should be considered. In this study, expectations for treatment efficacy were similar across treatment conditions. 10 QoL, as a subjective outcome measure, may be more susceptible to impacts of outcome expectancy than a clinician-rated measure of depression (i.e., MADRS). Placebo response may therefore be higher when investigating QoL improvements. 31 Second, it has been previously suggested that QoL impacts may be slower to develop than symptom change in the treatment of mood disorders: a trial of olanzapine in bipolar disorder showed depressive symptoms improved during the 3-week active treatment period, but QoL improvements in the domains of social functioning and general health emerged only during the 49-week open label extension. 32 Potentially, with a longer period of observation, treatment-specific effects on QoL may have emerged—future studies of a longer duration are necessary to determine the full impact of light therapy on QoL. Finally, although significant differences between treatment arms were not observed, effect sizes for change in QoL relative to placebo were promising for both the combination (Cohen’s d = 0.41) and light monotherapy arms (d = 0.28). These effect sizes are similar to effects on QoL reported in meta-analyses of pharmacotherapy (Hedge’s g = 0.31) and psychotherapy (g = 0.35) in depressive disorders. 19 The target sample size of 54 patients per condition arm (based on an a priori power analysis using end point change on the primary outcome, MADRS) 10 was not achieved, and as such, this study was not powered to detect significantly significant differences between intervention arms. Further large-scale studies of light therapy in MDD will be important for drawing stronger conclusions about the impact of this treatment on QoL outcomes.
The proportion of participants with significantly impaired QoL at the endpoint of this study, despite promising findings for change in depressive symptom, remission rates, and effect sizes for QoL relative to placebo for the light monotherapy and combination therapy arms, warrants further attention. Q-LES-Q-SF scores at the 8-week endpoint were approximately 50% of the potential maximum score and significantly lower than a general population sample, 30 suggesting QoL remains an unmet need in patients receiving light therapy for MDD. The finding that the “success” of light therapy in MDD differs depending on the outcomes considered accords with concerns about the limitations of solely relying on depressive measures as an indicator of treatment efficacy. 17 Future studies of light therapy in nonseasonal MDD should continue to evaluate QoL self-report alongside depressive symptoms in order to holistically appraise the impact of treatment. Clinicians working with individuals receiving light therapy for MDD should also be cognizant of whether the QoL-related treatment needs of these patients are being met: Potentially, such individuals may benefit from adjunctive treatments to address any persisting QoL deficits.
Unsurprisingly, reduction in depressive symptoms was a significant predictor of improvement in QoL. The final model examined in this study (including change in depressive symptoms) was able to explain just over half of the variance in QoL improvements, which aligns with other estimates of the variance in QoL scores accounted for by depressive symptoms. Symptom severity has been shown to account for only 48.1% of the variance in QoL in a sample receiving a community outpatient treatment for MDD. 3 In a longitudinal study in which QoL significantly improved in response to antidepressant treatment, change in depression scores accounted for 50% of the variance in improvement in QoL. 33 Further, a meta-analysis of the impact of psychotherapy showing that across studies, improvements in QoL were not fully explained by reduction in depressive symptom severity. 34 It is therefore important for trials of light therapy in nonseasonal MDD to explore additional predictors of change in QoL to better target both symptoms and subjective, patient-valued markers of recovery.
Several methodological limitations of the study need to be considered. First, the planned sample size was not achieved due to slow recruitment and expiration of funding, and therefore, the power to detect significant differences between conditions was limited. Small to moderate effect sizes were observed for change in QoL for the treatment arms relative to placebo (with the largest effect emerging for combination therapy), encouraging further investigation of the impacts of fluoxetine combined with light therapy on QoL in a larger sample. Second, as the Q-LES-Q-SF was used rather than the full 93-item version, we were unable to investigate the impacts of treatment across various life domains. Potentially, when viewed with more granularity, treatment-related differences may have emerged. Finally, as highlighted above, this study was limited in scope to investigating the impacts of light therapy during an 8-week period of active treatment.
In summary, although there is an emerging body of evidence to support the benefits of bright light therapy (both alone and in combination with antidepressant medication) for reducing symptoms of depression in nonseasonal MDD, its efficacy for improving QoL in this population requires further study. Although roughly half of the participants receiving light or combination therapy could be classified as achieving remission according to response on clinician-rated depression measures, 10 this analysis showed that the vast majority of participants still had significantly impaired QoL relative to the general population at the conclusion of the trial. This emphasizes the importance of assessing both clinical and patient-valued outcomes to best judge the impacts of a treatment. Unsurprisingly, reduction in depressive symptoms was a significant predictor of positive change in QoL; however, the final model accounted for little over half of the variance in this outcome. The findings of this study suggest that in patients receiving bright light therapy, antidepressant medication, or a combination of these strategies, adjunctive interventions to support QoL may be required. Further research is needed to illuminate predictors of change in QoL in MDD besides improvement in depressive symptoms, in order to better refine treatments.
Footnotes
Authors’ Note
E. Morton conducted the statistical analysis and drafted the manuscript. The original study concept and design is attributed to R. Lam, A. Levitt, R. Levitan, E. E. Michalak, A. Cheung, and L. Yatham. All authors provided critical revision of the manuscript for important intellectual content and contributed to acquisition of data. Funding was obtained by R. Lam, A. Levitt, A. Cheung. Administrative, technical, or material support was provided by R. Lam, A. Levitt, R. Levitan, E. E. Michalak, A. Cheung, R. Ramasubbu, E. Tam. Data are not publicly available in accordance with ethics approval given by the ethics board from the participating university. Interested investigators may submit inquiries to the corresponding author.
Acknowledgments
EM was supported by a postdoctoral award from the Marshall Scholars and Fellows Program in Mental Health, Institute of Mental Health, University of British Columbia.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Rajamannar Ramasubbu has received research grant from Alberta Innovates and Health Solutions (AIHS), and investigator-initiated grants from Astra Zeneca and Pfizer. Has received honoraria and speakers fees from Astra Zeneca and served in advisory boards for Astra Zeneca, Lundbeck, Janssen and Otsuka. Lakshmi Yatham has received research support from or served as a consultant or speaker for Alkermes, Allergan, CANMAT, CIHR, Dainippon Sumitomo, Forest, Janssen, Lundbeck, Otsuka, Sanofi, Sunovion, Teva and Valeant. Raymond Lam has received honoraria for ad hoc speaking or advising/consulting or received research funds from: Akili, Allergan, Asia-Pacific Economic Cooperation, BC Leading Edge Foundation, Canadian Institutes of Health Research, Canadian Network for Mood and Anxiety Treatments, Canadian Psychiatric Association, CME Institute, Hansoh, Healthy Minds Canada, Janssen, Lundbeck, Lundbeck Institute, Medscape, MITACS, Movember Foundation, Ontario Brain Institute, Otsuka, Pfizer, St. Jude Medical, University Health Network Foundation, and VGH-UBCH Foundation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grant MCT-94832 from the Canadian Institutes of Health Research. The funder had no role in the design and conduct of the study.
