Abstract
Objective:
The potential of clozapine in severe bipolar disorder is suggested by its efficacy in refractory schizophrenia, but the evidence is limited thus far. This report utilizes data from the standard care pathway of the Systematic Treatment Enhancement Program to examine the clinical impact of clozapine in bipolar disorder, comparing it to two groups, one that received olanzapine and an additional group that received neither drug.
Method:
A total of 4,032 outpatients were available for this analysis. Groups for longitudinal analyses are based on the medication used at each visit. Outcomes assessed were clinical status, symptoms subscales, hospitalizations, and death. We utilized mixed models and generalized estimating equations to adjust for baseline differences and investigate longitudinal differences in symptoms, clinical status, and hospitalization rates between groups.
Results:
During the study, 1.1% (n = 43) of the patients used clozapine at any time. Those on clozapine had significantly fewer manic and depressive symptoms during follow-up as compared with those on neither clozapine nor olanzapine, while those on olanzapine had more symptoms. The use of clozapine was not associated with an increased risk of hospitalization. No deaths were recorded for clozapine group during the trial.
Conclusions:
Although prescribed to very few patients, the impact of clozapine was notable, with fewer symptoms in patients who had more severe illnesses at baseline. Clozapine could prove to be as successful an intervention for late-stage bipolar disorder as it has been in schizophrenia.
Introduction
Bipolar disorder is a persistent, episodic, and debilitating condition with an estimated lifetime prevalence of over 2.0%. 1 Its course can be considerably heterogeneous—while some patients recover well even after several episodes, others have increasing illness severity from the first onset of symptoms. 2 For many patients, perhaps up to 50%, the illness has a progressively deteriorating course. 1,3 Treatment guidelines are now consolidated, with lithium, anticonvulsants, and mood stabilizers all recommended as first-line pharmacological treatment, often in combination. 4 –6
Different models have been put forth in an attempt to conceptualize refractory cases of bipolar disorder. 7 In a neuroprogression framework, there is a continuum of progressing disability that can lead in late stages to chronic symptomatology, treatment resistance, cognitive deterioration, and functional impairment. 3 Irrespective of the ultimate confirmation of this thesis, there is a significant treatment gap for those patients who are most refractory and disabled, with little evidence or guidance on how to select appropriate interventions. 8 In fact, most interventions tend to be less effective in highly recurrent illness, where complex interventions are typically needed, and polytherapy is the norm, often with suboptimal outcomes. 1,9 This clearly points to the need of validating interventions for the most severely affected patients. Atypical antipsychotics have become a cornerstone of the treatment of bipolar disorder, used in all phases of the illness, both as adjunctive treatments and monotherapy. 4,5 They are often prescribed for the most disabled patients, 9 but the specific evidence base for treatment-resistant bipolar disorder is scarce. Most studies are highly variable in the quality of their designs and are largely inconclusive. 10
Clozapine is unique among the atypical antipsychotics in its efficacy for treatment-resistant schizophrenia. This effect has been shown in both randomized studies and observational real-world data, in a variety of measures. 11 The use of clozapine reduces hospitalization rates and duration. 12 When compared to olanzapine, clozapine has been associated with reduced suicide rates and suicidality. 13 This suggests room for the use of clozapine in the treatment of bipolar disorder. The current evidence base in this condition for the use of clozapine, however, is largely based on small randomized trials, uncontrolled naturalistic trials, and retrospective studies. 14,15 For this reason, treatment guidelines using both study methodology and tolerability in their weighting tend to be cautious in recommending clozapine as an option for refractory bipolar disorder. 4,5
Taking into account the limitations of current clinical trials and the need for a greater body of evidence of the efficacy of clozapine, in this report, we utilized data from Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD). 16 Observational studies can be an important source of information for relevant decisions when no other data are available, in treatment of severe and refractory illness and funding for large randomized trials is unlikely. Several outcomes of clinical relevance were measured, and we investigated correlates of clozapine use, affective symptoms, hospitalizations, and death. Although we anticipated that few people would have been prescribed clozapine, this large data set offered the opportunity to examine the clinical impact of clozapine.
Methods
The STEP-BD was a nationwide multicenter long-term study for bipolar disorder funded by the National Institute of Mental Health (NIMH), recruiting patients from 2001 to 2007. STEP-BD had different pathways: standard care pathway (SCP) and randomized care pathway. 16 The SCP was open to all participants with a diagnosis of bipolar disorder and is felt to represent patients treated for bipolar disorder in the United States at that moment in time. All patients received the same ongoing assessments of treatment and outcome information.
Participants
Participating treatment centers offered STEP-BD enrollment to patients seeking outpatient treatment. The STEP-BD registry included patients older than 15 years, diagnosed with bipolar I disorder, bipolar II disorder, bipolar disorder not otherwise specified, or cyclothymia, in any phase of illness. The diagnosis was determined during the screening and baseline assessments with the Mini International Neuropsychiatric Interview and a standardized affective disorder evaluation (ADE). 16 In total, 4,360 patients from 22 sites in the United States were enrolled.
Interventions
The model practice procedures for pharmacotherapy were based on published treatment guidelines. 16 To investigate clozapine use during STEP-BD in the SCPs, we separated participants into three groups. In an attempt to minimize the inherent selection by indication bias present in naturalistic studies, we identified all patients prescribed olanzapine during STEP-BD as an active comparison group and created groups based on the participant ever using clozapine (n = 43, 1.1%), olanzapine (n = 851, 21.1%), or neither (n = 3,138, 77.8%). The group on neither clozapine nor olanzapine could be on any prescribed interventions, including other atypical antipsychotics.
For all the longitudinal analysis, groups were based on patients using these medications at any given time point, that is, groups could vary if patients were started, taken out of or moved from one drug to the other. Patients could be on any of the medications at entry of the study or had initiated any of them throughout it. For instance, if a patient began the study on olanzapine and was later taken out of it and prescribed clozapine, she would provide data to the analysis firstly on olanzapine and later on clozapine. They could also be using any other pharmacotherapy.
Follow-Up and Assessment of Outcomes
The standardized ADE was administered at study entry. The ADE includes a modified version of the mood and psychosis modules from the Structured Clinical Interview for DSM-IV (SCID), intended for routine use by practicing clinicians. The ADE also collects specific data on age of onset, estimate of number of prior episodes, current or past comorbid psychiatric disorders, medical conditions, current or past psychoactive substance use, medications, and demographic features.
Eligible and consenting patients received clinical care from an STEP-BD clinician, and all visits were recorded using the clinical monitoring form (CMF). 17 Visits were scheduled with the treating psychiatrist, as clinically indicated. As such, patients have a diverse number of clinical visits during the study, ranging from 1 to 106, with a median of 12. The last recorded observation occurred at 63 months, but 75% of data points were recorded up to 19 months. In the meantime, an independent evaluator administered formal assessments and self-rated scales quarterly in the first and semiannually thereafter.
The data for the analyses came from both CMF and scales fulfilled at independent evaluations. From the CMF, current clinical state was assessed by the treating physician as depression, mania, hypomania, mixed/cycling, roughening, continued symptoms, recovering, recovered, and other diagnosis. Summary scores for depressive and mood elevation symptoms were also assessed with the continuous symptoms subscales for depression (SUM-D) and mania (SUM-M). Scores for depression (SUM-D) range from 0 to 22, with higher scores indicating more severe symptoms. Symptom subscale for mood elevation (SUM-M) scores range from 0 to 16. Current clinical status was also rated longitudinally by clinicians as “depression,” “hypomania,” “mania,” “mixed,” “continued sx,” “recovering,” “recovered,” or “roughening” (defined as symptoms at a subsyndromal level after recovery). Clinical Global Impression Severity Scale and Global Assessment of Functioning (GAF) Scale were also part of the CMF.
Hospitalizations in STEP-BD could be recorded in two ways. The hospitalization form was applied at independent evaluations, with start in the third month of the study and continued quarterly in the first year and semiannual in the second year. Hospitalization criteria were fulfilled if the patient had any psychiatric hospitalization since his last STEP-BD evaluation. The date of hospitalization, reason, and number of days in the hospital were registered in the form. Alternatively, hospitalizations could be recorded as part of the serious adverse experience form. Both contribute to the hospitalization analysis discussed subsequently. Mortality was also recorded for the first 2 years of study.
Statistical Analysis
A total of 4,032 outpatients with bipolar disorder from the STEP-BD were available for the analysis presented here. Because of missing data for several sets of variables, sample sizes for the different analyses vary between 3,073 and 4023, and this is indicated in the Results section. For one set of data analyses, patients were grouped according to having any recorded prescriptions of clozapine or olanzapine (or neither) during the trial. From this set, we report factors associated with the patient ever being prescribed clozapine or olanzapine. We used descriptive statistics to analyze simple group differences. We employed logistic regression for the multivariable data, with gender, ethnicity, marital status, being on disability, unemployment, low education, bipolar type, rapid cycling, very early onset (before age 13), lifetime anxiety and substance use disorders, lifetime psychosis, medical comorbidity, and baseline medications—usual predictors of outcome 18 —as predictors of use of clozapine. We also reported death during the trial according to these medication groups and used Mc Nemar test to report significances.
For the longitudinal analyses, within-patient repeated observations are used for all outcomes. For rating scales, we used random intercept and random slope linear mixed models to make comparisons between patients using clozapine, olanzapine, or neither throughout the trial. Such models are useful in this naturalistic design situation where the design is unbalanced and patients are allowed to change groups during the study. 19 In this way, results of the mixed model represent the effect of either being on the drug and the clinical impact at that point in time.
All models reported in this analysis are adjusted for the baseline presence of some major baseline confounders: being on disability benefits, being in a depressive episode at intake, being on lithium, having a history of psychosis, or of a chronic medical comorbidity. In addition, we adjusted for the effect of both time and the number of visits recorded during STEP. All models used as random effects the intercept, time and medication group (clozapine, olanzapine or neither) and unstructured covariance matrixes. 19 Because of potential differential effects according to staging, we also tested models with treatment interactions with both number of episodes and being on disability benefits. All models were nonsignificant. Residuals from the final models were inspected for normality.
Effects of being on clozapine or olanzapine on psychiatric hospitalization and having a clinical status of mania or depression were also reported. To use all longitudinal data, including repeated hospitalizations, we employed generalized estimating equation models, 20 with the group (clozapine, olanzapine, or neither) and time as predictors, adjusted for all the variables mentioned for the linear mixed models above
Results
Clozapine use was recorded on at least one visit by 43 (1.1%) of the 4,032 patients included in the SCP pathway. Of these, 15 were using clozapine at intake and a further 28 started clozapine during the trial. The median maximum dose was 200 mg (interquartile range 75 to 350 mg).
Of the 43 patients, 29 used clozapine until they left the trial. A higher maximum dose of clozapine was associated with the patient completing the trial on clozapine (300 vs. 129 mg, F = 9.02, P = 0.005). For the 28 patients who started clozapine during the trial, clinical status was variable upon initiation: 12% were depressed, 15% manic or mixed, and 50% had either continued symptoms or recovering status, while 23% were recovered; median GAF was 60. There was a prior recorded use of olanzapine for 12 patients. In a before-and-after analysis of these patients, initiation of clozapine was associated with fewer depressive and manic symptoms (Figure 1).
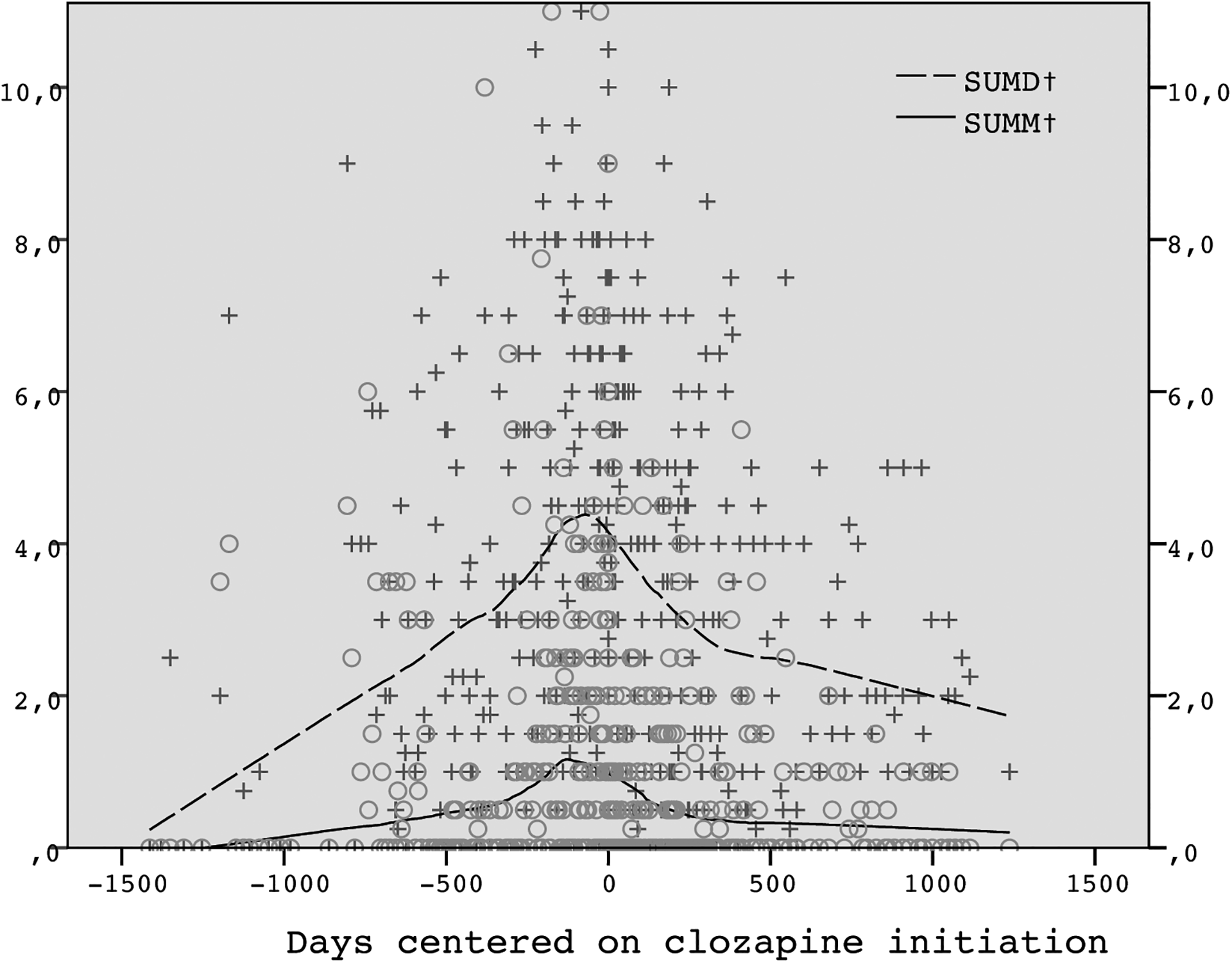
Changes in depressive (SUM-D) and manic (SUM-M) symptom scores before and after the initiation of clozapine (n = 28). Time is displayed in days and centered (i.e., day 0) upon initiation of clozapine.
Olanzapine was used at any point during the trial by 851 (21.1%) patients. Kaplan-Meier statistics indicated a median time using clozapine of 233.43 (standard error [SE] 119.93) weeks as compared to 74.00 (SE 4.51) weeks for those on olanzapine (χ2(1) = 22.80, P < 0.001. This significance was maintained when adjusted for baseline use of the drug. Those on clozapine also stayed longer on the drug while in the trial than those on olanzapine (B = 3.89; 95% confidence interval [CI], 2.13 to 9.09; P < 0.001). Significantly more patients on clozapine finished the trial on the drug than those on olanzapine (74.4% vs. 49.2%, χ2(1) = 10.34, P = 0.001). A maximum dose of clozapine over 200 mg was associated with a greater likelihood of the patient staying on clozapine while in the trial the longest (χ2(1) = 9.98, P = 0.026). Those on clozapine had a higher number of visits while in the study (a median of 31 vs. 20 for those on olanzapine and 10 for those on neither at any point, P < 0.001).
Several patient variables were associated with the use of clozapine at any point of the study (see Table 1 for bivariate and multivariate associations). Among the factors investigated, being on disability, meeting Diagnostic and Statistical Manual, fifth edition criteria for bipolar I disorder, a lifetime history of psychosis or of benzodiazepine use were significantly associated with clozapine use in the bivariate analysis. Age of onset younger than 13 years and presence of medical comorbidity were associated with a significantly less frequent use of clozapine. In a multivariable model, only lifetime psychosis (odds ratio [OR] = 4.16; 95% CI, 1.62 to 10.69) and being on disability (OR = 2.48; 95% CI, 1.03 to 5.96) predicted the use of clozapine. When compared with those everprescribed olanzapine, those everprescribed clozapine were more likely to be on disability (P = 0.048) and to have a lifetime psychotic episode (P = 0.007) but less likely to have had a very early onset (P = 0.045) or a chronic medical comorbidity (P = 0.046).
Characteristics of STEP-BD Participants (n = 4,032) at Entry and According to Being Prescribed Clozapine at Any Point During the Trial.
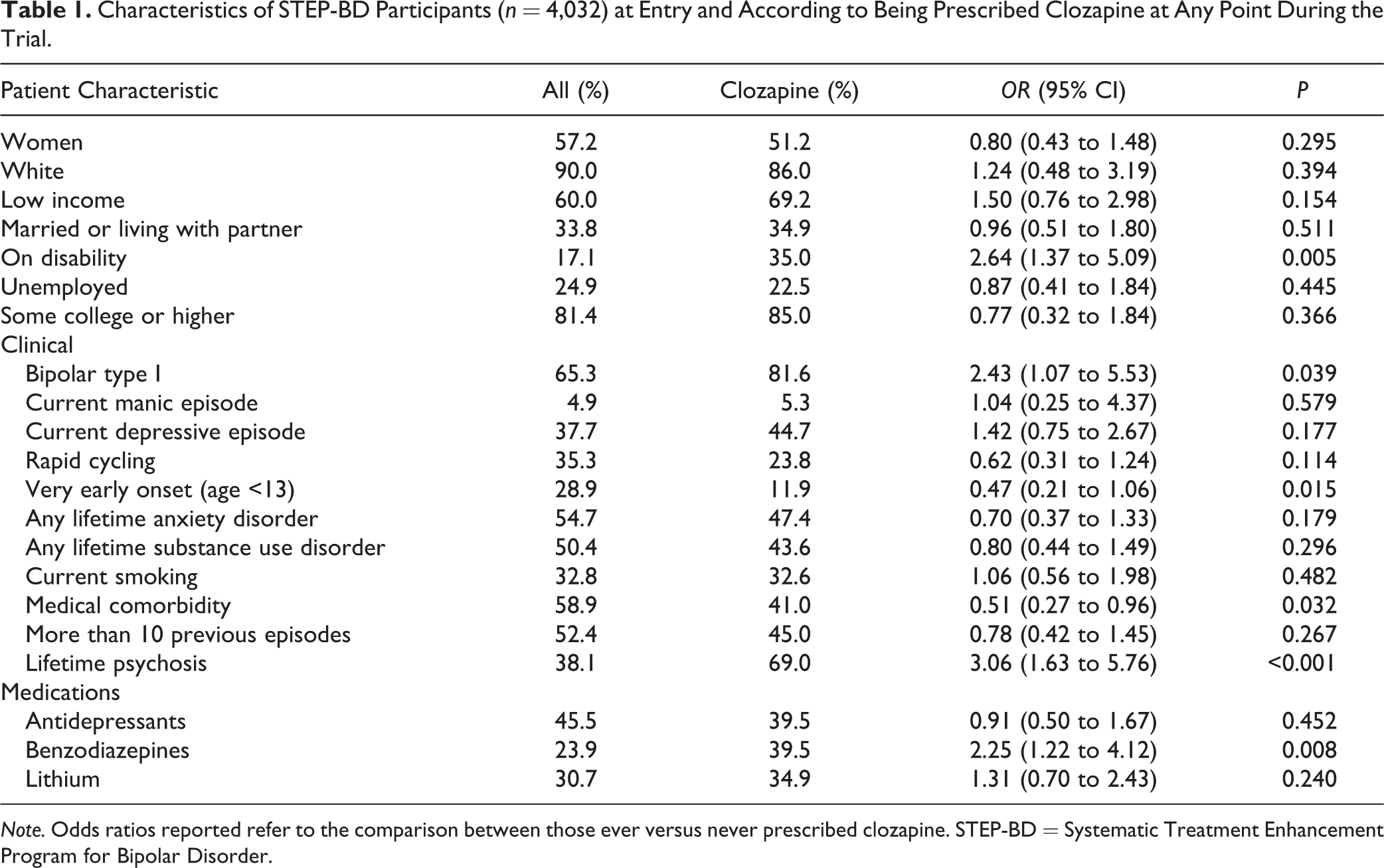
Note. Odds ratios reported refer to the comparison between those ever versus never prescribed clozapine. STEP-BD = Systematic Treatment Enhancement Program for Bipolar Disorder.
Clinical status recorded prospectively was significantly different in those using clozapine. When corrected for multiple comparisons, patients on clozapine were less likely to be classified as having as status “continued symptoms,” “recovering,” or hypomania, while they were more likely to be classified as recovered or roughening. When compared to those using olanzapine, they were less likely to have a depression, continued symptoms, or recovering status, while they were again more likely to have a recovered or roughening status. Longitudinally, a status of depression was recorded in 19.7% of visits, a status of mania in 5% of visits, and a recovery status in 27.2% of visits. The use of clozapine was associated with fewer ratings of depression in the multivariable model (n = 3,069; OR = 0.25; 95% CI, 0.11 to 0.59; P = 0.002), while the effect of olanzapine was not significant (OR = 1.09; 95% CI, 0.91 to 1.30; P = 0.364). There was an interaction between the use of clozapine and time (P = 0.002). Ratings of mania were not associated either with use of clozapine (OR = 0.49; 95% CI, 0.13 to 1.82; P = .284) or olanzapine (OR = 1.02; 95% CI, 0.77 to 1.35; P = 0.875).
The mixed models investigating longitudinal symptom score (in the SUM-D and SUM-M) differences between groups revealed different patterns for those using clozapine, olanzapine, or neither (n = 3,052). Those on clozapine had significantly fewer affective symptoms during the trial as compared with those on neither clozapine nor olanzapine, both depressive (diff = −0.64; df (32.61); 95% CI, −1.22 to −0.06; P = 0.030) and manic symptoms (diff = −0.39; df (26.68); 95% CI, −0.64 to −0.14; P = 0.004). Those on olanzapine had more depressive (diff = 0.35; df (737.51); 95% CI, 0.20 to 0.50; P < 0.001) and manic symptoms (diff = 0.13; df (593.41); 95% CI, 0.05 to 0.21; P = 0.002) as compared with those on neither (Figure 2).
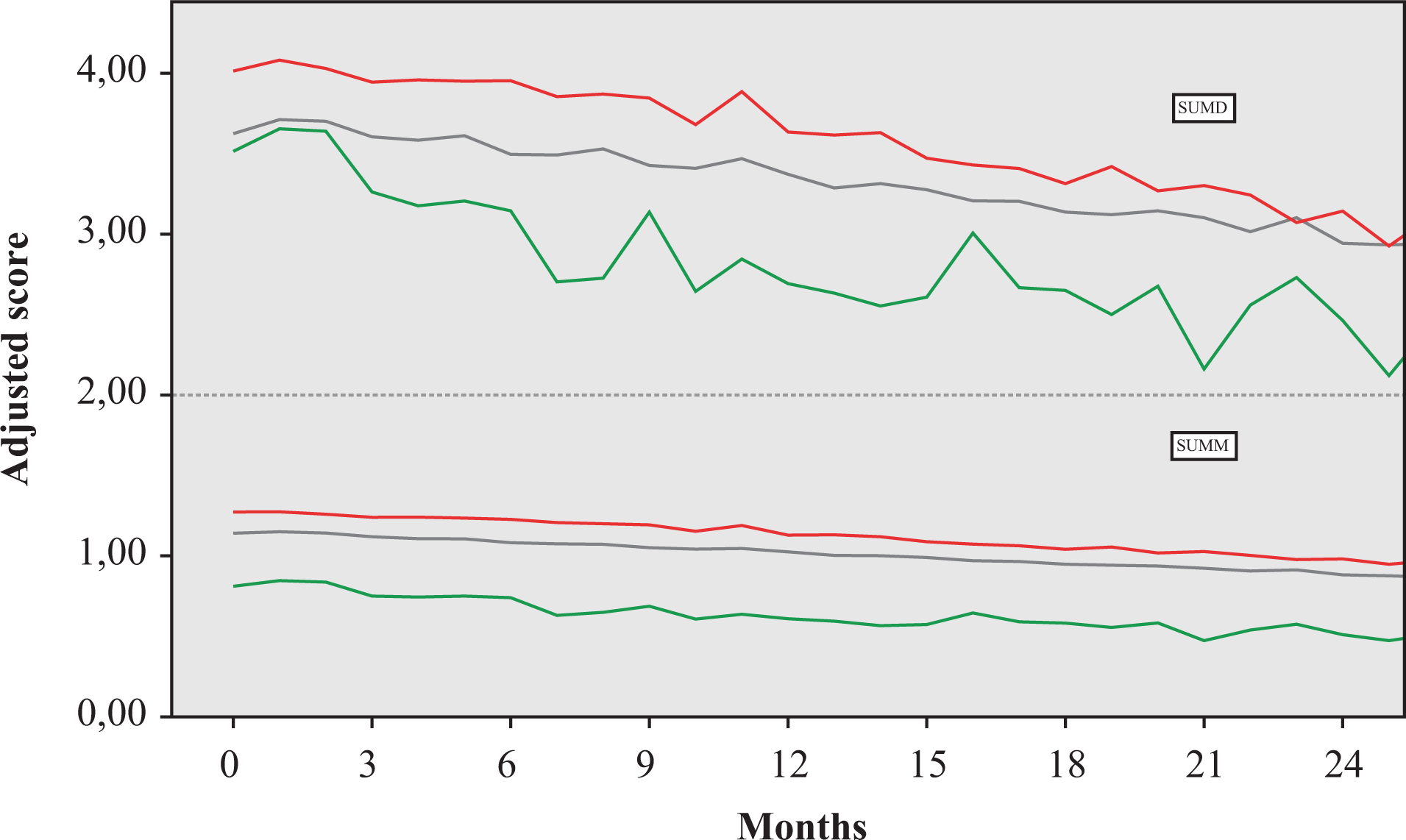
Changes in depressive (SUM-D, upper half of graph) and manic (SUM-M, lower half) symptom scores in patients receiving treatment with clozapine (green line), olanzapine (red line), or neither (black line). Scores are adjusted for being on disability, on a depressive episode at intake, on lithium, having a history of psychosis or of a chronic medical comorbidity, and the total number of visits during follow-up. Maximum sample sizes during the trial were 43 for clozapine, 851 for olanzapine, and 3,138 for neither drug.
For 781 patients (of 3,308), there was at least one recorded hospitalization. Those who used clozapine at any point during the trial were more likely to have any hospitalizations during the trial (55.8%) than those using olanzapine (31.0%) or those using neither (15.6%; P < 0.05 for all comparisons). When analyzed longitudinally and according to the medication the patient was using at the moment of hospitalization, however, in the adjusted model, only olanzapine was associated with greater risk of hospitalization (OR =1.53; 95% CI, 1.18 to 1.98), while clozapine was not (OR = 0.80; 95% CI, 0.29 to 2.21).
Finally, there were 25 recorded deaths during the follow-up—six due to suicide. No deaths were recorded for the patients who were prescribed clozapine at any point during the trial (P = 0.038 for the difference).
Sensitivity Analyses
The sensitivity analyses excluding the 15 patients (and corresponding 457 observations) who started the study on clozapine, that is, with only new clozapine users, did not alter significantly the results of the effect of clozapine on depression, mania, or hospitalizations. Clozapine was no longer significantly different on depressive symptoms (P = 0.17), but the effect on manic symptoms was maintained (P = 0.004).
Discussion
Perhaps unsurprisingly, only a very small percentage of patients in STEP-BD were prescribed clozapine. In all likelihood, this can be partially ascribed to the dearth of evidence supporting its use, perceived tolerability issues, as well as the lack of regulatory approval. Clozapine was used by patients with markers of a more severe disorder, such as living on disability and lifetime psychosis. When such severity is taken into account, the impact of clozapine was substantial. The use of clozapine was associated with fewer affective symptoms during the trial; the use of olanzapine, the relevant active comparator, was associated with more symptoms. Unlike those on olanzapine, those on clozapine were no more likely than those on neither drug to be hospitalized during the trial. Finally, no deaths were recorded for those using clozapine at any point of the study.
Prescription rates of clozapine for bipolar disorder are still very low, and it is the least prescribed antipsychotic for bipolar disorder. 21,22 This is all congruent with the data reported here for STEP-BD, which likely reflects a currently common pattern. There is indeed very limited randomized evidence of the impact of clozapine in bipolar disorder. 23 A recent systematic review of all previous extant data suggested clozapine may be an effective therapy, although the evidence base was severely limited in terms of number of patients and study quality. 14 The prospective trials that have been published—in only a few dozen patients—suggest improvement of symptoms is sustained for 12 months and that it may be efficacious in refractory mania. 24 Interesting observational data also suggest that the differential impact of clozapine in bipolar disorder may be akin—or even greater—to its impact in schizophrenia. 25 As noted, these are all tentative notions.
A few reasons for the enhanced efficacy of clozapine have been put forth and may well apply when it is used in bipolar disorder. Clinically, clozapine treatment is associated with reduced hostility and violence independently of its effects on psychosis, and positive symptoms, and of its nonspecific sedative effects. 26,27 An interesting finding was that no deaths were found in patients who were prescribed clozapine. Although chance and the very small sample have definitely played a role in this finding, it raises curious questions. There are studies that do suggest that clozapine is associated with a reduced overall mortality in relation to other antipsychotics, perhaps related the effect of improved efficacy on self-care and the requirement for greater clinical monitoring. 28
The strengths of this multicenter naturalistic study include its description of real-life practice and the initial coverage of more than 4,000 patients with bipolar disorder. Exclusion criteria were kept to a minimum to increase external validity. Nevertheless, the total number of patients prescribed clozapine was low. This poses problems both in terms of the representativeness of the sample and study power for certain comparisons. Treatment was also not blinded, and the patients were not randomly assigned to medication. There is, however, no reason to suspect that clozapine would be overprescribed to those with a better prognosis and that would be responsible for the differential impact. We also focused only on limited effectiveness outcomes, not on tolerability and safety, which can be an issue for those on clozapine. Nevertheless, the fact that those participants on clozapine tended to use it for a longer period than those on the comparators indicates tolerability in this population. One further point is to what extent outcomes for those using clozapine in general are a result of enhanced contact with health-care professionals due to mandatory monitoring, although we adjusted the final models for the greater number of office visits these participants had.
We selected olanzapine as an active comparator with the expectation that it would have the closest prescription pattern to that of clozapine. The comparison with a relevant active compound within the same class or prescribed in similar circumstances has been recommended as a way to attenuate differences in baseline characteristics, reducing unmeasured confounding. 29 We selected olanzapine for this role for a few reasons. It has been the comparison drug used to validate the superior effectiveness of clozapine in several key resistant-schizophrenia studies. 30 At least for a time, olanzapine was perceived to be potentially as effective as clozapine and considered a viable alternative for intolerant patients. 13 In bipolar disorder, studies on olanzapine have indeed shown earlier onset of response and reductions of hospitalization rates. 1,10
Although STEP-BD was not designed to test the efficacy of clozapine for bipolar disorder, its prospective design with several measures of clinical interest permitted the exploration of a few specific hypotheses. Even if prescribed at low rates, the impact of clozapine was notable, with fewer symptoms in patients with more severe illnesses. Although there are worries about clozapine safety, those on clozapine were no more likely to be hospitalized and—quite surprisingly—none was recorded deceased for any reason during the study. Clearly, these effects need to be confirmed in a randomized trial with relevant active comparators. Preferably, this would be conducted in those with the most recalcitrant illnesses, for whom the availability of clozapine could have the most significant impact, perhaps as great as it has had for those with refractory schizophrenia.
Supplemental Material
Supplemental Material, 19040-c-CJP-2019-187-OR.R1-abstract_for_translation - Clozapine and the Course of Bipolar Disorder in the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD)
Supplemental Material, 19040-c-CJP-2019-187-OR.R1-abstract_for_translation for Clozapine and the Course of Bipolar Disorder in the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD) by Marta H. Costa, Mauricio Kunz, Andrew A. Nierenberg, Thilo Deckersbach, Michael Berk and Pedro V. S. Magalhaes in The Canadian Journal of Psychiatry
Footnotes
Authors’ Note
Any opinions, findings, and conclusions expressed in this publication are those of the authors and do not reflect the views of the NIMH. The funder of the study had no role in this study design, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all data in the study and had the final responsibility for the decision to submit for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The STEP BD has been funded in whole or in part with Federal funds from the NIMH, National Institutes of Health, under Contract N01MH80001. M.B. is supported by a NHMRC Senior Principal Research Fellowship, 1059660. This work was supported in part by the Dauten Family Center for Bipolar Treatment Innovation. P.V.S.M. is supported by a National Council for Scientific and Technological Development—CNPq productivity fellowship.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
