Abstract
Objective:
The Yale Food Addiction Scale (YFAS) is the only questionnaire that assesses food addiction (FA) based on substance dependence criteria in the Diagnostic and Statistical Manual of Mental Disorders (DSM), Fourth Edition, Text Revision. Following recent updating of addiction criteria, a new DSM-5 version (YFAS 2.0) has been developed. Our study tested the psychometric properties of the French YFAS 2.0 in a nonclinical population.
Method:
We assessed 330 nonclinical participants for FA (French YFAS 2.0), eating behaviour, and eating disorder (Binge Eating Scale, Emotional Overeating Questionnaire, Three-Factor Eating Questionnaire-R18, Questionnaire on Eating and Weight Patterns-Revised, Eating Disorder Diagnostic Scale). We tested the scale’s factor structure (confirmatory factor analysis based on 11 diagnostic criteria), internal consistency, and construct and incremental validity.
Results:
Prevalence of FA was 8.2%. Our results supported a 1-factor structure similar to the US version. In both its diagnostic and symptom count versions, the YFAS 2.0 had good internal consistency (Kuder-Richardson alpha was 0.83) and was associated with body mass index (BMI), binge eating, uncontrolled and emotional eating, binge eating disorder, and cognitive restraint. FA predicted BMI above and beyond binge eating frequency. Females had a higher prevalence of FA than males but not more FA symptoms.
Conclusions:
We validated a psychometrically sound French version of the YFAS 2.0 in a nonclinical population, in both its symptom count and diagnostic versions. Future studies should investigate psychometric properties of this questionnaire in clinical populations potentially at risk for FA (that is, patients with obesity, diabetes, hypertension, or other metabolic syndrome risk factors).
Drug addiction is a chronic relapsing disorder characterized by compulsion to seek and take the drug, a loss of control over drug-seeking and drug-taking behaviours, and the continued use of the drug despite its adverse consequences. 1 It has recently been suggested that addictions should also include compulsive engagement in some pleasurable activity, such as gambling, gaming, Internet use, sex, exercising, eating, or shopping, in addition to pharmacologic rewards. 2 Apart from gambling disorder, which has been included in a new behavioural addiction category of the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5), more research is needed before considering the potential inclusion of these other activities as addictive disorders. 1
Despite the fact that food is widely available in Western societies, that eating is an essential and frequent human activity, and that certain foods have powerful addictive and rewarding effects similar to drugs, 3,4 the hypothesis that some individuals are addicted to food or to their eating behaviour has only recently been proposed. 5 Gearhardt et al 6 recently proposed the concept of food addiction (FA) as a specific phenotype defined by applying the DSM-IV-TR diagnostic criteria for substance dependence to certain foods, particularly processed foods high in added sugar, fat, and salt. This led to the development of the first self-administered questionnaire assessing FA symptoms: the Yale Food Addiction Scale (YFAS; hereafter called “original YFAS”). 7 Further studies demonstrated the excellent psychometric properties of the original YFAS, including a 1-factor structure, excellent internal consistency, and very good convergent validity with measures of binge eating. 7 This questionnaire has been cross-culturally validated, including Chinese, 8 French, 9 German, 10 Italian, 11 Spanish, 12 and Turkish 13 versions, allowing a growing body of research in this field. 14 FA was found to be more prevalent in single and obese individuals 15 ; in those with higher levels of depression, higher impulsivity, higher prevalence of posttraumatic stress disorder, higher prevalence rate for diagnosis of attention-deficit hyperactivity disorder in childhood 16 –19 ; and in patients with alterations in brain circuitry similar to those found in drug addiction (elevated activation in reward circuitry in response to food cues and reduced activation of inhibitory regions in response to food intake). 20
With the recent update of DSM-5 diagnostic criteria, an update of the original YFAS was needed to take into account 4 new criteria, namely, craving, use despite interpersonal or social consequences, failure in role obligations, and use in physically hazardous situations. An updated version could also help determine whether the DSM-5 criteria for addictive disorders could be applicable to food and whether FA could be included in the international diagnostic classifications as an addictive disorder. To this end, Gearhardt et al 21 designed the US version of the YFAS 2.0 and validated it in a nonclinical population: they used confirmatory factor analysis based on the 11 diagnostic criteria to compare a 1-factor model and a 2-factor model (7 DSM-IV-TR criteria plus craving compared with 3 DSM-IV-TR criteria of abuse recently added in the DSM-5). They finally retained a 1-factor structure based on fit indices (the 2-factor solution did not result in noticeably improved fit, and the 2 factors of the 2-factor model were highly correlated). This validation study demonstrated the excellent psychometric properties of the YFAS 2.0, with a 1-factor structure and high convergent validity with measures of disinhibited eating, obesity, and weight cycling. Regarding discriminant validity, the US version of the YFAS 2.0 was not significantly correlated with dietary restraint, and approximately half the participants with an FA diagnosis did not meet criteria for an existing eating disorder. In addition, the YFAS 2.0 exhibited incremental validity by accounting for variance in elevated body mass index (BMI) above and beyond binge eating frequency. To our knowledge, there is currently no French version of the YFAS 2.0 and no studies investigating the prevalence of FA and its associated factors as assessed by the DSM-5 criteria.
This study aimed to measure the psychometric properties of a French version of the YFAS 2.0 in a nonclinical sample by establishing its factor structure (confirmatory factor analysis), its internal consistency, its construct validity with eating disorder diagnosis, binge eating symptoms, uncontrolled eating, emotional eating and emotional overeating (convergent validity) and with cognitive restraint (discriminant validity), and its incremental validity (does the YFAS 2.0 symptom count explain unique variance in BMI above and beyond binge eating frequency?). We hypothesized that the French YFAS 2.0 had a 1-factor structure, good internal consistency, high convergent validity with BMI, binge eating, and emotional eating, and no correlation with cognitive restraint.
Methods
Participants and Procedures
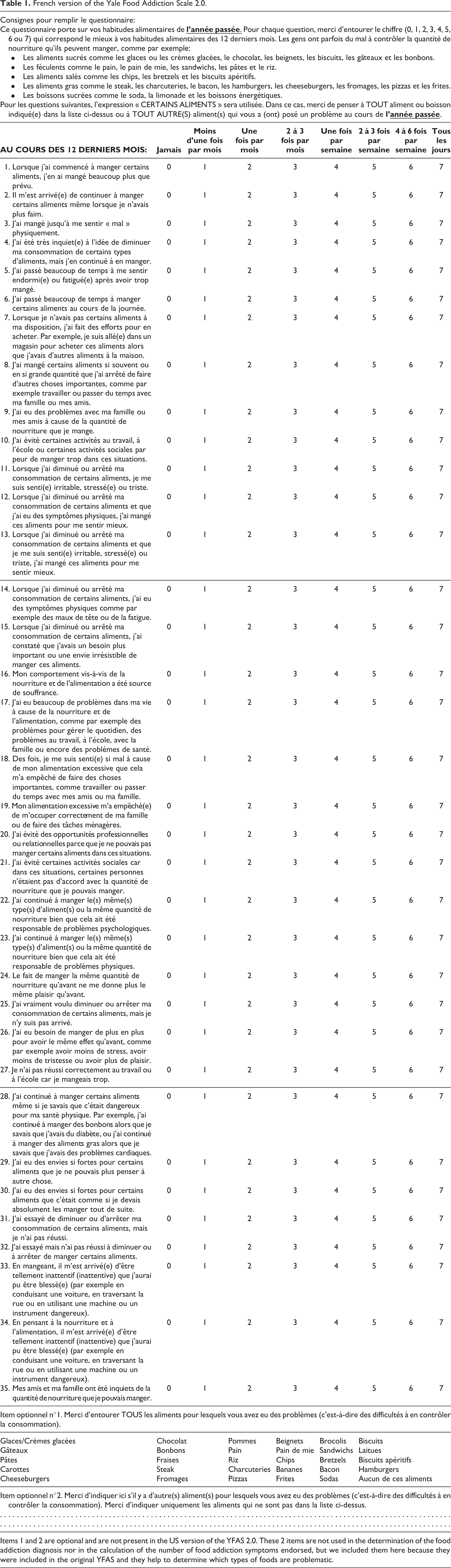
A total of 330 volunteers participated in our study between May 2014 and May 2015. They were recruited through a web-based questionnaire, created using Sphinx software (Sphinx Plus 2 version 5.1.0.4), 22 sent to students from the Department of Psychology and Medicine of Tours University (n = 164; 49.7% of the sample) and their families (n = 166; 50.3% of the sample). Students proposed the questionnaire to their family members who participated based on volunteering. We differentiated results from the students’ and family members’ answers. We ensured that only adults were included in the family sample. The YFAS translation procedure was the same as the one used in the validation of the French version of the original YFAS (see Brunault et al 9 for more details regarding the translation procedure): (1) translation, (2) blind-backward translation, (3) comparison between the 2 versions and modification if necessary, and finally (4) test of a pilot version in 10 controls and 10 patients and modification of the scale if necessary. The final French version of the YFAS 2.0 can be found in Table 1.
French version of the Yale Food Addiction Scale 2.0.
Measures
We used measures assessing the same constructs as Gearhardt et al 21 in their YFAS 2.0 validation study, collecting sociodemographic characteristics (i.e., age, sex, marital status), current BMI, and previous maximal BMI and using self-administered questionnaires assessing the same constructs (see below and Gearhardt et al 21 for more details).
Food Addiction (YFAS Version 2.0)
The YFAS 2.0 is a 35-item self-report scale designed by Gearhardt et al 21 to assess FA symptoms over the previous 12 months based on the 11 diagnostic criteria for substance-related and addictive disorders proposed in the DSM-5. 1 In this way, the YFAS 2.0 assesses 11 FA criteria (for a detailed description, see Supplementary Table S1), including the 7 previous food-related DSM-IV-TR criteria (consumption more than planned, persistent desire or unsuccessful efforts to cut down or control consumption of certain foods, great deal of time spent, important activities given up, use despite physical/psychological consequences, tolerance, withdrawal), 6 the 4 new criteria in the DSM-5 (i.e., craving, use despite interpersonal or social consequences, failure in role obligations, and use in physically hazardous situations), and significant distress in relation to food.
Each item is rated on an 8-point Likert-type scale ranging from 0 (never) to 7 (every day). Each item can be rated dichotomously (endorsed or not endorsed) depending on specific cutoffs that Gearhardt et al 21 defined for each item by examining the specificity for each response option based on receiver operator characteristic curves (see Gearhardt et al 21 for more details). Based on these thresholds, each of the 11 diagnostic criteria is met when 1 or more item representing that criterion is endorsed (see Supplementary Table S1 for the detailed scoring instructions for the YFAS 2.0). 21 The YFAS 2.0 provides 2 scoring options: a diagnostic version (FA is diagnosed when the participant reports 2 or more symptoms during the previous 12 months plus clinically significant impairment or distress) and a symptom count version (number of FA symptoms experienced in the previous 12 months, ranging from 0 to 11). FA is mild if there are 2 or 3 symptoms and clinically significant impairment/distress, moderate if there are 4 or 5 symptoms and significant impairment/distress, and severe if there are 6 or more symptoms and significant impairment/distress. 1,21
Details of the psychometric properties of the English version of the YFAS 2.0 are described above in the introduction.
Binge Eating (Binge Eating Scale)
The Binge Eating Scale (BES) is a 16-item questionnaire designed to assess the severity of binge eating using behavioural, affective, and cognitive symptoms (cutoff ≥18). 23 The BES is a reliable tool for assessing binge eating disorder and has been validated in French. 24 In our sample, Cronbach’s α was 0.89.
Eating Disorders (Questionnaire on Eating and Weight Patterns-R and the Eating Disorder Diagnostic Scale)
We screened for eating disorders based on DSM-5 criteria using the Questionnaire on Eating and Weight Patterns-R (QEWP-R) 25 (for binge eating disorder and bulimia nervosa) and the Eating Disorder Diagnostic Scale 26 (for anorexia nervosa).
Eating Behaviour Characteristics (Emotional Overeating Questionnaire, Revised 18-Item Version of the Three Factor Eating Questionnaire)
We assessed emotional overeating using the Emotional Overeating Questionnaire, which identifies the frequency of overeating in response to 6 emotions (anxiety, sadness, loneliness, tiredness, anger, and happiness) with high internal consistency (α = 0.85). 27 In our sample, Cronbach’s α was 0.81.
We used the revised 18-item version of the Three Factor Eating Questionnaire (TFEQ) 28,29 to assess uncontrolled eating (tendency to eat more than usual due to a loss of control over intake accompanied by subjective feelings of hunger; this subscale encompasses the previous “disinhibition” and “hunger” subscales of the 51-item TFEQ), emotional eating (inability to resist emotional cues), and cognitive restraint (conscious restriction of food intake to control body weight or to promote weight loss). The scores were calculated as described by de Lauzon et al 28 : the theoretical ranges for the items were 9 to 36 for uncontrolled eating, 3 to 12 for emotional eating, and 6 to 24 for cognitive restraint. In our sample, Cronbach’s α was 0.89.
Statistical Analyses and Ethical Considerations
Analyses were conducted using AMOS (SPSS Inc., Chicago, IL) 30 for confirmatory factor analyses and using the R statistical package version 2.15.2 31 with the psych package 32 for the other statistics. Statistical analyses included descriptive statistics and examination of the psychometric properties of the scale (factor structure, item statistics, internal consistency, construct validity, and incremental validity).
To test the factor structure of the French YFAS 2.0, we used the same procedure as Gearhardt et al 21 : we conducted confirmatory factor analyses based on the 11 diagnostic criteria to compare a 1-factor model and a 2-factor model (7 DSM-IV-TR criteria plus craving compared with 3 former DSM-IV-TR criteria of abuse recently added in the DSM-5). To assess the internal consistency of the scale, we used Kuder-Richardson alpha (KR-20) and McDonald’s omega. We used KMO statistics to assess the sampling adequacy.
We assessed the construct validity of both the diagnostic and the symptom count versions by examining the associations between FA (assessed using either the diagnostic or the symptom count version of the YFAS) and the following measures: sociodemographic characteristics, weight-related variables, eating disorder diagnosis, binge eating, emotional eating and emotional overeating (convergent validity), and cognitive restraint (discriminant validity). We used chi-square tests, parametric mean comparison tests (analysis of variance), or Pearson’s correlation tests, as appropriate.
We used hierarchical multiple regression to test incremental validity: does the YFAS 2.0 symptom count predict elevated BMI above and beyond binge eating frequency (item 3 of the QEWP-R)?
There were no missing data, as all of the questions required a response to proceed to the next page of the survey. Our study did not require institutional review board approval because it was not considered biomedical research under French law; however, it followed the tenets of the Declaration of Helsinki.
Results
Sample Characteristics
Table 2 presents the sociodemographic characteristics, weight-related variables, and prevalence of each FA symptom. Participants had a mean age of 28.9 years (SD 11.3, range = 18–76) and a mean current BMI of 23.3 kg/m2 (SD 4.9). Supplementary Table S2 presents and compares the sociodemographic characteristics of students and family members). Prevalence of FA was 8.2%. The mean number of FA symptoms was 1.1 (SD 1.9), with no significant difference between men and women (0.7 SD 1.3 compared with 0.8 SD 1.4; P = 0.65). Ten percent were underweight (BMI <18.5 kg/m2), 65.2% were normal weight (18.5 kg/m2 ≤ BMI < 25 kg/m2), 16.4% were overweight (25 kg/m2 ≤ BMI < 30 kg/m2), and 8.5% were obese (BMI ≥30 kg/m2). Table 3 presents the prevalence of eating disorders and the eating behaviour characteristics of our population. Sixteen of the 27 participants with FA (59.3%) had significant binge eating.
Descriptive statistics of the overall sample and comparison of participants with and without food addiction.
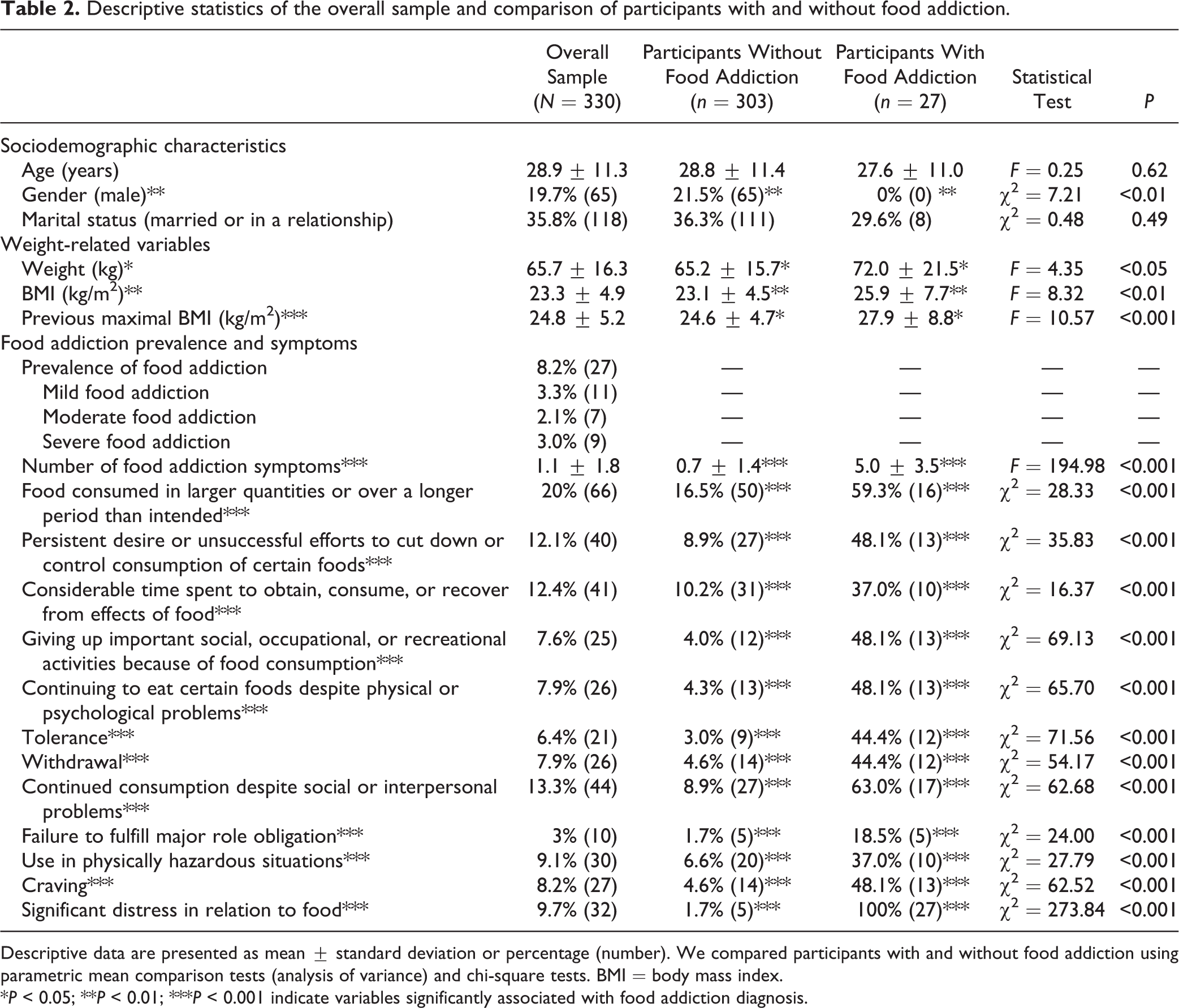
Descriptive data are presented as mean ± standard deviation or percentage (number). We compared participants with and without food addiction using parametric mean comparison tests (analysis of variance) and chi-square tests. BMI = body mass index.
*P < 0.05; **P < 0.01; ***P < 0.001 indicate variables significantly associated with food addiction diagnosis.
Prevalence of eating disorders and eating behaviour characteristics in the overall sample and comparison of participants with and without food addiction.
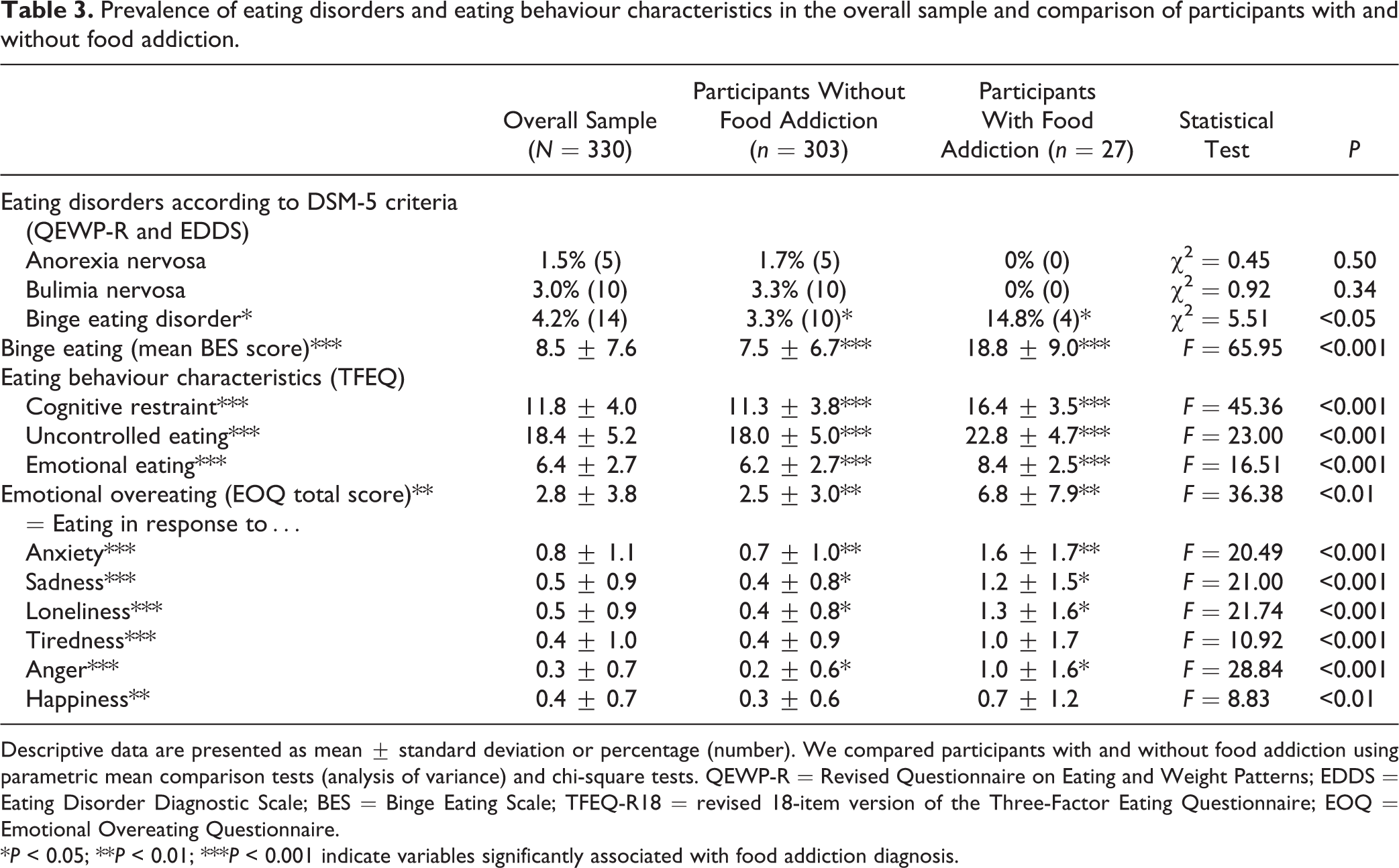
Descriptive data are presented as mean ± standard deviation or percentage (number). We compared participants with and without food addiction using parametric mean comparison tests (analysis of variance) and chi-square tests. QEWP-R = Revised Questionnaire on Eating and Weight Patterns; EDDS = Eating Disorder Diagnostic Scale; BES = Binge Eating Scale; TFEQ-R18 = revised 18-item version of the Three-Factor Eating Questionnaire; EOQ = Emotional Overeating Questionnaire.
*P < 0.05; **P < 0.01; ***P < 0.001 indicate variables significantly associated with food addiction diagnosis.
Item Statistics, Factor Structure, and Internal Consistency
Table 4 summarizes the item statistics, including the mean, standard deviation, and item-total correlation for each item.
Association between the Yale Food Addiction Scale 2.0 symptom score, sociodemographic characteristics, weight-related variables, and eating disorders and behaviours.
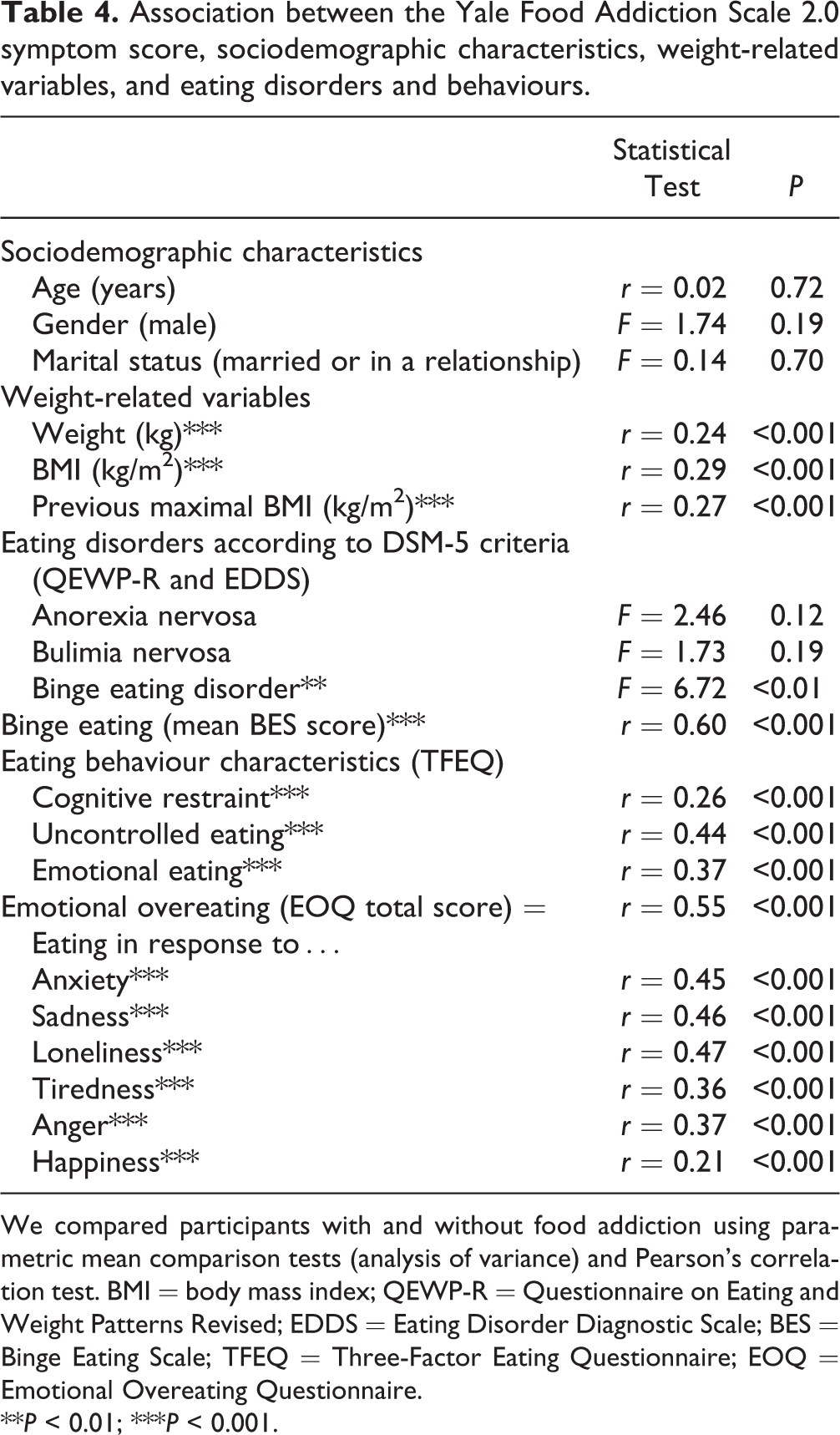
We compared participants with and without food addiction using parametric mean comparison tests (analysis of variance) and Pearson’s correlation test. BMI = body mass index; QEWP-R = Questionnaire on Eating and Weight Patterns Revised; EDDS = Eating Disorder Diagnostic Scale; BES = Binge Eating Scale; TFEQ = Three-Factor Eating Questionnaire; EOQ = Emotional Overeating Questionnaire.
**P < 0.01; ***P < 0.001.
Results and fit indices of confirmatory factor analyses are presented in Table 5. The 1-factor model had adequate fit indices (confirmatory fit index [CFI] was 0.887, root mean square error of approximation [RMSEA] was 0.083) with all factor loadings greater than 0.32. In line with Gearhardt et al 21 and YFAS 2.0 validation study and consistent with the examination by Gillespie et al 33 of the factor structure of substance use disorder symptoms, we retained a 1-factor solution because the 2-factor model did not result in noticeably improved fit (CFI = 0.891, RMSEA = 0.082) and the 2 factors of this 2-factor model were highly correlated (r = 0.69, P < 0.001). The internal consistency of the YFAS 2.0 (11 diagnostic criteria plus significant distress) was good (KR-20 = 0.83, McDonald omega = 0.86) and higher than the one observed with the DSM-IV-TR diagnostic criteria of our sample (7 diagnostic criteria plus significant distress; KR-20 = 0.74). Sampling adequacy was very good (KMO = 0.86). Mean interitem correlation was 0.26 for the 7 DSM-IV-TR criteria and r = 0.30 for the 11 DSM-5 criteria.
Factor loadings for the 1- and 2-factor structures of the Yale Food Addiction Scale 2.0 (confirmatory factor analyses).
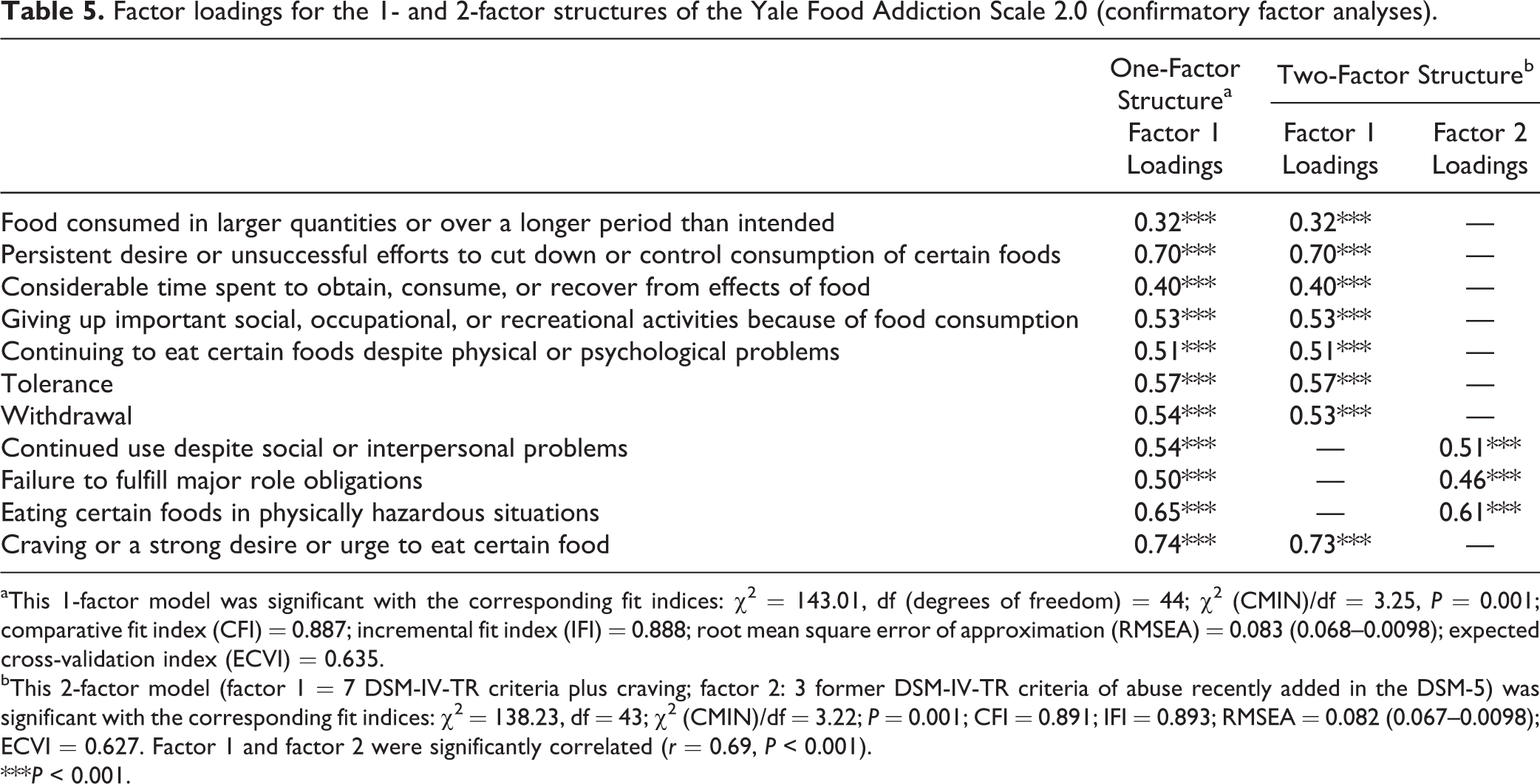
aThis 1-factor model was significant with the corresponding fit indices: χ2 = 143.01, df (degrees of freedom) = 44; χ2 (CMIN)/df = 3.25, P = 0.001; comparative fit index (CFI) = 0.887; incremental fit index (IFI) = 0.888; root mean square error of approximation (RMSEA) = 0.083 (0.068–0.0098); expected cross-validation index (ECVI) = 0.635.
bThis 2-factor model (factor 1 = 7 DSM-IV-TR criteria plus craving; factor 2: 3 former DSM-IV-TR criteria of abuse recently added in the DSM-5) was significant with the corresponding fit indices: χ2 = 138.23, df = 43; χ2 (CMIN)/df = 3.22; P = 0.001; CFI = 0.891; IFI = 0.893; RMSEA = 0.082 (0.067–0.0098); ECVI = 0.627. Factor 1 and factor 2 were significantly correlated (r = 0.69, P < 0.001).
***P < 0.001.
Convergent Validity of the YFAS 2.0 (Diagnostic Version)
Participants with FA (compared with participants without FA) were more frequently female and had higher current weight, BMI, and higher previous maximal BMI (Table 2). They had a higher prevalence rate for DSM-5 binge eating disorder but not for bulimia nervosa or anorexia nervosa (Table 3). Participants with FA had higher scores for binge eating, uncontrolled eating, and emotional eating (P < 0.001). They also ate more frequently in response to negative and positive emotions (i.e., anxiety, sadness, loneliness, anger, tiredness, happiness; Table 3).
Convergent Validity of the YFAS 2.0 (Symptom Count Version)
Table 4 presents the factors associated with the YFAS symptom score: the YFAS symptom score was associated with higher current BMI and higher previous maximal BMI but not with marital status, age, or gender. The YFAS symptom score was associated with diagnosis of binge eating disorder, higher scores for binge eating, uncontrolled eating, emotional eating, and eating in response to negative or positive emotions such as anxiety, sadness, loneliness, anger, tiredness, or happiness. It was not associated with diagnosis of anorexia nervosa or bulimia nervosa.
Discriminant Validity of the YFAS 2.0 (Diagnostic and Symptom Count Versions)
FA diagnosis and symptom count were associated with higher cognitive restraint (Table 4).
Incremental Validity of the YFAS 2.0
In simple linear regression, binge eating frequency was a significant predictor of BMI (t = 2.59, β = 0.14, P < 0.01), accounting for 1.7% of the variance. In hierarchical multiple regression, when YFAS 2.0 symptom count was entered in the model, binge eating was no longer a significant predictor (t = 0.61, β = 0.04, P = 0.55), and YFAS 2.0 symptom count was a significant predictor (t = 4.71, β = 0.27, P < 0.001), accounting for an additional 6% of the variance in BMI (F = 14.67, P < 0.001).
Discussion
This study demonstrated that the French version of the YFAS 2.0 is a reliable tool to diagnose FA and FA symptoms based on the DSM-5 criteria for Substance-Related and Addictive Disorders. It has a one-factor structure, good internal consistency, and high convergent validity with measures of binge eating, emotional eating, BMI, and with diagnosis of DSM-5 binge eating disorder. We did not confirm the discriminant validity of the YFAS with cognitive restraint found in the US validation study. The YFAS symptom count predicted BMI above and beyond binge eating frequency. These results demonstrate that the addiction model is relevant and applicable to food consumption and that FA seems to be a unitary concept and construct, related to but different from traditional eating disorders. 21
The 1-factor structure and good internal consistency of the French version of the YFAS 2.0 is in line with the results obtained with the DSM-5 version of the YFAS. 21 Interestingly, the addition of the 4 new criteria in the DSM-5 (including craving) did not modify the 1-factor structure of this scale and even improved its internal consistency. The YFAS 2.0 also corrected a limitation of the original YFAS: the lack of reliability of items related to “persistent desire or unsuccessful efforts to cut down or control use.” 34 In the French YFAS 2.0, this item was endorsed by 12.1% of the sample (compared with 90.1% in the original YFAS), indicating better assessment of this diagnostic criterion in the YFAS 2.0. Prevalence of FA was higher in the US sample than in the French sample (see Supplementary Table S3 for a comparison of FA prevalence and criteria in the original French YFAS, the French YFAS 2.0, and the US YFAS 2.0), probably because of cultural differences or differences in BMI (in our sample, 1 participant out of 4 was overweight or obese as compared with 1 out of 2 in the US sample 21 ). The adequate factor loadings observed for all of the 12 dichotomous diagnostic criteria (11 diagnostic DSM criteria plus significance questions) suggest that the DSM-5 addiction criteria applied to food assess a unitary concept.
Although the DSM-5 thresholds have been lowered for addictive disorders (i.e., 2 criteria out of 11 are necessary for diagnosis in the DSM-5, compared with 3 out of 7 in the DSM-IV-TR), we found no significant increase in the prevalence rate for DSM-5 FA (Supplementary Table S3). The lower thresholds for the 11 addiction criteria might have been counterbalanced by the higher YFAS 2.0 thresholds for each item.
Interestingly, FA was associated with emotional eating, which is in line with the high comorbidity between FA, emotional dysregulation, 35 and mood and anxiety disorders. 5,36 Our results are in line with Bruch and Kaplan’s psychosomatic theory, which postulates that food intake increases in response to an internal state of emotional arousal, such as anxiety or depression, and that this could partly explain weight gain. 37,38 Although FA was associated with higher BMI, it is still unclear whether FA could be a direct cause of weight gain and whether treatment for FA could improve weight. FA prevalence was higher among women than among men, but the mean number of FA symptoms did not differ between women and men. Because men have generally more difficulty than women at processing and expressing emotions, 39 use of the symptom count version (number of FA criteria met) may be more useful than the diagnostic version (diagnosis) for men.
We did not confirm the YFAS discriminant validity with cognitive restraint found in the US validation study, 21 probably because of cultural differences or differences in samples (by including young students, our participants may have had higher cognitive restraint and lower BMI).
This study has a number of limitations, including use of self-administered questionnaires (future studies should first design and validate a semistructured diagnostic instrument to test the sensitivity and specificity of the YFAS 2.0), use of self-report for height (often overestimated) and weight (often underestimated), no assessment of the test-retest reliability of the scale, and use of the YFAS 2.0 in a nonclinical population. Future research should test its psychometric properties in patients with eating disorders or with diseases potentially associated with FA (i.e., obesity, diabetes, hypertension, or other metabolic syndrome risk factors).
By validating the French version of the YFAS 2.0, this study provides an opportunity to study the food addiction phenotype and its associated factors in greater depth. Before including FA in the international classification of diseases—a subject that is still hotly debated—the similarities of this phenotype with other addictions in terms of disease course, phenomenology, comorbidity, neurobiological mechanisms, and response to treatment must be identified, as well as its specificities. 40 If it is included, it must then be determined whether FA should be conceptualized as a substance-related disorder, 41 as a behavioural addiction, 42 or as a “mixed” substance-related and behavioural addiction. This could have important practical implications, such as choosing the most appropriate therapeutic strategy for a given patient (including psychotherapy and substitution or substance replacement therapy) and developing effective harm reduction programs or even public health policies. It also highlights the importance of investigating each patient’s mental representations and values associated with food, which may explain how and why an individual chooses food rather than another substance or behaviour in a stressful situation.
Conclusions
This study demonstrated that the French version of the YFAS 2.0 is a reliable tool for assessing FA symptoms based on the DSM-5 criteria for substance-related and addictive disorders. We demonstrated that the YFAS 2.0 can be used either to diagnose FA or to assess the number of FA symptoms in a nonclinical sample. Future studies should test the validity of the YFAS 2.0 in other nonclinical and clinical samples. The assessment of a food addiction phenotype using this DSM-5–based questionnaire is an important preliminary step in identifying its risk factors and prior to discussing the pro and cons of considering FA as an addictive disorder. To contribute to this debate, future studies should investigate FA in patients who experience harm related to their FA, including obesity and other metabolic syndrome risk factors or metabolic complications (e.g., type 2 diabetes, hypertension, dyslipidemia, atherosclerosis, stroke, or coronary heart disease).
Footnotes
Acknowledgments
We thank Manon Barbet and Cynthia Beheydt for assistance with data collection. We thank William Revelle for helping us in using the psych statistical package. We thank Elizabeth Yates for correcting the English version of the article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Brunault reports personal fees from Lundbeck, personal fees from Astra-Zeneca, personal fees from DNA Pharma, outside the submitted work; Dr Ballon reports personal fees from Lundbeck, personal fees from Astra-Zeneca, personal fees from DNA Pharma, outside the submitted work; and the other authors declare that they have no conflict of interest to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
