Abstract
Objective:
A systematic review was conducted to examine the efficacy, tolerability, and acceptability of asenapine compared with other antipsychotics in the treatment of psychotic disorders.
Methods:
Four databases, 8 trial registries, and conference presentations were searched for randomized clinical trials of asenapine versus any comparator for the treatment of any psychotic illness. Primary outcome measures were changes in the Positive and Negative Syndrome Scale (PANSS) total score and the incidence of withdrawal due to adverse effects.
Results:
Eight randomized clinical trials, encompassing 3765 patients, that compared asenapine with placebo (n = 5) and olanzapine (n = 3) were included. No differences were found between asenapine and olanzapine in terms of changes to PANSS total or PANSS negative subscale scores. Patients taking asenapine were more likely to experience worsening schizophrenia and/or psychosis than were those taking olanzapine. No differences were found between asenapine and olanzapine in rates of discontinuation due to adverse drug reactions or lack of efficacy, but those taking asenapine had higher rates of withdrawal for any reason than those taking olanzapine. Asenapine caused less clinically significant weight gain or increases in triglycerides than olanzapine and was more likely to cause extrapyramidal symptoms than olanzapine. In comparison to placebo, either no difference or superiority was demonstrated in favour of asenapine on all efficacy measures.
Conclusion:
The current evidence is limited, as asenapine has been compared only with placebo or olanzapine. In the randomized clinical trials analysed, asenapine was similar or superior to placebo and similar or inferior to olanzapine on most efficacy outcomes. While asenapine demonstrated fewer adverse metabolic outcomes than olanzapine, rates of extrapyramidal symptom–related adverse effects were higher.
Affecting approximately 1% of the population, schizophrenia is associated with significant social and occupational dysfunction. 1 Various antipsychotic agents have been developed since the 1950s that have radically improved treatment of the positive symptoms of this disorder. 2 However, negative and cognitive symptoms are more difficult to treat and respond poorly to currently available treatment options. 2
While several new antipsychotics (such as asenapine, lurasidone, and iloperidone) have been developed in recent years, data are lacking that compare the efficacy and safety of these agents with established therapies in the treatment of psychotic disorders, making the decision to use the new agents difficult.
Asenapine (ASP) is the only antipsychotic available on the Canadian market for sublingual use, allowing for a more rapid onset of action than antipsychotics that are absorbed enterally. 3,4 This could allow for a faster de-escalation of acute psychosis and improve adherence with medication in patients who, for a variety of reasons, may be unwilling to swallow medications. 5 ASP is also of interest due to its broad range of receptor affinities, possibly enhancing its efficacy against negative and cognitive symptoms of schizophrenia. 6
Available reviews on this topic are limited in several ways. Szegedi et al 7 conducted a meta-analysis of placebo-controlled trials of 6 weeks’ duration and a multiple treatment meta-analysis to inferentially compare the relative efficacy of ASP to other antipsychotics, although direct comparisons may or may not have been performed. 8 This meta-analysis did not study the long-term safety and efficacy of ASP, and a direct comparison between ASP and other agents was not provided. Instead, the investigators incorporated their results for ASP into a previously published review by Leucht et al. 9 in order to compare its efficacy with that of other available antipsychotics. 7 A systematic review and meta-analysis by DeHert et al. 10 compared ASP, iloperidone, lurasidone, and paliperidone for metabolic adverse effects only. Leucht et al. 11 conducted a multiple-treatments meta-analysis including both direct and indirect comparisons of 15 different antipsychotics in patients requiring acute treatment. This review excluded the patients with primarily negative symptoms of schizophrenia, patients with concurrent medical illnesses, patients with treatment refractory disease, and those who were clinically stable. 11
The current review was conducted to examine the efficacy, tolerability, and acceptability of ASP in comparison to other currently available antipsychotics in the short- and long-term treatment of both acute and clinically stable psychotic disorders, in order to identify patient populations most likely to benefit from this medication.
Methods
Protocol and Registration
Methods of this review were specified in advance, and the protocol was registered with PROSPERO (CRD42014008970).
Search Strategy
Medline, EMBASE, PsychInfo, the Cochrane Central Register of Controlled Trials, and the Cochrane Schizophrenia Group Trials Registry were searched (2013 Oct 13) using the terms asenapine, asenapine maleate, Saphris®, and Sycrest®. No limits were placed on this search, and there were no language restrictions. Eight clinical trial registries and electronically available conference materials from the American Psychiatric Association, World Psychiatric Association, and Canadian Psychiatric Association (2009-2013) were searched for unpublished literature. Clinical experts, and the manufacturer of ASP, Lundbeck Canada, were also contacted to request unpublished trials. Reference lists of included and excluded articles were hand searched for randomized clinical trials matching the inclusion criteria.
Eligibility Criteria
Randomized clinical trials, regardless of publication status or language, were eligible for inclusion. We included patients of any age or disease severity (meeting criteria for the diagnosis of acute or chronic schizophrenia, psychosis, schizoaffective disorder, schizophreniform disorder, and other psychotic disorders as defined by the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition [DSM-IV], DSM-IV Text Revision [DSM-IV-TR], and/or ICD-10 Classification of Mental and Behavioural Disorders). Relevant interventions were ASP (any dosage form and dosing regimen) compared with any typical or atypical antipsychotic (any dosage form or dosing regimen) or placebo (PLC). The primary efficacy outcome measure was a change from baseline in the Positive and Negative Syndrome Scale (PANSS) total score. The primary safety outcome was the rate of discontinuation due to adverse effects. A list of secondary outcome measures can be found in Table 1.
Secondary and post hoc outcomes.

PANSS = Positive and Negative Syndrome Scale.
Data Extraction
Data extraction was performed in duplicate by 2 authors (C.O. and S.D.). When trials used both mixed model of repeated measures and last observation carried forward to impute missing data, the former was used; if only last observation carried forward was used for missing data imputation, this was used as the data source. For trials in which patients were assigned to 5-mg BID or 10-mg BID arms, the 10-mg BID arm was selected for primary comparison, as this is the maximum recommended dose of ASP for the indications in question. For trials that included more than 2 arms, only data from the arms for which the trial was powered were extracted for this review (to avoid unit-of-analysis errors). 12
Statistical Analysis
A random effects model was used to combine outcomes, using Review Manager 5.2. 13 Odds ratios with 95% confidence intervals were calculated for dichotomous outcomes, and weighted mean difference was used to report continuous outcomes. Results were subdivided based on the comparator. We used the I 2 statistic in Review Manager to assess heterogeneity, as described in the Cochrane Handbook. 14
Risk of Bias Assessment
The risk of bias from the included trials was assessed using the Cochrane risk of bias assessment tool. 14 All trials were randomized and double-blinded, but methods of randomization and allocation concealment were not mentioned. Additional sources of bias include the fact that all included articles were written in English and were sponsored by the manufacturers of ASP. Reporting bias was also possible as all trials reported frequent adverse reactions, omitting rare but potentially serious adverse effects. In addition, very few of the trials obtained through the initial search were available in published form, and very few of the unpublished trials were obtained from either the manufacturer or the authors. Publication bias may be present, and we speculate that some negative trials may be missing from our review. The decision was made by the authors not to include a funnel plot as it is very difficult to show true asymmetry when fewer than 10 articles are included. 15 There was also significant attrition in all studies, as shown by high withdrawal rates. The majority of the authors were current employees or representatives of the manufacturer of ASP. See Figure 1 for more details.
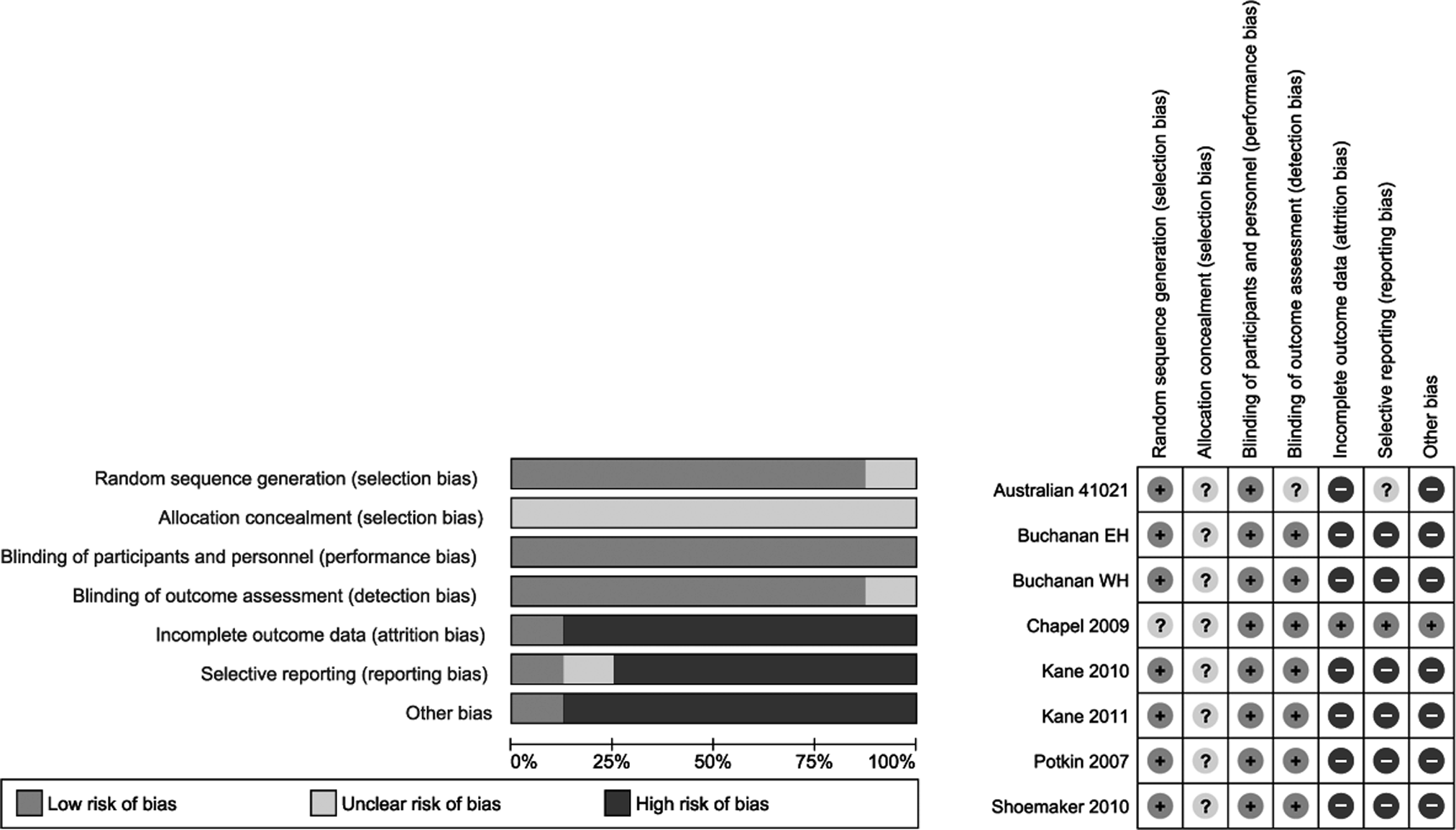
Cochrane risk of bias assessment.
Results
The search yielded 742 citations, of which 8 articles (3765 patients) met inclusion criteria. 16 –23 See the flow diagram in Figure 2 for additional details regarding trial selection and Table 2 for characteristics of the included studies. Table 3 lists the excluded trials and reasons for exclusion. Of the 8 trials included, 5 were placebo-controlled and 3 compared ASP with olanzapine (OZP). No unpublished studies were obtained from either the manufacturer or the expert authors. Data for the unpublished study 41021 were obtained through the FDA website and an Australian government document. All patients included had a diagnosis of either DSM-IV schizophrenia or schizoaffective disorder.
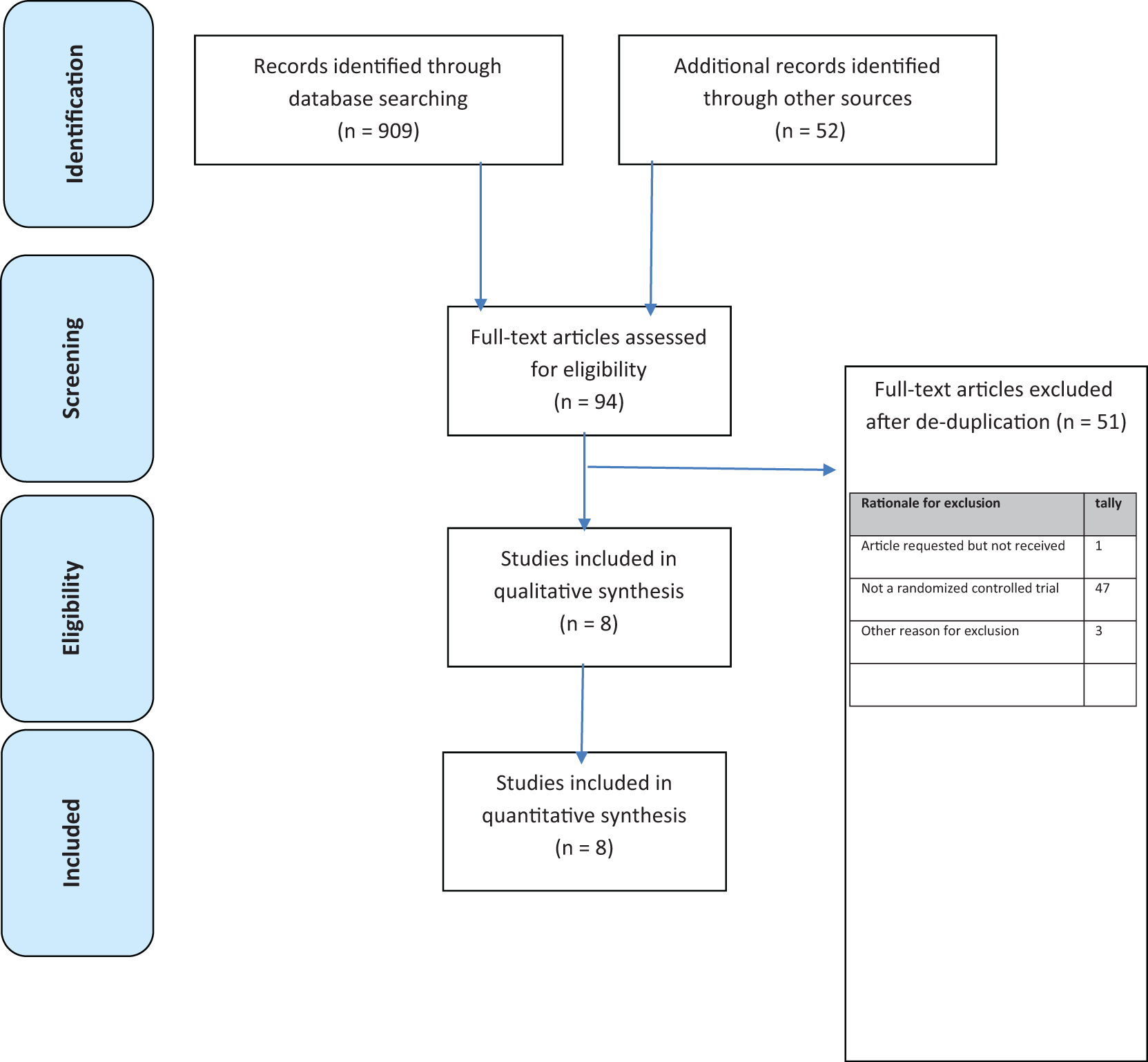
Study selection.
Characteristics of included trials.
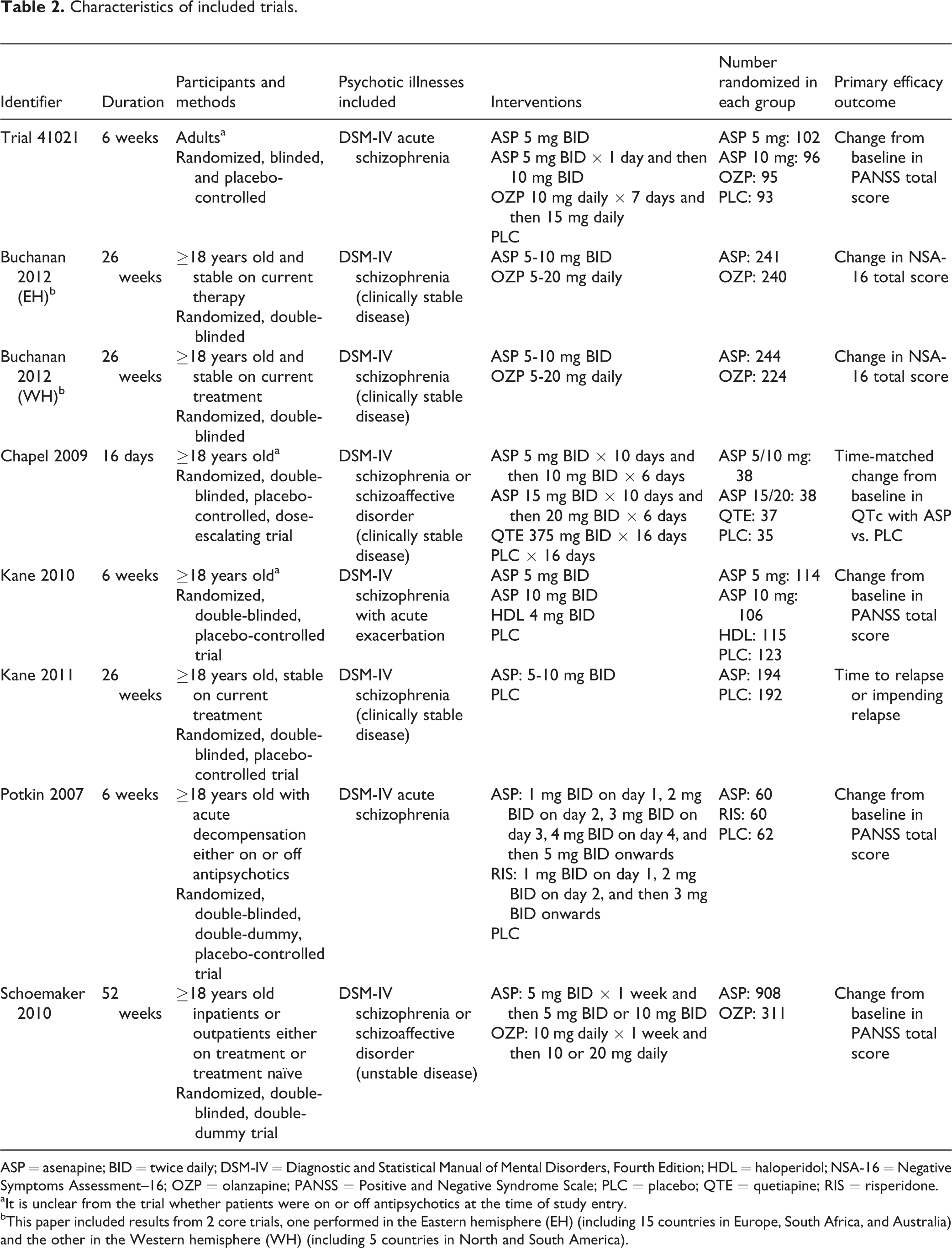
ASP = asenapine; BID = twice daily; DSM-IV = Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition; HDL = haloperidol; NSA-16 = Negative Symptoms Assessment–16; OZP = olanzapine; PANSS = Positive and Negative Syndrome Scale; PLC = placebo; QTE = quetiapine; RIS = risperidone.
aIt is unclear from the trial whether patients were on or off antipsychotics at the time of study entry.
bThis paper included results from 2 core trials, one performed in the Eastern hemisphere (EH) (including 15 countries in Europe, South Africa, and Australia) and the other in the Western hemisphere (WH) (including 5 countries in North and South America).
Trials excluded from the review with reasons for exclusion.

RCT = randomized clinical trial. Full text screening was performed in duplicate by C.O. and S.D.
Efficacy Outcomes
PANSS
Patients were considered to be PANSS responders if they achieved a ≥30% decrease in their PANSS total score from baseline at the end of the study. Results favoured ASP over PLC in terms of both change in PANSS total score (OR –8.98; 95% CI, –12.73 to –5.23) (Figure 3) and percentage of PANSS responders (OR 1.88; 95% CI, 1.24 to 2.84), No differences were found between ASP and PLC in terms of changes to the 3 PANSS subscale scores. No differences were noted between ASP and OZP in terms of changes to PANSS total (Figure 3), nor were there any differences between ASP and OZP in changes to PANSS negative subscale scores. In addition, ASP was found to be inferior to OZP regarding changes to PANSS positive and PANSS psychopathology subscale scores, with differences of 0.94 (95% CI, 0.46 to 1.42) and 1.38 (95% CI, 0.51 to 2.25), respectively. Both the ASP vs. PLC and the ASP vs. OZP comparisons demonstrated considerable heterogeneity (82% and 78%, respectively) for the outcome change in PANSS total score.
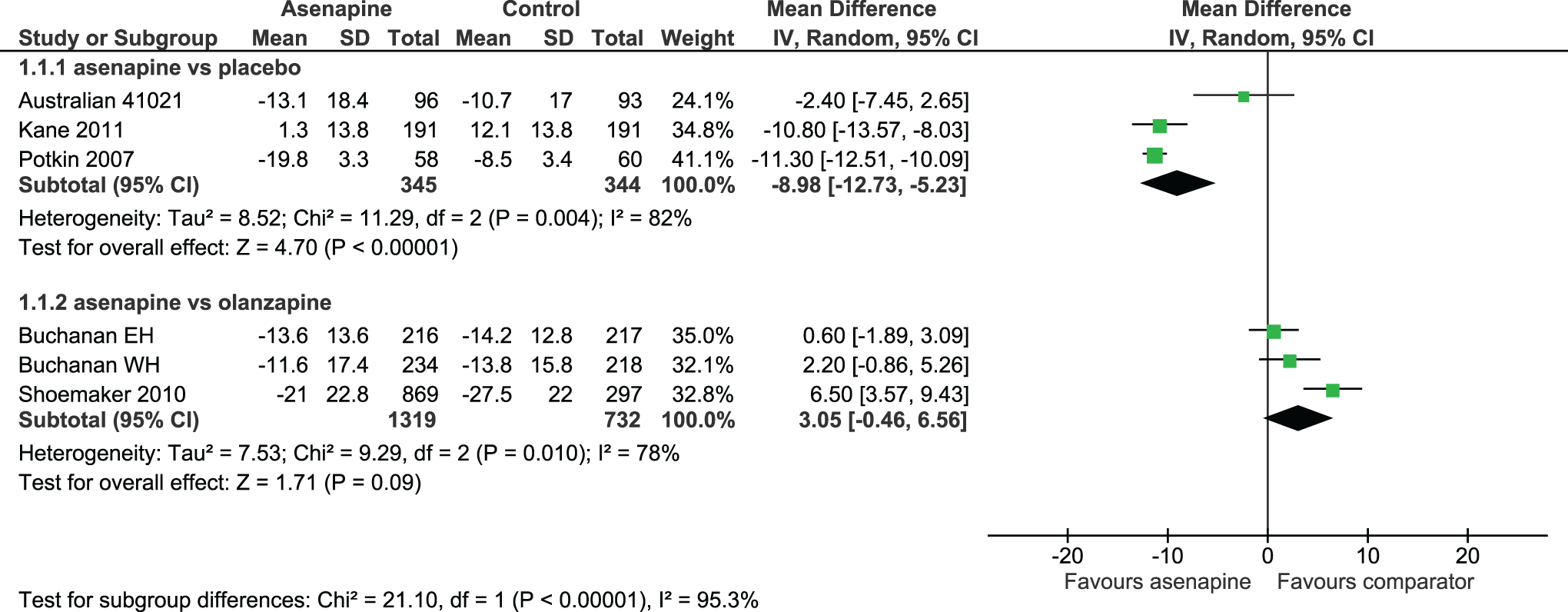
Changes in PANSS total score.
Clinical Global Impression (CGI)
Schoemaker et al. 20 and Kane et al. 17 considered CGI improvement scale (CGI-I) responders to be those with a CGI-I score of 1 (very much improved) or 2 (much improved) at study end. ASP was superior to PLC on improvement in CGI severity scale (CGI-S) scores, while no difference was observed between ASP and OZP in this outcome. There was no difference in the rates of CGI-I responders between those randomized to ASP and those randomized to PLC. ASP was inferior to OZP in this outcome (OR 0.66; 95% CI, 0.52 to 0.84).
Worsening schizophrenia and/or psychosis
Rates of worsening schizophrenia and/or psychosis did not differ between ASP and PLC. However, compared with those randomized to OZP, more patients randomized to ASP experienced this outcome (OR 1.38; 95% CI, 1.05 to 1.82). Refer to Table 4 for a summary of efficacy results.
Summary table of efficacy outcomes.

ASP = asenapine; BID = twice daily; CGI-I = Clinical Global Impression–improvement scale; CGI-S = Clinical Global Impression–severity scale; OR = odds ratio; OZP = olanzapine; PANSS = Positive and Negative Syndrome Scale; PLC = placebo; WMD = weighted mean difference.
Withdrawals
Rates of either withdrawal for any reason or withdrawal due to adverse effects were no different between patients receiving ASP and those receiving PLC (Figure 4). ASP-treated patients had lower rates of withdrawal due to lack of efficacy than those taking PLC (OR 0.37; 95% CI, 0.20 to 0.69). When ASP was compared with OZP, no difference was shown for rates of discontinuation due to adverse drug reactions (ADRs) (Figure 4) or lack of efficacy, but ASP had higher rates of overall withdrawal (OR 2.06; 95% CI, 1.71 to 2.49).
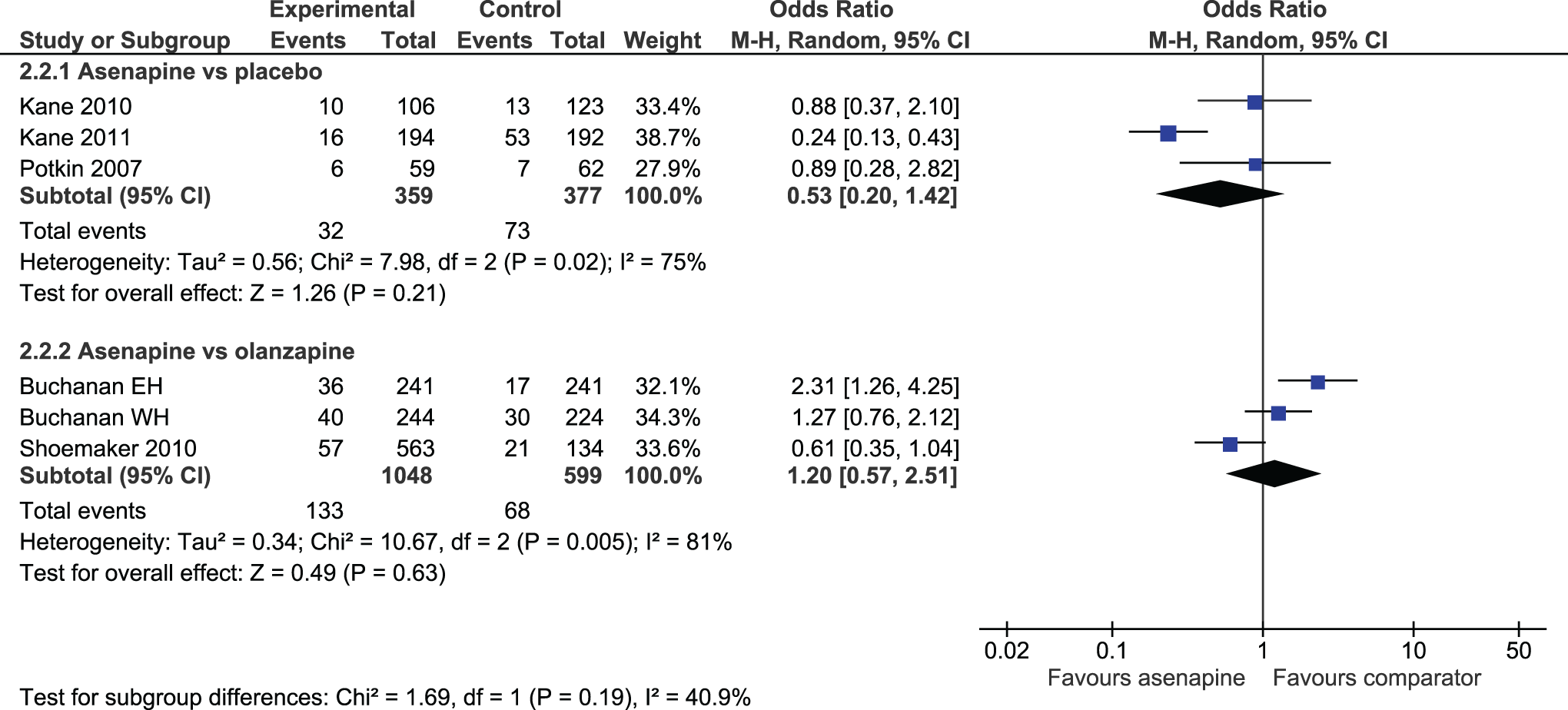
Rates of withdrawal due to adverse effects.
Safety and Tolerability Outcomes
Adverse effects
No differences were found between ASP and either PLC or OZP in rates of “any ADR.” Higher rates of serious ADRs were demonstrated in those receiving ASP compared with OZP (OR 1.84; 95% CI, 1.36 to 2.47). Trials did not specify which serious adverse effects occurred.
Effect on sleep
Rates of insomnia were comparable between ASP and PLC, whereas patients randomized to ASP were more likely to report this adverse effect than those randomized to OZP (OR 1.46; 95% CI, 1.13 to 1.90). Rates of sedation were similar between ASP and both comparators.
Metabolic adverse effects
The incidence of clinically significant weight gain was greater with ASP compared with PLC (OR 3.58; 95% CI, 1.13-11.31). Those taking ASP were less likely to have reported this outcome than those randomized to OZP (OR 0.35; 95% CI, 0.27 to 0.44). Changes from baseline in triglyceride values were similar between ASP and PLC. Based on results from 1 trial, patients receiving ASP had less change in triglyceride than OZP-treated patients (OR –0.45; 95% CI, –0.71 to –0.19). No significant differences were found between ASP and either comparator in changes in fasting blood glucose or new-onset National Cholesterol Education Program metabolic syndrome. 16
Extrapyramidal symptoms (EPS)
No differences were found between ASP and PLC in reported rates of EPS. Patients randomized to ASP were more likely to experience EPS and akathisia than were patients taking OZP: OR 2.06 (95% CI, 1.38 to 3.06) and OR 2.27 (95% CI, 1.45 to 3.55), respectively. No differences were noted between groups in changes to any of the EPS rating scales: Simpson Angus scale, Abnormal Involuntary Movement Scale, Extrapyramidal Symptoms Rating Scale, or Barnes Akathisia Rating Scale.
Patient Acceptance and Quality of Life Outcomes
Very few trials measured patient-reported outcomes, such as improvements in quality of life and subjective well-being while receiving neuroleptics. Schoemaker et al. 20 reported both patients’ and investigators’ subjective impressions of improvement of the patients’ disease condition in comparison to when they were taking previous neuroleptics. Those taking ASP were less likely to be considered “much improved” by the investigator than those receiving OZP (OR 0.65; 95% CI, 0.45 to 0.93).
Refer to Table 5 for a summary of additional clinically relevant results.
Summary of clinically important outcomes.
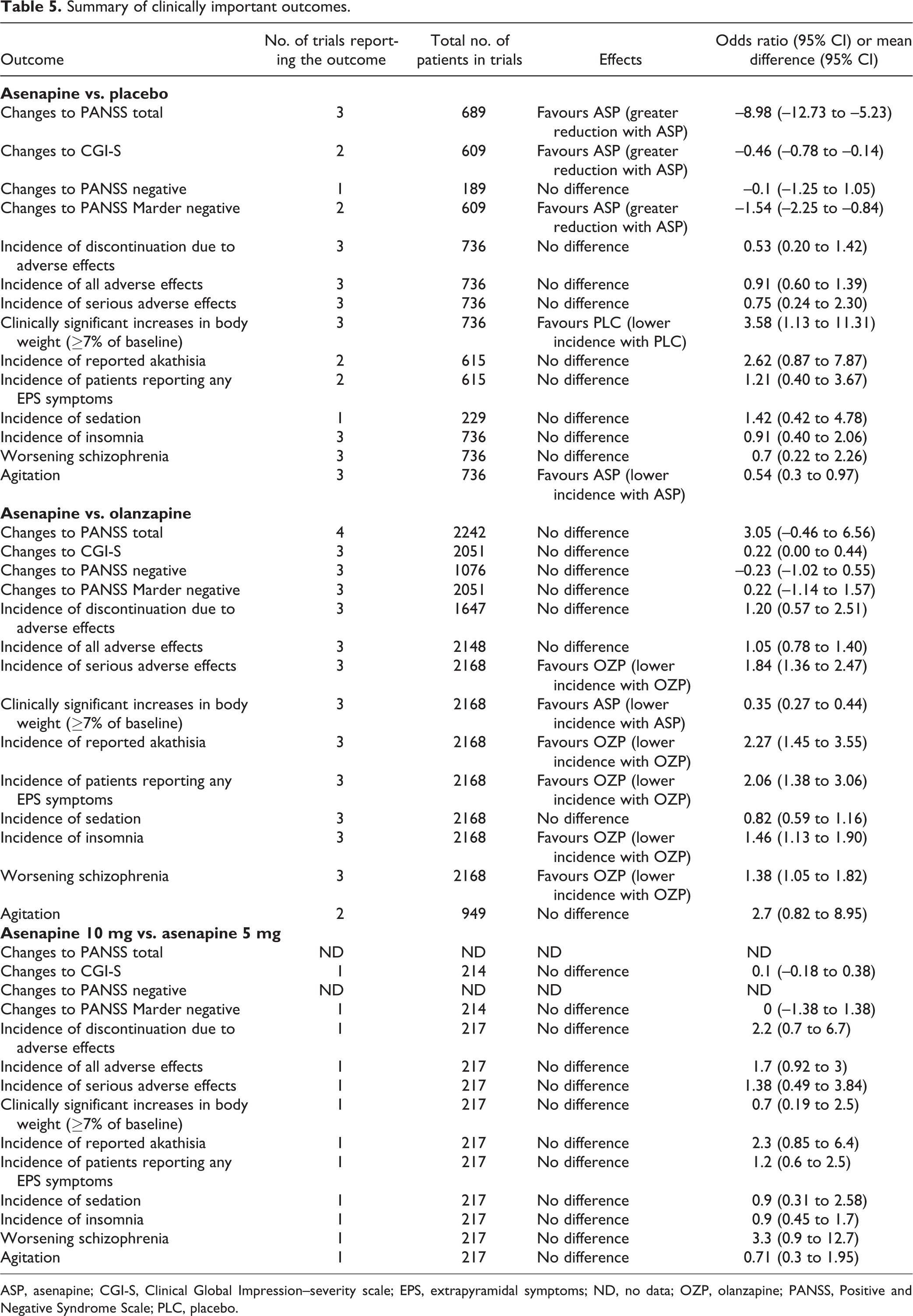
ASP, asenapine; CGI-S, Clinical Global Impression–severity scale; EPS, extrapyramidal symptoms; ND, no data; OZP, olanzapine; PANSS, Positive and Negative Syndrome Scale; PLC, placebo.
Additional Analyses
Two post hoc subgroup analyses were conducted, one comparing ASP 5-mg BID and 10-mg BID dosing and the other including only long-term trials (those ≥26 weeks long).
ASP 5 mg BID vs. 10 mg BID
No statistically significant differences were found for efficacy, rates of withdrawal, or tolerability outcomes between the 2 doses.
Long-term trials
Four trials were included in this analysis. 16,18,20 Trials with PLC comparator showed comparable results when limited to long-term trials. All trials with OZP comparator were long-term trials, so there was no change to those results.
Discussion
This review demonstrates that the current evidence for the use of ASP in psychotic disorders is limited. A total of 8 trials were included in this review: 5 were placebo-controlled and 3 compared ASP with OZP.
The results demonstrate that ASP 10 mg BID was superior to PLC on several efficacy outcomes but was not superior to OZP. ASP’s purported enhanced activity against negative and cognitive symptoms of schizophrenia was not confirmed by the results of this review. Nor was enhanced adherence with ASP demonstrated. In addition, patients taking ASP were more likely to withdraw from clinical trials, suggesting that adherence to ASP in the clinical setting may be inferior to OZP.
Second-generation antipsychotics have a documented adverse impact on the patient’s metabolic profile. 2 Results from this review favoured ASP over OZP in this regard, which suggests that ASP may be advantageous in patients at higher risk of cardiovascular morbidity and mortality.
ASP demonstrated a similar propensity towards sedation as both PLC and OZP, a surprising finding considering the well-known sedative effects of OZP. A lack of power to detect a difference in rates of sedation may be at play, reducing our ability to detect a significant difference when results were combined for this meta-analysis.
Rates of EPS were lower with OZP than ASP and comparable between ASP and PLC. The authors believe that these findings may be due to the number and duration of trials included in this review. Some forms of EPS may occur months to years after the initiation of an antipsychotic. All trials comparing ASP and OZP were long-term trials, while only 1 placebo-controlled trial was a long-term trial. It is possible that with time, a significant difference favouring PLC would have been detected for this adverse effect; however, the majority of the placebo-controlled studies included were likely too short in duration to reveal a significant difference in this outcome.
No conclusions can be drawn about the impact of oral hypoesthesia, a common adverse effect reported with ASP, 5 since only 1 placebo-controlled trial reported on this outcome. 17
The subgroup analysis comparing ASP 5 mg BID and 10 mg BID demonstrated no differences in efficacy, withdrawals, or tolerability outcomes between the 2 doses. Although not powered for this comparison, these findings suggest that the efficacy of ASP is not dose-dependent and that dose escalation is not warranted.
The strengths of this review include the fact that it was conducted in accordance with PRISMA (Table 6) and Cochrane guidelines and was registered a priori. 14,24 The breadth of the search strategy ensured that all relevant articles matching the inclusion criteria were found. All study selection and data extraction were done in duplicate, thus minimizing the risk of bias. 25
PRISMA checklist.

There are some limitations of this review. All included trials had strict inclusion and exclusion criteria such as the exclusion of those with concurrent psychiatric illnesses, including substance abuse, which may limit generalizability of the results to a broad patient population. All patients were included in this meta-analysis regardless of whether patients were treated as inpatient or outpatient and whether they were in florid psychosis or had stable disease. This may explain the heterogeneity observed in this review, such as in the change in PANSS total score with an I 2 of 95.3%. Trials handled missing data inconsistently, resulting in the combination of results obtained from either mixed model of repeated measures or last observation carried forward in the meta-analysis. This contributes further to the heterogeneity of results. Thirteen potentially eligible trials were unobtainable from authors and/or the manufacturer of ASP despite repeated requests. This large source of unknown data could have a significant effect on the outcomes reported in this review.
Conclusions
Insufficient evidence is available to make recommendations on preference of ASP over other antipsychotic medications in the treatment of psychotic disorders. Results from the included trials suggest that ASP is either comparable to or inferior to OZP for the majority of efficacy, tolerability, and acceptability outcomes. ASP demonstrated a safer metabolic profile than OZP with a trend towards less sedation; ASP was more likely than OZP to lead to EPS. With this in mind, ASP may be a treatment choice for those who cannot tolerate the sedative effects of OZP, such as drivers and night shift workers, as well as those who are at high risk of metabolic adverse effects.
ASP should also be considered an option in patients who will be able to follow the prescribed administration instructions.
There is a need for well-designed, randomized clinical trials that are adequately powered to compare the efficacy and acceptability of ASP with active comparators other than OZP. The outcome measures that are clinically relevant and merit further study include quality of life, cost, adherence with medication, and length of hospitalization. The comparisons of different doses of ASP may be beneficial in order to determine whether dosing flexibility affects treatment efficacy and safety.
Footnotes
Acknowledgments
We thank our clinical librarian, Sandra McKeown, for helping to establish a search strategy and library technician, Marshall Lemon, for help with article acquisition. We also thank our psychiatry pharmacists, Boris Tong, Bradley Linton, and Sharon Lawrence, for their clinical expertise during this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our project was funded by London Health Sciences Centre, Pharmacy Department.
