Abstract
Background:
The Canadian Network for Mood and Anxiety Treatments (CANMAT) conducted a revision of the 2009 guidelines by updating the evidence and recommendations. The scope of the 2016 guidelines remains the management of major depressive disorder (MDD) in adults, with a target audience of psychiatrists and other mental health professionals.
Methods:
Using the question-answer format, we conducted a systematic literature search focusing on systematic reviews and meta-analyses. Evidence was graded using CANMAT-defined criteria for level of evidence. Recommendations for lines of treatment were based on the quality of evidence and clinical expert consensus. This section on “Special Populations” is the sixth of six guidelines articles.
Results:
Recent studies inform the treatment of MDD in children and adolescents, pregnant and breastfeeding women, women in perimenopause or menopause, and the elderly. Evidence for efficacy of treatments in these populations is more limited than for the general adult population, however, and risks of treatment in these groups are often poorly studied and reported.
Conclusions:
Despite the limited evidence base, extant data and clinical experience suggest that each of these special populations can benefit from the systematic application of treatment guidelines for treatment of MDD.
Keywords
In 2009, the Canadian Network for Mood and Anxiety Treatments (CANMAT), a not-for-profit scientific and educational organization, published a revision of evidence-based clinical guidelines for the treatment of depressive disorders. 1 CANMAT has updated these guidelines in 2016 to reflect new evidence in the field.
The scope of these guidelines remains the management of adults with unipolar major depressive disorder (MDD), with a target audience of psychiatrists and mental health specialists. This section covers the treatment of depressive disorders in children and adolescents, women in the perinatal and menopausal stages, and the elderly, recognizing that these life stages carry distinct challenges for treatment. The section is 1 of 6 guidelines articles; other sections expand on principles of care and psychological, pharmacological, neurostimulation, and complementary and alternative medicine treatments. Treatment recommendations in this section will emphasize differences from the general guidelines for adults. These recommendations are presented as guidance for clinicians who should consider them in context of individual patients and not as standards of care.
Methods
The full methods have been previously described, 2 but in summary, relevant studies in English published from January 1, 2009, to December 31, 2015, were identified using computerized searches of electronic databases (PubMed, PsychInfo, Cochrane Register of Clinical Trials), inspection of bibliographies, and review of other guidelines and major reports. Each recommendation includes the level of evidence for each graded line of treatment, using specified criteria (Table 1). The level of evidence criteria now reflect the primacy of meta-analysis because of its increasing use in the evaluation of evidence.
Criteria for Level of Evidence and Line of Treatment.

RCT, randomized controlled trial.
aNote that Level 1 and 2 Evidence refer specifically to treatment studies in which randomized comparisons are available. Recommendations involving epidemiological or risk factors primarily arise from observational studies, and hence the highest level of evidence is usually Level 3. Higher order recommendations (e.g., principles of care) reflect higher level judgement of the strength of evidence from various data sources and therefore are primarily Level 4 Evidence.
bClinical support refers to application of expert opinion of the CANMAT committees to ensure that evidence-supported interventions are feasible and relevant to clinical practice. Therefore, treatments with higher levels of evidence may be downgraded to lower lines of treatment due to clinical issues such as side effects or safety profile.
In special populations, consideration of harm becomes a more prominent concern than in general adult populations, because of the unique vulnerabilities of these developmental windows. The recommendations for various treatment approaches therefore reflect an attempt to balance treatment benefit and potential risks in a way that is acceptable to clinicians and patients. As studies examining harm in the treatment of MDD are often of low quality, 3 the confidence of the treatment recommendations in these groups may be lower than in sections focused on general adult populations. The following sections provide an overview of the treatment challenges and options for children and adolescents; pregnant, postpartum, and menopausal women; and the elderly.
Childhood and Adolescence: A Unique Neurodevelopmental Period
In 2014, 11.4% of American youth aged 12 to 17 years reported at least 1 major depressive episode (MDE) in the past year. 4 Canadian statistics are limited, but 2012 Statistics Canada data found that 8.2% of surveyed youth aged 15 to 24 years reported mood disorders. 5 Most of the randomized-controlled trials (RCTs) of youth assess antidepressant effectiveness in 12- to 18-year-old participants, despite the rapid maturational changes during this period and the fact that a 12-year-old is developmentally distinct from an 18-year-old. 6 Some studies also combine children (<12 years) and adolescents (12-18 years); when recommendations are intended for a specific age group (pediatric or adolescent), this is explicitly stated.
6.1. What is the Initial Approach to a Child or Adolescent with Suspected Depression?
Use of standardized depression screening tools is recommended for assessing children and youth; different screening tools exist for these age groups. 7,8 When feasible, health care providers should use a semistructured approach to diagnostic assessment of children and adolescents who screen positive for MDD (e.g., Kiddie Schedule for Affective Disorders [K-SADS]). Given that a semistructured interview requires both time and training, this may be difficult in some settings but should be attempted (e.g., by appointing trained personnel for this purpose). Although diagnostic criteria for MDD are the same for children and adolescents, presenting symptoms may differ by age group; adolescents typically report more hypersomnia, fewer appetite and weight changes, and fewer psychotic symptoms than children. 9 As such, the patient’s age should be taken into account when assessing children/youth, selecting treatments, and tracking response. 10 Best clinical practice includes the use of various sources for diagnosis and symptom severity assessments, including a clinical interview and auxiliary information (i.e., from parents, teachers).
Supportive clinical care may be sufficient to reduce depression symptoms of a mild MDE. Supportive approaches include psychoeducation, active and empathetic listening, and lifestyle advice, including the benefits of good sleep hygiene, proper eating habits, and exercise. 11
6.2. Is Psychotherapy an Effective Treatment for Depressed Children/Adolescents?
Previous meta-analyses found that psychotherapy, largely in the form of cognitive-behavioural therapy (CBT), confers modest antidepressant effects in depressed children/adolescents relative to comparison conditions (e.g., waitlist, minimally-treated, active placebo), with more evidence for its use in adolescents.
12,13
A recent review of psychotherapeutic interventions in children/adolescents (52 studies,
The effectiveness of Internet-based psychotherapeutic interventions in children/adolescents has also been explored. One meta-analysis found no significant benefit to Internet-based interventions in 7- to 25-year-olds on depression symptoms (although anxiety was reduced) compared with waitlist controls. 17 Others found that computer/Internet-based CBT in children and youth was more effective than comparison conditions (e.g., waitlist, no treatment) in alleviating depression symptoms, particularly in adolescents. 18,19 As such, these interventions may be a promising treatment alternative when in-person/face-to-face treatment is not feasible or available. Most Internet-based interventions have a considerable component of parental and/or teacher involvement, as well as guidance from a therapist. Therefore, Internet-based therapies may be better conceived as a piece within a therapeutic intervention strategy rather than a stand-alone approach.
A Cochrane meta-analysis (11 trials,
Table 2 summarizes the treatment recommendations for MDD. In summary, as there is no clear comparative advantage for pharmacotherapy or psychotherapy in treating children/youth with non-treatment-resistant MDD, psychotherapy should be the first line of treatment in mild to moderate MDD. CBT and IPT should be considered ahead of other types of psychotherapies in treating depressed pediatric and adolescent populations.
Treatment of Major Depressive Disorder in Children/Youth.
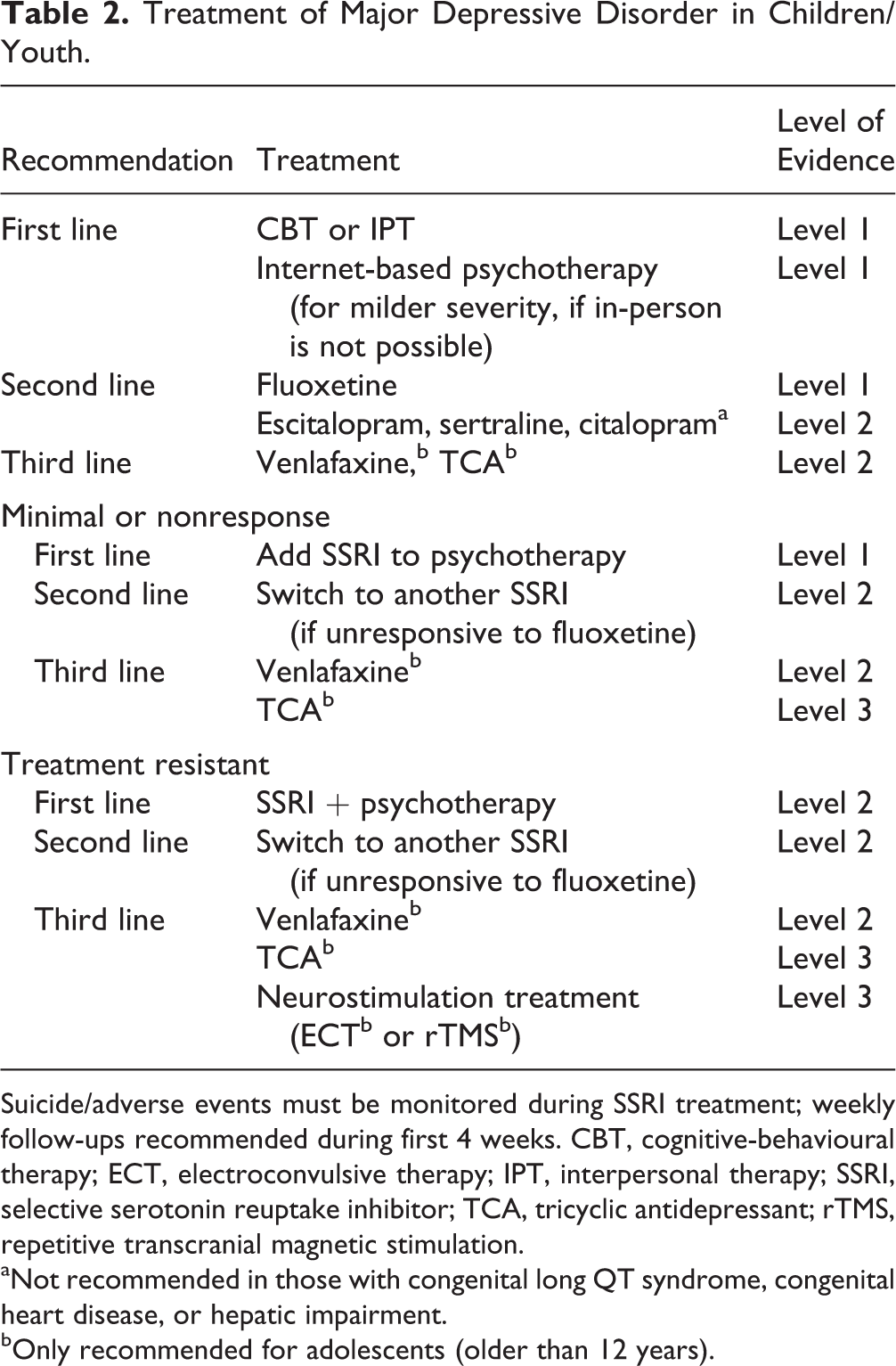
Suicide/adverse events must be monitored during SSRI treatment; weekly follow-ups recommended during first 4 weeks. CBT, cognitive-behavioural therapy; ECT, electroconvulsive therapy; IPT, interpersonal therapy; SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressant; rTMS, repetitive transcranial magnetic stimulation.
aNot recommended in those with congenital long QT syndrome, congenital heart disease, or hepatic impairment.
bOnly recommended for adolescents (older than 12 years).
6.3. What Antidepressant Medication Should Be Used in Depressed Children/Adolescents?
Selective serotonin reuptake inhibitors (SSRIs) are the most extensively studied medications for the treatment of MDD in children/youth. A Cochrane review (19 trials,
Tricyclic antidepressants (TCAs) are not useful in treating depression in children, and there is only marginal evidence to support their use in adolescents. 28 Monoamine oxidase inhibitors (MAOIs) are not recommended for depressed children/youth because there has been limited assessment of MAOI effectiveness in this population and because of the side effect burden as well as potential for difficulties with the tyramine-free diet.
In summary, if psychotherapy is not accessible, acceptable, or effective, pharmacotherapy should be considered in youth with depressive episodes of moderate severity (Table 2). Pharmacotherapy should be considered as a first-line intervention in more severe cases of depression. Fluoxetine is considered a first-choice antidepressant in children/youth while escitalopram, sertraline, and, to a lesser extent, citalopram are generally considered second-choice antidepressants. Paroxetine is not recommended. TCAs and MAOIs should only be considered in treatment-resistant depression.
6.4. How Should Children/Adolescents Be Monitored following Initiation of Pharmacotherapy?
The United States Food and Drug Administration (FDA) recommends that patients be seen on a weekly basis during the first 4 weeks of treatment, followed by visits every 2 weeks for a month, and then after 12 weeks of treatment to monitor adverse events/suicidality. 29 This is especially true in more severely depressed patients, those with high suicidal ideation, and those experiencing family conflict. 30 The Canadian Psychiatric Association also recommends that appointments or telephone contacts should be scheduled at least weekly within the first month of treatment for children and adolescents. 31 When starting antidepressant pharmacotherapy in youth, the initial dose is generally at the low end of the therapeutic range and continues for a minimum of 4 weeks before a dose increase is considered. If the patient continues to show only a partial response after 12 weeks despite adequate dosing, a change in treatment is warranted. 8,9
6.5. How Long Should Children/Adolescents Be Treated with Pharmacotherapy?
Relatively little is known about antidepressant maintenance strategies in children/adolescents. Based primarily on adult research, maintenance treatment for 1 year or more is recommended in children/youth with a history of at least 2 depressive episodes or 1 severe or chronic episode. 9 In individuals with no MDD history, maintenance strategies should persist for 6 to 12 months. Antidepressant discontinuation should consist of a slow taper and occur during a relatively stress-free time (e.g., summer months).
6.6. How Should Treatment-resistant Depression or Comorbidity Be Approached in Children or Adolescents?
If a child/adolescent is unresponsive to first-line treatment, the possibility of a misdiagnosis (e.g., undetected bipolar disorder, comorbid medical or psychiatric disorder) should be considered prior to a treatment switch. Treatment nonadherence should also be considered, as should psychosocial factors (e.g., bullying, sexual identity concerns, and family conflict).
Based largely on findings from the Treatment of Resistant Depression in Adolescents (TORDIA) study, following an adequate course with an initial SSRI, children/adolescents showing minimal response (<20% decrease in symptoms) should be switched to another SSRI. Although participants in the TORDIA trial were equally responsive to the serotonin and norepinephrine reuptake inhibitor (SNRI) venlafaxine as to another SSRI, venlafaxine was associated with a higher rate of self-harm events in those with higher suicidal ideation; venlafaxine is therefore less preferable than switching to another SSRI. 30 For youth with SSRI-resistant depression, combined treatment (antidepressant + psychotherapy) decreases the number of days with depression and may be cost-effective. 32
There is limited evidence for the use of neurostimulation treatments and other modalities in treating depression in pediatric/adolescent populations. Repetitive transcranial magnetic stimulation (rTMS) may hold some promise, 33 although large-scale randomized, sham-treatment controlled studies are lacking. Similarly, RCTs of electroconvulsive therapy (ECT) in children/adolescents are lacking, although ECT parameters in adolescents exist. 34 Case series indicate that ECT is effective in alleviating depression symptoms in adolescents with treatment-resistant MDD, although some individuals did report long-term cognitive/memory impairments. 35 Given the potential side effect profiles and lack of evidence, ECT is not recommended in children (<12 years of age) and is only recommended with extreme caution in adolescents with treatment-resistant and severe MDD (Table 2).
Finally, the presence of a comorbid psychiatric disorder may complicate treatment. There are sparse data to guide treatment of MDD in the context of psychiatric comorbidity in individuals younger than 18 years. Some limited evidence supports the use of fluoxetine in depressed youth with mild to moderate alcohol use disorders 36 and with oppositional symptoms. 37 In the TORDIA study, remission from depression, regardless of treatment, was associated with a greater reduction in measures of anxiety, attention-deficit/hyperactivity disorder (ADHD), and oppositional symptoms. 38 Although the evidence is limited, treating depression in children/adolescents may reduce comorbid disorder(s) symptoms.
6.7. What Are the Safety Concerns for Antidepressant Medications in Children/Adolescents?
Health Canada has not approved any antidepressant medications for use in individuals younger than 18 years. Fluoxetine is the only antidepressant approved by the FDA for preadolescents (8 years and older), but both fluoxetine and escitalopram are FDA-approved for children 12 years and older.
The FDA issued a black-box warning in 2003 on SSRI use in those younger than 24 years; other regulatory agencies, including Health Canada, followed suit. The Cochrane review of newer generation antidepressants (SSRIs and others) found that median baseline risk of suicide-related outcomes (behaviour and ideation) rose from 25/1000 to 40/1000. 24 These results were consistent with the FDA meta-analysis that showed an ∼1.5- to 2-fold risk of increased suicidal thoughts/behaviours (no suicide deaths reported) for newer antidepressants. 39 While epidemiological data do not demonstrate a relationship between prescriptions of antidepressants and suicide deaths in large populations of youth, 40 a systematic review of observational studies found a higher risk (odds ratio = 1.92) of suicidal acts (suicide and attempted suicide) with SSRI exposure in adolescents but a reduced risk in older age groups. 41 Given that these were observational studies, it is possible that the adolescents with SSRI exposure were more severely depressed and at higher risk of suicidality. While recognizing the risks associated with SSRI use, the consequence of untreated depression in children/adolescents is more likely to result in harm; therefore, treatment with SSRIs may be appropriate with careful monitoring.
Perinatal Depression
Unipolar MDEs occurring during pregnancy and in the first year postpartum are frequently referred to as
Up to 7.5% of women will have a unipolar MDE during pregnancy, and 6.5% will experience one in the first 3 months postpartum. When cases of minor depressive disorder are considered, these rates increase to 18.4% and 19.2%, respectively. 42,43 If left untreated, MDEs can affect infant development, future depression risk, and family and vocational functioning. Timely treatment is therefore essential to optimizing outcomes for women and their families.
6.8. What Are the Principles of Management for Perinatal Depression?
Up to 50% of pregnancies are unplanned. 44 Discussions about a woman’s intent to become pregnant and the safety of selected treatment strategies if a pregnancy (planned or unplanned) occurs should therefore comprise a part of the assessment and documentation of all depressed women of childbearing age.
The treatment of MDD during pregnancy and the postpartum period is marked by a number of unique challenges. These include the known risks of fetal and infant exposure to pharmacologic treatments during pregnancy and lactation, as well as those posed by untreated depression. Unfortunately, the evidence upon which our understanding of these risks is based remains limited. The
6.9. How Should Depression during Pregnancy Be Treated?
Decision making around the treatment of depression during pregnancy must balance the risks associated with fetal medication exposure with those of untreated depression. Left untreated, MDEs during pregnancy are not only associated with poorer nutrition and prenatal medical care, smoking, and recreational substance misuse, 45,46 but also with significant suffering for women. Depression is linked to an increased risk of poor obstetrical outcomes, 47 small neonates for gestational age, 48 neonatal intensive care unit admission, 49 increased rates of neonatal complications, 50 impairments in mother-infant bonding, infant sleep difficulties, 51 mild developmental delays, 52 and cognitive, behavioural, and emotional problems in offspring. 53
The recommendations for MDD in pregnancy are summarized in Table 3. The efficacy of first-line treatments for mild to moderate depression, including CBT and IPT delivered in either individual or group format, is supported by meta-analyses. 54,55 Given the established efficacy of SSRIs as first-line treatments in MDD outside of the perinatal period, citalopram, escitalopram and sertraline are recommended based on efficacy and safety; combination treatment with an SSRI and CBT or IPT can also be considered. Other SSRIs (except paroxetine) and newer antidepressants are less preferred options given the relative absence of reproductive data and limited antenatal clinical use. Despite increased risks of fetal cardiovascular (CV) malformations (outlined below), paroxetine and clomipramine may be discussed with women where there is a compelling reason to consider it, such as a previous good response or ongoing stability on the medication. Doxepin should be avoided during pregnancy given its high rate of passage into breast milk and accompanying complications. MAOIs are not recommended during pregnancy given their propensity to interact with certain analgesic and anaesthetic agents. When MAOIs must be used, early consultation with anaesthesia is recommended.
Treatment of Mild to Moderate Major Depressive Disorder during Pregnancy.
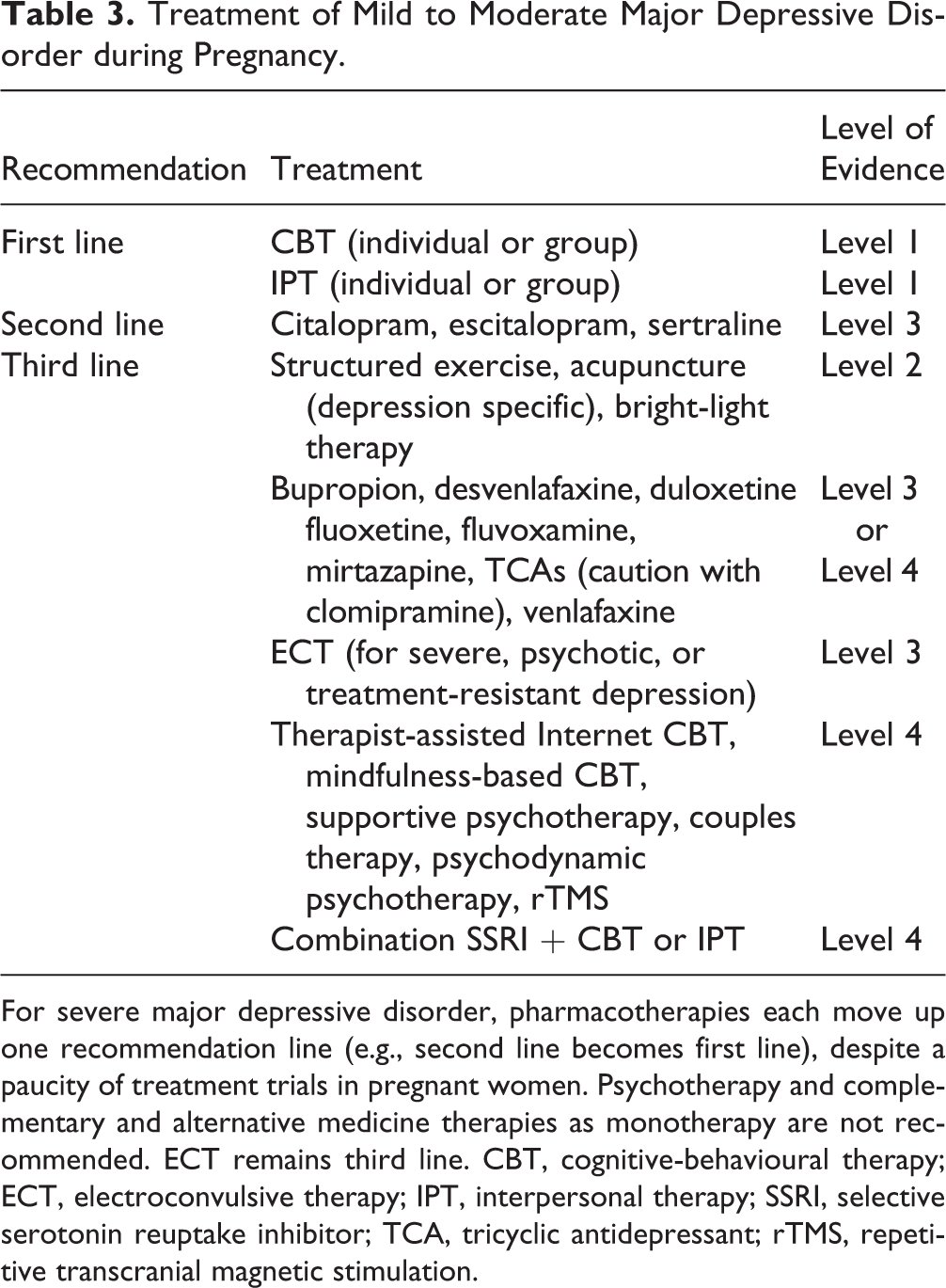
For severe major depressive disorder, pharmacotherapies each move up one recommendation line (e.g., second line becomes first line), despite a paucity of treatment trials in pregnant women. Psychotherapy and complementary and alternative medicine therapies as monotherapy are not recommended. ECT remains third line. CBT, cognitive-behavioural therapy; ECT, electroconvulsive therapy; IPT, interpersonal therapy; SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressant; rTMS, repetitive transcranial magnetic stimulation.
Other treatments, including neurostimulation and complementary and alternative medicine strategies, can also be considered as third-line recommendations. 56 Recognizing the need for rapid treatment during pregnancy, interventions that have previously been effective for that woman may be worth discussing as potential second-line strategies, as long as they are not contraindicated.
In keeping with recommendations in general population samples, the use of antidepressants in the perinatal period should continue until 6 to 12 months after remission in low-risk women, although treatment for longer periods of time should be considered in those at high risk of relapse.
6.10. What Is the Approach to Treating Severe Depression during Pregnancy?
For severe depression during pregnancy, pharmacotherapy with particular agents is a first-choice treatment, either alone or in combination with CBT or IPT. The remaining SSRIs (except paroxetine), newer generation antidepressants, and TCAs are second line. ECT can also be considered. 57 Combination pharmacotherapy (see Section 3) 58 may be cautiously considered, but little is known about short- and long-term risks to the fetus with this approach.
6.11. What Are the Risks of Using Antidepressant Medications in Pregnancy?
Unfortunately, studies examining the risks of antidepressants during pregnancy are limited by the presence of exposures (e.g., maternal depression, substance or prescription misuse, poor prenatal care, maternal physical health problems) that confound associations between antidepressants and these risks. Available studies cannot fully adjust for these factors, and so the magnitude and specific nature of the risks associated with antidepressants are not completely understood. 59
Most antidepressants have not been linked to an increased risk of major congenital malformations. An increased risk of CV malformations (odds ratio ∼1.5) has been found with first-trimester paroxetine exposure, 59 although a number of these complications resolve spontaneously and do not pose significant functional impairment. 60 Reports have linked fluoxetine use early in pregnancy to a small increase in congenital malformations as well. 61 Significant evidence has not yet accrued that supports increased risks with the other SSRIs, bupropion, mirtazapine, SNRIs, or TCAs (except for clomipramine, which may be associated with an elevated risk of CV malformations). However, antidepressant risk is an active area of study, and discussions with patients should take into account the most recent data. Consultation by patients and/or physicians with Motherisk (www.motherisk.org) can support these conversations.
There may be a very modest link between gestational SSRI use and clinically recognized spontaneous abortion (odds ratio ∼1.5). 62 However, neither this nor the risk of malformations is in excess of the 2-fold increase in risk that is accepted as clinically significant in the field. 63 Studies have also linked SSRIs to a 4-day shortened gestational duration and reduced birth weight (74 grams). 62
At delivery, fetuses exposed to SSRI antidepressants in the third trimester are at elevated risk of developing a syndrome of poor neonatal adaptation marked by jitteriness, irritability, tremor, respiratory distress, and excessive crying. Occurring in 15% to 30% of infants, these symptoms are most often time-limited (typically resolving in 2-14 days), are not associated with an increased risk of mortality or longer-term neurodevelopmental problems, and resolve with supportive care. 64 This risk may be highest with paroxetine, venlafaxine, and fluoxetine. 64 Limited data also suggest that SSRIs taken late (but not early) in pregnancy may be associated with an increased risk of persistent pulmonary hypertension of the newborn (PPHN). The absolute risk is 2.9 to 3.5 per 1000 infants compared to a general population risk of 2 per 1000. 65
The limited data on the longer-term postnatal effects of fetal intrauterine exposure to SSRIs report no lasting cognitive, language, emotional, or behavioural problems in offspring. 66 Finally, despite the fact that a small number of studies have suggested that fetal SSRI exposure may be associated with autism-spectrum disorder in offspring, these studies have significant methodological limitations, have wide confidence intervals, and require further replication before evidence-based recommendations can be made. 67
6.12. How Is Depression Treated during the Postpartum Period?
The deleterious effects of untreated postpartum depression (PPD) on women and their families can be significant. PPD has been linked to impaired mother-infant attachment 68 and cognitive, emotional, and behavioural problems in offspring. 69 Successful treatment of maternal depression may reduce these risks. 70
Breastfeeding is not contraindicated during treatment with an antidepressant medication. Concerns about breastfeeding during medication treatment include short-term adverse reactions and longer-term neurodevelopmental effects. Treatment recommendations for PPD are given for use in women who are breastfeeding. Women with PPD who are not breastfeeding should follow the general CANMAT guidelines.
For women with a mild to moderate PPD who are breastfeeding, first-line recommendations again include IPT and CBT
54,55
(Table 4). Second-line treatments include citalopram, escitalopram, and sertraline, which have data for effectiveness during the postpartum period, minimize risk during lactation, and pose the least known risk during the childbearing years.
70
Structured exercise and depression-specific acupuncture are complementary and alternative treatments that have some evidence in the postpartum period.
71
–73
An increasing body of evidence also supports the use of therapist-assisted Internet-based behavioural activation and CBT, whereas the effectiveness of
Treatment of Mild to Moderate Postpartum Depression during Breastfeeding.
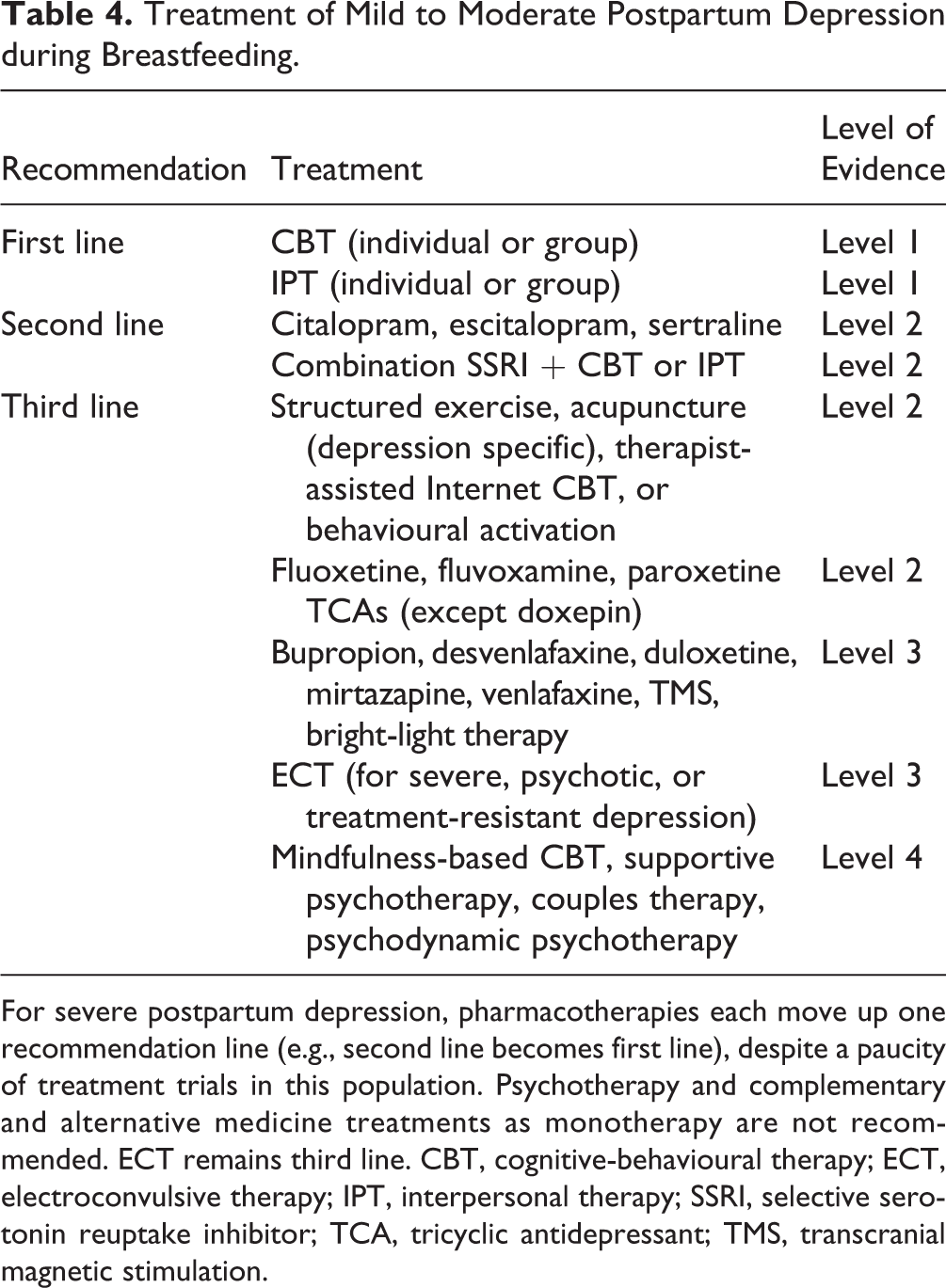
For severe postpartum depression, pharmacotherapies each move up one recommendation line (e.g., second line becomes first line), despite a paucity of treatment trials in this population. Psychotherapy and complementary and alternative medicine treatments as monotherapy are not recommended. ECT remains third line. CBT, cognitive-behavioural therapy; ECT, electroconvulsive therapy; IPT, interpersonal therapy; SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressant; TMS, transcranial magnetic stimulation.
Despite the presence of RCT support for fluoxetine and paroxetine, they are recommended as third-line choices, the former because of its long half-life and slightly higher rates of minor adverse reactions in breastfed infants, 77 and the latter because of its association with CV malformations in subsequent pregnancies. Other second-generation antidepressants are categorized as third-line treatments because of limited evidence in lactating women. Among the TCAs, nortriptyline has the most evidence in the postpartum setting and a solid track record in lactation. 78 Doxepin should be avoided in the postpartum period because of reports of significant adverse reactions in infants with breastfeeding. 79,80 Finally, rTMS 79,81 and bright-light therapy 81,82 may be effective for mild to moderate PPD.
6.13. What Is the Approach to Treating Severe PPD?
For severe PPD, pharmacotherapy should be used first line, with or without psychotherapy. First-choice medications are citalopram, escitalopram, and sertraline. Other antidepressants are second-choice treatments for women who are more severely depressed. ECT is also an effective treatment that is listed as third line because of its side effect profile, but it can be considered a first-choice treatment for severe depression, especially with psychosis; women can also continue to breastfeed during ECT. 83
6.14. What Are the Risks of Antidepressants during Breastfeeding?
Exposure to antidepressants in breastfed infants is 5 to 10 times lower than exposure in utero. Serum levels in preterm infants or those with liver and/or kidney impairment may be higher, and so consultation with a pediatrician should help guide decisions in these cases. Relative infant doses (RID) of medication <10% are generally safe, and all of the SSRIs and SNRIs tested to date appear to meet this criterion. 84 Sertraline, fluvoxamine, and paroxetine have the lowest RID and “milk-to-plasma” ratios. Minor reactions have been noted in case studies of over 200 infants with breastfeeding exposure to sertraline or paroxetine. Citalopram and fluoxetine have had higher rates of infant reactions (4%-5%), but these are reversible and generally limited to short-lived increases in irritability, restlessness, somnolence, or insomnia. 78 Given its relatively low relative infant dose, nortriptyline can be a good choice if women prefer or require treatment with a TCA. Unfortunately, next-to-no data exist on MAOIs during lactation. There is a paucity of data on the long-term neurodevelopmental outcomes of infants who receive antidepressants in breast milk, but there is currently no evidence of significant long-term neurodevelopmental effects.77
Perimenopausal Depression
The transition to menopause (or perimenopause, the beginning of ovarian failure) starts when menstrual cycles become 7 days longer or shorter than usual and extends to the early postmenopausal years. 85 Perimenopause is a period of increased risk for depression compared to premenopausal years. Notably, in epidemiological studies, both increased depressive symptoms and diagnosis of an MDE occurred more frequently in perimenopausal relative to premenopausal women. 86 –89 Perimenopause is associated with risk for both depressive recurrence and new-onset depression. 87,88 Along with increased rates of depression and anxiety, this period is also associated with emergence of menopausal symptoms such as hot flashes, night sweats, decreased libido, vaginal dryness, sleep disturbances, and memory complaints, all of which may negatively affect mood. Hot flashes and night sweats have been identified as independent predictors of perimenopausal depression. 90
Table 5 summarizes the current evidence for treatment of MDD in perimenopausal women.
Current Evidence for Treatment of Perimenopausal Depression.
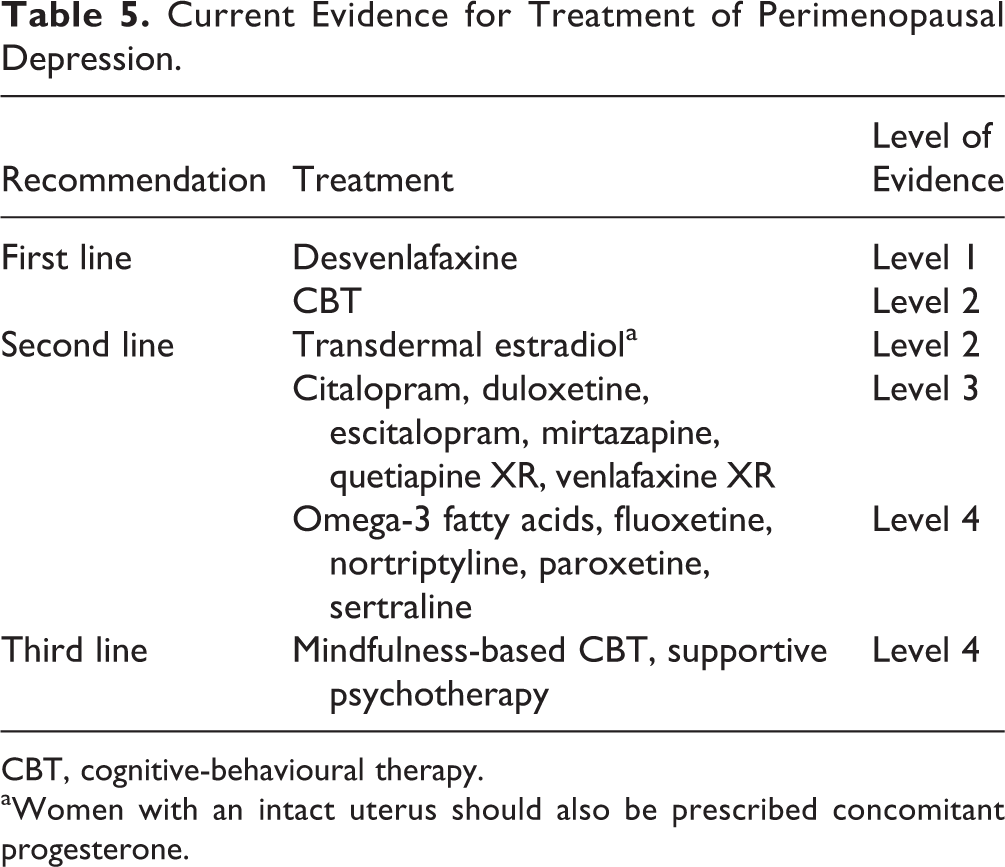
CBT, cognitive-behavioural therapy.
aWomen with an intact uterus should also be prescribed concomitant progesterone.
6.15. Is Antidepressant Medication Effective during Menopause?
Only desvenlafaxine has been specifically evaluated through randomized, placebo-controlled trials for antidepressant efficacy in peri- and postmenopausal depressed women; the 2 trials found that desvenlafaxine (50 mg daily,
6.16. Are Hormonal Agents Effective as Monotherapy or Adjunctive Treatment with Antidepressants?
Transdermal estradiol has been evaluated as both monotherapy and adjunctive posttherapy to treat perimenopausal depression. In a comparative trial of 3 hormone replacement therapies as adjuncts to venlafaxine XR in postmenopausal women, methyltestosterone but not estradiol was superior to placebo. 94 In 2 other small RCTs, estrogen augmentation was superior to placebo in perimenopausal women, 95,96 while there was no difference between transdermal estradiol and placebo in late postmenopausal women. 97 Hormonal agents are recommended as second-line agents for women who understand the risks and have no contraindications to hormonal therapy.
6.17. Are There Effective Nonpharmacologic Treatments for Depression during Menopause?
Only 1 study (
In contrast, adjunctive acupuncture conferred no advantage when added to self-care versus self-care alone for the treatment of hot flashes and depressive symptoms in postmenopausal women. 100
Late-Life Depression
Late-life depression (LLD) can be defined as MDD occurring in adults 60 years and older. When discussing LLD, it is important to differentiate early adult-onset depression recurring in late life from late-onset depression. Compared to patients with earlier onset of MDD, late-onset depression has a worse prognosis, a more chronic course, a higher relapse rate, and higher levels of medical comorbidity, cognitive impairment, and mortality. 101 The vascular depression hypothesis posits that cerebrovascular disease predisposes, precipitates, or perpetuates some depressive syndromes in older age. This vascular burden affects fronto-striatal circuitry, resulting in depression and associated cognitive impairment, especially executive dysfunction. 102,103 Evidence also suggests that late-onset depression or depressive symptoms may be a prodrome for dementia; hence, monitoring of cognition at initial assessment and over time is warranted. 104,105
6.18. What Is the Role of Nonpharmacological Treatments in LLD?
Meta-analyses have demonstrated efficacy for psychological treatments of depression in older adults, 106 with even higher effect sizes when minor depression and dysthymia were included. 107 Newer meta-analyses have addressed some methodological issues in earlier studies—namely, the need for randomization of treatment and the need to assess the effect of the type of control group on the magnitude of psychotherapy effects. A meta-analysis of 27 RCTs including 2245 participants demonstrated great variability in standardized mean differences of 0.05 to 1.36 depending on the control group. 108 In this meta-analysis, psychotherapies (including bibliotherapy) yielded large effects compared with waitlist and attention controls but small to moderate effects compared with supportive therapy or treatment as usual. The authors suggested that supportive therapy best controlled for the nonspecific elements of psychotherapy and should be used as the control for future studies and that problem-solving therapy (PST) has the strongest evidence base using supportive therapy as a control. 108 A recent meta-analysis assessed the efficacy of PST in MDD in older adults, demonstrating that PST significantly reduced depression rating scale scores and reduced disability. The authors also noted that PST is one of the few therapies studied in older people with cognitive impairment and executive dysfunction. 109
6.19. What Are the Principles of Pharmacological Treatment of LLD?
The adage of “start low and go slow (and keep going)” is relevant in LLD. Divisions into young-old (<75 years) and old-old (≥75 years) can be helpful, with a greater degree of vigilance required in treating the old-old. Overall, there are pharmacokinetic changes with aging that may decrease the rate of absorption, modify bioavailability, increase half-life for lipid-soluble drugs, and increase relative concentration for water-soluble drugs and metabolites. 110 As comorbid medical burden and polypharmacy expand, the risk for pharmacokinetic and pharmacodynamic drug interactions increases (see Section 3). 58 In addition, rare antidepressant side effects in adults such as bone loss, serotonin syndrome, extrapyramidal side effects, and neuroleptic malignant syndrome are more common in the elderly. 111 Particular attention should be paid to falls, hyponatremia, and gastrointestinal bleeding, which are associated with SSRIs in general 112,113 and to QTc prolongation with citalopram. 114 Standard principles of conservative prescribing should be applied to minimize adverse drug outcomes. 115 Meta-analyses also suggest that longer antidepressant treatment trials (10-12 weeks) are required in LLD. 116
6.20. What Is the Pharmacological Approach to LLD?
An inherent paradox in the treatment of LLD stems from the dissonance between routine clinical practice and RCT evidence. For example, while citalopram and escitalopram are generally considered by clinicians to be first-line treatments for LLD due to tolerability and fewer drug interactions, 117 –119 none of the RCTs involving these drugs demonstrated superiority over placebo in the elderly, 120 –122 with the exception of citalopram in a subset of old-old (>75 years) patients with severe depression (Hamilton Depression Rating Scale score > 24). 120 In fact, a meta-analysis of 7 studies demonstrated no difference between citalopram and other antidepressants for depression remission or trial withdrawal for adverse effects. 123 In contrast, geriatric clinicians are reluctant to prescribe paroxetine due to anticholinergic effects and fluoxetine due to drug interactions, yet these same SSRIs have positive RCT evidence in the treatment of LLD. 124,125 Thus, treatment recommendations for LLD have been evidence-informed, rather than evidence-based. 119
Overall, recent systematic reviews and meta-analyses support the efficacy of antidepressants in LLD, with no difference between SSRI and SNRI classes,
126
and in adult-onset MDD where episodes recurred in LLD.
127
A subsequent meta-analysis, in adult and geriatric populations, demonstrated that antidepressants are efficacious for depression in adults 55+ years of age.
128
However, drug-placebo differences for studies with an entry criterion of 65+ years were modest and nonsignificant. Heterogeneity, small study number, physical comorbidity, and chronicity were all considered to affect the ability of a trial to separate drug from placebo effects.
128
A recent network meta-analysis, with response as an outcome (>50% reduction in depression score from baseline), demonstrated relative risks compared to placebo of greater than 1.2 for only 3 drugs: sertraline, paroxetine, and duloxetine.
129
A meta-analysis of moderators of treatment response in LLD suggests older adults with longer illness duration and moderate to severe depression benefit from antidepressants compared to placebo, whereas short illness duration does not show antidepressant response.
130
Furthermore, executive dysfunction, especially in the subdomains of planning and organization, has been associated with poor antidepressant treatment response in LLD, which may be a factor in trial heterogeneity.
131
One can speculate that vascular depression, associated with executive dysfunction, may be more resistant to traditional pharmacotherapeutic approaches, and may be related to depressive syndromes that are in fact early manifestations of dementia. These are important considerations when assessing lack of response to initial treatment approaches. Among new antidepressants, vortioxetine and agomelatine have been evaluated in LLD. An RCT (
There is also evidence to support efficacy of continuation and maintenance treatment in LLD. A meta-analysis of 8 double-blind RCTs found antidepressants effective in preventing relapses and recurrences in the elderly, with similar tolerability for TCAs and SSRIs. 134
6.21. Is There a Role for Atypical Antipsychotic Medication in LLD?
In a post-hoc analysis pooling clinical trial data of the 61- to 67-year age group, adjunctive aripiprazole and antidepressants showed a large effect size of 0.8 compared to placebo; the most common side effects were akathisia and dizziness.
135
A recent National Institute of Mental Health–funded RCT (
An RCT (
When prescribed for dementia, antipsychotic medications are associated with increased risk of all-cause mortality, with greater risks for typical than atypical antipsychotics; the risk is less well elucidated in cognitively intact elderly populations. 139 Antipsychotic medications may be considered in selected elderly individuals, recognizing that the risk profile in cognitively intact individuals has not been confirmed.
6.22. What Is the Recommended Sequential Approach to Pharmacological Treatment of LLD?
There is support for a stepwise approach to treatment of LLD in providing the best likelihood of achieving response and remission.
119
In 2 large studies, IMPACT
140,141
and PROSPECT,
142,143
elderly depressed patients randomized to a stepwise algorithmic approach were much more likely to improve than if they were randomized to usual care. Specifically, the odds ratio for IMPACT versus usual care was 3.45 (response rate 45% vs. 19%;
A systematic review and meta-analysis of treatment-resistant depression (defined as failure to respond to at least 1 treatment) in adults aged >55 years identified a dearth of randomized trial data for this patient population. Half of the participants responded to a switch or augmentation strategy, with lithium augmentation demonstrating the most consistent data for all approaches. 144 Of all studies included in the analysis, a sequential treatment strategy provided the highest response rates. 145
For LLD, RCT data generally only assess an individual step in an algorithmic or stepwise approach. Given the challenges in interpreting the evidence in LLD, therefore, an evidence-informed sequential treatment approach is recommended, rather than simply extrapolating from individual trials (Table 6). While good clinical judgement suggests choosing antidepressants to avoid mechanisms that may be harmful in the elderly (e.g., avoiding anticholinergic antidepressants to minimize confusion and delirium risk), there is yet little evidence over the long term to support ad-hoc tailoring of antidepressant choices to target symptom clusters or to leverage specific side effects for therapeutic benefit. For example, evidence does not necessarily support that using a sedating medication to optimize sleep in a depressed patient improves overall outcomes over the course of treatment or longer. It is possible, for example, that when depression has remitted and sleep has normalized that the ongoing sedating effects of medications contribute to noncompliance or lack of tolerability. Hence, use of medications in a consistent and algorithmic manner is suggested, leveraging the extensive evidence for this approach to optimize depression outcomes. 119
Algorithmic Pharmacological Treatment of Late-Life Depression.
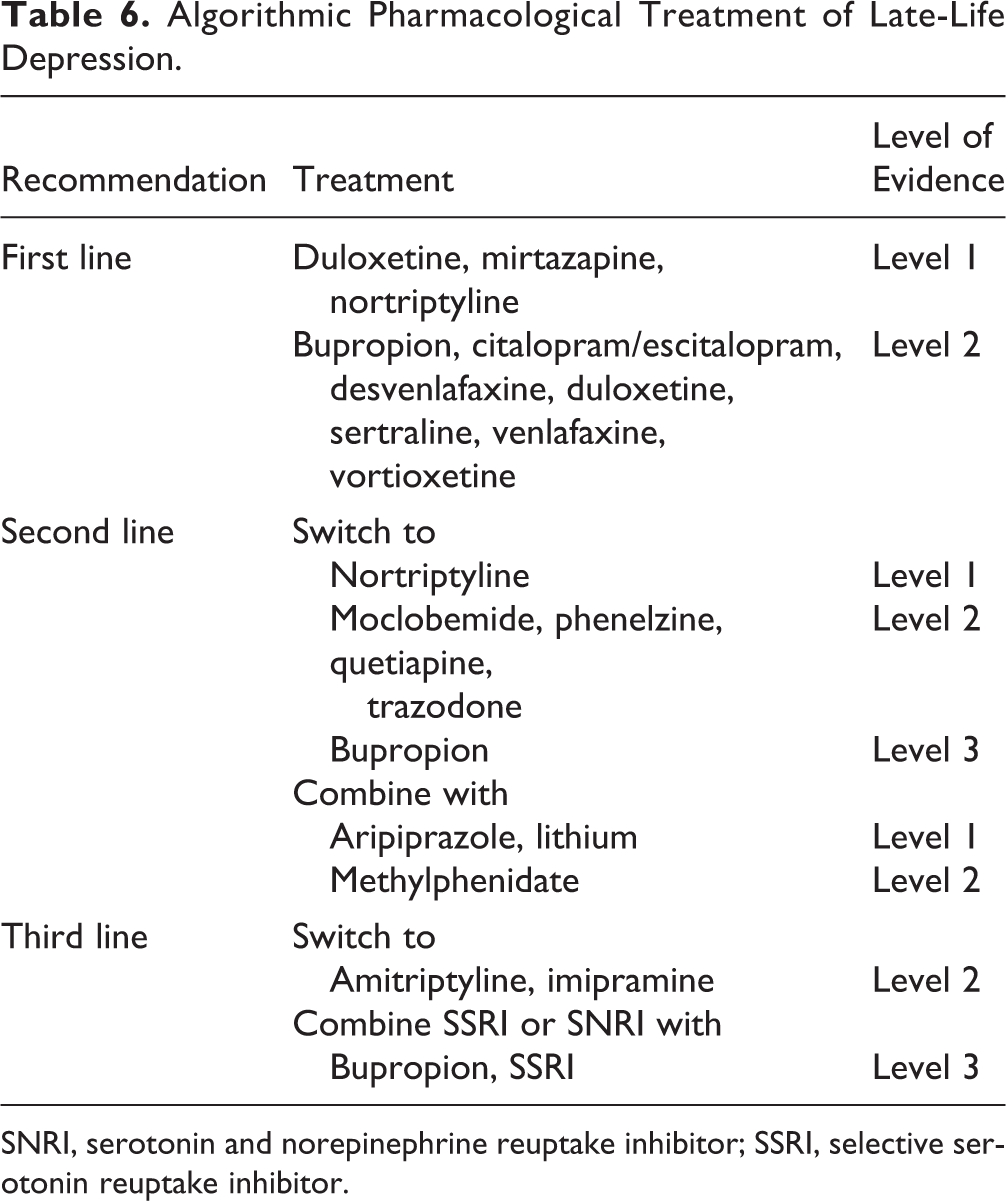
SNRI, serotonin and norepinephrine reuptake inhibitor; SSRI, selective serotonin reuptake inhibitor.
Summary
Depression is common across the life span. While special populations (children and youth, women in the perinatal or menopausal period, and older adults) bring unique challenges, the essential approach to depressive episodes is similar to that of the general adult population. Careful diagnosis, evidence-based evaluation of the risk-benefit ratios of specific treatment strategies, and careful monitoring of outcomes are universal elements of optimal treatment. Evidence for efficacy of treatments in these populations is often more limited than for the general population, and risks of treatment in these groups are often poorly studied and reported. Despite the limited evidence base, extant data and clinical experience suggest that each of these special populations can benefit from the systematic application of treatment guidelines for treatment of depression.
Footnotes
Disclosures
The guidelines process and publication were funded entirely by internal CANMAT funds; no external support was sought or received. No honoraria were paid to authors, and no professional editorial assistance was used. All members of the CANMAT Depression Work Group disclosed potential conflicts of interest (available at ![]() ). CANMAT is a project-driven organization governed by a volunteer, unpaid advisory board, with no permanent staff or dedicated offices. CANMAT has a conflict of interest policy that includes disclosures by all participants, and all continuing professional development (CPD) projects are accredited by academic institutions. CANMAT has diverse funding, but in the past 5 years (2011-2015), sources of CANMAT revenue (excluding CIHR and research funding) included national/international scientific conferences (28% of revenue), publications (26%), industry-supported CPD projects (26%), and academic projects (18%).
). CANMAT is a project-driven organization governed by a volunteer, unpaid advisory board, with no permanent staff or dedicated offices. CANMAT has a conflict of interest policy that includes disclosures by all participants, and all continuing professional development (CPD) projects are accredited by academic institutions. CANMAT has diverse funding, but in the past 5 years (2011-2015), sources of CANMAT revenue (excluding CIHR and research funding) included national/international scientific conferences (28% of revenue), publications (26%), industry-supported CPD projects (26%), and academic projects (18%).
The CANMAT guidelines are not officially endorsed by the Canadian Psychiatric Association.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
GMM has been on advisory boards or a speaker for Janssen, Lilly, Lundbeck, and Pfizer.
BNF has received honoraria for ad hoc speaking or advising/consulting or received research funds from Alternative Funding Plan Innovations Award, AstraZeneca, Brain & Behavioral Foundation, Bristol-Myers Squibb, Canadian Institutes of Health Research, Canadian Network for Mood and Anxiety Treatments, Canadian Psychiatric Association, Daiichi Sankyo, Eli Lilly, Hamilton Health Sciences Foundation, J. P. Bickell Foundation, Lundbeck, Lundbeck International Neuroscience Foundation, Ontario Brain Institute, OMHF, Ontario Ministry of Research and Innovation, NSERC, Pfizer, Servier, Society for Women’s Health Research, Sunovion, and the Teresa Cascioli Charitable Foundation.
ZI has received honoraria for ad hoc speaking or advising/consulting or received research funds from Canadian Biomarker Integration Network for Depression, Canadian Consortium for Neurodegeneration and Aging, Canadian Institutes of Health Research, Janssen, Joan and Clifford Hatch Foundation, Katthy Taylor Chair in Vascular Dementia, Lundbeck, National Institute of Aging, Ontario AFP Innovation Fund, Otsuka, Pfizer, and Sunovion.
NJ has no financial conflicts to declare.
MS has no financial conflicts to declare.
RJV has no financial conflicts to declare.
SHK has received honoraria for ad hoc speaking or advising/consulting or received research funds from Allergan, Brain Canada, Bristol-Myers Squibb, Canadian Institutes of Health Research, Janssen, Lundbeck, Ontario Brain Institute, Pfizer, St. Jude Medical, Servier, and Sunovion.
RWL has received honoraria for ad hoc speaking or advising/consulting or received research funds from Asia-Pacific Economic Cooperation, AstraZeneca, Brain Canada, Bristol-Myers Squibb, Canadian Institutes of Health Research, Canadian Depression Research and Intervention Network, Canadian Network for Mood and Anxiety Treatments, Canadian Psychiatric Association, Coast Capital Savings, Johnson & Johnson, Lundbeck, Lundbeck Institute, Medscape, Pfizer, St. Jude Medical, Takeda, University Health Network Foundation, and Vancouver Coastal Health Research Institute.
RVM has received speaker and consultant honoraria or research funds from Allergan, Bristol-Myers Squibb, Canadian Institutes of Health Research, Canadian Network for Mood and Anxiety Treatments, Canadian Psychiatric Association Eli Lilly, Johnson & Johnson, Lallemand, Lundbeck, Merck, Ontario Brain Institute, Ontario Mental Health Foundation, Otsuka, Paladin, Pfizer, Queen’s University, Sunovion, Takeda, the University Health Network Foundation, and Valeant.
SVP has been a consultant to Bristol Myers Squibb, Lundbeck, and Takeda; has had a research contract with Assurex; and has equity in Mensante.
AVR has received speaker and consultant honoraria or research funds from Bristol-Myers Squibb, Canadian Depression Research and Intervention Network, Canadian Foundation for Innovation and the Ministry of Economic Development and Innovation, Canadian Institutes of Health Research, Grand Challenges Canada, Janssen, Lundbeck, Ontario Mental Health Foundation, Pfizer, and Sunovion.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
