Abstract
The Ediacaran Period was a time when life got big with confirmed fossils of megascopic algae, lichens, sponges, and worms. But which of these kinds of organisms are the most likely biological affinities for enigmatic vendobionts such as
Keywords
INTRODUCTION
The Ediacaran Period was a turning point in the evolution of life on Earth, as the time when life got big, meters long rather than millimetres (Payne et al., 2009). Most of the classical Ediacaran fossils from South Australia were originally (Glaessner & Wade, 1966) interpreted as highly mobile animals (Figure 1). Later reassessment objected that they could not have been animals for a variety of reasons, such as asymmetric segmentation (Seilacher, 1992). The discovery of palaeosols beneath the fossils (Retallack, 2012, Seilacher, 1992). The discovery of palaeosols beneath the fossils (2013a) was evidence for an alternative interpretation as some kind of terrestrial vegetation (Figure 2). Only a few Ediacaran fossils were undisputed animals that could move of their own accord, as demonstrated by trace fossils, such as
Classical reconstruction of Ediacaran fossil assemblages of the Ediacara Member of the Rawnsley Quartzite of South Australia at the Museum of the Earth, Ithaca, New York. The museum label in 2015 described this as a ‘legacy diorama’, showing outdated ideas of highly mobile animals promoted by Glaessner and Wade (1966): l-r, Dickinsonia costata, Arborea arborea, Medusinites asteroides, Praecambridium sigillum, Parvancorina minchami, Kimberella quadrata and Cyclomedusa davidi . All but Arborea were then considered highly mobile ocean swimmers.

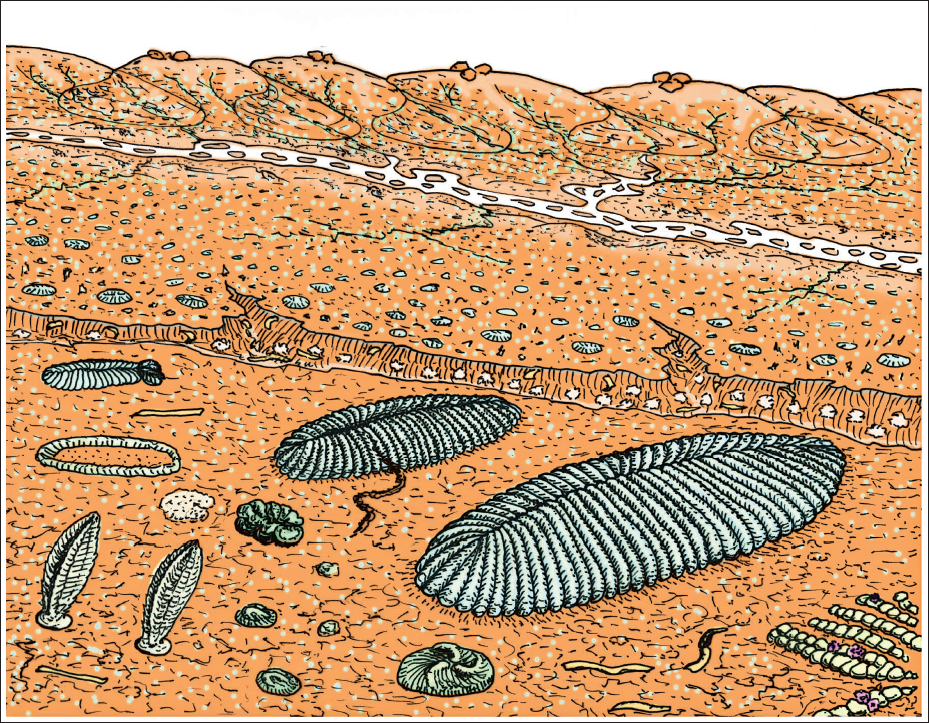
Revised reconstruction of Ediacaran fossil assemblages of the Ediacara Member of the Rawnsley Quartzite based on occurrence in Gypsid paleosols in South Australia, showing l-r, Spriggina floundersi , fragments of Aulozoon soliorum, Phyllozoon hanseni, Dickinsonia costata, Quaestio simpsonorum displaced from former location by wind, Parvancorina minchami, Obamus coronatus , trail of Helminthoidichnites tenuis feeding on Dickinsonia costata, Tribrachidium heraldicum , and Funisia dorothea .
ALTERNATIVE BIOLOGICAL INTERPRETATIONS OF EDIACARAN FOSSILS
While the biological affinities and mobility of Ediacaran vendobionts remain controversial, a variety of biological affinities are plausible because they are represented by other Ediacaran fossils of undisputed taxonomic position. Not all Ediacaran burrows and trails are necessarily from animals because flaring trails (Meyer et al., 2014), such as
ALTERNATIVE PALAEOENVIRONMENTAL INTERPRETATIONS OF EDIACARAN FOSSILS
On first discovery, Ediacaran megafossils were identified as shallow marine invertebrates (Glaessner & Wade, 1966). Even though the biological affinities of these fossils remain highly problematic (Seilacher, 1992), marine habitats are still urged without corroborating evidence (Runnegar, 2022; Weyland & Droser, 2025). Within the past decade, the distinction between marine and non-marine rocks of Ediacaran age has been tested by various techniques. It began with recognition of palaeosols within red beds with Ediacaran fossils (Retallack, 2012, 2013a), some of them with well-developed calcareous nodules (calcic or Bk horizons) and desert rose pseudomorphs (gypsic or by horizons). The profiles also showed weathering of feldspar to clay, geochemical tracers of that reaction (molar ratio of alumina over bases), and oxidation on land (molar ratio of ferric to ferrous iron). Ediacaran palaeosols are more deeply weathered in nutrient bases and phosphorus than Mesoproterozoic palaeosols, and comparable with early Palaeozoic palaeosols in calculated rate of weathering of nutrient bases (Ca, Mg, Na, and K) and of phosphorus (Retallack, 2022b). Ediacaran isotopic depletion of carbon and oxygen in pedogenic versus marine carbonate is also greater than in Mesoproterozoic palaeosols, as additional evidence of enhanced biogenic weathering on land (Retallack & Bindeman, 2024). Other geochemical discriminators of marine versus non-marine conditions successfully applied to Ediacaran rocks are C/S ratios (Retallack, 2014), Ge/Si ratios (Retallack, 2017), B/K ratios (Retallack, 2020), and light rare earth to heavy rare earth ratios (Retallack, 2024a). Other indications of non-marine conditions are drab-haloed filament traces (Retallack, 2012, 2013a) and the microbial fabric widely identified as ‘old elephant skin’ (
Marine beds of Ediacaran age are also known, and are grey coloured with reduced (ferrous) iron like Phanerozoic marine rocks, rather than red with oxidised (ferric) iron (Retallack, 2014). Ediacaran marine beds are proven by the same molecular ratios and enrichments of nutrient bases and phosphorus mentioned above (Retallack, 2014, 2020, 2024a). Marine Ediacaran rocks contain stromatolites (Walter et al., 1979) and the tubular fossils attributed to animals such as
Ediacaran palaeosols have been especially useful for evaluating Ediacaran paleoclimate, which, with one exception, was temperate to periglacial rather than tropical. The exception is the Baltic paleosol, a deeply weathered tropical soil (Oxisol) on flood basalt at the unconformable base of Vendian shales dated to 560 Ma around the Baltic and White Seas, once considered anomalous for its high latitude (Driese et al., 2018; Liivamägi et al., 2014). However, the paleolatitude of Baltica has been reassessed at 557 Ma as 17.6° ± 5.7° (Domeier et al., 2023), so the Baltic palaeosol is no longer a climatic anomaly. Sand wedges, ice wedges, and freeze-thaw banding have been documented from both Cryogenian (Retallack, 2023; Retallack et al., 2015) and Ediacaran palaeosols (Retallack, 2022d). Ediacaran periglacial palaeosols are associated with a series of glacial advances evident from tillites and solifluction flows, labelled Gaskiers (580 Ma), Fauquier (672 Ma), Bou Azzer (565 Ma), and Hankalchough (550 Ma; Retallack, 2022d). These glacial advances are associated with glacioeustatic sea level drawdowns creating paleovalleys as deep as 600 m (Giles et al., 2024; Retallack et al., 2014). Between these stratigraphic levels of glacial facies and periglacial palaeosols are many other palaeosols with weathering ratios and salinisation as evidence of temperate paleoclimatic conditions. The global distribution of Ediacaran palaeosols included gypsic and calcic palaeosols of temperate aridlands (Retallack, 2012, 2013a, 2022e), as well as deeply leached palaeosols of temperate humid paleoclimate (Retallack, 2014, 2016c). The transition from Cryogenian Snowball Earth to the Ediacaran was not an abrupt paleoclimatic change from frigid to tropical, because the Ediacaran Period remained cool with glacial advances (Retallack, 2022d).
MOBILITY OF DICKINSONIA ?
Movement of
The mechanism of movement of
Evidence against the movement of
Alternative hypotheses for
Crustose lichen is an alternative interpretation of
MOBILITY OF QUAESTIO?
The occurrence of
On land, lateral displacement may have been caused by wind gusts of frozen individuals on melting ice (Retallack, 2021). Such ‘snow mice’ of displaced polsters of mosses or vagrant lichens are common in periglacial regions today (Hotaling et al., 2020; Pérez, 1997). Other evidence of periglacial conditions in the same beds as
MOBILITY OF KIMBERELLA ?
These structures represent a variety of ground ice. Bundles of these ice crystals are like frost flowers (Plate 4.5) with sharp, faceted terminations like monoclinic ice crystals with helical twinning (Plate 4.3–4). They are unusually sharp and parallel for radular scratches, and commonly displace small (1–2 mm) clods of sediment (Plate 4.1–2). The needles radiate from a segment of a terminal flange (Plate 4.4), like that of
MOBILITY OF FUNISIA ?
Chief among the problems for this interpretation is the way in which
An alternative view of
CONCLUSIONS
Putative evidence that the iconic quilted Ediacaran fossils known as vendobionts could move is reexamined and found wanting. Supposed ‘footprints’ of
Terrestrial red beds, unlike intertidal to marine grey beds, are now widely recognized in Ediacaran rocks using a variety of petrographic (Retallack, 2012, 2013a), sedimentological (McMahon et al., 2020), paleopedological (Driese et al., 2018), and geochemical evidence (Retallack, 2020, 2024a). These studies highlight a difference between marine grey beds with stromatolites and tubular fossils such as
A variety of Ediacaran megascopic life: algae (4), slime mould (3), worm tube (2), worm burrow (1), lichens (6–10), and enigmatic vendobionts (11–15): (1) Helminthoidichnites tenuis burrow cutting across Dickinsonia costata from Nilpena Member of Rawnsley Quartzite, Nilpena, South Australia; (2) Corumbella werneri from Tamengo Formation in Itaú quarry, Ladário, Brazil; (3) Lamonte trevallis from Shibantan Member of Denying Formation near Wuhe, China; (4), Longifuniculum sp. cf. dissolutum from Nilpena Member of Rawnsley Quartzite, Nilpena, South Australia; (5) Nilpenia rossi from Nilpena Member of Rawnelsy Quartzite Nilpena, South Australia; (6–8) Ganarake scalaris , hand specimens dark in matrix and details of thallus structure, spherical photobionts, tubular hyphae, and skeletal bipyramidal crystals of pruina whewhellite, from the Noonday Formation of Galena Canyon, Death Valley, California; (9), Ceratosphaeridium mirabile , Wilari Dolomite Member, Tanana Formation, Observatory Hill no., 1 well, northern South Australia; (10) Schizofusa zangwenlongii , Dey Dey Mudstone, Observatory Hill bore, northern South Australia; (11) Rangea scheiderhoehni from Kliphoek Member of Dabis Formation on farm Aar, near Aus, Namibia; (12) Dickinsonia costata from Ediacara Member of Rawnsley Quartzite Ediacara Hills, South Australia; (13) Charnia masoni from Bradgate Formation in Pocketgate quarry, England; (14) Pteridinium simplex from Aarhauser Sandstone on Aar farm, southern Namibia; (15) Ernietta plateauensis from Kliphoek Member of the Dabis Formation in Farm Aar, Aus, Namibia. Specimens are (1) South Australian Museum P35740 from J.G Gehling, (2) Ohio State University Geology Museum 46402 from Gabriel de Matos, (3) Virginia Polytechnic Institute Geoscience Museum 4670 from M. Meyer, (4) South Australian Museum P48767 from S. Xiao, (5) Nilpena RO8-155 from M.Droser, (6–8) Museum of Natural and Cultural History, University of Oregon F116110A ; (9–10) Geoscience Australia Canberra Paleontological Collection slides 36417 and 36355 from K. Grey, (11) Geological Survey of Namibia Museum, Windhoek F182, (12) South Australian Museum F137460, Retallack photo, (13), Leicester Museum LEIUG 2328, Retallack photo; (14) Retallack field photo; (15) Geological Survey of Namibia Museum Windhoek F1244, Retallack photo.

Dickinsonia costata , ‘Epibaion costatum’, Aulozoon soliorum , and comparable modern vagrant mosses and lichens: (1–2) Body fossil Dickinsonia costata , with presumed resting traces ‘Epibaion costatum’ from Ediacara Member of Rawnsley Quartzite in Ediacara Hils, South Australia; (3) detail of Aulozoon soliorum tubes (arrow) connected to Dickinsonia costata deep within slab, and below other specimens of Phyllozoon hanseni from Nilpena Member of Rawnsley Quartzite, Bathtub Gorge, South Australia; (4), thin section of Dickinsonia ornata below ripple-marked sandston from Ediacara Member of Rawnsley Quartzite in Brachina Gorge, South Australia; (5), Thin section of Aulozoon soliorum below basal impression of Dickinsonia costata from Nilpena member of Rawnsley Quartzite near Hookapunna Well, South Australia; (6) Grimmia pulvinata on alpine soil near Salzburg, Austria; (7) Xanthoparmelia reptans on red soil between red mallee (Eucalyptus socialis ) near Damara station, inland New South Wales; (8) Dimelaena oreina on Steens Basalt at Paisley Caves, Oregon. Fossil specimen sources are (1–2) South Australian Museum P14359, Retallack photo; (3) South Australian Museum P35665–86; (4–5) Museum of Natural and Cultural History University of Oregon specimens F115735 and F117937; (6) Wikimedia Darkone; (7–8) Retallack photos.

Quaestio simpsonorum and comparable modern vagrant and jelly lichens: (1) body fossil (right arrow) and trace impression (left arrow) of Quaestio simpsonorum from the Nilpena Member of the Rawnsley Quartzite, Nilpena, South Australia; (2) reconstruction of Quaestio simpsonorum and its trace on ‘old elephant skin’ (Rivularites repertus ) microbial earth on a Wadni pedotype; (3) tumbleweed shield lichen Xanthoparmelia vagans from Cortez, Colorado; (4) jelly lichen Collema tenax near Worland, Wyoming; (5) rock tripe lichen Umbilicaria torrefacta , Wells Gray Park, British Columbia Canada. Fossil specimen (1–2) NENP LV-FUN 591s 1287e (Evans et al., 2024); (3) photo Dale Ward; (4) photo Tyrell Perry; (5) photo Jason Hollinger.

Kimberella quadrata , ‘Kimberichnus teruzzi’, and comparable modern ice needles and ground ice: (1–2) specimen and annotated sketch of Kimberella quadrata , and supposed radular scratches given invalid name ‘Kimberichnus teruzzi’, here interpreted as ice needles, from Zimnie Gory Formation at Zimnie Gory, White Sea, Russia; (3) frost flower (‘Kimberichnus teruzzi’ from Zimnie Gory Formation at Solza River near Archangelsk Russia; (4) Kimberella quadrata and ‘Kimberichnus teruzzi’ from Ediacara Member of Rawnsley Quartzite in Brachina Gorge, South Australia; (5) frost flower growing from water-filled hollow in plant stalk from roadside pond near Frisco, Colorado (2 May, 2011); (6) Frost palisades uplifting soil clods southwest of Granges–sur–Vologne, France (28 December, 2008). Fossil specimen (1–2) in Paleontological Institute Moscow 3493/5137, photo Dmitry Grazhdhankin; (3) Paleontological Institute Moscow 3993/5619, photo Dmitry Grazhdhankin; (4) South Australian Museum P35657, photo Retallack; (5) photo Robert Berwyn; (6) photo Thomas Bresson.

Funisia dorothea , and comparable living lichens Stellarangia namibiensis and S. elegantissima : (1–2) Funisia dorothea from the Nilpena Member of the Rawnsley Formation at Nilpena, South Australia; (3–4) Stellarangia namibiensis from 15 km east of Swakopmuind, Namibia; (5) Stellarangia elegantissima with prominent apothecia, from 20 km northeast of Swakopmund, Namibia. Fossils (1–2) South Australian Museum P40275 and P40309; lichen photos (3–5) by Ingeborg van Leeuwen.

Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Sandal Society of the Museum of Natural and Cultural History of the University of Oregon.
