Abstract
The Himalayan orogeny played a pivotal role in shaping South Asia’s climatic and ecological evolution, leading to the deposition of approximately 6000 m of molasse sediments in the Himalayan Foreland Basin, known as the Siwalik Group. While fossil flora of the Siwalik sediments in western and eastern Himalaya has provided valuable insights into past vegetation shifts, the plant fossil record from eastern Nepal remains poorly documented. In this study, we describe fossil fern of Aspleniaceae and a leaf of
INTRODUCTION
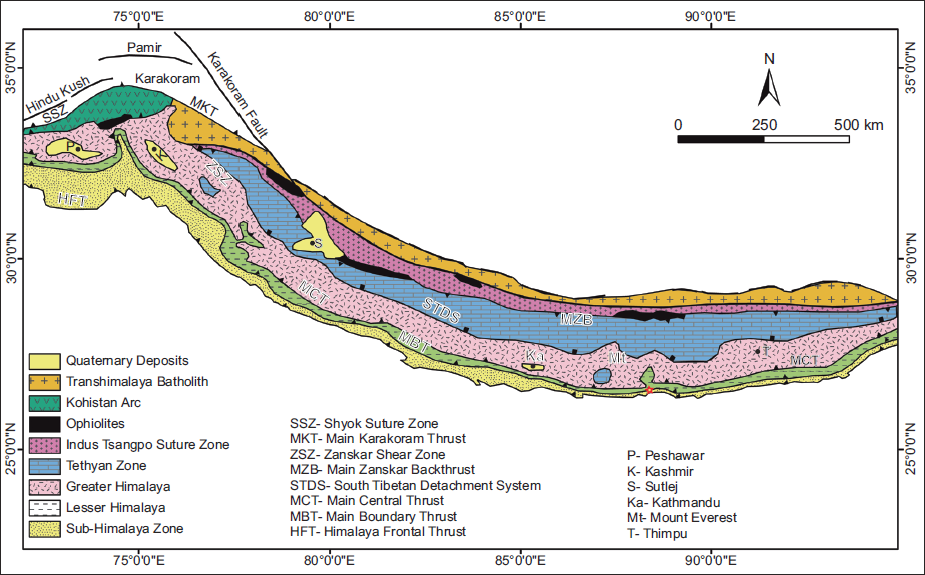
The deformation and uplift of the Himalayan belt led to the accumulation of approximately 6000 m of molasse sediments in the Himalayan Foreland Basin, extending ~2400 km from Pakistan in the west to Arunachal Pradesh in the east. The sediments in this basin, known as the Siwalik Group, lie between the Main Boundary Thrust (MBT) to the north and the Himalayan Frontal Thrust (HFT) to the south (Gansser, 1964; Valdiya, 2002). The Siwalik sediments, deposited from the Middle Miocene to the Lower Pleistocene, are classified into Lower, Middle and Upper Siwalik (Parkash et al., 1980; Pilgrim 1910, 1913). These units exhibit a coarsening-upward fluvial succession, while individual beds display a fining-upward pattern (Nakayama & Ulak, 1999; Rai & Yoshida, 2021).
The Siwalik flora is essential for understanding the impacts of Himalayan orogeny on climate, vegetation shifts, monsoon patterns and elevation changes in the Himalayan‒Tibetan region (Ding et al., 2017; Srivastava et al., 2018). A transition from evergreen to deciduous forests has been documented in the Lower and Middle Siwalik sediments of both eastern and western Himalaya (Adhikari et al., 2024c; Bhatia et al., 2021, 2022; Srivastava et al., 2018), likely driven by rising temperatures and declining precipitation (Bhatia et al., 2021, 2022; Hoorn et al., 2000; Srivastava et al., 2018; Wu et al., 2014). However, the floral diversity of eastern Nepal remains poorly documented, with existing fossil records being limited (Adhikari et al., 2018, 2022, 2023, 2024a, b; Srivastava et al., 2017). This makes the region crucial for understanding the evolutionary history of its plant communities.
In this study, we describe two new fossil impressions from the Lower Siwalik sediments of the Kankai Mai River section in eastern Nepal and discuss their phytogeographical and climatic significance.
GEOLOGY OF THE STUDY AREA
The study area is located within the Siwalik Group of the Kankai Mai River section in eastern Nepal (26.68° N, 87.9° E) (Figures 1 and 2). The Siwalik Group in this region is divided into southern and northern belts, separated by the Central Churia Thrust (CCT) (Ulak, 2009, 2016). Lithostratigraphically, it comprises three litho units—Lower Siwalik, Middle Siwalik and Upper Siwalik—differentiated based on lithological variations and the thickness of sandstone and mudstone (Ulak, 2009, 2016).
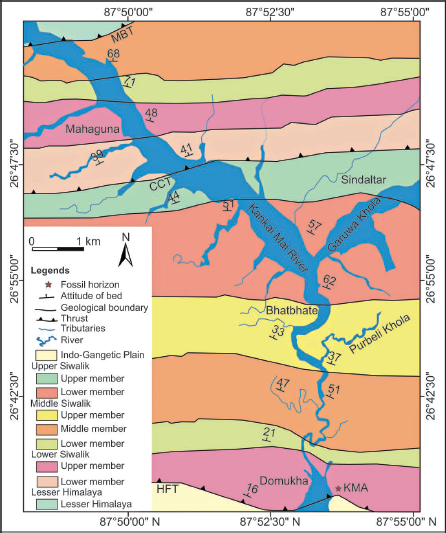
The Lower and Upper Siwalik are further divided into lower and upper members, whereas the Middle Siwalik comprises lower, middle and upper members. The Lower Siwalik is characterised by fine- to coarse-grained sandstone interbedded with variegated to dark grey mudstone and dark to light grey siltstone. The Middle Siwalik comprises medium- to very coarse-grained ‘salt and pepper’ sandstone to pebbly sandstone, dark grey to black mudstone and light grey siltstone. Less indurated sandstone beds are predominantly found in the middle part of this unit. The Upper Siwalik consists of poorly to well-sorted cobble-pebble-boulder conglomerates, reddish-brown sandstone, dark grey siltstone and mudstone. These Siwalik successions were deposited in a fluvial system (Ulak, 2009, 2016).
The Lower Siwalik and the lower member of the Middle Siwalik were formed under a meandering river system. In contrast, a braided river system deposited the middle to upper members of the Middle Siwalik and the Upper Siwalik (Ulak, 2009, 2016). The fossil leaf impressions were excavated from grey siltstone beds within the upper member of the Lower Siwalik (Figure 3).
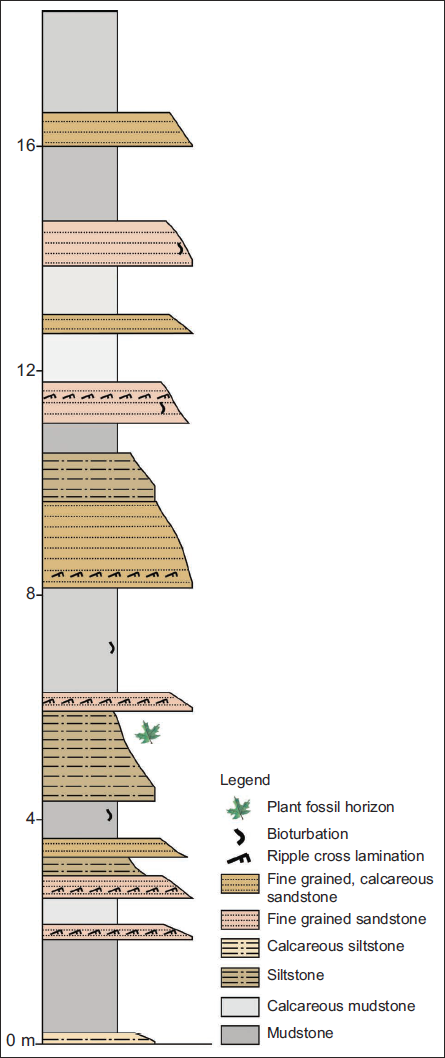
MATERIALS AND METHODS
A soft brush, hammer and fine chisel were used to carefully expose the fossil leaf impressions, which were then photographed under low-angle natural sunlight using a digital camera (Canon G7X Mark II). Minute features of the leaf impressions were examined using a stereoscopic microscope (Leica S8APO). The descriptive terminology follows the leaf architecture classification of Dilcher (1974) and Ellis et al. (2009). Fossil identification was carried out at the herbarium of the Forest Research Institute (FRI), Dehradun and the Central National Herbarium (CNH), Howrah, India. All fossil impressions are housed in the Department of Geology, Birendra Multiple Campus, Bharatpur, Chitwan, Nepal.
SYSTEMATICS
Holttum et al. (1970) analysed morphologically similar genera, including
Joshi and Mehrotra (2003) established the organ genus
Additionally, comparisons were made with various taxa within the Lythraceae family, such as
Fossil leaves of
DISCUSSION
The Aspleniaceae, the largest family of ferns, comprises 24 genera and approximately 950 species, predominantly distributed across tropical and subtropical regions worldwide (POWO, 2024; Smith et al., 2006). Notably, species diversity is highest in low-latitude Malesia (~440 species), followed by the Neotropics (~300 species) and Africa and its neighboring islands (55 species) (Smith, 1990). Most family members thrive in damp, primary undisturbed forests, typically at low to mid-elevations within or near rainforest edges (Smith, 1990).
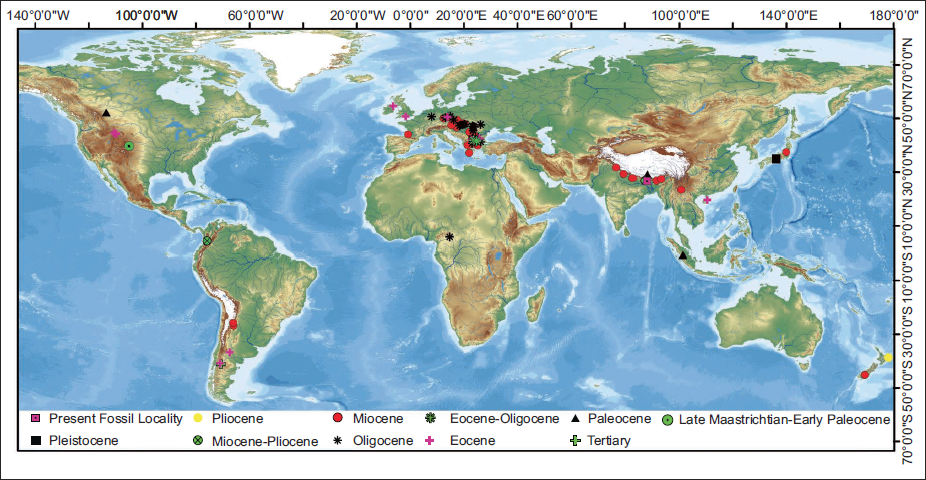
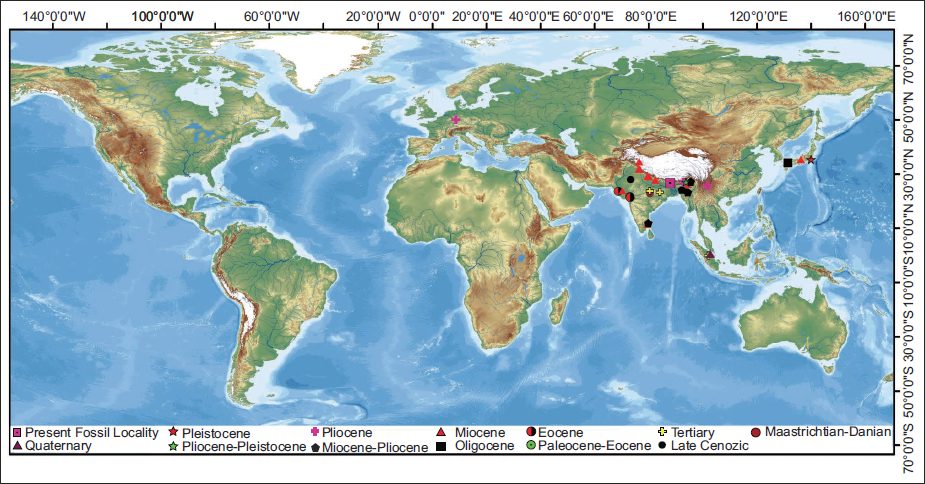
Megafossils of the Aspleniaceae family are well-documented from the Cretaceous and continue throughout the Cenozoic (Figure 4; Supplementary Table 1). The earliest known fossil of this family has been reported from the late Maastrichtian–early Paleocene sediments of central Colorado, USA (Berry, 2019). During the Paleogene, Aspleniaceae across Africa, China, Europe, North America, South America and Southeast Asia (Figure 4). In the Neogene, their fossil records indicate occurrences in Europe, New Zealand, South America, East Asia and South Asia (Figure 4), with evidence of their presence in South Asia during the Miocene. Fossilised remains of an Aspleniaceae genus from the Paleocene sediments of Indonesia (Kräusel, 1929) suggest that the family might have migrated to South Asia from Southeast Asia, possibly after the collision of the Eurasian and Indian plates (Chatterjee et al., 2013); however, other possible routes cannot be ruled out.
The genus
Sterile frond of the fossil and the modern fern. (1) Fossil fronds of Thelypteridaceophyllum tertiarum Joshi & Mehrotra showing shape, size, and venation pattern such as mid vein of the pinna (costa) (red arrows), fused pinnule margin (dark green arrows), midrib of pinnules (yellow arrows), and lateral veins of pinnules (pink arrows). (2) and (3) Fossil pinna showing shape, size, and venation pattern such as mid vein of pinna (red arrow), fused pinnule margin (dark green arrow), midrib of pinnules (yellow arrows), and lateral veins of pinnules (pink arrows). (4) Modern pinna of Cyclosorus proliferous Tard. and Chr. showing shape, size, and venation pattern such as mid vein of pinna (red arrows), fused pinnule margin (dark green arrows), midrib of pinnules (yellow arrows), and lateral veins of pinnules (pink arrows) (Scale bar = 1 cm).

Fossil leaf of Lagerstroemia himalayaensis Srivastava, Gaur et Mehrotra and modern leaf of Lagerstroemia parviflora Roxb. (1) Fossil leaf of Lagerstroemia himalayaensis showing shape, size and venation pattern such as primary vein (white arrows), secondary vein (yellow arrows), base (light blue arrow) and apex (red arrow). (2) Modern leaf of Lagerstroemia parviflora showing shape, size, and venation pattern such as primary vein (white arrows), secondary vein (yellow arrows), base (light blue arrow), and apex (red arrow) (Scale bar = 1 cm).

The current megafossil record of
CONCLUSIONS
The discovery of Aspleniaceae and
Footnotes
Acknowledgements
PA is grateful to the Head of Department, Central Department of Geology, Tribhuvan University, Kathmandu and Campus Chief, Birendra Multiple Campus, Tribhuvan University, Bharatpur, Chitwan. HB, S and GS are thankful to the Director of the Birbal Sahni Institute of Palaeosciences, Lucknow, for providing necessary facilities and encouragement during the research work. The authors are grateful to the Director of the Central National Herbarium (CNH), Howrah, and the Forest Research Institute (FRI), Dehradun, India, for providing access to herbarium sheets for consultation. We thank Lalit Kumar Rai and Ashok Poudel for their assistance during our fieldwork.
Data Availability Statement
All data used are available in the manuscript and the supplementary files.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest regarding the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The present research work was supported by the University Grants Commission (UGC), Sanothimi, Bhaktapur, Nepal [Grant Number PhD-76/77-S&T-8] to PA.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
