Abstract
Coral reef environments are known for the proliferation of algal symbiont-bearing foraminifera, which are sensitive indicators of water-quality conditions that support reef development. Keeping this in mind, 18 sediment samples were collected in the Kavaratti lagoon of Lakshadweep in 2004. The foraminiferal distribution and abundance of the major genera Amphistegina, Calcarina, Amphisorus and miliolids were compared with those reported nearly three decades ago. The aim was to identify changes in the foraminiferal data within the lagoon over time, through time-gap-sampling, to be able to assess environmental stress if any. The total foraminiferal number (TFN) in the lagoon ranges between 45 and 983 specimens per gram of dry sediments. The average TFN in the northern part of the lagoon (506/g) is lower than that in the southern part of the lagoon (612/g). Amphistegina and Calcarina, both being algal-symbiont-bearing foraminifera, show the tendency to occupy shallower depths of the lagoon. Amphisorus prefer the northern part of the lagoon, due to the prevalence of comparatively stronger currents. Over the past three decades, TFN in the lagoon has on an average gone down from 65,667 in 1976 to 56,100 (present study) specimens per 100 g of sediments. The larger foraminiferal distribution in the Kavaratti lagoon has slightly but undisputedly undergone quantitative and distributional changes over the past three decades. The FORAM Index suggests conducive environment for coral growth in the lagoon. However, definite deterioration due to dredging in the northern lagoon is indicated by the assemblage. The study also records signatures of domestic discharge and predicts a larger impact on the corals since 2004 to 2024.
Introduction
Coral reef communities are threatened worldwide by stresses ranging from localised impacts to global change. Resource managers require indicators of the biological condition of reef environments that can relate data acquired through remote sensing, water quality and benthic-community monitoring to stress responses in reef organisms (Crevison & Hallock, 2007).
Larger benthic foraminifera (LBF) are unicellular, shell-bearing organisms that occur abundantly in reef ecosystems and use all available surfaces as substrate. Reef-dwelling foraminifera have been widely employed for the assessment and health monitoring of coral reefs. Specially, the larger taxa which host symbiotic algae are potential indicators of reef vitality (Cockey et al., 1996; Hallock, 2002; Toler & Hallock, 1998). This is because zooxanthellate of corals and symbiotic algae of foraminifera show similar response to stress conditions and have similar environmental requirements like limited supply of fixed-nitrogen, essential for maintaining host-symbiont relationship (Hallock, 2000).
Cockey et al. (1996) suggest that larger foraminiferal populations are immune to coral-specific diseases and recover much more quickly from physical impacts than long-lived coral populations. Thus, they are sensitive indicators of water-quality conditions that support reef development. The life-span of foraminifera is short as compared to that of long-lived corals, making them promising bio-indicators which can discriminate between the cause of reef decline, that is, long-term reef decline correlated with reduced water quality and temporary reef decline analogous to natural occurring episodic mortality events (Hallock, 2000). Where larger foraminifera quickly decline as a result of reduced water quality, foraminiferal assemblages would also recover much faster after irregular mortality events like hurricanes or extreme high temperatures. The positive point for this kind of assessment is that foraminifera are relatively very small and abundant, thus from a handful of sediment samples, statistically significant data can be generated quickly and at low cost (Hallock, 2000).
India is blessed with some rich coral biodiversity within its exclusive economic zone. Of these, Lakshadweep in the Arabian Sea consists of 12 atolls, 3 reefs, 5 submerged banks and 36 islands covering an area of 32 sq. km and lagoon area of 4,200 sq. km. A total of 105 coral species have been reported (Muley et al., 2000). Major threats to these coral atolls are from the increased population, developmental activities, sewage dumping, pollution, dredging for making shipping lanes, sedimentation, siltation, etc. Corals in this region are threatened by anthropogenic activities like coastal development, removal of vegetation, dredging for boat, sewage discharge and coral mining (Sarkar, 2019). In February 2020, the Lakshadweep Administration notified three new marine protected areas, with a total area of 645 sq. km (WCS-India, 2020).
Though several studies (Gadi et al., 2016; Guptha & Sankarnarayana, 1973; Srivastava et al., 1985) have documented foraminiferal populations from different island areas, no studies have attempted to employ them to ascertain the health of these sensitive ecosystems. Rao et al. (1987) is the only qualitative and quantitative description of foraminifera based on sampling (in 1976) from four coral lagoons of the Lakshadweep. Thus, the present study is an attempt to use these reef-dwelling foraminifera for monitoring the environment of the lagoon of the capital island, Kavaratti, and detect changes if any over a three-decade sampling interval (1976–2004).
Study Area
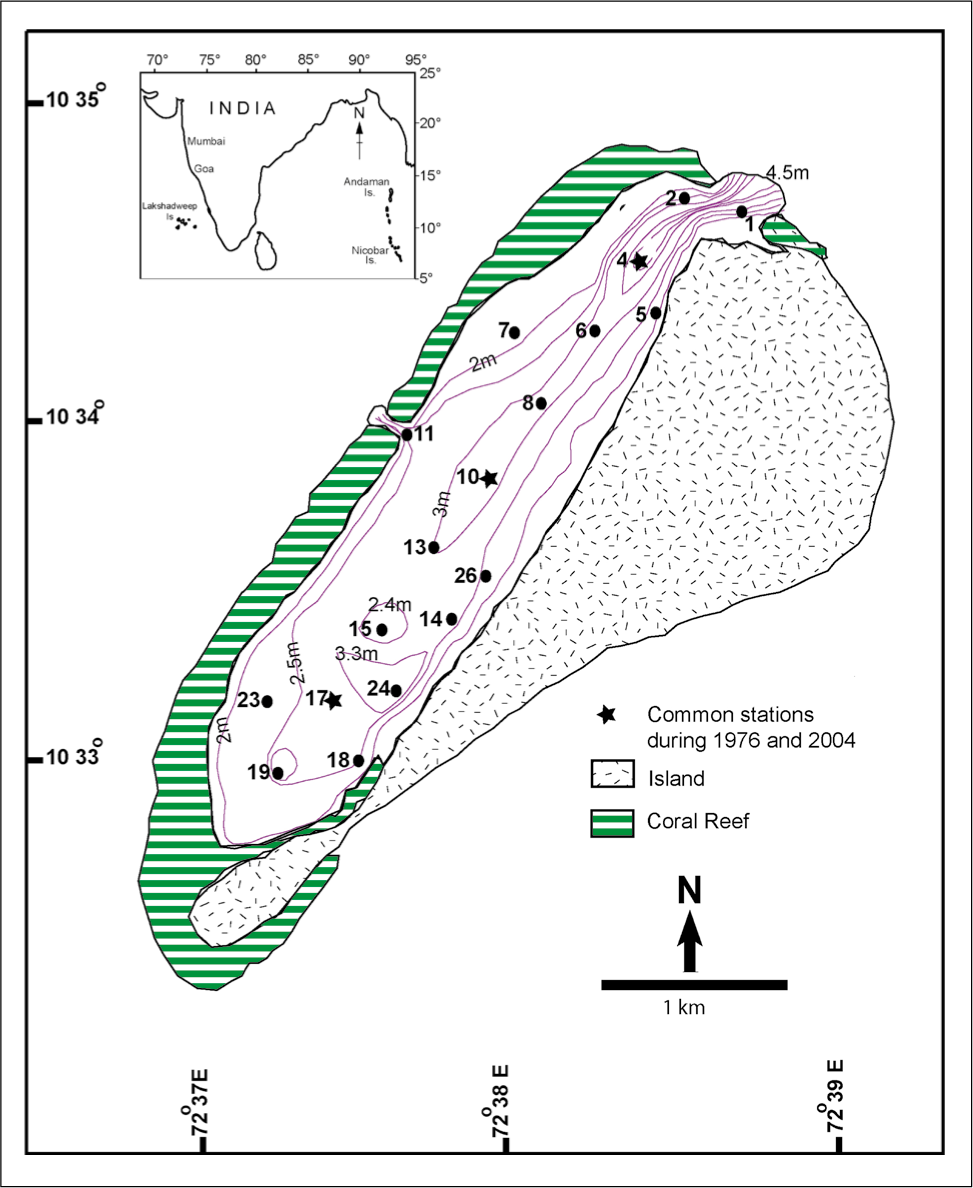
The study area, namely the Kavaratti Island, is the capital of the Union Territory of Lakshadweep in the southeast Arabian Sea and is bounded by 10˚32′N and 10˚35′N latitudes, and 72˚37′E and 72˚39′E longitudes. It was surveyed in 1987 and included in the Naval Hydrographic Chart No. 2023. Kavaratti is a ‘perfect atoll’ (Gardiner, 1903), and it is broadly divided into three physiographic divisions, viz. the island, lagoon and reef. The coral island, Kavaratti has a ‘chicken-leg’ shaped outline with a length of 5.8 km, a maximum width of 1.5 km and an area of 3.45 sq. km (Nasnolkar et al., 1997). The island along with its lagoon is aligned in the NNE-SSW direction (Figure 1). It rises not more than 3–4 m above mean sea level. The lagoon is shallow with an average depth of 2 m (Nasnolkar et al., 1997), and it is 4.5 km long and 1.2 km wide (Figure 1). Its bottom is not smooth because of the presence of pinnacles and knolls (Rao et al., 1987). The lagoon has two connections with the Lakshadweep Sea. The main inlet of the lagoon is at the northeastern end, while the secondary inlet is situated on the northwestern (leeward) flank of the lagoon. The coral reef is situated at a depth of 2 m on the leeward side of the Kavaratti lagoon and surrounds the lagoon on three sides. It is an elongated reef of organic limestone that encircles the shallow lagoon and is partly, intermittently or completely covered by water, depending upon the tide. The reef width varies between 100 m on the northwestern side and 750 m towards the southwestern tip of the lagoon.
Location map of the Kavaratti lagoon Lakshadweep showing the 18 locations under present study.
Methodology
The Kavaratti lagoon was sampled in May 2004 onboard a small mechanised fishing boat. The surface sediments were scooped up in appropriately pre-labelled plastic jars by skin diving. Using the Naval Hydrographic Chart No. 2023, sampling stations were selected following a grid pattern encompassing the entire lagoon. The sampling locations approximately 300–500 m apart were progressively numbered from 1 to 28. The geographical position of each location was determined by means of hand held Meggalin NAVDLX-10 Geographical Positioning System (GPS). The temperature and salinity of the water in the jars at each location were recorded using a thermometer and a hand-held refractometer.
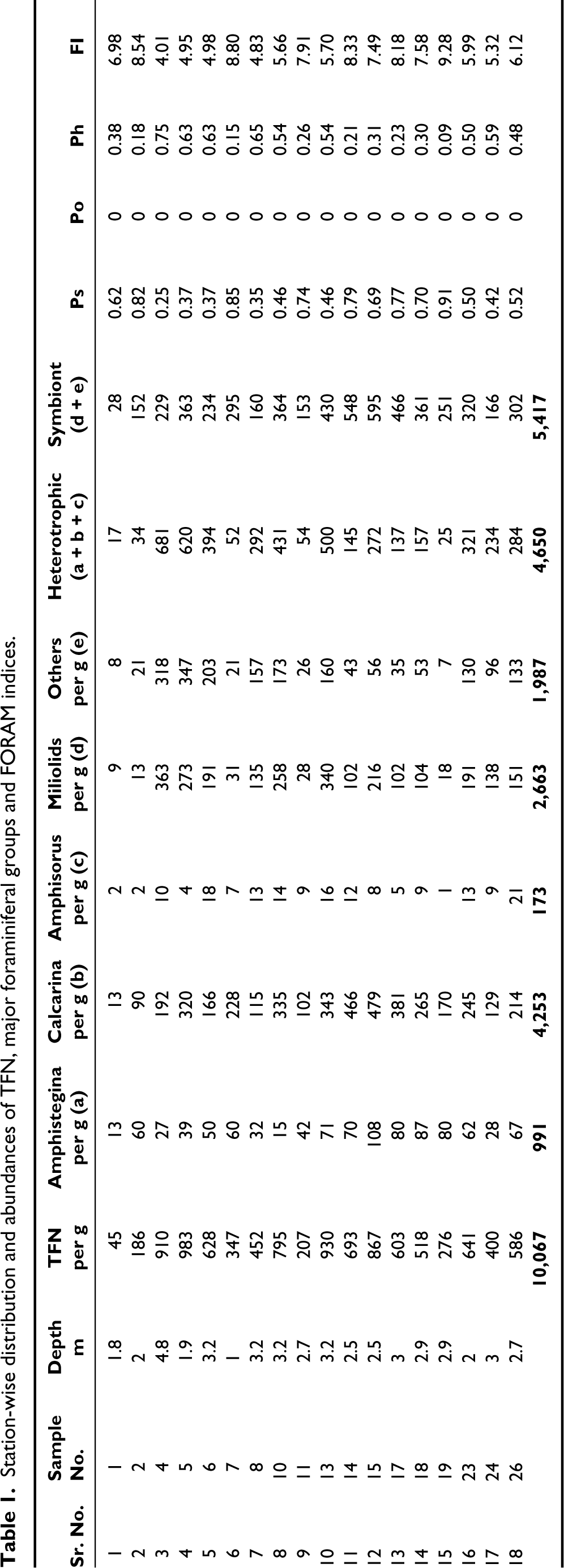
18 samples out of the 28 samples collected were selected for foraminiferal studies. The sediments being coarse were just washed with water to remove salt encrustations. The sediment samples were coned and quartered to obtain a representative fraction from which approximately 300 foraminiferal specimens were picked. After picking, the total foraminiferal specimens in each sample were counted. They were identified up to the generic level and their abundances were recorded. The different genera were grouped into three different functional groups as symbiont, opportunistic and smaller heterotrophic taxa, following Hallock et al. (2003) and Natsir and Subkhan (2011). Total foraminiferal number (TFN) and abundance of major foraminiferal groups in per gram sand fraction of each sample has been tabulated in Table 1. The FORAM (Foraminifera in Reef Assessment & Monitoring) Index (FI) was computed using the following formula proposed by Hallock et al. (2003) and listed in Table 1:
Station-wise distribution and abundances of TFN, major foraminiferal groups and FORAM indices.
FI = FORAM Index = FI = (10xPs) + (Po) + (2xPh)
Po = No/T = (total no. of symbiont bearing foraminifera/total no.of specimen counted)
Ph = Nh/T = (total no. of opportunistic foraminifera/total no.of specimen counted)
Ps = Ns/T = (total no. of small heterotrophic foraminifera/total no. of specimen counted)
Results
The water temperature in the Kavaratti lagoon ranges between 30.2 and 32.4˚C with no trend. The average salinity in the lagoon is 34.3–34.5‰.
Symbiotic genera Amphistegina, Calcarina, Amphisorus and the Miliolid group were the most abundant in the lagoon. The less abundant foraminifera were clubbed together as ‘Others’, which were constituted by the genera Textularia, Gaudryina, Bolivina, Discorbina, Cyclogyra, Rosalina, Heterostegina, Eponides, Cibicides, Planorbulina, Cymbaloporetta, Spirillina, Loxostomum and some specimens that could not be identified due to excessive polishing.
The spatial distribution in the lagoon is very patchy with no predictable trend. None of the samples yielded planktonic forms. The foraminiferal numbers are comparatively low at stations close to the inlets (Station No. 1 and 2 towards the north and Station No. 11, towards the west). The TFN in the lagoon ranges between 45 and 983 specimens per gram of dry sediments. The average foraminiferal number of the Kavaratti lagoon presently is 561 specimens per gram of sediments. It is also clear that the average TFN in the northern part of the lagoon (506/g) is lower than that in the southern part of the lagoon (612/g). The abundance of the different groups in the lagoon varies as follows: Calcarina > Amphistegina > Miliolids > Others > Amphisorus > Heterostegina. The abundance of Hyaline forms (mostly Calcarina, Amphistegina and ‘Others’) exceeds that of the porcellaneous forms (Miliolids and Amphisorus). The arenaceous forms are negligible (Textularia and Guadryina).
As no trend was evident in the spatial distribution of foraminiferal groups, depth contours were plotted in the study area map (Figure 1) to assess if any relationship existed between the foraminifera and depth. Amphistegina and Calcarina both seem to decrease in numbers with an increase in depth. Abundance of Calcarina group which also constitutes the hyaline forms increases towards the centre of the lagoon. Rare and juvenile specimens were encountered at the deepest station 24. It is also very evident that except for Amphisorus, the TFN and abundance of other groups are high in the south of the lagoon beyond the station numbers 26 and 10. The arenaceous forms are very low in numbers. Opportunistic species were not recovered in any of the samples.
Symbiont Calcarina is the most abundant in the lagoon followed by Miliolids, which are known to be common inhabitants of coral environments. Even though Miliolids and ‘Others’ together contribute to create the small heterotrophic foraminiferal functional group, they make up only 46% of the total foraminiferal in the lagoon while the symbiont-bearing functional group contribute to 54% (Table 1).
Discussion
The present environmental assessment of the lagoon is based on the sampling and reconnaissance of the lagoon done in 2004. The environment within the lagoon is characterised by a set of natural phenomenon and anthropogenic activities. Stronger currents prevail in the north of the lagoon and weaken out towards the south. This could be attributed to the fact that the inlets to the lagoon control the current circulation within the lagoon (Chandramohan et al., 1993, 1996) and both the inlets are in the northern half of the lagoon. The main inlet is periodically dredged to maintain the navigational channel (Chandramohan et al., 1996; Website of the Lakshadweep Economic Infrastructure). Thus, over the years the navigation channel has deepened (Figure 1 & Table 2). Previous studies have proven that even if the sediments get disturbed, they settle down fast to the bed due to their coarse size (Chandramohan et al., 1996). The opening in the north is relatively large. The sediments in suspension and bed load are transported southwards during the flood tide causing deposition along the southern part (Chandramohan et al., 1993).
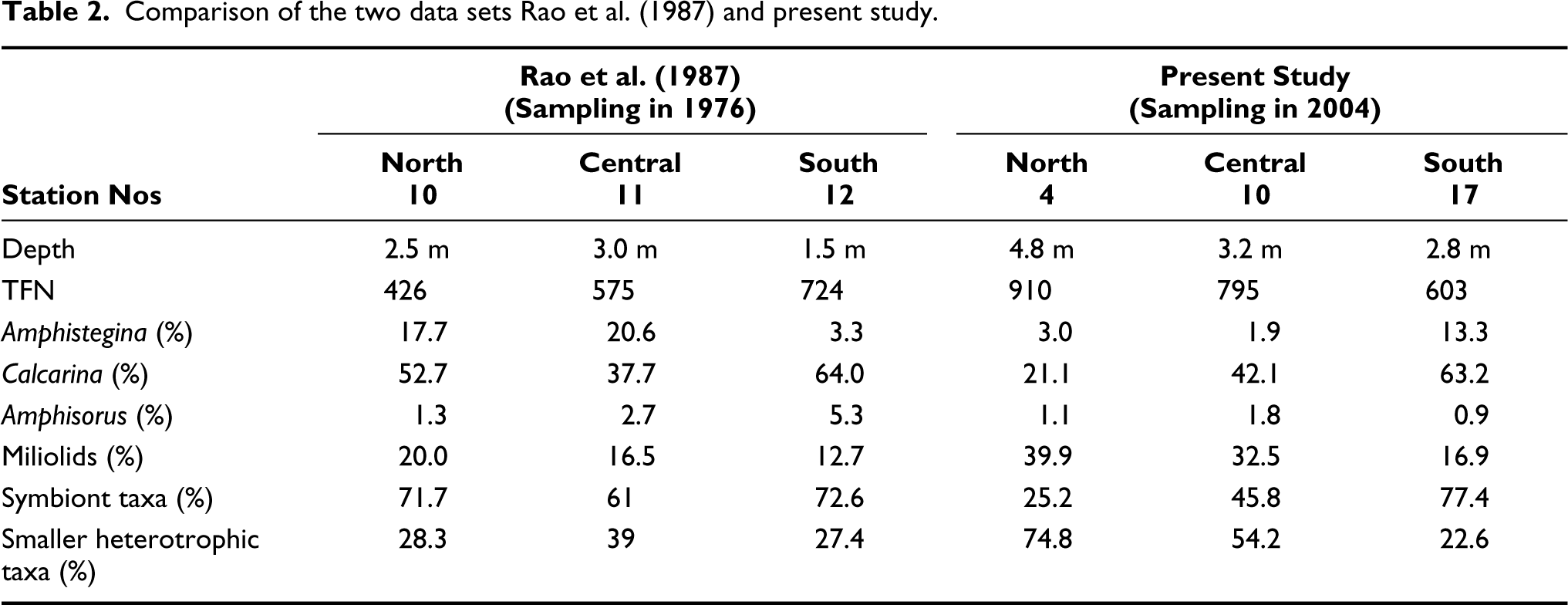
Comparison of the two data sets Rao et al. (1987) and present study.
During fieldwork, non-point domestic discharge into the lagoon was observed south of the lagoon beyond Station 26. Local islanders revealed that though each house on the island had a septic tank, islanders released their kitchen and domestic wastes directly into the lagoon, on which the authorities had no control. Although it is believed that natural ocean circulation is enough to flush the wastes out of the lagoon, the current dynamics prevalent in the lagoon do not favour it. The southern part of the lagoon is very shallow with weak currents less than 0.5 m/s (Chandramohan et al., 1996).
Water samples collected at Kavaratti showed high BOD, low DO, high suspended solid concentration and most viable in terms of microbial activity. The highest biomass was also recorded in the Kavaratti waters apart from reports of maximum coliforms and fecal coliforms, indicators of anthropogenic pollution at Kavaratti (Ramachandran & Varma, 1997). Interestingly, no opportunistic foraminiferal fauna is found in the present study, underscoring the intactness of the robust self-cleansing potential of the lagoon.
Abundant Calcarina and Amphistegina show an inverse relationship with depth, i.e., their abundances reduce with an increase in depth. Though LBF like Amphistegina develop thicker shells in environments exposed to wave action, they are also known to be diatom-bearing (Hallock, 1979; Hallock et al., 1986; Hallock & Hansen, 1979; Hansen & Buchardt, 1977; Larsen, 1976; Reiss & Hottinger, 1984). This could explain the preference of these symbiont-bearing foraminifera to occupy shallower portions of the lagoon.
In the present study, abundance of Amphisorus is highest towards the North and reduced southwards, in contrast to the trend of other groups. According to Saraswati (2002), discoidal Miliolids (Amphisorus, Sorites and Marginopora) are evidence of high-energy environments. Amphisorus is most common amongst those inhabiting highly energetic environments by taking shelter in seagrass and spaces between coral rubbles. Their lower numbers in the south thus indicate that the energy conditions in the south are lower as compared to that in the north, which has also been reported before (Chandramohan et al., 1996). During the present fieldwork, abundant sea grass was observed along the lagoon coast North of the lagoon, more close to the jetty explaining the higher abundance of Amphisorus in the region.
The relatively low TFN (average 506/g) in the northern part of the lagoon, especially at stations close to the inlets (1, 2 and 11) can be attributed to the water currents and periodic dredging of the navigational channel. However, then high TFN at stations 4, 5 and 10 can be debated. This could be because of high numbers of smaller hyaline forms constituting ‘Others’ and Miliolids that are neither symbiotic nor averse to currents.
Sewage and waste disposal are known to serve as nutrients to foraminifera (Watkins, 1961; Zalesny, 1959). Thus, even in the present study, the higher foraminiferal abundance in the south (612/g) could be attributed to the non-point domestic discharge in the lagoon. Chandramohan et al. (1993) report that suspended material in the lagoon is also reported to be diverted to and deposited in the south. Additionally, the weak currents in the south could be the cause of poor flushing and cause of accumulation of the waste.
Station 24 shows the lowest TFN (400 specimens per gram) in comparison to the other stations in the south. Samir and El-Din (2001) have reported a reduction in abundance and diversity in the face of nutrient pollution. Hallock (2000) has reported that large, symbiont-bearing foraminifera lose dominance to small, fast-growing herbivorous and detritivore species when nutrient supply increases in tropical reef-associated environments. The small heterotrophic benthic foraminifera constitute almost 60% of the assemblage at this station. This could be indicative of a contaminated/stagnated location within the lagoon. Hyaline and perforate forms have been reported to increase near the sewage aureoles (Bandy et al., 1964). This station also shows maximum depth in the south, which along with stagnation seems to be the probable reason for low Calcarina, at this station. It is noteworthy, that this is also the only location where Heterostegina depressa are encountered (though low in abundance). This species is known to be adapted to low light intensities and are active feeders of algae (Röttger et al., 1990).
The low numbers of arenaceous forms in the lagoon can be attributed to the coarse nature of sediments in the lagoon that cannot be taken up by foraminifera to build their test (Rao et al., 1987).
FORAM Index
As per Hallock et al. (2003), FORAM indices higher than 4 are all indicative of environments conducive for the growth of corals. The FI computed for all the stations in the lagoon are higher than 4. Sampling location 4 situated at the northern inlet shows a critical threshold value of 4.01, which is suggestive of guaranteed environmental change since 1976, which is discussed in the next section. The other locations in the lagoon that are slightly above 4 and appear vulnerable are locations 5, 6 and 8, all in the path of dredging in the northern half of the lagoon. FI lower than 2 would suggest that the environment would not support coral growth.
Comparison with Previous Data
Rao et al.’s (1987) work was based on sampling conducted in 1976. The present work is based on sampling conducted in 2004, that is, nearly 3 decades after the previous studies. This gave an opportunity to monitor the lagoon and detect any change in the lagoon over time. Rao et al. (1987) had only three sampling stations in the lagoon. In the present study, samples were collected at comparable stations (locations 4, 10 and 17) to enable comparison of the two data sets (Table 2).
According to Rao et al. (1987), the average TFN of the Kavaratti lagoon was 65,667 specimens per 100 g sand fraction, whereas, in the present study, it was recorded as 56,100 specimens per 100 g of sediments, indicating a 15% fall in the average TFN of the lagoon.
There is a definite deepening of all sampling locations being compared (Table 2). There is a substantial rise in TFN in the Northern and Central location while there is a 17% drop in TFN in the Southern station. However, this rise and fall in TFN can be attributed to the drop in symbiont taxa in the north and central stations and a rise in symbiont forms in the southern location. Though the southern location seems to have got slightly more conducive for the growth of corals, the northern and central locations show substantial deterioration in the lagoonal environment and that can be attributed to the periodic dredging and continuous navigational activities in the lagoon.
In Rao et al.’s (1987) data, the southern station showed maximum abundance of Amphisorus (5.3%). However, the comparable station in the present study indicates a tremendous drop in Amphisorus abundance (0.9%). The reasons for this change have already been discussed above. At the same station, Rao et al. (1987) had found very low numbers of Amphistegina (3.3%). In the present study, these numbers have significantly gone up (13.3%). This could be because the northern and central locations have now deepened in comparison to the southern location and Amphistegina prefers shallow depths. Even Calcarina shows a similar pattern of change over the years. Miliolids show an overall increase in the lagoon; a two-fold increase at the first two stations and a slight increase in the southernmost station.
The Kavaratti is a busy island with maximum human and developmental activities in the Lakshadweep, which have only intensified over the past two decades. Faecal and nutrient pollution and human-induced metal contamination have been reported from coral sediments and coastal and groundwater of the Kavaratti (Antony et al., 2022; Madhusoodanan Pillai & Ouseph, 2002; Sudhanandh et al., 2013). The present study detects the changes in the Kavaratti lagoon that have occurred between 1976 and 2004. There is a need to repeat the environmental impact assessment of the coral health using the LBF, which could hold great potential to manage and conserve the coral lagoon.
Conclusions
Amphistegina, Calcarina, Amphisorus and the Miliolid group are the most abundant foraminiferal taxa in the Kavaratti lagoon of Lakshadweep. The less abundant to rarely occurring taxa are represented by the genera Textularia, Gaudryina, Bolivina, Discorbina, Cyclogyra, Rosalina, Heterostegina, Eponides, Cibicides, Planorbulina, Cymbaloporetta, Spirillina and Loxostomum. The FI of all the 18 locations analysed in the lagoon suggest that the lagoon had an environment conducive for coral growth as of 2004. It shows a definite environmental change at the northern inlet and deterioration of environment in the northern and central regions of the lagoon. This can be attributed to deepening of the lagoon due to periodic dredging of the lagoon to support navigational activities. The southern portion of the lagoon has shown a slight improvement since 1976. Location 24 was the only location in the lagoon where smaller heterotrophic taxa exceeded the symbiont taxa attributable to domestic discharge and poor flushing in the region. New changes have been anticipated in the lagoon over the past two decades.
Footnotes
Acknowledgements
The authors thank the Director, the National Institute of Oceanography, Dona Paula Goa and the Directorate of Science and Technology at Lakshadweep to support the study logistically and financially. Mr T.P. Amanulla, Mr Anwer Rabodiya and Mr Saheer are thanked for their help and assistance in sampling the Kavaratti lagoon. R.P. is thankful to the Ministry of Earth Sciences (Department of Ocean Development), Government of India for a Research Fellowship on the BENFAN Project. L.V.N. acknowledges the Science and Society Division of the Department of Science & Technology, New Delhi, for the financial assistance in the form of a Women Scientist Scholarship (WOS-B).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The present work was institutionally funded by CSIR-National Institute of Oceanography, Goa. R.P. is thankful to Ministry of Earth Sciences (Department of Ocean Development), Government of India for Research Fellowship on the BENFAN Project. L.V.N. acknowledges Science and Society Division of Department of Science & Technology, New Delhi for the financial assistance in the form of Women Scientist Scholarship (WOS-B).
