Abstract
Objectives
To investigate the distinctive features of lymphocytes promoting inflammation in ulcerative colitis.
Methods
We performed flow cytometric analysis of peripheral blood mononuclear cells (PBMCs) and colorectal mucosa lymphocytes in ulcerative colitis patients (n = 13) and control patients (n = 5).
Results
CD62L+/CD3+CD4+ (35.7 ± 14.0% vs. 19.9 ± 6.4%) and CD62L+/CD3+CD4− cells (17.1 ± 17.4% vs. 2.4 ± 3.9%) were higher in the rectum of ulcerative colitis patients than in control patients. Subpopulation analysis revealed that CD45RA−CD62L+/CD3+CD4+, that is, central memory T cell fraction in CD4+ T cells, was significantly increased in the rectum of ulcerative colitis, compared to that in control patients (23.3 ± 10.5% vs. 8.2 ± 4.0%). Comparison of rectum and colon samples in ulcerative colitis patients indicated that CD56+/CD3+ was decreased in the rectum compared to that in the colon (11.3 ± 12.5% vs. 21.3 ± 16.5%). The ratio of CD56+/CD3+ was also decreased in the rectum of active ulcerative colitis patients compared to that in ulcerative colitis patients at the endoscopic remission stages (2.8 ± 1.7% vs. 18.5 ± 13.3%).
Conclusion
We demonstrated that CD62L+ T lymphocytes, particularly the CD45RA−CD62L+ T cell subset that represents central memory T cells, were increased in the rectum of patients with ulcerative colitis. In addition, the CD56+/CD3+ subset (natural killer T cells) was decreased in the rectum compared to that of less inflamed colonic mucosa. These results suggest that the enrichment of central memory T lymphocytes and the reduction of natural killer T cells in the gut mucosa are involved in the pathogenesis of ulcerative colitis.
Keywords
Background
Ulcerative colitis is an inflammatory bowel disease that causes prolonged inflammation primarily in the colorectum, which leads to mucosal erythema, friability, erosions, and/or ulcer formation. Although the exact pathophysiology of this disease has not been elucidated yet, immune-mediated tissue destruction secondary to a combination of barrier dysfunction, genetic risk, and dysbiosis is essential for the development and deterioration of inflammation in the gut.1–3 Since T lymphocytes play a central role in cell-mediated immunity, they are thought to be involved in the pathogenesis of ulcerative colitis.
Flow cytometry enables the typing of immune cells based on the presence of specific cell surface markers, expression of cytokines, and phosphorylation of key proteins. Therefore, flow cytometry can analyze lymphocytes in patients with ulcerative colitis. In previous studies, peripheral blood mononuclear cells (PBMCs) 4 and colorectum-resident lymphocytes5,6 were primarily used as sample cell sources for flow cytometric analysis. The composition of T lymphocytes in intestinal tissues differs from that in the blood due to intestinal microbiota responses and homing receptor/ligand interactions 7 ; however, there is a lack of data on the in-depth surface marker profiles of PBMCs and colorectum-resident T lymphocytes in patients with ulcerative colitis. This study aimed to perform a comprehensive flow cytometric characterization of T lymphocytes in ulcerative colitis and reveal the distinctive features of lymphocytes that promote inflammation in this disease.
Materials and methods
Overview
To characterize the lymphocyte surface markers of PBMCs and the colorectum and reveal the distinctive features of lymphocytes that promote inflammation in ulcerative colitis, comparative analyses were performed between (i) rectum-resident lymphocytes in patients with ulcerative colitis and those in control patients, (ii) PBMCs in patients with ulcerative colitis and those in control patients, (iii) lymphocytes from the inflamed mucosa (rectum) and those from less-inflamed mucosa (colon) in patients with ulcerative colitis, and iv) rectum-resident lymphocytes in the active stage and those in the remission stage in patients with ulcerative colitis.
To comprehensively analyze the profiles of T lymphocytes in patients with ulcerative colitis using flow cytometry, the expressions of helper T-lymphocyte marker CD4, cytotoxic T-lymphocyte marker CD8, regulatory T (Treg) lymphocyte markers CD25 and CD127, natural killer cell marker CD56 (also known as neural cell adhesion molecule), mature T-lymphocyte marker CD7, central inhibitory receptor programmed death receptor-1 (PD1, also known as CD279), activated T-lymphocyte marker CD30 (also known as tumor necrosis factor receptor superfamily member 8), human leukocyte antigen DR isotype (HLADR), and type-2 helper T-lymphocyte marker C-C chemokine receptor type 4 (CCR4, also known as CD194) were assessed. The expressions of CD45RA and CD62L were assessed to categorize lymphocytes into subpopulations such as effector memory T cells, central memory T cells, and naïve T cells.
Ulcerative colitis patients
Flow cytometry was prospectively performed between April and November 2020 at Okayama University Hospital (Okayama, Japan) on endoscopic biopsy specimens obtained from 14 patients who had been diagnosed with ulcerative colitis. The inclusion criteria for ulcerative colitis patients were: (i) individuals who had never undergone surgical resection of the colorectum, and (ii) those who had not been taking biologic drugs, including infliximab, adalimumab, golimumab, vedolizumab, and ustekinumab, or the Janus kinase inhibitor, tofacitinib, for at least 6 months prior to study enrollment. Disease activity was determined according to the Mayo endoscopic subscore (MES). 8 Briefly, normal or inactive ulcerative colitis indicated MES 0; mild disease showing erythema, decreased vascular pattern, and/or mild friability indicated MES 1; moderate disease with marked erythema, absent vascular pattern, friability, and/or erosions indicated MES 2; and severe disease with spontaneous bleeding and/or ulceration indicated MES 3. As no patients presented an MES of 3, all patients in this study were classified as having MES 0, 1, or 2, as described below.
Follow-up endoscopy and biopsy were performed as part of standard care for screening neoplasia, mostly on a yearly basis. In one of the ulcerative colitis patients, flow cytometry analysis was solely performed on two samples from the sigmoid colon mucosa, as specimens were not taken from the rectum. Therefore, this patient was excluded from the study. For each of the remaining 13 ulcerative colitis patients, a single specimen was obtained using disposable biopsy forceps from the rectal mucosa during endoscopy, and another specimen was obtained from the colon, where inflammation was absent or mild. Lymphocytes were isolated from each biopsied specimen using the one-step lymphocyte isolation procedure, which was reported in our earlier work.9–11 In addition, PBMCs were isolated from 5 mL of peripheral venous blood by density centrifugation using Lymphoprep (Alere Technologies AS, Oslo, Norway).
Patients with colon polyp
Flow cytometry analysis was also performed in five patients who underwent endoscopic mucosal resection (EMR) for colorectal polyps. The inclusion criteria for patients with colorectal polyps were as follows: (i) patients without known inflammatory bowel disease or other autoimmune disorders, (ii) patients without colitis, (iii) patients who were not taking immunosuppressive or anticancer drugs, and (iv) patients who had never undergone surgical resection of the colorectum.
We isolated lymphocytes from endoscopically obtained specimens from the rectum of each of the five patients with colon polyps. PBMCs were isolated from 5 mL of peripheral venous blood using Lymphoprep.
Flow cytometry
PBMCs or lymphocytes isolated from the colon or rectal mucosa were utilized for flow cytometry. Monoclonal antibodies against CD45 (clone J33; Beckman Coulter, Pasadena, CA, USA), CD3 (UCHT1, Beckman Coulter), CD4 (13B8.2; Beckman Coulter), CD8 (B9.11; Beckman Coulter), CD7 (8H8.1; Beckman Coulter), CD25 (B1.49.9; Beckman Coulter), CD30 (HRS4; Beckman Coulter), CD45RA (2H4; Beckman Coulter), CD56 (N901; Beckman Coulter), CD62L (DREG56; Beckman Coulter), CD127 (R34.34; Beckman Coulter), CCR4 (i.e., CD194; L291H4; BioLegend), HLADR (Immu-357; Beckman Coulter), and PD1 (CD279; PD1.3; Beckman Coulter) were employed. The immunostained cells were analyzed using FACScan (Navios flow cytometer, Beckman Coulter) and Kaluza analysis software (version 1.3; Beckman Coulter). Lymphocytes were separated by flow cytometry based on high CD45 antigen expression and forward and side scatter properties. Subsequently, the flow cytometry data were analyzed according to the percentage of cell populations detected in each quadrant on two-dimensional scatterplots. We calculated the percentages of CD4+, CD8+, CD56+, CD7+, PD1+, CCR4+, CD30+, and HLADR+ cells among CD3+ cells. We also assessed the percentages of Treg, CD45RA+, and CD62L+ cells among CD3+CD4+ cells and percentages of CD45RA+ and CD62L+ cells among CD3+CD4− cells. In this study, we defined CD3+CD4+CD25+CD127low/- cells as Treg cells.
Analysis
First, to assess the differences between the lymphocyte population of patients with colon polyps (control) and that of ulcerative colitis patients, we compared the flow cytometry results for PBMC samples and rectal samples between the two groups. Second, to reveal the characteristics of the rectum-resident lymphocytes in ulcerative colitis patients, we contrasted the flow cytometric results from rectum samples with those of colon samples or PBMCs. Third, to evaluate the relationship between inflammation severity and lymphocyte composition in ulcerative colitis, we compared the flow cytometry results of PBMC, rectum, and colon samples between MES 0 patients and MES 1 and 2 patients. Fourth, to evaluate the relationship between the clinical courses of patients within 6 months after colonoscopy and the lymphocyte composition in ulcerative colitis, we compared the flow cytometry results of rectal samples between patients with and without relapse. The relapse group was defined as patients in whom clinical symptoms, such as increased bloody stool, diarrhea, abdominal pain, uncontrolled fever, and increased levels of inflammatory markers, worsened within 6 months of colonoscopy or those who required induction of biologic drugs or tofacitinib. The non-relapse group was defined as patients without worsening of clinical symptoms within 6 months following colonoscopy.
Statistical analyses were performed using JMP 14.0.0 software (SAS Institute Inc., Cary, NC, USA). Significant differences between samples were determined using paired or unpaired t-tests, where indicated. Differences were considered significant if the p value was less than 0.05.
Ethics approval
The patients were prospectively registered and analyzed in this study. Written informed consent was obtained from all participants. This study adhered to the principles of the Declaration of Helsinki and was approved by the ethics committee of Okayama University Hospital (approval number: 2003-011 and 2009-014). The study protocol was registered in the UMIN Clinical Trials Registry (UMIN000039868).
Results
Patients’ characteristics
Characteristics of the enrolled patients.
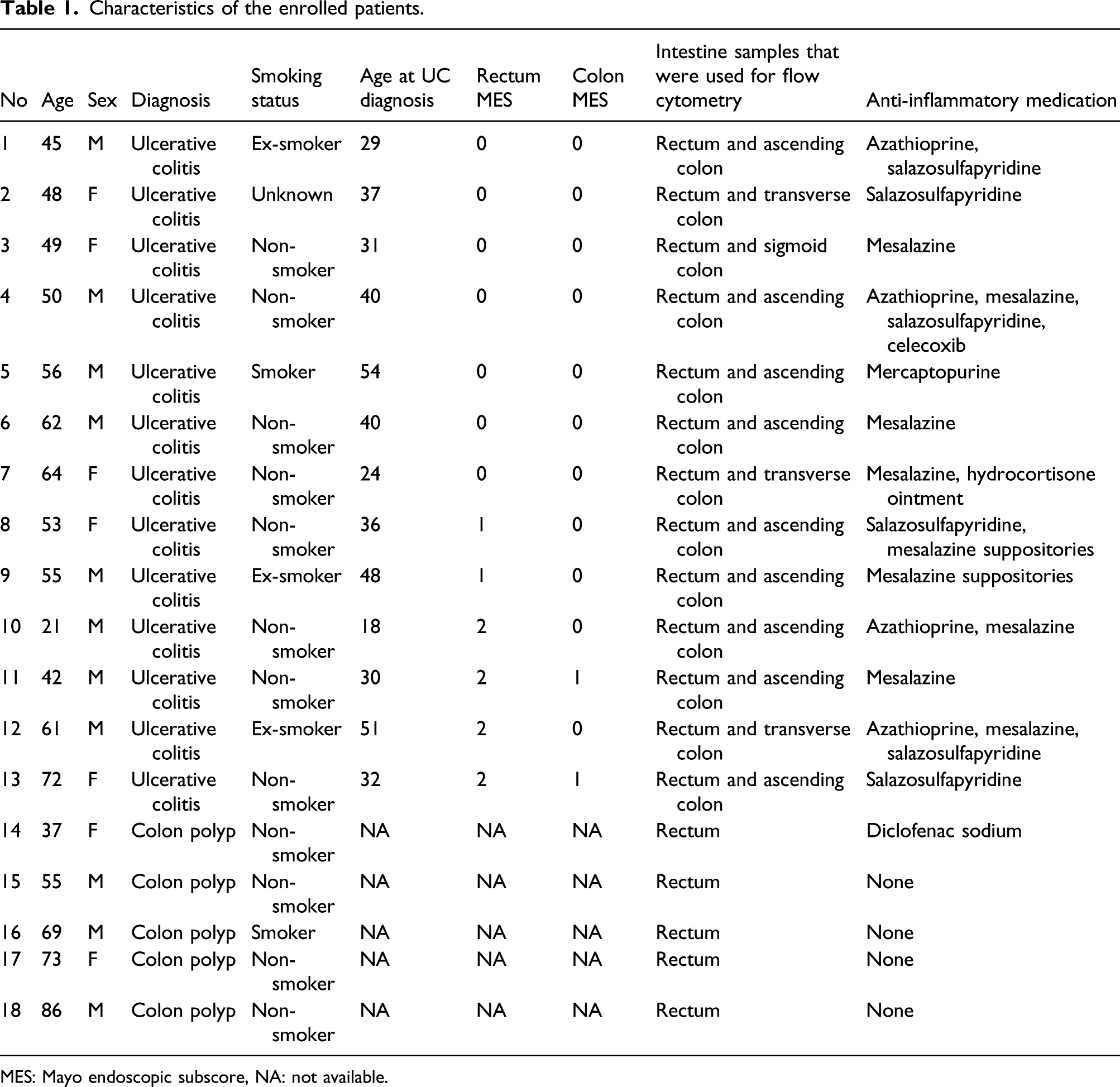
MES: Mayo endoscopic subscore, NA: not available.
In the control group, which consisted of five patients with colon polyps, there were three men and two women; average age was 64.5 years (range, 37–86 years). All patients, except one who was a current smoker, were non-smokers. One patient had been taking diclofenac sodium, whereas the other patients had not been taking anti-inflammatory agents.
Statistical differences were not observed in terms of sex and age between the ulcerative colitis and control groups. Further, significant differences were not observed in terms of patient characteristics between the inactive (MES 0) and active (MES 1 and 2) ulcerative colitis subgroups.
Lymphocyte composition in control and ulcerative colitis patients
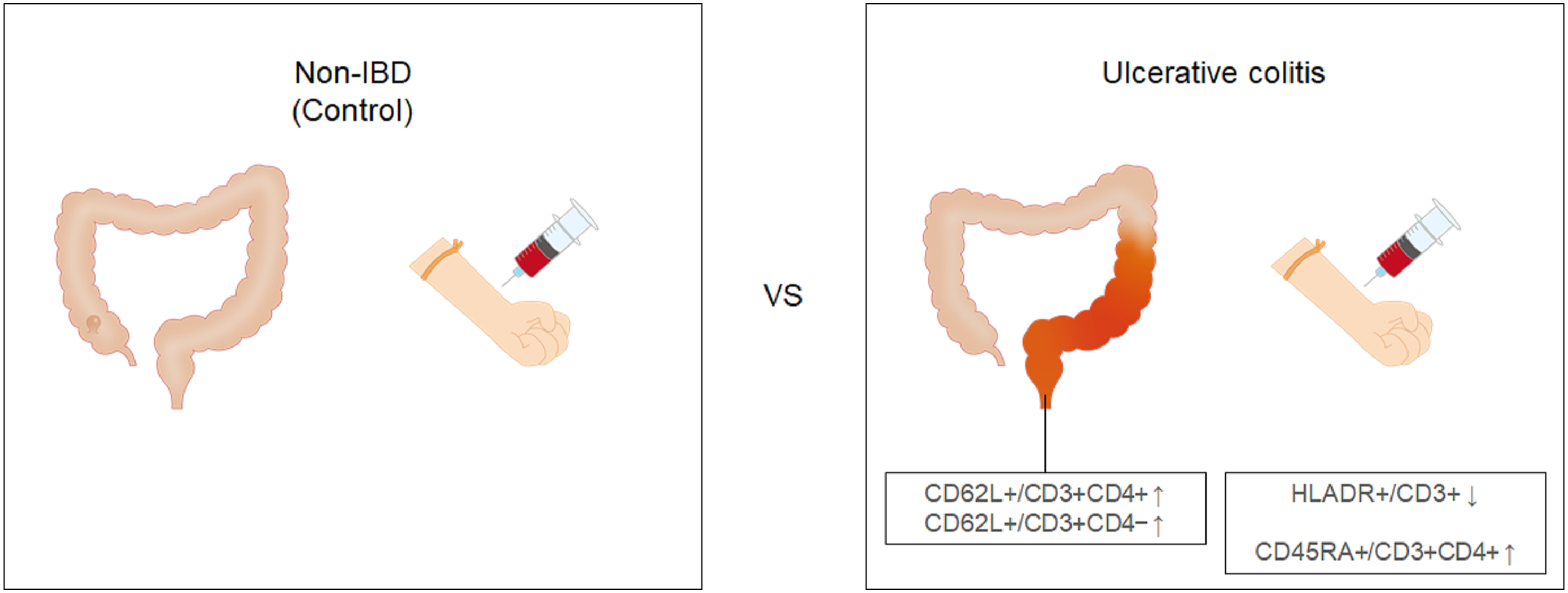
The flow cytometry results for all samples are shown in Figure 1. Comparison between control and ulcerative colitis groups revealed that ratios of CD62L+/CD3+CD4+ (35.7 ± 14.0% vs. 19.9 ± 6.4%) and CD62L+/CD3+CD4− cells (17.1 ± 17.4% vs. 2.4 ± 3.9%) were higher in the rectum of patients in the ulcerative colitis group than those of the control group. In PBMCs from ulcerative colitis patients, HLADR+/CD3+ ratio was decreased (5.1 ± 6.1% vs. 19.6 ± 7.3%), while CD45RA+/CD3+CD4+ ratio was increased (55.1 ± 15.1% vs. 43.9 ± 5.0%) compared to PBMCs from control patients (Figure 2). Flow cytometry analysis. Lymphocyte composition was analyzed in samples from control and ulcerative colitis patients. Regulatory T (Treg) cells were defined as CD3+CD4+CD25+CD127low/− cells. *, p < 0.05 (paired t-test); **, p < 0.01 (paired t-test); †, p < 0.05 (unpaired t-test); ††, p < 0.01 (unpaired t-test); PBMC, peripheral blood mononuclear cells; UC, ulcerative colitis. Images depicting differences in lymphocyte composition between patients with colon polyps (control) and ulcerative colitis patients. In the rectum of ulcerative colitis patients, CD62L+/CD3+CD4+ and CD62L+/CD3+CD4− were increased, compared to those of control patients. HLADR+/CD3+ was decreased and CD45RA+/CD3+CD4+ was increased in peripheral blood mononuclear cells (PBMCs) of ulcerative colitis patients, compared to those of control patient PBMCs.
Characteristics of the rectum-resident lymphocytes in ulcerative colitis
Comparison between the rectum and colon samples in ulcerative colitis patients revealed that CD56+/CD3+ ratio was decreased (11.3 ± 12.5% vs. 21.3 ± 16.5%), while CD4+/CD3+ (71.2 ± 11.0% vs. 59.7 ± 19.4%) and PD1+/CD3+ (78.1 ± 6.6% vs. 69.1 ± 10.8%) ratios were increased in the rectum compared to those in the colon mucosa (Figure 3). Furthermore, lymphocyte composition from the rectum and from PBMC samples in ulcerative colitis patients differ in various aspects: the ratios of CCR4+/CD3+, CD45RA+/CD3+CD4+, CD62L+/CD3+CD4+, CD45RA+/CD3+CD4−, and CD62L+/CD3+CD4− were significantly lower, whereas the ratios of CD8+/CD3+, PD1+/CD3+, CD30+/CD3+, and HLADR+/CD3+ were significantly higher in the rectum than in PBMCs. An image illustrating differences between lymphocyte composition of the less-inflamed mucosa (colon) and that of the rectum in ulcerative colitis patients. Although the CD56+/CD3+ fraction was decreased, CD4+/CD3+ and PD1+/CD3+ were enriched in the rectum compared to those in the colon of ulcerative colitis patients.
Disease activity and lymphocyte composition in ulcerative colitis
A comparative analysis between the flow cytometry results of MES 1 and 2 ulcerative colitis patients (n = 6) and those of MES 0 patients (n = 7) showed that CD8+/CD3+ (28.7 ± 10.7% vs. 52.7% ± 18.2%) and CD56+/CD3+ (2.8 ± 1.7% vs. 18.5 ± 13.3%) ratios were decreased in the rectum of MES 1 and 2 patients, compared to those of MES 0 patients (Figure 4). With respect to the colon mucosal lymphocytes, we found a lower ratio of CD30+/CD3+ (3.1 ± 2.0% vs. 7.9 ± 3.3%), and a higher ratio of CD7+/CD3+ (91.8 ± 4.0% vs. 80.4 ± 12.2%) in MES 1 and 2 patients than in MES 0 patients. In addition, CCR4+/CD3+ ratio was increased in PBMCs from MES 1 and 2 patients compared to that from MES 0 ulcerative colitis patients (28.7 ± 7.9% vs. 15.5 ± 5.6%) (Figure 5). Flow cytometry analysis comparing samples from ulcerative colitis patients with Mayo endoscopic subscore (MES) of 0 and those from patients with an MES of 1 or 2. * p < 0.05, unpaired t-test; PBMC, peripheral blood mononuclear cells. Images depicting differences in lymphocyte composition between ulcerative colitis patients with Mayo endoscopic subscore (MES) of 0 and those of MES 1 or 2. Compared to patients with MES 0, CD30+/CD3+ was decreased and CD7+/CD3+ was increased in the colon, CD8+/CD3+ and CD56+/CD3+ were decreased in the rectum, and CCR4+/CD3+ was increased in the peripheral blood mononuclear cells (PBMCs) of ulcerative colitis patients with MES 1 or 2.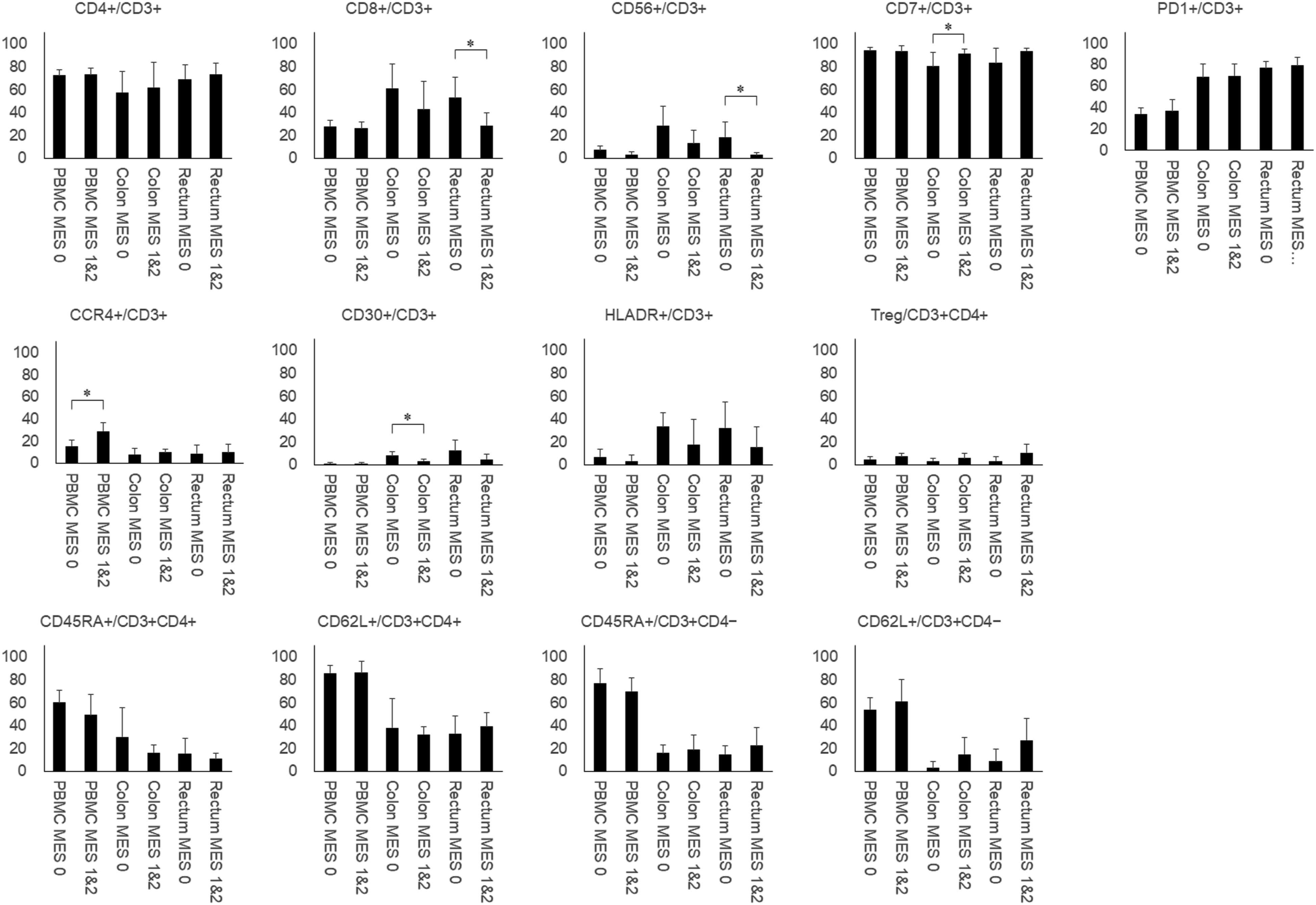

Central memory T cells among the rectum-resident lymphocytes in ulcerative colitis
As described above, we discovered that ulcerative colitis patients had increased ratios of CD62L+/CD3+CD4+ and CD62L+/CD3+CD4− cells in the rectal mucosal lymphocytes compared to those in patients from the control group. To explore the changes in lymphocyte composition in depth, we further separated CD3+CD4+ and CD3+CD4− cells into four subpopulations based on their CD45RA and CD62L expression: CD45RA+CD62L− cells were considered as effector memory T cells, CD45RA−CD62L+ cells as central memory T cells, CD45RA+CD62L+ cells as naïve T cells, and CD45RA−CD62L− cells (Figure 6). As shown in Figure 7, the CD45RA−CD62L+/CD3+CD4+ fraction was increased in the rectum of ulcerative colitis patients, compared to that of control patients (23.3 ± 10.5% vs. 8.2 ± 4.0%). Although the ratio of CD45RA−CD62L+/CD3+CD4− was also higher in the rectum of ulcerative colitis patients than in control patients (8.1 ± 6.7% vs. 1.5 ± 2.9%), this difference was not statistically significant (p = 0.051). Representative flow cytometric results of the rectal lymphocytes from an ulcerative colitis patient. Two-dimensional scatterplots using CD45RA and CD62L antibodies show that CD45RA−CD62L−, CD45RA+CD62L−, CD45RA−CD62L+, and CD45RA+CD62L+ fractions account for 43.7%, 0.4%, 36.8%, and 19.1% of CD3+CD4+ cells, respectively. Among the CD3+CD4− cells, CD45RA−CD62L−, CD45RA+CD62L−, CD45RA−CD62L+, CD45RA+CD62L+ fractions account for 64.6%, 5.0%, 14.9%, and 15.5%, respectively. Subpopulation analysis based on CD45RA and CD62L expression. CD45RA+CD62L− cells represent effector memory T cells, CD45RA−CD62L+ cells represent central memory T cells, and CD45RA+CD62L+ cells represent naïve T cells. *, p < 0.05; paired t-test, versus colon; ††, p < 0.01 (unpaired t-test, vs. rectum of the control patients).

Comparison between MES 1 and 2 ulcerative colitis patients and MES 0 ulcerative colitis patients revealed that the CD45RA−CD62L+/CD3+CD4− fraction was higher in the MES 1 and 2 group than in MES 0 group (12.6 ± 4.8% vs. 4.3 ± 5.8%) (Figure 8). Despite the CD45RA−CD62L+/CD3+CD4+ fraction being higher in MES 1 and 2 than in MES 0 (29.2 ± 7.6% vs. 18.2 ± 10.4%), this difference was not statistically significant (p = 0.052). Subpopulation analysis based on CD45RA and CD62L expression comparing the rectum-resident lymphocytes between ulcerative colitis patients with Mayo endoscopic subscore (MES) of 0 and those of MES 1 or 2. CD45RA+CD62L− cells represent effector memory T cells, CD45RA−CD62L+ cells represent central memory T cells, and CD45RA+CD62L+ cells represent naïve T cells. †, p < 0.01 (unpaired t-test).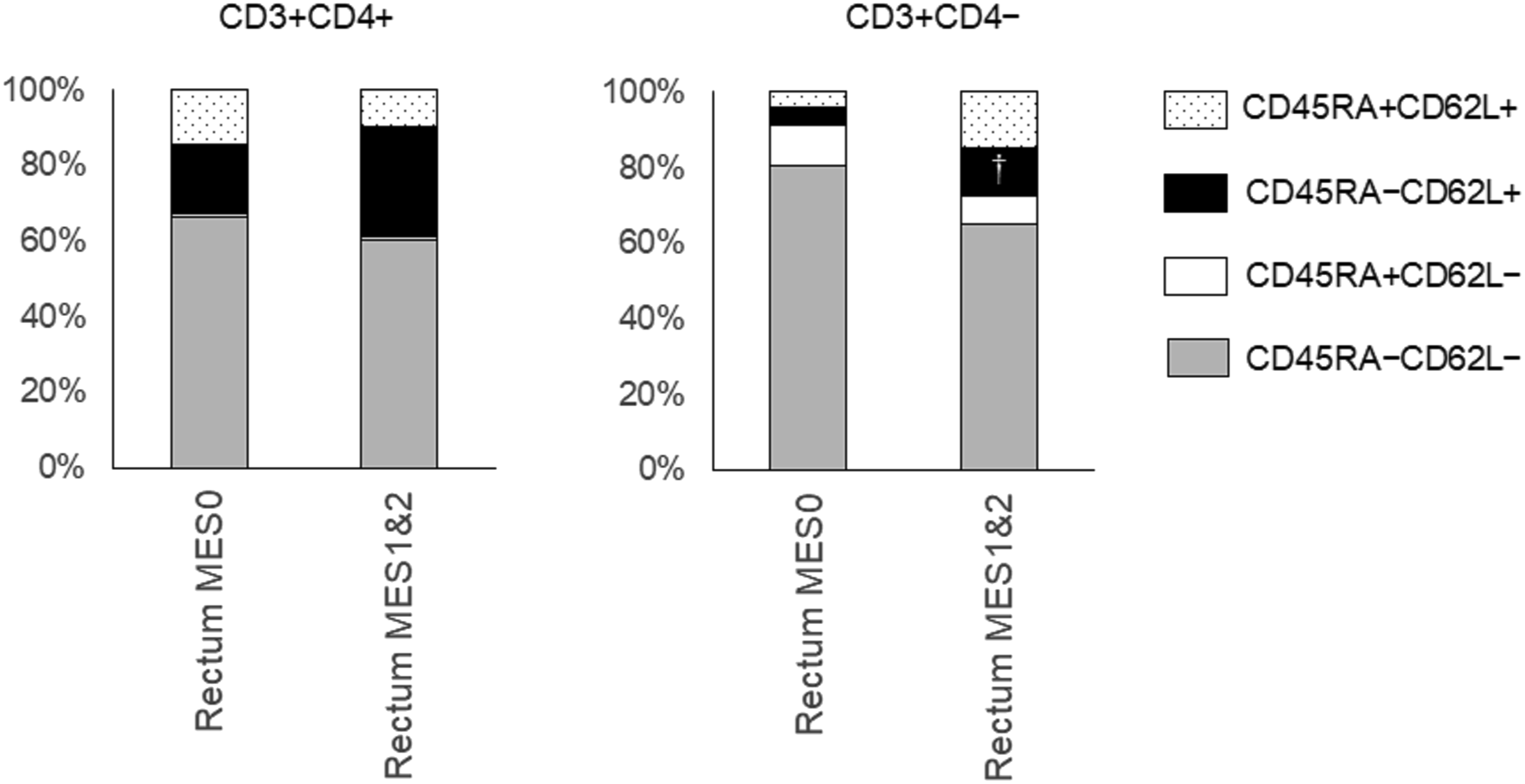
Central memory T cells and natural killer T cells as potential predictors of a relapsing clinical course
Within 6 months after the colonoscopy, one patient (Case 8) developed bloody stool, one patient (Case 11) developed worsening diarrhea, and another patient (Case 10) required administration of vedolizumab to control intestinal inflammation. Thus, the relapse group comprised these three patients, while the non-relapse group comprised the other 10 patients.
Based on the results obtained, we investigated the ratio of central memory T cells (CD45RA−CD62L+ cells) and natural killer T (NKT) cells (CD3+CD56+ cells) as a potential predictor of relapsing clinical course in patients with ulcerative colitis (Figure 9). The CD45RA−CD62L+/CD3+CD4+ fraction was higher in the relapse group than in the non-relapse group (31.1 ± 3.8% vs. 21.0 ± 10.9%, p = 0.03). Furthermore, the CD45RA−CD62L+/CD3+CD4− fraction was higher (15.1 ± 4.4% vs. 6.0 ± 5.9%, p = 0.04), while the CD3+CD56+ fraction was lower in the relapse group than in the non-relapse group (2.8 ± 1.5% vs. 13.8 ± 13.2%, p = 0.03). A cutoff value of CD45RA−CD62L+/CD3+CD4− fraction ≥10% versus <10% provided a sensitivity of 100% and specificity of 80%. In addition, CD45RA−CD62L+/CD3+CD4+ fraction ≥25% had a sensitivity of 100% and specificity of 60% for differentiating patients with relapse from those without relapse. CD3+CD56+ fraction <5% had a sensitivity of 100% and specificity of 60% for differentiating patients with relapse. Flow cytometry analysis of rectum samples from ulcerative colitis patients with relapse within 6 months after colonoscopy and those without relapse. *, p < 0.05 (unpaired t-test).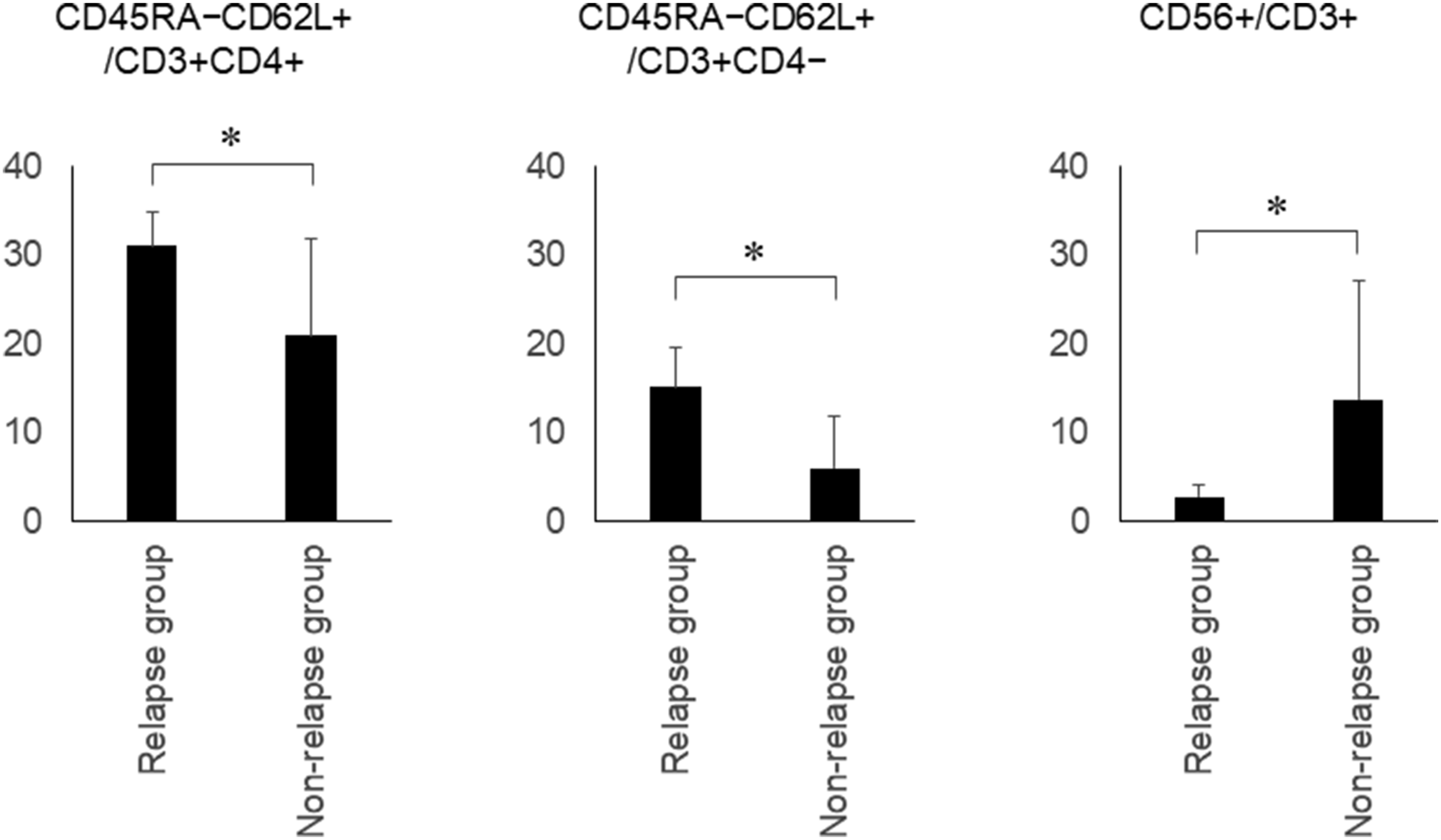
Discussion
Comprehensive flow cytometry analysis revealed that the composition of T lymphocytes is different in various aspects between PBMCs, lymphocytes from the less-inflamed mucosa (colon), and lymphocytes from inflamed mucosa (rectum) in patients with ulcerative colitis (Figure 1). The present results reinforce that sample cell sources for flow cytometric analysis should be selected cautiously in research studies on ulcerative colitis. We consider that isolation of lymphocytes from the rectal mucosa and its use for flow cytometric analysis is the most desirable to discover essential features involved in the pathogenesis of ulcerative colitis.
In this study, we performed flow cytometric analysis of colorectal lymphocytes and PBMCs, primarily to reveal distinctive features of the rectal lymphocyte composition in ulcerative colitis patients, by comparing the rectum samples of ulcerative colitis patients to those of control patients. The results showed that CD62L+/CD3+CD4+ and CD62L+/CD3+CD4− cells were increased and further subpopulation analysis revealed that the CD45RA−CD62L+/CD3+CD4+ fraction was increased in the rectum of ulcerative colitis patients compared to those of control patients. Additionally, we found a trend toward an increased ratio of CD45RA−CD62L+/CD3+CD4− in ulcerative colitis patients compared to control patients. Furthermore, CD45RA−CD62L+/CD3+CD4− and CD45RA−CD62L+/CD3+CD4+ fractions tended toward being increased in more inflamed rectal mucosa (MES 1 and 2) than in the rectal mucosa in remission (MES 0). Based on these results, we speculate that central memory T lymphocytes (CD45RA−CD62L+CD3+ cells) are involved in the pathogenesis of ulcerative colitis.
Central memory T cells express the adhesion molecule CD62L, also known as L-selectin or leukocyte adhesion molecule 1, 12 and mainly reside in the T cell areas of the secondary lymphoid organs. In response to antigenic stimulation, these cells produce IL-2, rapidly proliferate, and efficiently differentiate into effector cells after proliferation. Long-term maintenance of the memory T-cell response is the hallmark of immune protection, allowing the activation of a rapid response against re-exposure to the pathogen. Horjus Talabur Horje et al. investigated T lymphocyte subsets in patients with inflammatory bowel disease and defined the CD45RA+CD27+ population as naïve T cells and CD45RA−CD27+ as central memory T cells. 5 Patients with inflammatory bowel disease had a higher frequency of central memory cells than control patients (42% in ulcerative colitis and 37% in Crohn’s disease vs. 25% in the control group). In addition, naïve T cell subsets were larger in ulcerative colitis (28%) and Crohn’s disease (31%) than in controls (19%). Furthermore, patients with endoscopic active inflammatory bowel disease had increased percentages of central memory T cells compared to those in healthy controls and patients with endoscopic inactive inflammatory bowel disease. 13 Moreover, high endothelial venules, which express specific adhesion molecules named addressins that attach to CD62L and anchor lymphocytes, were increased in the inflamed colon of patients with ulcerative colitis compared to those in healthy controls. 6 An increased number of colonic high endothelial venules was associated with enhanced infiltration of naïve T cells and central memory T cells in the inflamed gut, compared to the low density of colonic high endothelial venules in control patients. These results support our speculation of central memory T cells possibly contributing to the development and worsening of ulcerative colitis.
PBMCs of ulcerative colitis showed decreased HLADR+/CD3+, while CD45RA+/CD3+CD4+ cells were increased, compared to PBMCs of control patients. As HLADR is a marker of T lymphocyte activation, the number of cytotoxic T lymphocytes expressing HLADRs is known to increase in autoimmune diseases. 14 CD4+CD45RA+ cells were defined as suppressor-inducers of T lymphocytes. Suppressor-inducer T cells induce cytotoxic T cells to become suppressor cells, that is, Treg cells, which in turn reduce inflammation. Therefore, the results of our study imply that inflammatory reactions in circulating T-cell populations may be suppressed in ulcerative colitis patients. As most of the patients had been taking 5-aminosalicylic acid and/or immunomodulatory drugs, such as azathioprine and mercaptopurine, these drugs might have affected the circulating T-cell compositions.
We also demonstrated that, CD56+/CD3+ in the rectum is lower than that in the colon in ulcerative colitis patients. In addition, MES 1 and 2 patients had a lower ratio of CD56+/CD3+ in the rectum, compared to MES 0 patients. This is relevant because CD3+CD56+ cells are known as NKT cells, which share properties of both T cells and natural killer cells. NKT cells possess multiple functions, including enhancement of microbial and tumor immunity, suppression of autoimmune disease, and promotion of tolerance. For instance, NKT cell deficiencies have been reported in systemic lupus erythematosus mouse models. 15 A decreased number of NKT cells has also been described in peripheral blood from patients with autoimmune diseases, such as rheumatoid arthritis, systemic sclerosis, type 1 diabetes mellitus, and systemic lupus erythematosus. In ulcerative colitis patients, a selective decrease in colonic CD56+ T lymphocytes in the inflamed mucosa has been reported. 16 Taken together, our results reinforce that gut inflammation in ulcerative colitis patients is inversely correlated with the number of NKT cells.
A comparison between control (patients with colon polyp) and patients with ulcerative colitis with low or high disease activity revealed that the CD56+/CD3+ ratio was lower in the rectum of patients with MES 1 and 2 ulcerative colitis than in the rectum of control (p = 0.03); however, there was no difference between patients with MES 0 ulcerative colitis and control. Similarly, the CD45RA−CD62L+/CD3+CD4− ratio was higher in the rectum of patients with MES 1 and 2 ulcerative colitis than in the rectum of controls (p < 0.01); however, no difference was observed between patients with MES 0 ulcerative colitis and controls. Our results complement the findings of Smids et al., who reported that intestinal T lymphocyte subsets of inactive ulcerative colitis were partly comparable to those of healthy controls. 13 In contrast, the CD45RA−CD62L+/CD3+CD4+ ratio was higher in the rectum of patients with MES 1 and 2 (p < 0.01) and MES 0 (p < 0.05) than in the rectum of controls. These memory cells may exist in a functionally quiescent state in inactive disease states and rapidly expand the effector functions on antigen stimulation.
A comparison between the relapse and non-relapse groups revealed that the ratio of central memory CD4+ T cells and central memory CD4− T cells was higher and the ratio of NKT cells was lower in the rectum samples of patients who relapsed, as assessed from flow cytometry. Thus, these fractions may be potential biomarkers for predicting future relapse in patients with UC. However, as the relapse group consisted of MES 1 and 2 patients, underlying inflammation in this group might have affected the results. Future studies incorporating a larger number of patients and their clinical courses may reveal the key to flow cytometry results to predict relapse of this disease.
Our study had several limitations. First, all ulcerative colitis patients had been diagnosed prior to enrollment in this study and had been taking aminosalicylates and/or immunosuppressive drugs. Although ultimate ideal objects are treatment-naïve ulcerative colitis patients, such studies are difficult to conduct, considering the low incidence of this disease. Therefore, medications used in our study groups might have affected lymphocyte composition. The second limitation is the small number of participants. However, we found several significant differences in lymphocyte composition despite the small sample size. Third, diet and other lifestyle factors can affect the gut microbiota, which may in turn alter the immune system of the host. However, except smoking, no other lifestyle factors were included in this study. Fourth, although flow cytometry analyzed T lymphocyte markers using a panel of 13 commercially available antibodies, other markers may be involved in the pathogenesis of this disease. The tissue-resident memory T cells that mediate the local immune protection against infections and cancer have attracted attention. Dendritic cells, which are responsible for the initiation of adaptive immune responses, have also been implicated in pathogenesis of ulcerative colitis based on genetics and their central role in controlling microbial interactions. Therefore, further research is needed to reveal the role of these lymphocytes in ulcerative colitis.
Conclusions
Our study showed that CD62L+ T lymphocytes, particularly the CD45RA−CD62L+ T cell subset, were increased in the rectum of patients with ulcerative colitis. In addition, CD56+/CD3+ was lower in the rectum than in the less-inflamed mucosa. These results suggest that the enrichment of central memory T lymphocytes and depletion of NKT cells in the gut mucosa are involved in the pathogenesis of ulcerative colitis. These findings offer potentially useful information for this patient population. Further investigations with a greater number of patients, including treatment-naïve individuals, will reveal the true nature of colorectum lymphocytes in ulcerative colitis.
Footnotes
Authors’ contributions
MI designed and performed the research, interpreted the data, and wrote the manuscript; T Takahashi and NW contributed to the flow cytometry analysis; KO, T Tanaka, TI, SH, and FO substantively revised the manuscript; and HO supervised the research. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Ethical approval
Written informed consent was obtained from all participants. This study adhered to the principles of the Declaration of Helsinki and was approved by the ethics committee of our institution (approval number: 2003-011 and 2009-014). The study protocol was registered in the UMIN Clinical Trials Registry (UMIN000039868).
Consent for publication
All authors consented to the publication of the manuscript.
