Abstract
The increase of CC chemokine ligand 2 (CCL2) is associated with multiple sclerosis (MS), but the relationship between gene promoter region 2518A/G and the pathogenesis of MS is still not obvious. Collected 54 cases of relapsing-remitting MS patients and 54 healthy controls. By detecting the CCL2-2518A/G polymorphism of MS patients and analyzing the plasma CCL2 level. High levels of A/A genotype and A allele frequency in serum CCL2 and PBMC were found in MS patients. The serum CCL2 of MS patients with A/A genotype is higher than other genotypes. Lipopolysaccharide stimulated PBMC, CCL2 levels in the supernatant of all genotypes were higher, and the A/A genotype levels of MS patients were the highest. Finally, CCL2-2518A/G polymorphism is related to the pathogenesis of MS.
Introduction
Multiple sclerosis (MS) is an autoimmune disease of the nervous system. Demyelination of the central nervous system is the main pathological change. So far, the etiology and pathogenesis of the disease have not been fully elucidated. 1 Recruitment of inflammatory cells to the injury site is a key step in the inflammatory process, and chemokines are the main factors that mediate this process. The chemokine CC motif ligand 2 (CCL2), also known as monocyte chemotactic protein 1 (MCP-1), is a subfamily of chemokine CC, with a relative molecular weight of 13,000 and consisting of 60 amino acids. 2 Structurally expressed CCL2 plays a key role in the transformation of PBMC into tissues and macrophages, and abnormally highly expressed CCL2 can recruit monocytes to inflammatory sites, promote Th cell differentiation, and play a role in inflammatory diseases. Important role. 3 In animals with autoimmune cerebrospinal meningitis and MS patients, the expression of CCL2 increases. 4 The human chemokine CCL2 gene is located on chromosome No. 17, and there is a single nucleotide polymorphism (CCL2-2518A/G polymorphism) in the promoter region 2518 of the gene encoding CCL2. The polymorphism of this locus is believed to be related to the genetic susceptibility of a variety of autoimmune diseases, such as systemic lupus erythematosus, Crohn’s disease, diabetes and asthma. 5 In this study, the CCL2-2518A/G polymorphism in MS patients was detected, and the expression of CCL2 in plasma was analyzed. Observe the expression of CCL2 in different genotypes of PBCLs CCL2-2518A/G PBMCs under the stimulation of lipopolysaccharide to study the correlation between CCL2-2518A/G polymorphism and the pathogenesis of MS and its effect on CCL2 expression.
Methods and materials
Object
Collected patients with relapsing-remitting MS diagnosed in the Neurology Department of the Third Affiliated Hospital of Qiqihar Medical University from January 2017 to December 2019, and met the 2014 McDonald’s diagnostic criteria as subjects in the MS group. Detailed collection of each patient ’s clinical data, serological indicators, the number of brain MRI-enhancing lesions and the Expanded Disability Status Scale 6 (EDSS). EDSS has been verified for use. 7 A total of 54 MS patients were between 23 and 42 years old, with an average age of 35.43 ± 7.17 years, and a course of 5.90 ± 1.97 years. Among them, 12 are males and 42 are females. 54 healthy controls were selected from the physical examination center and trained students of our hospital, who matched the gender and age of the MS group. The entire study complied with the requirements of the “Declaration of Helsinki” and was approved by the hospital ethics committee, and obtained informed consent signed by all subjects in the study.
Plasma CCL2 level test
The CCL2 level in the plasma of the subjects was detected by ELISA. The kit was purchased from RD Systems Inc. Follow the instructions for the kit to perform specific operations. PBMC were extracted by Ficoll (purchased from sigma) density gradient and resuspended with 10% heat-inactivated FBS (made by RPMI 1640) to a cell density of 2 × 106/mL. After resuspending 0.5 mL, PBMC was co-cultured with penicillin at a final concentration of 80 U/mL, streptomycin at a final concentration of 100 μg/mL, and with or without lipopolysaccharide at a final concentration of 5 μg/mL (LPS, Sigma Inc.) In a 48-well culture plate (Corning). After 72 hours, the cell culture supernatant was collected, and the CCL2 level of the supernatant was detected by ELISA.
CCL2-2518A/G polymorphism genotype detection
Peripheral EDTA anticoagulant was collected from all patients and controls. Collect about 750μL of mixed whole blood, and extract genomic DNA according to the specifications of the peripheral blood genomic DNA extraction kit (Qiagen, Germany). The CCL2-2518A/G polymorphism of the sample was tested according to the PCR-restriction fragment length polymorphism (RFIJP) method reported in the literature. 8 The PCR primer sequence is as follows: forward primer 5′-CCGAGATGTTTTCCCAGCACAG-3′, reverse primer 5′-TGCTTTGCTTGTGCCTCTT-3′, 100 ng genomic DNA used for PCR amplification reaction: denatured at 94°C for 2 minutes; at 94°C Cycle down for 30 seconds, cycle at 60°C for 30 seconds, and cycle at 72°C for 45 seconds for 35 cycles. The extension reaction was carried out at 72°C for 10 minutes. After the PCR reaction was completed, a 930 bp product was produced. Since allele A has no restriction endonuclease Pvu II action site, the gene of the sample to be tested can be determined based on the size of the digested product fragment after digestion with Pvu II (Fermentas, USA) overnight at 37°C type. 1% agarose gel electrophoresis (Oxoid Limited, UK).
Statistical analysis
Use SPSS19.0 statistical software (SPSS INC. Chicago, USA). All data are expressed in the form of mean ± standard deviation. One-way analysis of variance (ANOVA) is used for comparison between multiple groups.
Result
Plasma CCL2 levels in MS patients and controls
The expression level of CCL2 in the plasma of MS patients was 382.61 ± 67.40 pg/mL, which was higher than the control group’s 241.11 ± 42.01 pg/mL (
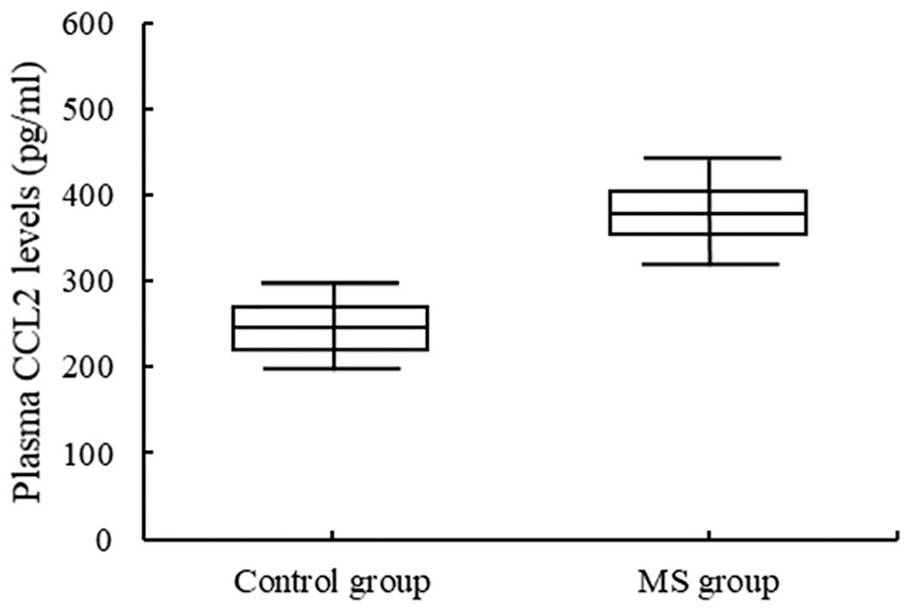
Plasma CCL2 levels in MS patients and controls.
Clinical and laboratory control results for patients with elevated plasma CCL2 levels and normal MS.

Distribution and genotypes of CCL2-2518A/G alleles in MS group and control group
Table 2 shows the genotype and allele frequency of the CCL2-2518A/G polymorphism in MS patients and controls . The distribution of CCL2-2518A/G genotype and alleles in the control group and MS group was in Hardy-Weinberg H equilibrium. The results showed that the genotype and allele frequency of CCL2-2518A/G polymorphism were significantly different between the control group and the MS group, and the A/A genotype and A allele frequency of MS patients were higher than those of the control group.
The distribution of CCL2-2518A/G alleles and genotypes in the control group and MS group.

Effect of CCL2-2518A/G gene polymorphism on plasma CCL2 expression in MS patients and healthy controls
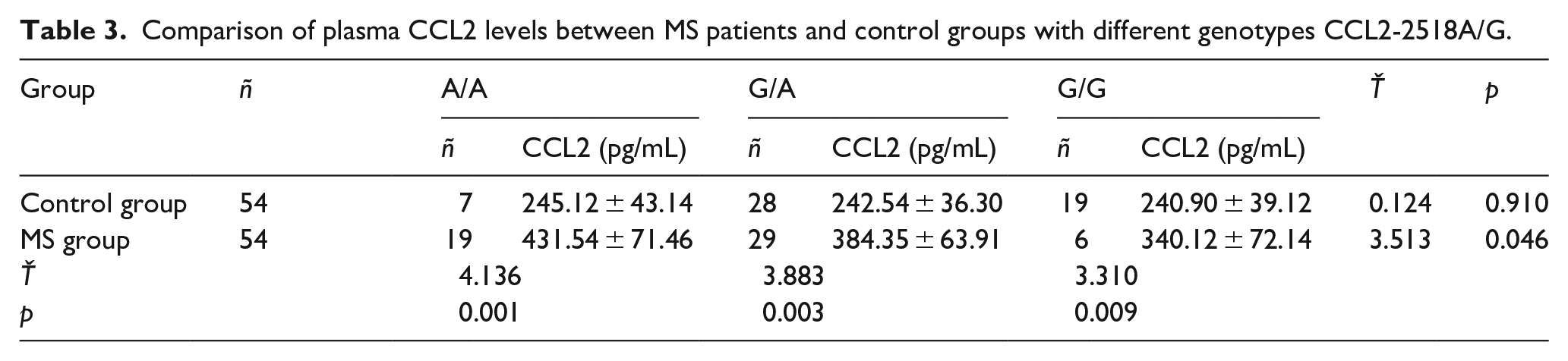
Literature 9 reported that the 2518 polymorphism in the CCL2 promoter region may affect CCL2 gene expression. Therefore, we analyzed the CCL2 levels in the plasma of MS patients and control groups with different CCL2-2518A/G genotypes. In healthy controls with three genotypes, there was no statistically significant difference in serum CCL2 levels. However, among MS patients, plasma ACL/A homozygotes have the highest plasma CCL2 levels, followed by the G/A genotype. Individuals who are homozygous for G/G have the lowest serum CCL2 levels, see Table 3. These results indicate that the A allele of the CCL2-2518A/G locus may affect the expression of CCL2.
Comparison of plasma CCL2 levels between MS patients and control groups with different genotypes CCL2-2518A/G.
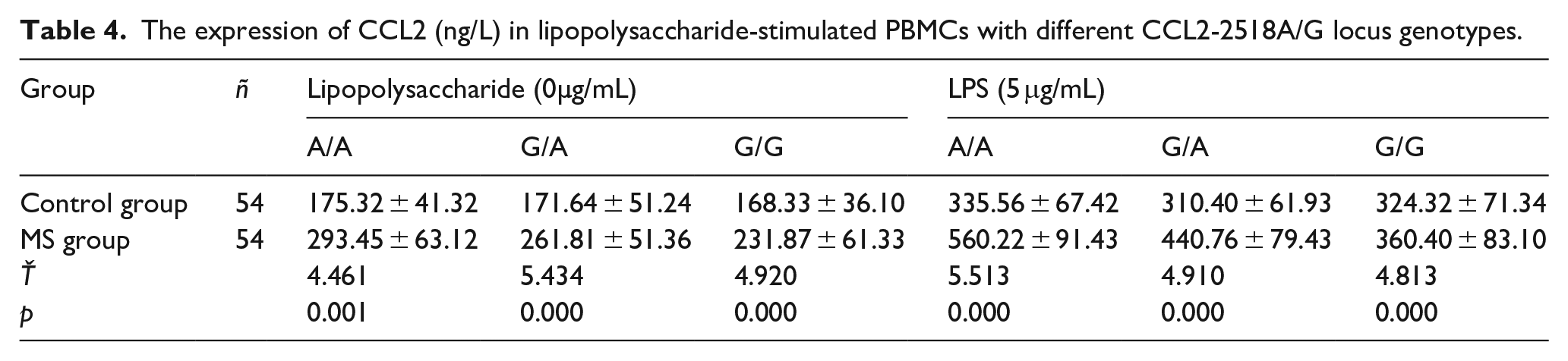
In order to confirm the effect of the A allele of the CCL2-2518A/G locus on CCL2 expression, the CCL2 levels produced by PBMC under inflammatory stimulation were measured in the control group and MS patients, respectively. As the previous study found that the CCL2 level in the cell culture supernatant increased with the increase of the culture time, it was the highest at 72 h. Therefore, the CCL2 level in the supernatant was detected 72 h after the lipopolysaccharide stimulated PBMC. MS patients The levels of CCL2 in PBMCs of different genotypes were higher than those in the control group, while the A/A homozygotes produced the highest CCL2 after stimulation of PBMCs in MS patients. Please refer to Table 4.
The expression of CCL2 (ng/L) in lipopolysaccharide-stimulated PBMCs with different CCL2-2518A/G locus genotypes.
Forum
Chemokines are small molecules capable of directional migration of chemotactic cells. According to the position and number of cysteine residues at the N-terminal of chemokines, chemokines are now divided into four subgroups: C, CC, CXC and CX3C. CCL2, also known as MCP-1, belongs to the subfamily of CC chemokines. As indicated by the CCL2 nomenclature, this molecule mainly targets monocytes (such as T cells, monocytes and macrophages). 10 CCL2 exerts its extensive physiological or pathological effects by binding to its specific receptor CCR2, which is a G protein-coupled receptor on the cell surface. The association between CCL2-2518A/G polymorphism and MS susceptibility has not been reported. We examined the CCL2-2518A/G polymorphism in MS patients and controls to investigate whether the CCL2-2518A/G polymorphism directly affects the pathogenesis of MS. The results showed that the allele frequency and genotype frequency of CCL2-2518A/G polymorphism in MS patients and the control group were significantly different, indicating that CCL2-2518A/G polymorphism may be related to the pathogenesis of MS.
We found that CCL2 gene polymorphism may be related to the pathogenesis of MS, and studies have shown that CCL2 gene polymorphism may affect the expression of CCL2. 11 In order to explore whether the CCL2-2518 polymorphism has an effect on the expression of circulating CCL2 protein in MS patients, we compared the plasma CCL2 expression levels of MS patients with different genotypes, and found that plasma CCL2 levels were higher in MS patients carrying two A alleles The highest (A/A homozygous), followed by MS patients carrying an A allele (G/A homozygote), CCL2 plasma levels were the lowest in patients without A allele (G/G homozygous). However, no similar findings were observed in the control group. According to reports, in patients with pulmonary tuberculosis and coronary atherosclerosis, the expression level of CCL2 protein in individuals carrying the A allele is significantly higher than that of individuals without the A allele. 12 These results indicate that the G allele of the CCL2-2518A/G locus may up-regulate CCL2 protein expression.
To further verify the up-regulation of CCL2 expression by the A allele of the CCL2-2518 locus, we examined the CCL2 levels produced by PBMCs from MS patients (with different CCL2-2518A/G genotypes) after inflammatory stimulation. When choosing PBMC as the research cell, it is considered that CCL2 is mainly secreted by monocytes. The results showed that after lipopolysaccharide stimulation, the A/A homozygous gene PBMCs had the highest CCL2 level in the culture supernatant, followed by the A/G and G/G genotypes.
MS is usually divided into relapsing remitting type, secondary progressive type, primary progressive type and progressive recurrence type. However, the relapse-remitting type (approximately 85%) is the most common type in clinical practice, followed by the primary progressive type (approximately 10%). The other two types are relatively rare in clinical practice. Due to the limited number of cases in this study, MS patients with relapse and remission are the only research subjects, and whether CCL2 plays a vital role in the other three types of MS still needs further research.
This study also has some limitations. This is a single-center case-control study. Although we have separated all eligible cases, there are only 54 patients. We will further expand the sample size and conduct in-depth research.
Summary
The peripheral blood CCL2 protein level of MS patients in the high CCL2 expression group was significantly increased, and the repetition interval was shortened, and the CCL2-2518A/G gene polymorphism was related to the pathogenesis of MS . The A allele may affect the pathological process of MS by up-regulating the expression of CCL2, thereby enhancing the expression of CCL2 in PBMCs in a mortality environment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethics approval
The clinical approval for this study has been obtained from the Clinical Committee of the Third Affiliated Hospital of Qiqihar Medical University (201611003).
Funding
The author(s) disclosed the receipt of the following financial support for the research, authorship, and/or publication of this article: The research is supported by the Qiqihar Academy of Medical Sciences (No. QMSI2019M-24).
Informed consent
Obtain written informed consent from all orientations before the study.
