Abstract
Objective
SARS-CoV-2 infection has been shown to result in increased circulating levels of adenosine triphosphate and adenosine diphosphate and decreased levels of adenosine, which has important anti-inflammatory activity. The goal of this pilot project was to assess the levels of soluble CD73 and soluble Adenosine Deaminase (ADA) in hospitalized patients with COVID-19 and determine if levels of these molecules are associated with disease severity.
Methods
Plasma from 28 PCR-confirmed hospitalized COVID-19 patients who had varied disease severity based on WHO classification (6 mild/moderate, 10 severe, 12 critical) had concentrations of both soluble CD73 and ADA determined by ELISA. These concentrations were compared to healthy control plasma that is commercially available and was biobanked prior to the start of the pandemic. Additionally, outcomes such as WHO ordinal scale for disease severity, ICU admission, needed for invasive ventilation, hospital length of stay, and development of thrombosis during admission were used as markers of disease severity.
Results
Our results show that both CD73 and ADA are decreased during SARS-CoV-2 infection. The level of circulating CD73 is directly correlated to the severity of the disease defined by the need for ICU admission, invasive ventilation, and hospital length of stay. Low level of CD73 is also associated with clinical thrombosis, a severe complication of SARS-CoV-2 infection.
Conclusion
Our study indicates that adenosine metabolism is down-regulated in patients with COVID-19 and associated with severe infection. Further large-scale studies are warranted to investigate the role of the adenosinergic anti-inflammatory CD73/ADA axis in protection against COVID-19.
Highlights
• Soluble CD73 is decreased during COVID-19 infection • Soluble Adenosine deaminase is decreased during COVID-19 and correlates with decreased soluble CD73 • Decreased soluble CD73 during COVID infection correlates with severity of disease • Decreased soluble CD73 and Adenosine deaminase are associated with thrombus formation during COVID-19 infection • CD73 is a potential molecular target that may lead to advances in therapeutics that decrease severity of disease
Introduction
SARS-CoV-2 infection has a wide spectrum of presentations from asymptomatic carriage to multi-organ failure and the development of acute respiratory distress syndrome (ARDS). 1 The pathophysiology of this varied presentation is poorly understood, but it is thought that severe cases of SARS-CoV-2 infection are due to a dysregulated pro-inflammatory state. 1 Among the severe complications of SARS-CoV-2 are deep vein thrombosis and pulmonary emboli. 2
CD39 and CD73 catalyze the conversion of adenosine triphosphate (ATP) to adenosine, with CD39 converting ATP and adenosine diphosphate (ADP) into adenosine monophosphate (AMP), and CD73 converting AMP to adenosine. 3 CD73 is the rate-limiting enzyme that converts AMP to adenosine; it exists in both cell membranes and as a soluble form. 3 Similar to CD73, CD39 has both a membrane-bound and soluble form, and recent studies have shown that increased CD39 during SARS-CoV-2 infection is positively associated with disease severity. 4 The soluble form of CD73 has been shown to be a major producer of adenosine under normal physiologic conditions. 5 Adenosine has many functions but most important in the context of SARS-CoV-2 infection is its role as an anti-inflammatory molecule acting directly on target cells and indirectly through the induction of anti-inflammatory T-regulatory cells. 6
SARS-CoV-2 infection induces a relatively adenosine-low state with excess ATP, which is both pro-inflammatory and pro-thrombotic.4,7 Adenosine may play an important role in the inhibition of the thrombo-inflammatory cascade, decreasing thrombus formation during pro-inflammatory states.4,8 Adenosine signaling is regulated through multiple mechanisms including adenosine deaminase which converts adenosine to inosine.5,9 Inosine has both pro and anti-inflammatory effects with the latter mediated through adenosine receptors.10-12 Studies examining inosine during SARS-CoV-2 infection have shown increased plasma levels of inosine which correlate with severity. Interestingly inosine pranobex has been used as a treatment for SARS-CoV-2 infection in clinical trials. 13
The objective of this study was to examine circulating levels of CD73, the major enzyme responsible for the production of adenosine, and adenosine deaminase in the context of SAR-CoV-2 infection and the severity of the disease.
Materials and methods
Patients
This observational study taking place at Maine Medical Center Portland Maine and informed consent process were approved by the Maine Medical Center Institutional Review Board May 8th 2020 approval number 1598969. Informed written consent was obtained from the patient or the legally authorized representative (LAR) using secure electronic consent (to prevent disease transmission), either in-person (with the patient) or by telephone (with the LAR). Consent to participate in the study included consent to publish the data collected during enrollment in the study. Consent from the LAR was utilized in cases where the patient was unable to consent for themselves (i.e., patient was intubated). Patients were enrolled between July and December 2020 within 72 hours of being hospitalized with PCR-confirmed SARS-CoV-2 infection (real-time RT-PCR test, NorDx Laboratories, Portland, ME, USA). Patients were excluded if they were less than 18 years of age, those who represented vulnerable populations, or had hemoglobin <8 g/dL, or from whom informed consent was not obtained. Our cohort included a few asymptomatic patients admitted for other reasons who were determined to have incidental SARS-CoV-2 infection on administrative testing, although most had severe or critical COVID-19 disease.
COVID-19 disease severity was defined using the ordinal scale for SARS-CoV-2 disease severity developed by the World Health Organization (WHO). 14 Other factors used to represent disease severity were admission to the Intensive Care Unit (ICU), need for invasive ventilation, and hospital length of stay.
Diagnosis of deep vein thrombosis (DVT) and/or pulmonary embolus (PE) was done via review of hospital charts post-discharge for evidence of clinical thrombosis. Screening for DVT and PE were based on clinical need and was not routinely performed in all patients.
Single patient plasma samples collected in EDTA tubes from 11 males and 9 females ages 34–68 were purchased from Innovative Research (Novi, MI, USA) and used as control; samples were collected prior to 12/2019 predating the first diagnosed case of SARS-CoV-2 in the United States. This step was taken due to the prevalence of SARS-CoV-2 after that date, the presence of patients with asymptomatic carriage, and the unknown long-term effects of SARS-CoV-2 on patients that may have been infected prior to study commencement.
Standards of care
Patients generally received remdesivir and dexamethasone; many received systemic anticoagulation with unfractionated heparin or enoxaparin, and antibiotics for concurrent bacterial pneumonia. IL-1 or IL-6 inhibitors were not administered. Mechanically ventilated patients received lung-protective ventilation, proning for refractory hypoxemia, and neuromuscular blocking agents. 15 Patients may have received continuous or intermittent renal replacement therapy or underwent arteriovenous or venovenous extracorporeal membrane oxygenation, as clinically indicated.
Blood sample Collection
Subjects underwent phlebotomy on the day of enrollment and, when feasible, at day 4 and day 7 post-enrollment. Blood (8.5 mL per sample) was collected from SARS-CoV-2 positive participants using BD Vacutainer ACD tubes. Platelet-free plasma (PFP) was isolated using 2-step centrifugation, each at 2000 x g for 20 minutes. Plasma was stored at −80°C. No more than one freeze/thaw cycle was allowed for PFP samples to avoid degradation.
Plasma analysis
The concentration of soluble CD73 and IL-6 was measured using a CD73 duoset ELISA kit (Bio-Techne/R&D Systems) spike/recovery validation was performed in EDTA samples per manufacturer’s recommendations with all samples being diluted 1:10 or 1:2 in PBS prior to analysis. The concentration of circulating adenosine deaminase was determined using ADA ELISA kit (ThermoFisher Scientific) with plasma samples being diluted 1:2 per manufacturer’s recommendations prior to analysis. All samples were assayed in duplicate and plasma concentration was calculated based on standard curve per manufactures recommendations, calculations performed in Microsoft Excel.
Statistical analysis
Comparisons between the healthy controls and SARS-CoV-2 positive subjects were done using Mann–Whitney. Comparisons between multiple groups were done using one-way ANOVA Kruskal–Wallis with Dunn post hoc analysis. Correlation analysis was performed using a Spearman (skewed distribution) correlation.
Results
Subjects
Patient demographics for COVID-19 positive patients. WHO COVID severity classification mild/moderate: symptomatic but not requiring supplemental oxygen, severe: patients requiring supplemental oxygen, critical: patients with respiratory failure, septic shock, or multi-organ failure. (SD: standard deviation).

Soluble CD73 and adenosine deaminase are decreased during SARS-CoV-2 infection
Soluble CD73 in the SARS-CoV-2 population was significantly decreased (Figure 1(a)) compared to healthy controls. This difference was more profound when comparing healthy controls to patients either requiring ICU level of care or those requiring invasive ventilation (Figure 1(b) and 1(c)). Similarly, soluble adenosine deaminase was decreased during SARS-CoV-2 infection, and lower levels correlated with the need for ICU level of care and invasive ventilation (Figures 1(d)-(f)). The decrease in soluble CD73 associated with lower adenosine deaminase levels in patients with SARS-CoV-2 [Figure 1 G]. Interestingly, higher levels of both CD73 and ADA were associated with increased levels of IL-6 on hospital day 1 but did not show any association with C-reactive protein (CRP) or lymphocyte count all of which are altered during COVID-19 infection [Figure 2 A-F]. Plasma CD73 and ADA are decreased during COVID-19 infection. (A–C) Levels of CD73 in the peripheral circulation of healthy controls and (A) patients with COVID-19 on different hospital days, (B) patients requiring ICU, and (C) patients requiring invasive ventilation. (D–F) Levels of soluble ADA in healthy controls and (D) patients with COVID-19, (E) patients requiring ICU, and (F) patients requiring invasive ventilation. (A–F) Kruskal-Wallis with Dunn post hoc analysis (HC n = 24, HD1 n = 28, HD4 n = 19, HD7 n = 14); * Soluble CD73 and Adenosine Deaminase associated with markers of inflammation during COVID-19 infection. (A–F) Spearman correlation between soluble CD73 and Adenosine deaminase compared to IL-6 hospital day 1 (a–b), lymphocyte count hospital day 1 (c–d), and C-reactive protein levels (e–f).

Alterations in soluble CD73 and adenosine deaminase are associated with disease severity during SARS-CoV-2 infection
Lower levels of soluble CD73 in COVID-19 patients were significantly associated with both the ICU level of care and invasive ventilation during admission (Figure 3 A and B). We also found that soluble CD73 concentration on admission was inversely correlated with peak Ordinal Scale severity score during admission (Figure 3(c)) and decreased soluble CD73 correlated with increased hospital length of stay (Figure 3 D and E). Decreased plasma CD73 is associated with need for ICU level of care, invasive ventilation and increased length of stay in patients hospitalized with COVID-19. (a) Blood plasma levels of CD73 in patients with COVID-19 requiring ICU level of care (HD1 n = 12; HD4 n = 10; HD7 n = 8) and non-ICU subjects (HD1 n = 16; HD4 n = 9; HD7 n = 6). (b) Circulating CD73 in non-ICU patients compared to patients requiring invasive ventilation (HD1 n = 8; HD4 n = 7; HD7 n = 6). (c, d) Associations between levels of CD73 and (c) hospital day 1 with peak ordinal scale value during admission, and (d) total hospital length of stay (LOS). Spearman coefficients and 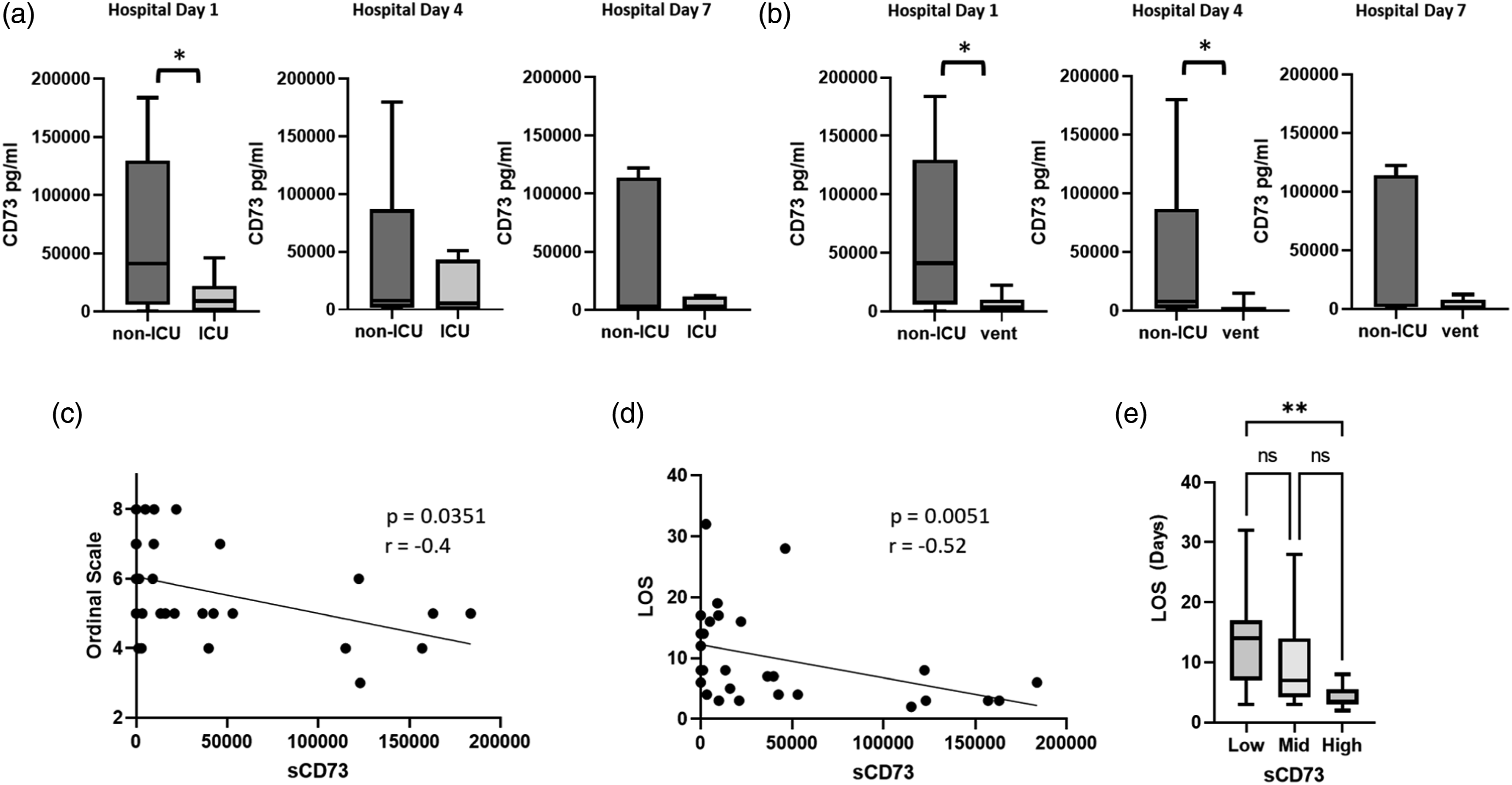
Adenosine deaminase concentrations were associated with the need for invasive ventilation (Figures 4(a) and (b)). They did not correlate with the Ordinal Scale severity score but were associated with hospital length of stay (Figure 4 C and D). Similar to soluble CD73, lower levels of ADA were assoicated with longer hospital length of stay (Figure 4(e)). Decreased plasma ADA is associated with need for invasive ventilation and increased length of stay in patients hospitalized with COVID-19. (a) Levels of circulating ADA in patients with COVID-19 non-ICU (HD1 n = 16; HD4 n = 9; HD7 n = 6) and those requiring ICU level of care (HD1 n = 12; HD4 n = 10; HD7 n = 8). (b) Levels of circulating ADA in patients with COVID-19 non-ICU (HD1 n = 16; HD4 n = 9; HD7 n = 6) and those requiring invasive ventilation (HD1 n = 8; HD4 n = 7; HD7 n = 6). (c) Spearman correlation between ADA levels during hospital day 1 and peak ordinal scale value during admission. (d) Spearman correlation between ADA level hospital day 1 and total hospital length of stay (LOS). (e) Median hospital length of stay in patients with ADA levels low: below the median <50%, medium 50–75%, high >75% on hospital day 1. Panels (a, b) Mann Whitney analysis, *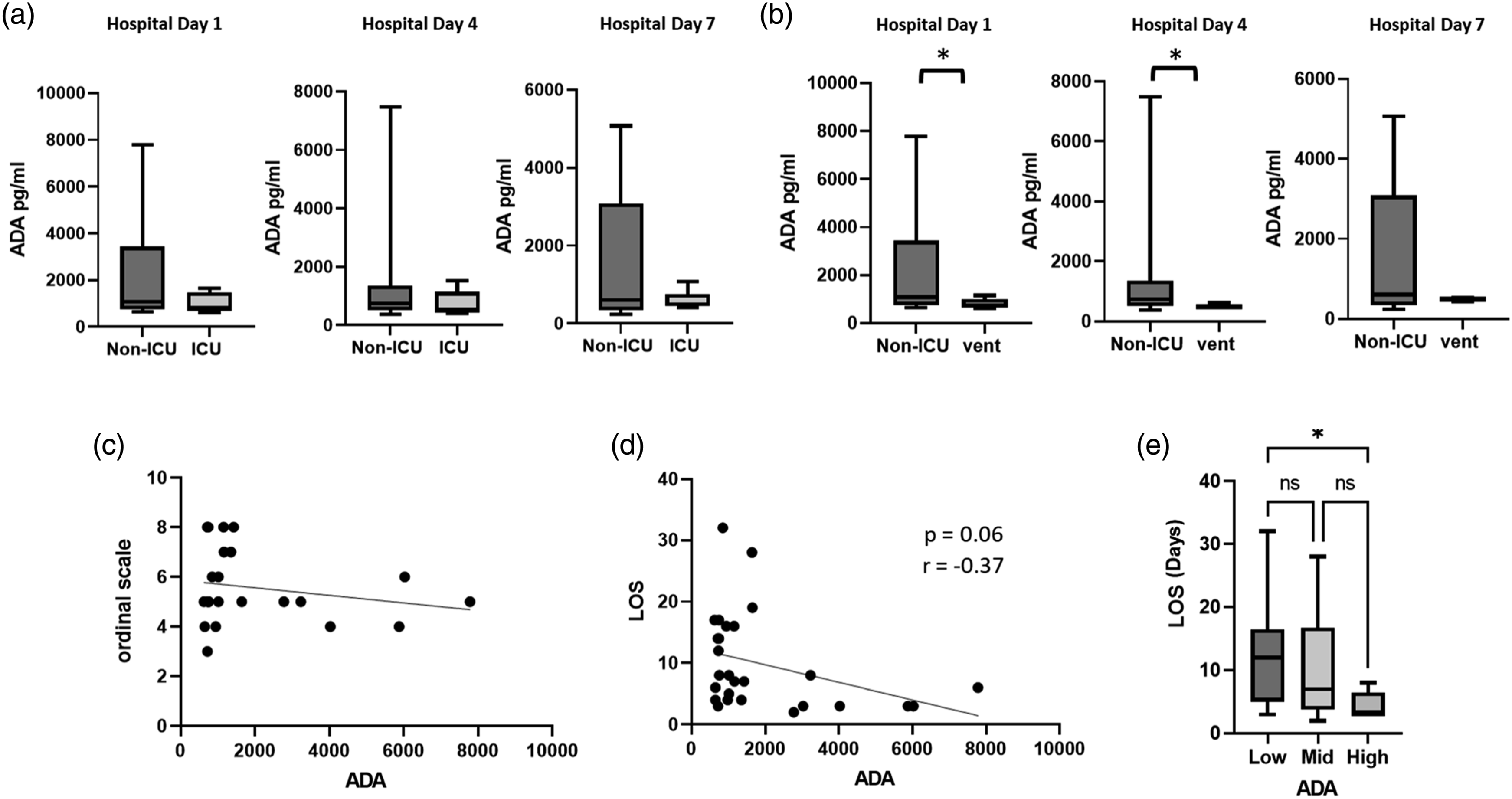
Changes in soluble CD73 and adenosine deaminase during thrombotic complications and mortality during SARS-CoV-2 infection
We examined concentrations of both soluble CD73 and adenosine deaminase in SARS-CoV-2 patients that developed image-proven clinical thrombosis and those whose death was associated with SARS-CoV-2. Both soluble CD73 and adenosine deaminase were significantly decreased in patients who developed a clinical thrombosis event during hospitalization (Figure 5 A and C). Soluble CD73 was decreased in patients who died due to COVID-19 during their hospitalization, with ADA concentrations being similar between survivors and non-survivors (Figure 5 B and D). Plasma CD73 and ADA are significantly decreased in hospitalized patients with COVID-19 who develop deep vein thrombosis or pulmonary embolism. (a) CD73 in patients with COVID-19 without (n = 22) or with (n = 6) imaging confirmed deep vein thrombosis or pulmonary embolism. (b) CD73 in patients with COVID-19 who survived to hospital discharge (n = 24) and those who died (n = 4). (c) Levels of ADA in patients with COVID-19 without (n = 22) or with (n = 6) imaging confirmed deep vein thrombosis (DVT) or pulmonary embolism (PE). (d) ADA level in patients with COVID-19 who survived to hospital discharge (n = 24) and those who died (n = 4). Mann Whitney test, *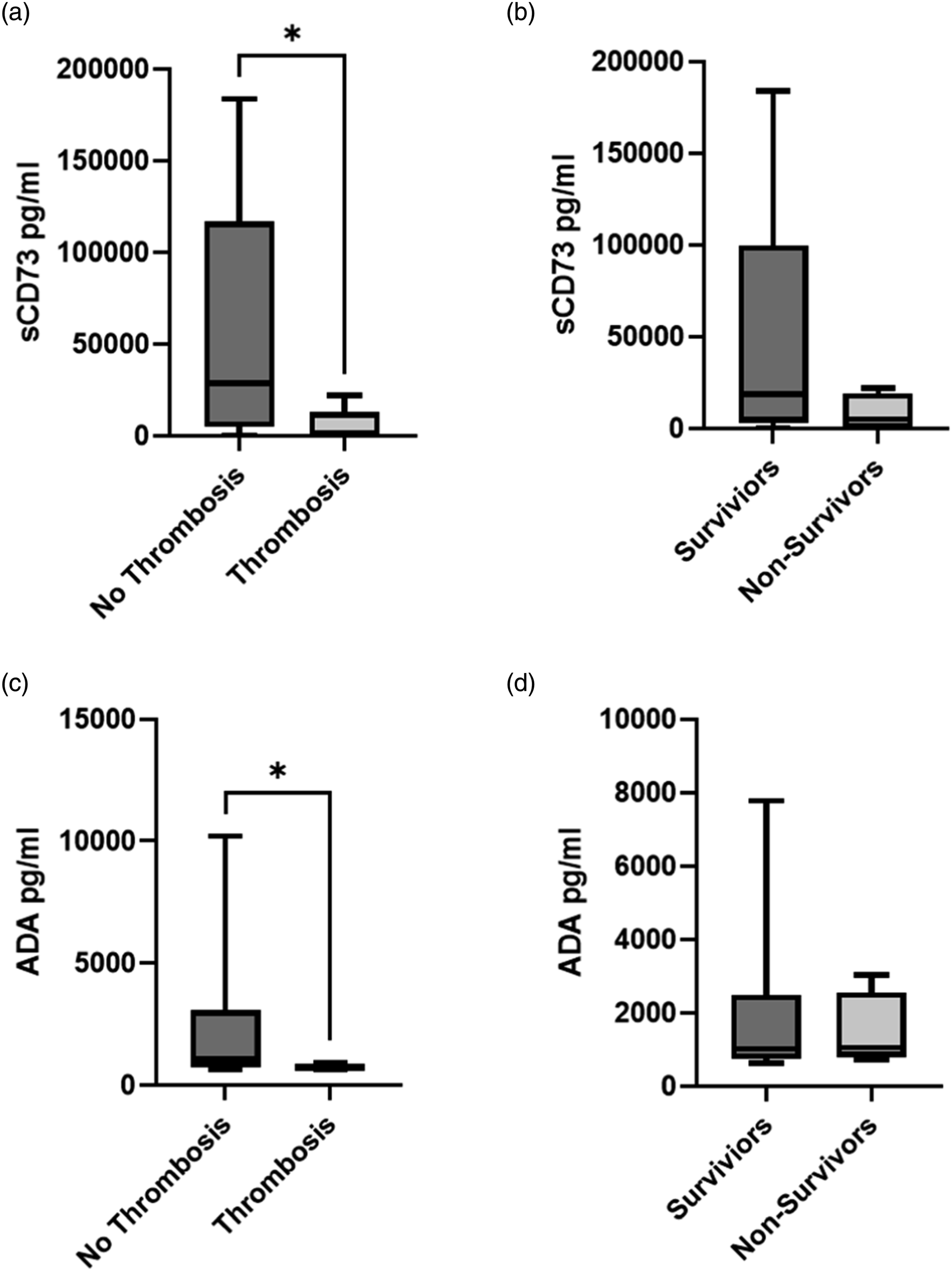
Discussion
We demonstrated the association of low concentrations of soluble CD73 and adenosine deaminase with multiple markers of COVID-19 severity, including need for ICU admission, hospital length of stay, need for mechanical ventilation, and thrombosis. These findings suggest that CD73 and ADA may have a role in modulating disease severity, and could be used to either predict disease severity, or as treatment targets themselves.
Numerous studies examining CD73 expression on T-cell and B-cell populations have showed that decreased membrane bound CD73 is associated with cytotoxic T-cell phenotype, disease severity and suggests the presence of a pro-inflammatory state.16-18 Our study examining cell-free soluble CD73 provides additional support to the importance of CD73 in COVID-19 and suggests a role for Adenosine Deaminase in potential control of inflammation during COVID-19 infection.
Previous publications have shown that children have higher level of adenosine and lower levels of adenosine deaminase when compared to adults. 5 Recent publications have shown that SARS-CoV-2 is associated with high levels of adenosine triphosphate, adenosine diphosphate and low levels of adenosine.4,7 These same studies showed that membrane bound CD73 is increased on PBMCs during SARS-CoV-2 infection and that it is associated with disease severity. 7 Our data show that SARS-CoV-2 infection results in a decrease in soluble CD73 which could explain the overall decrease of adenosine described in SARS-CoV-2 infection. Our examination of soluble CD73 and soluble ADA also showed some temporal differences with CD73 decreasing rapidly on hospital day 1 and ADA decreasing steadily after. The decrease in adenosine deaminase was highly correlated with decreased soluble CD73, one could reasonably believe that this delayed response is an attempt to preserve adenosine signaling in a high pro-inflammatory state. Though our study did not examine the pediatric population, it does raise the question of whether children are less susceptible to severe SARS-CoV-2 due to their higher levels of adenosine and lower relative amounts of adenosine deaminase. Interestingly inborn error in interferon-beta signaling has been proposed as a risk factor associated with severe disease from SARS-CoV-2 in the pediatric population. 19
SARS-CoV-2 has been shown to utilize multiple mechanisms to evade interferon signaling, and the development of anti-interferon beta auto antibodies during SARS-CoV-2 infection is positively associated with disease severity.20,21 One therapeutic that continues to be studied in the treatment of SARS-CoV-2 is interferon beta, though a full review of the action of interferon beta is outside the scope of this article, one published mechanism of interferon beta signaling is the upregulation of CD73.22,23 Similar publications were done examining the role of Interferon beta in the treatment of acute respiratory distress syndrome (ADRS) which is the most severe result of SARS-CoV-2 infection.24-26 Our data suggest that the rapid decrease and sustained depression of soluble CD73 is associated with disease severity and is more profound in patients that need ICU level of care or invasive mechanical ventilation. Recent randomized controlled trials suggested benefit to interferon-beta-1b administration early in the disease course. 27 Although these trials did not examine soluble CD73 or adenosine deaminase, one possible mechanism could be through the restoration of adenosine signaling via upregulation of CD73.
Inosine signaling is poorly understood with conflicting publications showing both pro and anti-inflammatory effects in various in vitro and in vivo models.10,11 Our data show a decrease in soluble adenosine deaminase, the major enzyme responsible for the generation of inosine from adenosine, during SARS-CoV-2 infection. Previous studies have shown that inosine is increased during SARS-CoV-2 infection and that it is positively associated with disease severity, and inosine pranobex is currently being examined as a potential therapeutic agent in SARS-CoV-2 infection.12,13 Previous findings that inosine concentration is increased in SARS-CoV-2 could be seen as contradictory to our finding, though our study does not examine the contribution of membrane bound adenosine deaminase which was shown to be increased and related to disease severity. 7
Taken together, our data show that SARS-CoV-2 infection is associated with a rapid decrease in soluble CD73 and adenosine deaminase, and that the decrease in soluble CD73 is positively associated with severity of disease. These results suggest that soluble CD73 and adenosine deaminase potentially could be used as markers for risk assessment and prediction of severe disease. Also, as discussed earlier, it is possible that a therapeutic agent targeting the restoration of adenosine signaling in SARS-CoV-2 could be beneficial in the early treatment of SARS-CoV-2 and result in a decrease in progression to severe disease particularly in more high-risk populations.
The results of this study are limited by the small cohort size of 28 patients, as a pilot study appropriate power calculation was not done at the start of this study with the results likely being underpowered the presence of clinical confounders such as pre-existing conditions and possible differences between viral variants. Similarly, though the results of the study might provide information about the pathophysiology of SARS-CoV-2 infection, it does not provide direct evidence of causation of the clinical presentations seen in COVID-19.
Conclusion
The goal of our study was to examine soluble CD73 and adenosine deaminase during COVID-19 infection and determine if changes in these factors are associated with disease severity and the complex pathophysiology of COVID-19. Though the results of this study have limitations we showed an association between decreased levels of soluble CD73 and Adenosine Deaminase in relation to severity of disease during COVID-19 infection. We also showed decreased levels of these soluble factors may be involved in thromboinflammation and thrombus formation during COVID-19 infection. Future large-scale appropriately powered studies are warranted as both soluble CD73 and Adenosine Deaminase represent potential therapeutic targets.
Footnotes
Acknowledgments
We are grateful to David Groft and the NorDx lab, Dr. Anne Breggia, Sue LaPierre, and the MMC BioBank for subject recruitment, and to Ivette F. Emery and Meghan P. Searight for their contributions and logistical support. The authors would like to thank Natalia Kimball for her critical review of the manuscript. We would like to thank our research subjects and their families for their contribution to this project and to offer condolences to the families of those that did not survive.
Author Contributions
Study design: Dr. David Seder and Dr.Sergey Ryzhov. Patient Screening and Consent: Dr. David Seder, Dr. Teresa May, Dr. Richard Riker, Ashley Eldridge, and Christine Lord. Critical review of data and manuscript: Dr. David Seder, Dr. Teresa May, Dr. Richard Riker, Dr. Douglas Sawyer, Dr. David Gagnon, Ashley Eldridge, Christine Lord, Joanne DeKay.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Maine Medical Center Mentored Research Grant Program, and by the Maine Medical Center Cardiovascular Research Institute 2015 Pilot Project Program, NIH/NIGMS under Grants P30GM106391, COBRE in Stem and Progenitor Cell Biology, and Regenerative Medicine and U54GM115516, Northern New England Clinical and Translational Research Network (Translational Technologies Core and Clinical Research Design, Epidemiology & Biostatistics Core), P20GM139745, COBRE in Acute Care Research and Rural Disparities, NIH/NHLBI OT2HL161847 Researching COVID to Enhance Recovery (RECOVER) Initiative. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Ethical approval
Ethical approval for this study was obtained from Maine Medical Center Internal Review Board May 8th 2020 approval number 1598969.
Informed consent
Informed written consent was obtained from the patient or the legally authorized representative (LAR) using secure electronic consent (to prevent disease transmission), either in-person (with the patient), or by telephone (with the LAR). Consent to participate in the study included consent to publish the data collected during enrollment in the study. Consent from the LAR was utilized in cases where the patient was unable to consent for themselves (i.e., patient was intubated).
Trial registration
Not applicable.
