Abstract
Objective
Understand the progress of inflammation over time caused by multi-walled carbon nanotubes (MWCNT).
Methods
Two types of MWCNTs were administered to C57BL/6N mice via intraperitoneal administration at low and high doses (0.05 and 1.0 mg/mouse, respectively). Inflammation was evaluated until 6 months after administration based on cytokine levels and pathological observations. The abdominal cavity lavage fluid was collected and analyzed 1 week, 1, 3, and 6 month(s) after administration. IL-6 expression markedly increased 3 months after the administration of high-dose MWCNT-7.
Results
Notable inflammation was observed in the groups administered with one of the MWCNT, MWCNT-7. On the other hand, inflammation in another MWCNT-treated group was milder than that in the MWCNT-7-treated group. MWCNT-7 induced pronounced inflammation but did not induce tumor formation during the experimental period. Inflammation reaction is one of the most important biological responses to MWCNT.
Conclusion
Three months post-exposure becomes a turning point for the harmful effects of the intraperitoneally administered MWCNT-7.
Introduction
Carbon nanotubes (CNTs) are one of the materials with industrially beneficial properties, and are expected to be used for the realization of a sustainable society. CNTs are composed from carbon and are classified as nano-objects. CNTs have characteristic electric conducting property and are applied in fuel cells for improving their performance. CNTs have a fiber-like structure with diameter in the nanoscale range from 1 to 100 nm. Generally, CNTs are classified into the following two types based on their structure: single-walled carbon nanotubes (SWCNT) and multi-walled carbon nanotubes (MWCNT). The SWCNT structure can be thought of as a graphene sheet rolled into a cylinder and joined edge to edge. Meanwhile, MWCNT can be thought of as a long sheet of graphene rolled into a spiral, giving it a morphology relatively similar to that of nested SWNCTs of different diameter. Although CNTs have industrial advantages, they exhibit hazardous properties. The toxic activity of CNTs depends on the individual material structure. In some cases, the biological effects including the toxicity of CNTs are more pronounced in MWCNTs than in SWCNTs.1,2
Among the CNTs which were investigated previously, one of the MWCNT, “MWNT-7,” showed remarkable toxicity. Lung carcinoma formation was observed in rats following the inhalation of MWCNT-7. 3 Epithelial hyperplasia, granulomatous changes, and localized fibrosis were also observed following the inhalation of MWCNT-7. Based on these results, MWCNT-7 was classified as Group 2B agents (possibly carcinogenic to humans) on the list of International Agency for Research on Cancer (IARC). On the other hand, carcinogenicity has not been reported for other MWCNTs. It remains unclear why only MWNT-7 is found to be carcinogenic. The difference in bioresponsiveness between MWNT-7 and other MWCNTs and its cause are still unclear. One of the representative biological responses triggered by MWNT-7 is proinflammatory.4–6 Although MWCNT-7 is carcinogenic, its proinflammatory effect is also an important toxic property. Since chronic inflammation promotes carcinogenesis,7,8 proinflammatory effect is important property on carcinogenesis. It is considered that carcinogenicity of MWNT-7 is related to its proinflammatory property.4,9 Therefore, understanding the proinflammatory behavior of MWCNT is important for understanding the hazard assessment of MWCNT. Induction of inflammation was observed upon inhalation of MWCNT-7. The count of macrophages and neutrophils in the bronchoalveolar lavage fluid was increased significantly by inhalation of 2 mg/m3 of MWCNT-7 for 104 weeks. 3 Our previous investigation reported that MWCNT-7 induced notable inflammation in mouse lungs by pharyngeal aspiration. 5 Additionally, MWNT-7 markedly induced the production of chemokines such as interleukin-8 (IL-8) in macrophage. 5 These proinflammatory effects by MWNT-7 were stronger than another different MWNCT examined at the same time. The biological responses induced by MWCNT will depend on the individual type of MWCNT. Although MWCNT-7 is more toxic than other MWCNTs, the reason for this high toxicity remains unclear. Furthermore, the factors affecting the biological activities of MWCNT-7 and other MWCNTs remain to be elucidated.
Previously, the pulmonary effects caused by MWCNT-7 and other MWCNTs with very low iron content were compared by pharyngeal aspiration in the mouse lung. 5 Both cellular effects and pulmonary toxicity caused by MWCNT-7 were stronger than those caused by other low-iron MWCNTs. These previous studies suggest that phagocytosis of MWCNTs by phagocytes, such as macrophages and neutrophils, and induction of inflammation are important factors in MWCNT-induced pulmonary toxicity. The biological responses of MWCNTs differ depending on their type. The iron content may be one of the critical toxicity determining factors of MWCNTs. Among the various types of asbestos, crocidolite, which has high iron content, demonstrates a highly harmful effect. Crocidolite is classified as an IARC Group 1 agent carcinogenic to humans. It has been suggested that a high iron content and induction of oxidative stress are associated with the harmful effects of crocidolite. 10 Even though the principal constituent element of the MWCNT is obviously carbon, in many cases, MWCNTs include metals, such as iron, nickel, and yttrium, derived from synthetic catalysts. In fact, the content of iron as an impurity in MWCNT-7 is comparatively high. Compared with low-iron-content MWCNTs, high-iron-content MWCNTs induced IL-8 expression via an increase in cellular Ca2+ levels. 11 Thus, residual iron present in the MWCNTs may be a contributing factor in the toxic response to them.
According to previous studies, among the MWCNTs, MWCNT-7 causes severe inflammation. However, these were only observed at the endpoint or at the previous stage. 5 Substantivity and time-dependent changes in inflammation caused by MWCNT-7 remain unclear. Additionally, the long-term effect of low-iron-content MWCNT remains to be elucidated. Understanding the time-dependent changes in inflammation caused by MWCNT-7 is important for the prevention of its harmful effects. In this study, time-dependent changes in inflammation caused by intraperitoneal administration of MWCNT-7 and low-iron-content MWCNT were evaluated.
Materials and methods
Preparation of MWCNT suspensions
MWCNT-7 was obtained from Hodogaya Chemical Co., Ltd. (Tokyo, Japan). MWCNT-B was obtained from Showa Denko K.K. (Tokyo, Japan). According to manufacturer’s data, MWNT-7 has a diameter of 60 nm, a length of 10 μm, and a specific surface area of 25–30 m2/g; and MWCNT-B has a diameter of 176 nm, a length of 5.2 μm, and a specific surface area of 14 m2/g. The iron content of MWCNT-7 and MWCNT-B are 4206 ppm and 9 ppm, respectively. 5 The MWCNTs were suspended in 0.5% Pluronic F-68 (Sigma-Aldrich Co., St Louis, MO, USA) solution to a concentration of 1.0 mg/mL and sonication in a sonic bath. For low-dose administration, the MWCNT suspension was diluted to 0.05 mg/mL with 0.5% Pluronic F-68 solution.
Intraperitoneal administration of MWCNT suspensions to mice
Animal experiments were approved by the Institutional Animal Care and Use Committee of the National Institute of Advanced Industrial Science and Technology (AIST). Six-week-old male C57BL/6N mice were obtained from The Jackson Laboratory Japan, Inc. (Yokohama, Japan). The mice were fed a standard diet and allowed to acclimatize to their environment for 1 week before experimentation. The number of mice was determined considering animal welfare and statistical significance. For long-term experiments, mice were divided into five experimental groups, with each group consisting of 5 mice for 1 week, 1 month, and 3 months or 9 mice for 6 months (n = 5 or 9). For short-term experiments, mice were divided into five experimental groups, each group consisting of three mice. Namely, total number of mice used in this study was 120 for long-term experiments and 27 for short-term experiments. These numbers of experimental animals were determined from a statistical and toxicological perspective. The suspensions of MWCNT were intraperitoneally administered to the abdominal cavity of the mice at a concentration of 1.0 or 0.05 mg/mouse (1 mL/mouse). One milliliter of 0.5% Pluronic F-68 solution alone was administered to the control group. For long-term experiments, 1 week, 1 month, 3 months, and 6 months after administration, mice were anesthetized by inhalation of isoflurane (Pfizer Japan Inc., Tokyo, Japan). For short-term experiments, 1 day, 3 days and 1 week after administration, mice were anesthetized by inhalation of isoflurane. The abdominal cavity was lavaged with 5 mL cold physiological saline (0.9% sodium chloride), and the lavage fluid was collected. After centrifugation at 1500 rpm for 10 min, the supernatant of the lavage fluid was collected and used for cytokine determination, and precipitated cells were used for cellular examinations. During autopsy, blood was collected from the heart and the liver was excised. The liver tissue was fixed in 10% formalin (Wako Pure Chemical Industries, Osaka, Japan). Histological sections were prepared and subjected to hematoxylin and eosin (H&E) staining. Histological observations were performed at the Shikoku Cytopathological Laboratory (Takamatsu, Japan).
Observation of intraperitoneal macrophages
The cells collected from the abdominal cavity lavage fluid were suspended in the RPMI 1640 medium supplemented with 10% FBS. The cell suspension was added to a 6-well plate. For polarizing microscopy, a cell suspension was added to a round cover glass (Matsunami Glass Ind.,Ltd., Kishiwada, Japan) in a 12-well plate. The plate was then incubated for 2 h at 37°C in an atmosphere containing 5% CO2. Subsequently, the culture medium was removed and the cells were washed once with PBS. Next, the adherent cells were used as intraperitoneal macrophages. The intraperitoneal macrophages were fixed with 100% methanol and stained by Giemsa stain (Kanto Chemical Co., Inc., Tokyo, Japan). The cells were observed using an all-in-one microscope (BZ-X700, Keyence Corporation, Osaka, Japan) and a polarizing microscope (ML9300, Meiji Techno Co., Ltd., Miyoshi, Saitama, Japan).
Determination of cytokine, osteopontin (OPN), mesothelin, and aspartate aminotransferase (AST) concentrations
The concentrations of IL-6, IL-1β, and TNF-α in the serum and abdominal cavity lavage fluid were determined using IL-6, IL-1β, and TNF-α Mouse Uncoated ELISA kits (Thermo Fischer Scientific, Waltham, MA, USA), respectively. The concentrations of Cxcl-1, Cxcl-2, mesothelin, and OPN in the serum and abdominal cavity lavage fluid were determined using a CXCL1 ELISA Kit (Proteintech Group, Inc., Rosemont, IL, USA), MIP-2/CXCL2 Mouse ELISA Kit (Thermo Fischer Scientific), mouse mesothelin ELISA kit (Abcam, Cambridge, UK), and mouse OPN (Osteopontin) ELISA Kit (Elabscience, Inc., Wuhan, Hubei, China), respectively. The concentration of AST in the serum and abdominal cavity lavage fluid was determined using a Mouse AST ELISA Kit (Abcam).
Statistical analysis
Data are expressed as the mean ± standard error (SE). Statistical analyses were performed using analysis of variance (ANOVA), followed by Dunnett’s test or Newman–Keuls’s test for multiple comparisons.
Results
Body weight change and survival rate after intraperitoneal administration of MWCNTs
Two types of MWCNT were administered to the abdominal cavity at dose of 0.05 mg/mouse (low dose) and 1.0 mg/mouse (high dose), and the mice were reared for a maximum of 6 months. There were no differences in body weight between the MWCNT-B-administered and control groups during the experimental period, t (Figure 1). However, the body weight of the MWCNT-7-administered group was significantly lower than that of the MWCNT-B-administered and control groups. The body weight of the low-dose MWCNT-7-administered group did not increase from 3 months (12 weeks) after administration. The body weight of the high-dose MWCNT-7-administered group did not increase from 2 months (8 weeks) onward. One of the high-dose MWCNT-7-administered mice died 3 months after administration. The experiment with high-dose MWCNT-7 animals was canceled at 3 months after administration because the average body weight of the mice had dropped to 81.8% of the control group. One of the low-dose MWCNT-7-administered animals also died 5 months (20 weeks) after administration. Six months after administration, two of the ten mice died. However, MWCNT-B-treated mice did not die during the feeding period. Body weight and survival rate of multi-walled carbon nanotubes (MWCNT)-administered mice. Two kinds of MWCNT (MWCNT-B and MWNT-7) were administered to abdominal cavity of the mice by intraperitoneal administration at a concentration of 1.0 mg (high dose) or 0.05 mg (low dose) per mouse. One milliliter of 0.5% Pluronic F-68 solution was also administered as control. Body weight and survival rate was measured every 3 weeks until 6 months.
Comparison of biomarker level in the serum and the abdominal cavity lavage fluid
About 0.5–0.9 mL of serum can be recovered from one mouse, whereas 3–4 mL of abdominal cavity lavage fluid can be recovered from one mouse. Therefore, abdominal cavity lavage fluid contains more biomarkers that can be measured. We confirmed the biomarker levels in the serum and abdominal cavity lavage fluid at 6 months after the administration of MWCNTs. Although absolute values of biomarkers were different between the serum and abdominal cavity lavage fluid, the trend was similar (Figure S1). Therefore, we chose to evaluate biomarkers in the abdominal cavity lavage fluid in the present study.
Cytokine level in the abdominal cavity lavage fluid
The concentration of cytokines in the abdominal cavity lavage fluid was measured 1 week, 1, 3, and 6 month(s) after administration (Figure 2). Remarkable induction of Cxcl-1 was observed 1 week after MWCNT-B administration. The concentration of Cxcl-1 at 6 months after treatment with a high dose of MWCNT-B and low dose of MWCNT-7 was significantly higher than that in control. MWCNT-B and MWNT-7 differed in bioresponsiveness. Regarding Cxcl-1, MWCNT-B tended to show a biological response in the acute phase, while MWNT-7 tended to show a biological response in the chronic phase. The IL-6 levels in mice administered with a high dose of MWCNT-B were significantly higher than those in control mice from 1 week to 6 months after administration. Compared with the MWCNT-7-administered group, induction of IL-6 expression in the high-dose MWCNT-B-administered group was not remarkable. Low doses of MWCNT-B did not induce IL-6 production. The concentration of IL-6 in low-dose MWCNT-7-administered mice was significantly high, but not remarkable. By contrast, the concentration of IL-6 in mice administered with a high dose of MWCNT-7 was markedly higher than that in control mice at 3 months after administration. In the MWCNT-7-administered group, there were mice that died after 3 months. At 6 months after administration, the IL-6 concentration in surviving mice that were administered a high dose of MWCNT-7 did not increase. Concentrations of IL-1β, TNF-α, and Cxcl-2 in the abdominal cavity lavage fluid were under analytical sensitivity (IL-1β,8 pg/mL; TNF-α,8 pg/mL; Cxcl-2.10 pg/mL) at any time point. Cytokine level in the abdominal cavity lavage fluid. Two kinds of MWCNT (MWCNT-B and MWNT-7) were administered to abdominal cavity of the mice by intraperitoneal administration at a concentration of 1.0 mg (high dose) or 0.05 mg (low dose) per mouse. One milliliter of 0.5% Pluronic F-68 solution was also administered as control. The abdominal cavity lavage fluid was collected at 1 week, 1, 3 and 6 month(s) after intraperitoneal administration. Suspensions IL-6 and Cxcl-1 concentrations were determined using ELISA. The data are represented as mean ± SE. High dose of MWCNT-7 at 6 month was not tested (NT). *p < 0.05, **p < 0.01 (vs. each time point of control mice, ANOVA and Dunnett’s test). # indicates under the analytical sensitivity (Cxcl-1: 6.5 pg/mL, IL-6: 4 pg/mL).
Osteopontin level in the abdominal cavity lavage fluid
The gene expression of osteopontin in rat lungs was enhanced by intratracheal instillation of SWCNTs.
12
Therefore, osteopontin concentration in the abdominal cavity lavage fluid was measured (Figure 3). In mice that were administered a high dose of MWCNT-B, osteopontin levels were higher than those in control mice at 1, 3, and 6 months after administration. Low doses of MWCNT-B did not affect the osteopontin levels. Meanwhile, osteopontin levels were higher in both low- and high-dose MWCNT-7-administered mice. Additionally, as a result of multiple comparison tests by Newman–Keuls method, there were no significant differences among MWCNT-B high, MWCNT-7 low, and MWCNT-7 high groups at 1 month after administration. The MWCNT-B high group showed similar osteopontin induction levels to the MWNT-7 low group. Osteopontin and mesothelin concentration in the abdominal cavity lavage fluid. Two kinds of MWCNT (MWCNT-B and MWNT-7) were administered to abdominal cavity of the mice by intraperitoneal administration at a concentration of 1.0 mg (high dose) or 0.05 mg (low dose) per mouse. One milliliter of 0.5% Pluronic F-68 solution was also administered as control. The abdominal cavity lavage fluid was collected at 1 week, 1, 3, and 6 month(s) after intraperitoneal administration of MWCNTs or 0.5% Pluronic F-68. Osteopontin and mesothelin concentrations were determined using ELISA. The data are represented as mean ± SE. High dose of MWCNT-7 at 6 month was not tested (NT). *p < 0.05, **p < 0.01 (vs. each time point of control mice, ANOVA and Dunnett’s test). Means with different letters differ significantly at p < 0.05 (ANOVA and Newman–Keuls’s Test).
Mesothelin level in the abdominal cavity lavage fluid
Mesothelin is a known biomarker for mesothelioma formation. 13 Mesothelin concentration in the abdominal cavity lavage fluid was measured (Figure 3). The mesothelin concentration significantly increased 1 month after MWCNT administration. Furthermore, the mesothelin concentration significantly increased 6 months after MWCNT administration in the high-dose MWCNT-B-administered group. Mesothelin concentration was not affected by high-dose MWCNT-7 administration. As a result of multiple comparison tests by Newman–Keuls method, there were no significant differences between MWCNT-B low and MWCNT-7 low groups at 1 month after administration. Mesothelin levels decreased at all doses including controls at 3 months after administration. It may be a transitional period between the acute and chronic stages, but the cause is unknown at this time.
Observation of intraperitoneal macrophages
Phagocytized MWCNTs were observed in intraperitoneal macrophages of MWCNT-B-treated mice. Although MWCNT-containing macrophages were observed during the experimental period, they were most frequently observed 1 week after administration. Observation of macrophages by polarizing microscopy clearly showed phagocytized MWCNT-B (Figure 4). In contrast, phagocytized MWCNTs were unclear in the MWCNT-7-administered mice. Thus, it was difficult to determine the presence of internalized MWCNT-7 1 and 3 months after administration. Observation of intraperitoneal macrophages. Two kinds of MWCNT (MWCNT-B and MWNT-7) were administered to abdominal cavity of the mice by intraperitoneal administration at a concentration of 1.0 mg (high dose) or 0.05 mg (low dose) per mouse. 1 milliliter of 0.5% Pluronic F-68 solution was also administered as control. The intraperitoneal macrophages were harvested from abdominal cavity lavage fluid which was collected at 1 week, 1, 3 and 6 month(s) after intraperitoneal administration of MWCNTs or 0.5% Pluronic F-68. (a) Observation of intraperitoneal macrophages using light microscopy. The arrows indicate internalized MWCNTs. The scale bars are 50 μm. (b) Observation of intraperitoneal macrophages using polarizing microscopy.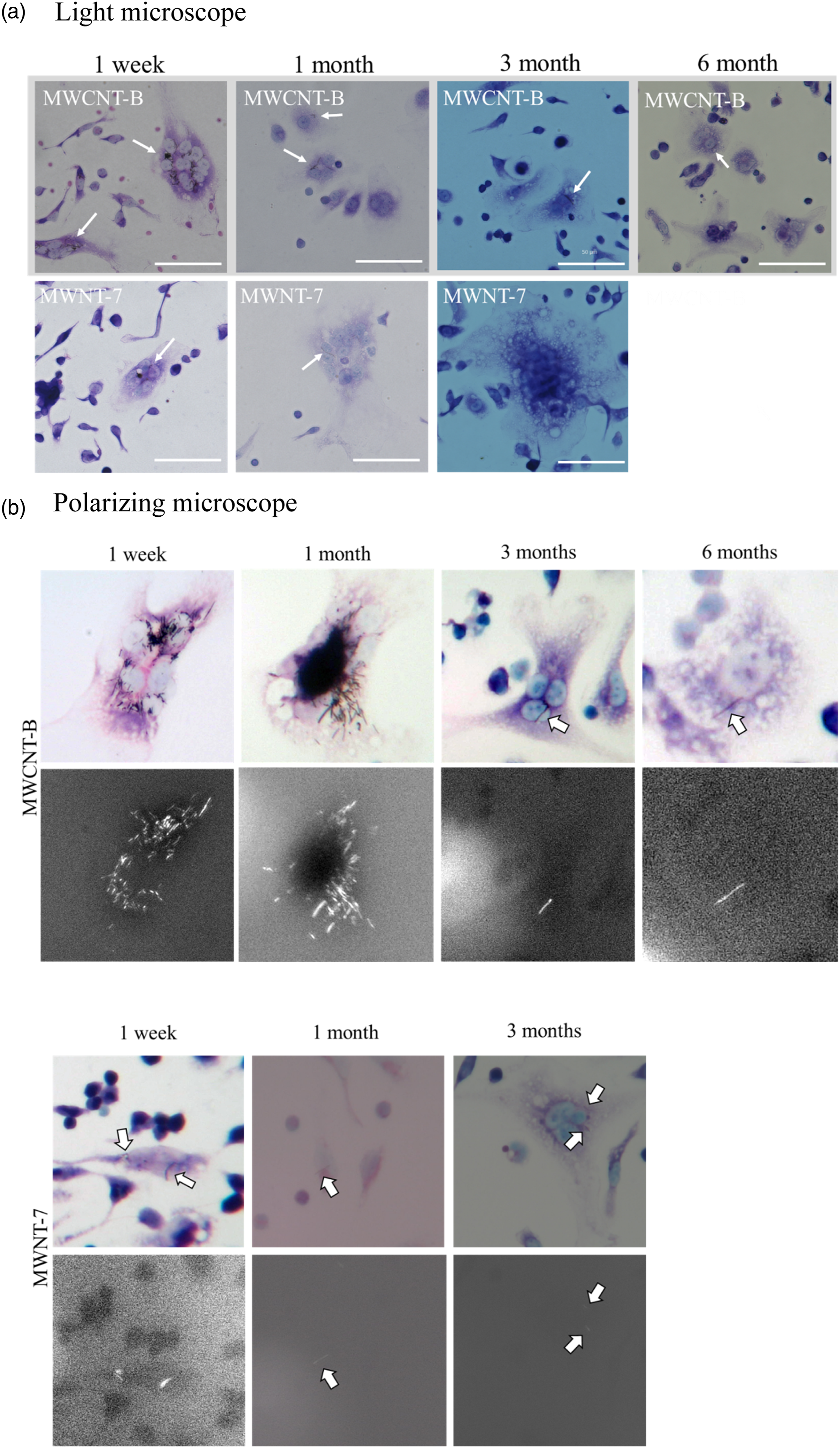
Pathological observations of hepatic damage upon intraperitoneal administration of MWCNTs
Granulomas were observed on the surface of the liver after intraperitoneal administration of MWCNT-7 and MWCNT-B (Figure 5). A change in the liver shape was observed in the high-dose MWCNT-B group. The liver became round edged and atrophic at 3 and 6 months after administration of a high dose of MWCNT-B. However, the expression of AST, a hepatic damage marker, did not increase in these mice (Figure 6). In both MWCNT-7- and MWCNT-B-administered mice, adhesion of MWCNTs on the serosal membrane and granuloma formation were observed. Pathological observations of the liver are summarized in Table 1. Histopathological observations of the liver of MWCNT-administered mice. Two kinds of MWCNT (MWCNT-B and MWNT-7) were administered to abdominal cavity of the mice by intraperitoneal administration at a concentration of 1.0 mg (high dose) or 0.05 mg (low dose) per mouse. 1 milliliter of 0.5% Pluronic F-68 solution was also administered as control. 1 week, 1, 3, and 6 month(s) after intraperitoneal administration, the mice were anesthetized, and the liver tissue was fixed in 10% (v/v) formalin. The histological sections were prepared and subjected to hematoxylin and eosin (H&E) staining. Aminotransferase (AST) concentration in the abdominal cavity lavage fluid. Two kinds of MWCNT (MWCNT-B and MWNT-7) were administered to abdominal cavity of the mice by intraperitoneal administration at a concentration of 1.0 mg (high dose) or 0.05 mg (low dose) per mouse. 1 milliliter of 0.5% Pluronic F-68 solution was also administered as control. The abdominal cavity lavage fluid was collected at 1 week, 1, 3 and 6 month(s) after intraperitoneal administration of MWCNTs or 0.5% Pluronic F-68. AST concentration was determined using ELISA. The data are represented as mean ± SE. High dose of MWCNT-7 at 6 months was not tested (NT). *p < 0.05, **p < 0.01 (vs. each time point of control mice, ANOVA and Dunnett’s test). Pathological observations of liver. aInfiltrating neutrophils was also observed at 1week. The total number of animals observed was 4 in each group.


Short-term effects of intraperitoneal administration of MWCNT suspensions
The effect of intraperitoneal administration of MWCNTs was examined for 1 week (Figure 7). The suspensions of MWCNT were intraperitoneally administered to the abdominal cavity of the mice at a concentration of 1.0 mg/mouse (high dose). Physiological saline was administered as negative control. Cytokine levels in the abdominal cavity lavage fluid were measured. In the MWCNT-7 group, IL-6 levels were significantly higher 24 and 72 h after administration. The maximum level of IL-6 was observed at 72 h after administration. IL-6 levels 1 week after administration were not different from those in the negative control group. In the MWCNT-B group, IL-6 levels were significantly elevated 24 h after administration. Cxcl-2 levels were significantly higher in the MWCNT-7 group 6, 24, and 72 h after administration. 1 week after administration, both IL-6 and Cxcl-2 showed no significant differences among any of the groups. Cytokine level in the abdominal cavity lavage fluid in early phase. Two kinds of MWCNT (MWCNT-B and MWNT-7) were administered to abdominal cavity of the mice by intraperitoneal administration at a concentration of 1.0 mg (high dose) per mouse. One milliliter of 0.5% Pluronic F-68 solution was also administered as control. The abdominal cavity lavage fluid was collected at 1, 3 days and 1 week after intraperitoneal administration. IL-6, Cxcl2, and TNF-α concentrations were determined using ELISA. The data are represented as mean ± SE *p < 0.05, **p < 0.01 (vs. each time point of control mice, ANOVA and Dunnett’s test). # indicates under the sensitivity (Cxcl-2: 10 pg/mL).
Discussion
Mesothelioma was formed by intraperitoneal administration of MWCNT-7 to p53 heterozygous C57BL/6 mouse. 14 In this study, the dose of MWCNT-7 was 3, 30, and 300 μg/mouse, and the mice died because of a mesothelioma 150 days after administration. However, tumor formation upon intraperitoneal administration of MWCNTs was not observed in the present study, which employed normal C57BL/6 mice, even when the dose of MWCNT was higher (50 and 1000 μg/mouse) than that in the previous study. In the case of asbestos, when 200 μg of crocidolite was administered intraperitoneally to mice, 76% of animals developed mesothelioma after 44 weeks in p53+/− mice. In contrast, wild-type mice had a mesothelioma incidence of 32% after 67 weeks. Mesothelioma was not found before 30 and 60 weeks in p53+/− and normal C57BL/6 mice, respectively.15,16 Therefore, the results of the present study are considered to be a pre-stage biological response leading to mesothelioma. The inflammatory response may be important as a precursor to MWCNT-induced mesothelioma. However, an overdose of harmful MWCNT will result in death due to severe inflammation. Compared with the previous study, the mice died 3 months or later after the administration of MWCNT-7. Survival rate of the low dose of MWCNT-7 administered mice at 6 months (approximately 180 days) after administration was 80%. Meanwhile, the mice administered MWCNT-B did not die. Additionally, body weight did not decrease after administration of MWCNT-B. According to pathological observations, the cause of death in MWCNT-7-administered mice was multiple organ failure caused by severe inflammation. In the present study, because two mice died, there were 7 mice in low-dose of MWNT-7 group at 6 months after administration. This is not sufficient for the evaluation of carcinogenesis. Although mesothelioma was not found in MWCNT-7-administered mice at this time point, the survival rate of these animals decreased in the present study. Both carcinogenic and proinflammatory properties may contribute to the harmful effects of MWCNT-7. In contrast, MWCNT-B-administered mice did not die for at least 6 months after administration. Although the shape of the liver changed in MWCNT-B-administered mice, expression of GPT, a marker for hepatic damage, did not increase. No remarkable pathological findings were noted. The cause of the shape change in the liver is unknown; it may not directly affect survival.
How does MWCNT-7 induce inflammation? There were two events until 3 days and 3 months after intraperitoneal administration of the MWCNT suspensions. We focused on the cytokines that mediate inflammation. IL-6 and Cxcl-2 levels were increased by intraperitoneal administration of MWCNT-7 suspensions at a very early phase, 3 days after administration. These cytokines are chemotactic factors of macrophages or neutrophils. These phagocytes may accumulate in MWCNTs and attempt to remove MWCNTs. MWCNT are degraded by hypochlorous acid. 17 Considering that neutrophils generate hypochloric acid for the digestion of organic substances by myeloperoxidase, 18 it is possible that phagocytized MWCNTs are degraded by neutrophils. Additionally, macrophages can degrade the stability of carbon nanotube. 19 However, the mechanistic details of in vivo degradation of phagocytized MWCNT remains unknown. Additionally, neutrophils probably cannot degrade all administered MWCNTs. When MWCNTs were injected into the lungs of mice via intratracheal administration, phagocytosed MWCNT in the macrophages were observed 6 months after administration. 5 Cytokine levels, which increased in the very early phase, were decreased 1 week after administration. In MWCNT-7-administered mice, IL-6 levels remarkably increased again 3 months after administration. These results suggest that MWCNT-7-induced inflammation is exacerbated 3 months after administration. After 3 months, the inflammation continued and was aggravated. Consequently, the mice died. If the dose of the MWCNT-7 is below the lethal dose, relatively minor inflammation continues, which may lead to mesothelioma. There was no difference in body weight among control, low-dose MWCNT-7, and MWCNT-B groups until 3 months after administration. However, after the third month, the body weight of the MWCNT-7-administered groups tended to be lower than those of the control and MWCNT-B groups. The point 3 months after exposure seems to be a turning point for the harmful effect of intraperitoneally administered MWCNT-7. Thus, it would be interesting to explore the effects 3 months after exposure to MWCNT-7. Moreover, the dose-dependent induction of IL-6 expression at 3 months after administration is intriguing. The remarkable induction of IL-6 expression was not observed in the MWCNT-B-treated groups. Overall, the MWCNT-induced biologic responses were stronger with MWNT-7 than with MWCNT-B. Previous studies have shown that the diameter and rigidity of MWCNTs is related to their toxicity. 20 MWCNTs with a diameter of 50 nm showed stronger cytotoxicity than MWCNTs with a diameter of 150 nm in MeT5A mesothelial cells. Also, the thin MWCNTs induced mesothelioma by intraperitoneal administration to Fischer 344/Brown Norway F1 rat, whereas the thick MWCNTs did not induce mesothelioma. Diameters of MWNT-7 and MWCNT-B were 60 nm and 176 nm, respectively. Therefore, MWCNT diameter may be an important factor for inflammation induction in this study. However, the responses of macrophage and lung epithelial A549 cells to other MWCNT whose diameter was 48 nm were minimal.21,22 This MWCNT induced lung inflammation in rats by inhalation but did not cause mesothelioma. 23 The iron content of this MWCNT was 53 ppm. Another major difference between MWCNT-7 and MWCNT-B is the iron content; MWCNT-B containing almost no iron and MWCNT-7 being relatively iron rich. It is generally considered that the iron included in MWCNT-7 is related to its harmful effect. It is suggested that iron may lead to induction of oxidative stress and subsequent inflammation. However, the contribution of oxidative stress to the harmful effects of MWCNT-7 is small. MWCNT-7 caused an increase in IL-8 expression due to iron overload and a subsequent increase in intracellular Ca2+ levels in neutrophil-like HL-60 cells. 11 There is a possibility that inflammation is enhanced by phagocytes, such as neutrophils, 3 months after administration because an increase in cytokine secretion was observed in the early phase, the quantity of neutrophils in the abdominal cavity lavage fluid slightly increased 3 months after administration. The iron content of MWCNTs may be related to the enhancement of inflammation via intracellular Ca2+ increase, IL-8 induction, and neutrophil accumulation. Additionally, it is possible to predict the inflammation caused by MWCNT at an early time by evaluating cytokine levels.
In this study, the induction of inflammation was shown as a biological response of MWCNT MWCNTs induce inflammation. The degree of inflammation induction differed depending on the type of MWCNT. In this study, it is not clear what properties of MWCNTs lead to differences in their bioresponsiveness. In addition, the biological response process leading to inflammation as an endpoint is also unclear. In the present study, we used the number of animals considering the individual differences and statistical significance, but the calculation and justification of the sample size was not done. These are the limits of this research and should be clarified in the future.
Conclusion
An inflammatory reaction occurs as a biological response of MWCNT. Inflammatory responses are divided into acute and chronic responses. These inflammatory responses differ depending on the type of MWCNT. MWNT-7 which is known as harmful MWCNT caused strong inflammation response.
Supplemental Material
Supplemental Material - Follow the time course of inflammation caused by intraperitoneal administration of multi-wall carbon nanotubes in mice
Supplemental Material for Follow the time course of inflammation caused by intraperitoneal administration of multi-wall carbon nanotubes in mice by Masanori Horie, Sakiko Sugino and Tomoki Ohno in International Journal of Immunopathology and Pharmacology
Footnotes
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Institutional Animal Care and Use Committee of the National Institute of Advanced Industrial Science and Technology (AIST) (Approval number: Dou2019-0216).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
