Abstract
Background
Alzheimer’s disease (AD) is the most common cause of dementia. About one million new cases of AD develop every year. AD commonly occurs after age 65. Early-onset AD occurs in less than 5% of cases and develops before 65 years. 1 AD symptoms may include memory loss, executive dysfunction, language difficulties, cognitive dysfunction symptoms, behavioral disturbances, and psychiatric illness as non-cognitive dysfunctions. 2
Valsartan is an angiotensin II type-1 (AT1) receptor blocker that has an influential role in the treatment of hypertension and heart failure. It can decrease neurodegeneration induced by reactive oxygen species (ROS), inflammatory responses, and apoptotic signals mediated by activation of the AT1 receptor. 3
Neuronal dysfunction associated with AD is mainly related to the accumulation of amyloid-beta (Aβ) plaques. Many enzymes are involved in the degradation of Aβ as neprilysin, endothelin converting enzyme-1, and insulin-degrading enzyme. 4
Neprilysin is an enzyme found in many tissues, especially the brush border of proximal tubules and glomerular epithelium. It is known as membrane metalloendopeptidase (MME) and neutral endopeptidase (NEP)
Sacubitril is a neprilysin inhibitor that can improve heart failure through vasodilatation and sodium loss and decrease cardiac and vascular hypertrophy and remodeling. 6 Sacubitril-valsartan combination was approved to manage patients with heart failure with preserved ejection fraction. 7
Patients who have used the sacubitril-valsartan combination for a long period for treatment of heart failure may be at risk for the development or acceleration of AD, especially if they are elderly patients, as neprilysin inhibitors may decrease the degradation of Aβ in the brain. Even younger patients receiving sacubitril/valsartan have the potential for longer-term exposure and consequent increased risk of young-onset Alzheimer’s disease (YOAD), described in subjects less than 65 years of age and has a more rapid progression than the typical late-onset AD. 8
The aim of the present study was to investigate the effect of treatment with valsartan alone or in combination with sacubitril on a rat model for AD by behavioral tests, biochemical measurement of rat brain malondialdehyde and amyloid-beta level, and also a histopathological examination of the hippocampus of rats’ brains.
Methods
Drugs, reagents, and solutions
Aluminum chloride powder, valsartan (Amoun Pharmaceuticals & Chemical Industries), sacubitril/valsartan combination; 97 mg/103 mg (Novartis), all chemicals were dissolved in distilled water.
Animals used: In this study, 72 male adult Wistar albino rats of matching age and weight (200–230 g) were used. Animals were housed in two a cage and were kept under hygienic conditions at a room temperature of 22°C and 55% humidity with a 12-h light/12-h dark schedule. Rats were fed standard rat pellets, and water was freely provided. This study was approved by Cairo University’s Institutional Animal Care and Use Committee (IACUC), medical sciences sector. The experiments were carried out in accordance with the ethical guidelines of the animal welfare application number (CU/III/F/28/19).
Experimental design
Animals were randomly assigned into the following groups (12 rats per each group):
** Doses of the drugs are calculated from human doses according to the body surface area (BSA) formula. 10
Assessment of behavioral changes
Morris water maze test
This test is utilized to assess the learning and spatial memory of the animals. The Morris water maze is a swimming-based model in which animals learn to escape a pool of water (150 cm in diameter, 45 cm in height, filled to a depth of 30 cm with water at 28 ± 1°C) by a hidden platform. The tank was divided into four equal quadrants by fixing two threads at the right angle to each other on the rim of the pool. A black platform (10 cm2) was placed in the northern quadrant center 1 cm below the surface of the water.
The platform position was maintained throughout the training session. Rats were subjected to training trials for four consecutive days. The animals were gently put in the water at one of the four randomly selected positions. Rats were allowed 2 min to locate on the submerged platform. Upon reaching the platform, rats were allowed to stay on it for 30 s. On day 5, the platform was removed, and each rat was allowed to explore the pool for 120 s. The escape latency time (ELT) to locate the hidden platform in the water maze was noted as an index of acquisition or learning. The mean time spent in the target quadrant searching for the platform was noted as an index of retrieval. The test was done every 2 weeks. 11
Novel object recognition test
This test shows that rodents will spend more time exploring a novel object than a familiar one
In the testing session, the animal was exposed to two objects: one identical to the object used in the familiarization session and one novel. These objects were placed in the same locations as during the familiarization session. The time taken by the animals to explore the objects was recorded, and the discrimination score was calculated
12
Assessment of blood pressure
SBP was monitored for all animals at the start of the experiment, second,, fourth, and sixth weeks using a tail-cuff blood pressure measuring system (Harvard Apparatus Ltd, Edenbridge, Kent, England). 13
Assessment of biochemical parameters
The excised hippocampus (6 rats from each group) was homogenized in ice-cold 50 mm sodium phosphate buffer (pH 7.4) containing 0.1 mm ethylenediaminetetraacetic acid (EDTA) by using a glass-Teflon homogenizer (Heidolph Silent Crusher M, Germany). The supernatant was separated USING centrifugation at 1000×g for 20 min at 4°C and then frozen at −80°C until UTILIZED for biochemical analysis. 14
Thiobarbituric Acid Reactive Substances (TBARS) Assay Kit is used for spectrophotometrical quantitative measurement of rat brain Malondialdehyde (MDA). Enzyme-Linked Immunosorbent Assay (ELISA) was used to measure Aβ1–42 level.
Pathological assessment
The isolated hippocampus of the brains (6 rats for each group) was fixed in 10% formalin, embedded in paraffin wax, sectioned, and stained. The sections were examined under a light microscope, and photomicrographs were done to assess histopathological changes.
Morphometric study
Data were obtained using (Leica Qwin 500 C) image analyzer computer system Ltd. (Cambridge, England). For each group, five slides of six different specimens were examined. Ten non-overlapping fields were measured in a standard measuring frame for each slide. The mean number of pyknotic nuclei of the neurons in the hippocampus was calculated using the interactive measuring menu.
Statistical analysis
Data were coded and entered using the statistical package SPSS (Statistical Package for the Social Sciences) version 25. Data were summarized using mean and standard deviation. Comparisons between groups were made using ANOVA with post hoc test in normally distributed quantitative variables, while the non-parametric Kruskal–Wallis test and Mann–Whitney test were used for non-normally distributed quantitative variables. Repeated measures ANOVA was used in normally distributed quantitative variables to compare serial measurements within each group, while the non-parametric Friedman test was used for non-normally distributed quantitative variables. p-values less than .05 were considered statistically significant. 15
Results
Behavioral tests
Morris water maze test
Time spent in the target quadrant
Time spent in target quadrant in Morris water maze test in studied groups.
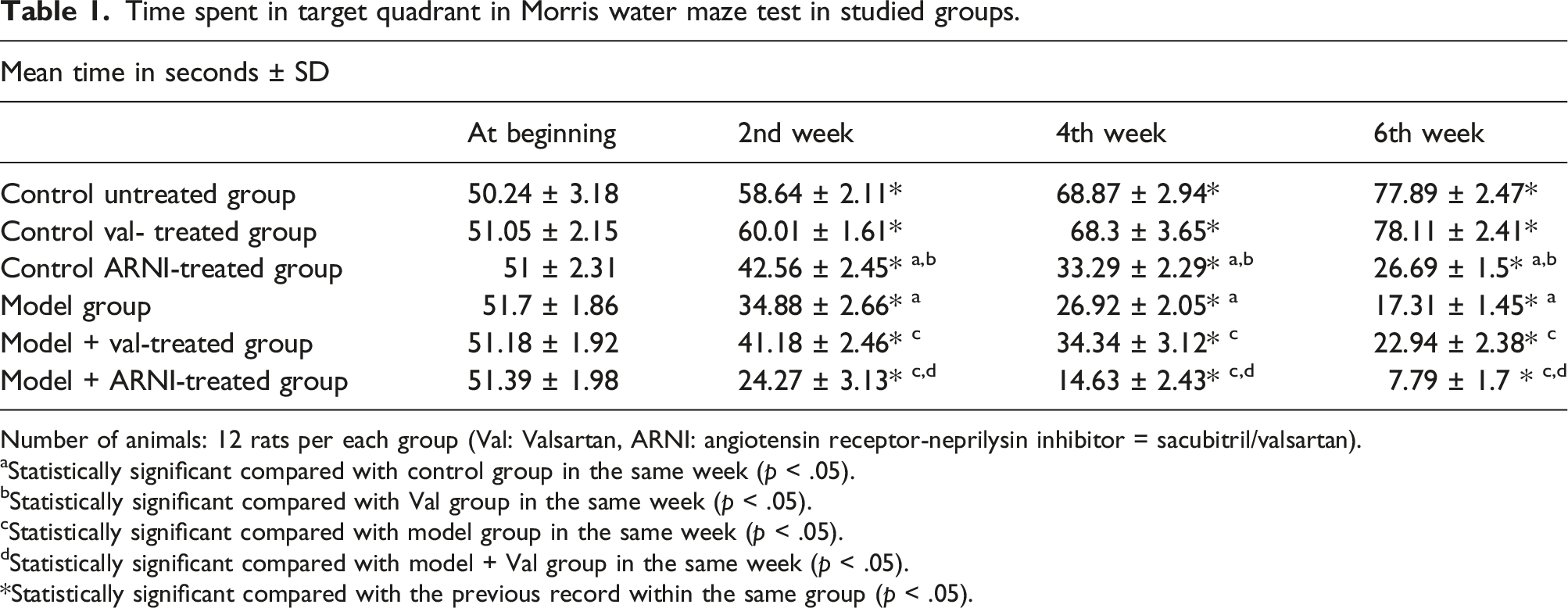
Number of animals: 12 rats per each group (Val: Valsartan, ARNI: angiotensin receptor-neprilysin inhibitor = sacubitril/valsartan).
aStatistically significant compared with control group in the same week (p < .05).
bStatistically significant compared with Val group in the same week (p < .05).
cStatistically significant compared with model group in the same week (p < .05).
dStatistically significant compared with model + Val group in the same week (p < .05).
*Statistically significant compared with the previous record within the same group (p < .05).
Rats of the model group (Group IV) showed significant deterioration in mean time spent in the target quadrant at the second, fourth, and sixth week compared to the beginning time and when compared to their corresponding values in the control untreated group (p < .05). Model animals treated with valsartan for 6 weeks (Group V) showed significant improvement in mean time spent in the second, fourth,, and sixth week compared to their corresponding values in the model untreated group (p < .05). Conversely, model animals treated with ARNI (Group VII) showed a significant progressive reduction in the meantime at the second, fourth,, and sixth week compared to model and model valsartan-treated groups (p < .05) (Table 1).
Escape latency time
Escape latency time in Morris water maze test in studied groups.

Number of animals: 12 rats per each group (Val: Valsartan, ARNI: angiotensin receptor-neprilysin inhibitor = sacubitril/valsartan).
aStatistically significant compared with control group in the same week (p < .05).
bStatistically significant compared with Val group in the same week (p < .05).
cStatistically significant compared with model group in the same week (p < .05).
dStatistically significant compared with model + Val group in the same week (p < .05).
*Statistically significant compared with the previous record within the same group (p < .05).
Rats in the model group showed a significant progressive increase in mean escape latency time at the second, fourth, and sixth week compared to the beginning time and when compared with the control group (p < .05). This increase was improved by valsartan treatment in Group V (p < .05) and was worsened by ARNI treatment in Group VII (p < .05) (Table 2).
Novel object recognition test
Novel object recognition test in studied groups.

Number of animals: 12 rats per each group (Val: Valsartan, ARNI: angiotensin receptor-neprilysin inhibitor = sacubitril/valsartan).
aStatistically significant compared with control group in the same week (p < .05).
bStatistically significant compared with Val group in the same week (p < .05).
cStatistically significant compared with model group in the same week (p < .05).
dStatistically significant compared with model + Val group in the same week (p < .05).
*Statistically significant compared with the previous record within the same group (p < .05).
Biochemical tests
The mean brain MDA and Aβ levels in control ARNI-treated group showed significant elevation when compared with the control untreated and control valsartan-treated group (p < .05). The model group induced a significant elevation in both levels compared to untreated rats (p < .05). Valsartan in model rats significantly reduces this elevation (p < .05), while ARNI treatment in the model group produces more elevation in mean brain MDA and Aβ levels compared with the model untreated group (p < .05) (Figures 1 and 2). Supplementary Figure 1 shows graphical abstract. Rat brain malondialdehyde (MDA) in different studied groups. Number of animals: 12 rats per each group (Val: Valsartan, ARNI: angiotensin receptor-neprilysin inhibitor = sacubitril/valsartan). *Statistically significant compared with the previous record within the same group (p < .05). a: Statistically significant compared with control group in the same week (p < .05). b: Statistically significant compared with Val group in the same week (p < .05). c: Statistically significant compared with model group in the same week (p < .05). d: Statistically significant compared with model + Val group in the same week (p < .05). Rat brain amyloid beta 1-42 (Aβ 1–42) in different studied groups. Number of animals: 12 rats per each group (Val: Valsartan, ARNI: angiotensin receptor-neprilysin inhibitor = sacubitril/valsartan). *Statistically significant compared with the previous record within the same group (p < .05). a: Statistically significant compared with control group in the same week (p < .05).b: Statistically significant compared with Val group in the same week (p < .05). c: Statistically significant compared with model group in the same week (p < .05). d: Statistically significant compared with model + Val group in the same week (p < .05).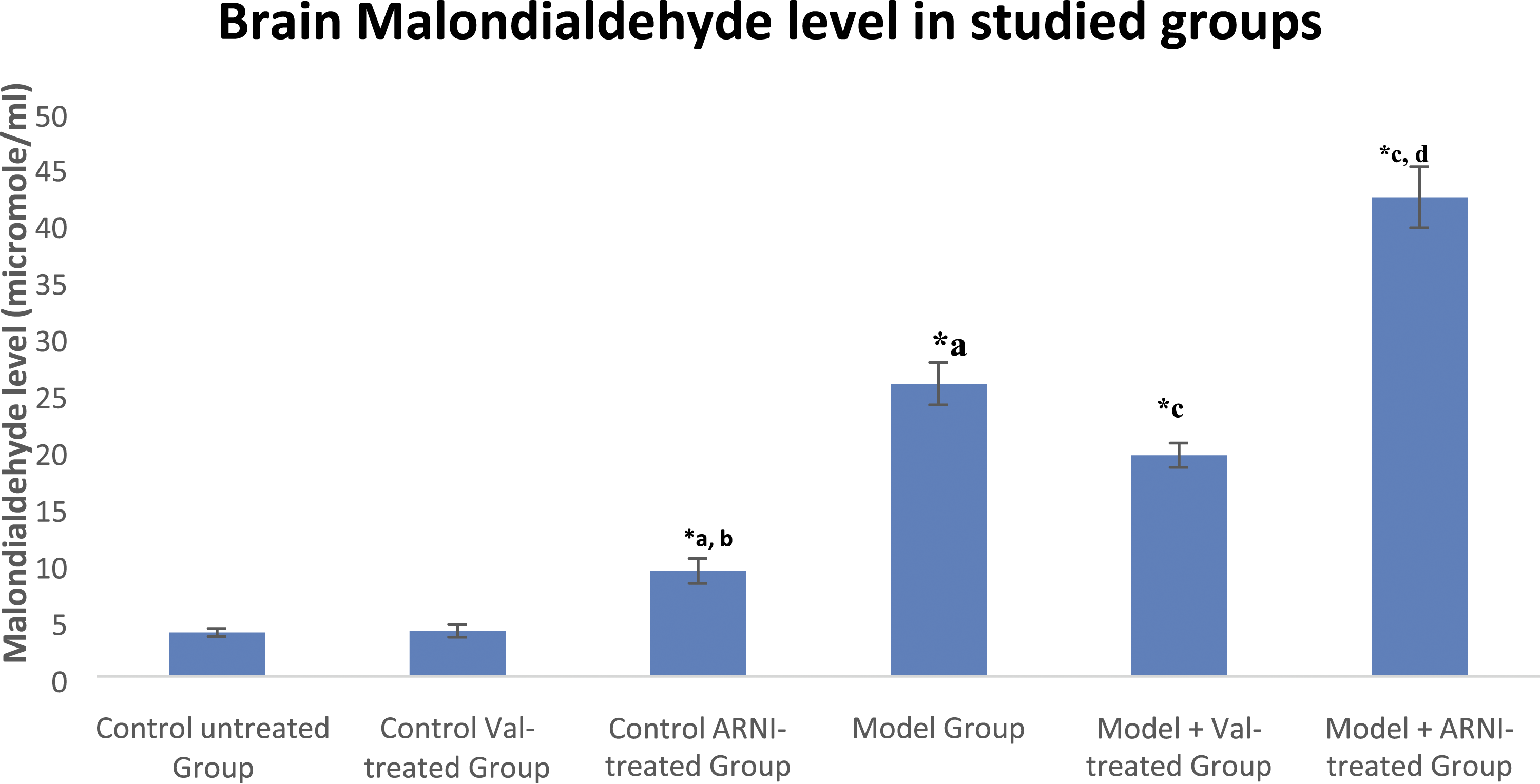

Blood pressure measurements
Control rats treated with either valsartan (Group II) or ARNI (Group III) produced a significant progressive decrease in mean SBP at the second, fourth, and sixth week of treatment when compared to the beginning and to the corresponding value in control untreated group (p < .05) with a significant decrease in Group III compared with Group II (p < .05). The model group showed a significant progressive increase in mean systolic blood at the second, fourth, and sixth week when compared to the beginning and values in control untreated, control valsartan treated, and control ARNI treated groups (p < .05) (Figure 3) Mean systolic blood pressure at beginning, second, fourth, and sixth week in different studied groups. Number of animals: 12 rats per each group (Val: Valsartan, ARNI: angiotensin receptor-neprilysin inhibitor = sacubitril/valsartan). *Statistically significant compared with the previous record within the same group (p < .05). a: Statistically significant compared with control group in the same week (p < .05). b: Statistically significant compared with Val group in the same week (p < .05). c: Statistically significant compared with model group in the same week (p < .05). d: Statistically significant compared with model + Val group in the same week (p < .05).
Valsartan treatment in the rats model group showed a significant increase in mean SBP compared to the model group in the sixth week (p < .05). ARNI treatment in Group VII showed a significant decrease in mean SBP at the second, fourth, and sixth week compared to model and model valsartan-treated groups (p < .05) (Figure 3).
Histological examination of hippocampal sections
Sections from control untreated rats showed that the hippocampus formed adequately from three layers; polymorphic layer (OL), pyramidal layer (PL), and molecular layer (ML). The pyramidal layer was formed of several layers of closely arranged neurons with large rounded vesicular nuclei with prominent nucleoli and scanty basophilic cytoplasm. Few dark, irregular shrunken cells were detected. These histological features were not disrupted in control valsartan-treated rats, while control treated showed some pyramidal cells with shrunken pyknotic nuclei. In addition, some of the neurons present in the dentate gyrus showed pyknotic shrunken nuclei, and others showed vacuolated cytoplasm (Figure 4). Photomicrograph of a section in the hippocampus of an albino rat in different studied groups (H&E, x100). (a) Group I (Control untreated Group): Neurons with large rounded vesicular nuclei with prominent nucleoli (arrowheads), few dark irregular shrunken cells (spiral arrow). (b) Group II (Control Valsartan Treated Group): Neurons with large rounded vesicular nuclei with prominent nucleoli (arrowheads). (c) Group III (Control Sacubitril/valsartan Treated Group): Many neurons present large rounded vesicular nuclei with prominent nucleoli (arrow heads) some possess shrunken pyknotic nuclei (arrows). (d) Group IV (Model Group): Neurons possess irregular shrunken pyknotic nuclei (spiral arrows). (e) Group V (Model Valsartan Treated Group): Neurons with large rounded vesicular nuclei with prominent nucleoli (arrowheads), some of the neurons possess shrunken pyknotic nuclei. (f) Group VI (Model Sacubitril/valsartan Treated Group): Most neurons in the Pyramidal layer showed shrunken pyknotic nuclei with vacuolated cytoplasm (arrow).
Histological examination of the model group showed many Pyramidal cells with irregular shrunken pyknotic nuclei. Most of the neurons present in the dentate gyrus show pyknotic shrunken nuclei. Sections obtained from the model valsartan-treated group showed a pyramidal layer formed of several layers of closely arranged neurons with large rounded vesicular nuclei with prominent nucleoli and scanty basophilic cytoplasm as some of the neurons present in the dentate gyrus, showed pyknotic shrunken nuclei. In contrast, others showed large rounded vesicular nuclei with prominent nucleoli. The pyramidal layer in ARNI treated model group possessed shrunken pyknotic nuclei with vacuolated cytoplasm, and the dentate gyrus possessed shrunken irregular pyknotic nuclei (Figure 4).
The mean number of pyknotic nuclei of neurons in hippocampus layers significantly increased in control ARNI-treated, model, model valsartan-treated, and model ARNI-treated groups compared to the control (Figure 5). Model rats treated with valsartan showed a significant decrease compared to the model. In contrast, there was a significant increase in ARNI-treated model rats compared to the model group. The mean number of pyknotic nuclei of neurons in hippocampus region in different studied groups. GI: Control untreated Group, GII: Control Valsartan Treated Group, GIII: Control Sacubitril/valsartan Treated Group, GIV: Model Group, GV: Model Valsartan Treated Group, GVI: Model Sacubitril/valsartan Treated Group. *significant when compared to the control. ●significant when compared to the model. β significant when compared group VI.
Discussion
In the present study, the induction of AD in rats using aluminum chloride for 6 weeks resulted in deterioration in behavioral tests, a significant increase in MDA and amyloid beta 1- 42 levels, and a significant increase in the mean number of pyknotic nuclei of the hippocampus compared with control untreated rats. The model group treated with valsartan for 6 weeks showed improvement in all tested parameters compared to untreated model rats. On the contrary, model animals treated with ARNI for 6 weeks showed significant impairment in all tested parameters compared to model and model valsartan-treated groups.
The current study’s results align with the results obtained by Kumar et al., 16 who observed that mice were treated with aluminum chloride for 90 days. Animals showed significant impairment in learning and memory ability indicated by cognitive deficits in the Morris water maze test that was paralleled by a significant increase in acetylcholinesterase (AChE) activity, decrease in superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) activity, as well as an increase in the level of MDA in cortex and hippocampus.
The elevation of AChE activity may be due to a direct neurotoxic effect on the plasma membrane caused by increased lipid peroxidation that may affect the integrity and functionality of the cholinergic system. Thus, alterations in the lipid membrane could be a decisive factor in changing the conformational state of the AChE molecule, which would result in learning and memory deficits. 17
Yang et al. 18 found that valsartan attenuated aluminum chloride-induced cognitive decline. This study suggested a potential role of valsartan as a neuroprotective agent against neurotoxicity due to its regulation of AChE activity in the brain and elevation of the antioxidant ability by increasing the activities of SOD, GSH-Px and reducing the formation of ROS by reducing the level of MDA.
Saviano et al. 19 found a correlation between the accumulation of A1–42 peptides in the brains of rats and cognitive decline. They also demonstrated that intracerebroventricular injection of Aβ1–42 peptides in mice led to cognitive decline that was improved by Ribodiet. Valsartan treatment might prevent Aβ-related spatial memory deficits by reducing Aβ aggregation into high molecular weight Aβ, increasing Aβ degradation by insulin-degrading enzymes, and promoting of movement of Aβ peptides from the brain to the periphery. 20
Abdallah et al. 21 and Goel et al. 22 illustrated that angiotensin-converting enzyme inhibitors (ACEIs) inhibit the degradation and deposition of Aβ1-42 in the brain of animal models. Davies et al. 23 and Ho and Nation 24 detected that patients taking angiotensin receptor blockers (ARBs) had lower risks of AD than those taking other antihypertensive medications. This beneficial effect of valsartan occurred at a dose approximately 2-fold lower than the equivalent recommended clinical dose for the treatment of hypertension. 25
Brain renin-angiotensin system (RAS) acts mainly through angiotensin 1, 2, and 4 receptors (AT1R, AT2R, and AT4R, respectively). The AT1R promotes inflammation, mitochondrial ROS generation, neurodegenerative pathways, and apoptotic signals. AT2R increases nitric oxide. AT4R is essential for dopamine and acetylcholine release and may mediate memory acquisition and recall and regulates blood flow. 26 An AT4R binds angiotensin II and a fragment of angiotensin II, referred to as angiotensin IV (AngIV). This AT4 site is prominent in the cerebral cortex, hippocampus, basal ganglia, cerebellum, and spinal cord. 27
Valsartan can decrease the risk of AD development as it selectively blocks AT1R 28 as RAS inhibitors have been associated with the reduction of brain damage in different experimental and clinical models of neurodegenerative diseases. 29 The implication of AT1R in the pathogenesis of AD was emphasized by Cosarderelioglu et al., 30 who showed that postmortem samples obtained from the brains of patients with AD showed higher AT1R levels and activity.
The present results are consistent with those detected by Royea et al., 31 who demonstrated the beneficial role of losartan treatment in improving the manifestation of AD in mice models. Furthermore, they suggested that this effect occurred through AT4R activation and was independent of changes in blood pressure, amyloidosis, and oxidative stress.
The potential effect of activation of AT4R was approved by Royea et al., 32 who injected AngIV intracerebroventricularly in APP-transgenic mice. It was found that AngIV restored short-term memory and spatial learning through cerebral vasodilatation and cerebrovascular nitric oxide bioavailability without alteration of blood pressure, neuroinflammation, or Aβ pathology. Even when AngIV is administered systemically, cognitive benefits appear to be maintained. 33
Valsartan could protect and improve AD through the unopposed action of angiotensin II on AT2R and AT4R activation as AT1R is blocked as ACE inhibitors were reported to be involved in the breakdown of Aβ peptides but did not control the manifestation of AD compared with ARBs. 34 Li et al., 35 in a large epidemiological study, and Ouk et al. 36 reported that ARBs use was associated with a significant reduction in the incidence and progression of AD compared to ACEIs.
The results of the present study are in agreement with Kim et al., 37 who recorded the effects of valsartan on oxidative stress in type 2 diabetic patients with hypertension, and Cheng et al., 38 who describe the protective effects of valsartan in decreasing oxidative stress in doxorubicin-induced myocardial injury in rats
The previous results contradict what Ferrington et al. 39 detected, that valsartan treatment did not alter the accumulation of Aβ in transgenic mice. These differences in results could be reconciled by the difference in animal models, the age of the animals, and the dose of the drugs.
Sacubitril (a neprilysin inhibitor) and its active metabolite can cross the blood-brain barrier. Neprilysin inhibitor interferes with the breakdown and clearing of beta-amyloid peptides, raising concern for AD development, especially in patients at risk of AD. 40
Iwata et al. 41 suggested that catabolic down-regulation of Aβ could be more influential than anabolic up-regulation in the pathological cascade leading to AD. Neprilysin is a rate-limiting enzyme involved in Aβ42 catabolism as alternative proteolytic pathways such as lysosomal proteolysis are inefficient to fully compensate for the suppression of the neprilysin degradation. Neprilysin can degrade monomeric forms of Aβ and its more toxic oligomers. 42
Despite the presence of valsartan in sacubitril/valsartan combination, the result of the use of the combination may be towards the increased deposition of Aβ in the brain as Aβ resulted from an inhibition of neprilysin induces the formation of oligomers of AT2R in the hippocampus that disrupts angiotensin II-mediated signaling. The Aβ-induced AT2R oligomerization was associated with enhanced neurodegeneration. 43 Also, the protective effect of valsartan is mainly due to the inhibition of the anabolic pathway, which is weaker in its effect than the catabolic pathway, through reducing Aβ aggregation into high molecular weight Aβ species. 20
The results of the present study are contradicted by Vodovar et al.,
44
who showed that no difference in Aβ in the CSF and amyloid positron emission tomography
McMurray et al. 45 disclosed that sacubitril/valsartan treatment in cynomolgus monkeys led to an increase in CSF Aβ 1-42 but not in the brain, and a 2-week administration in healthy human volunteers did not modify Aβ 1-42 levels in CSF. McMurray et al. 46 indicated that dementia-related adverse effect was not increased in healthy subjects treated with sacubitril/valsartan combination for 2 weeks and confirmed that by cognitive tests.
Henderson 47 measured the cognitive function using the CogState global cognition composite score (GCCS), which includes assessing attention, episodic memory, and executive function. The change in GCCS from baseline did not differ between patients treated with sacubitril/valsartan compared to those treated with valsartan. He suggested that other Aβ clearance pathways exist in the brain that compensates for any decreased clearance related to neprilysin inhibition. Grewal et al. 48 detected that the incidence of neurocognitive diagnoses was lower among sacubitril/valsartan combination recipients in a large-cohort, propensity-matched analysis of systolic heart failure patients.
These results did not exclude an increased risk of AD with long-term sacubitril/valsartan use, as most studies involved young monkeys and normal human volunteers. In both groups, the blood-brain barrier functions as it should. However, hypertension and other vascular conditions frequently compromise the blood-brain barrier in heart failure patients, allowing drugs to enter the central nervous system. 49
In addition, specific tests were not performed to assess whether early changes in either Alzheimer-specific cognitive function or macular degeneration had occurred. 50 Also, these studies did not investigate the drug for long. 51 Data showed that sacubitril/valsartan was not associated with increased cognition, and dementia-related adverse effects were not linked to a disproportionate rate of these events compared to other medications. 52
Furthermore, Yu et al. 53 reported that sacubitril/valsartan might lower inflammation and oxidative stress in the doxorubicin-induced heart failure model in rabbits. Nevertheless, measuring here was in cardiac muscle that may benefit from an increased level of natriuretic peptides.
In the present study, the control valsartan-treated group and the control ARNI-treated group showed a significant decrease in mean SBP compared with the control untreated group with a significant decrease in the control ARNI-treated group compared control valsartan treated group. The model group showed a significant increase in mean SBP compared with the control untreated group. Significant improvement in mean SBP was detected in model groups treated by valsartan or ARNI, with a much significant lowering in the model ARNI-treated group.
Martinez et al. 54 studied the effects of a 60-day aluminum exposure at doses similar to human dietary levels (8.3 mg/kg for 60 days) on the cardiovascular system. Moreover, they found that aluminum chloride exposure increased SBP, decreased nitric oxide bioavailability, increased ROS production and contractile prostanoids and increased lipid peroxidation, and altered the antioxidant status in plasma, aorta, and mesenteric resistance arteries.
The results of the present study can be explained by Vodovar et al., 44 who recorded that neprilysin inhibition leads to increase levels of neuropeptides and vasodilators that are beneficial in hypertension and heart failure. Kusaka et al. 55 reported that sacubitril/valsartan is more efficacious in improving ACh-induced relaxations than valsartan alone in rats fed a high-salt diet which explained its beneficial effects on vasodilator function.
The present results agree with Seki et al., 56 who found a significant difference in SBP between sacubitril/valsartan and valsartan-treated spontaneously hypertensive rats. In the large, randomized, double-blind PARADIGM-HF trial, there was a greater incidence of hypotension in the group treated with sacubitril/valsartan combination than in the group treated with enalapril. 57
Sacubitril/valsartan decreased plasma N-terminal pro-b-type natriuretic peptide (NT-proBNP) (not a neprilysin substrate) and increased plasma brain natriuretic peptide (BNP) (a neprilysin substrate) leading to enhanced urinary sodium excretion and suppressed sympathetic activity. 58
In a 7-day valsartan-controlled study in heart failure with reduced ejection fraction patients (HFrEF), administration of sacubitril/valsartan resulted in a significant non-sustained increase in natriuresis and decreased plasma mid-regional pro-atrial natriuretic peptide (MR-proANP) and NT-proBNP compared to valsartan. 59 In a 21-day study in patients with HFrEF, sacubitril/valsartan significantly increased urine ANP and decreased plasma NT-proBNP, aldosterone, and endothelin-1. 60
Limitation
The main limitation of this study was that we did not conduct any power analysis to calculate the sample size selected for this study.
Conclusion
Sacubitril/valsartan combination increased the risk of development of AD in control healthy rats and increased progression of the disease in a rat model of AD induced by aluminum chloride compared with valsartan alone, which decreased the risk of development and progression of AD in control healthy rats and a rat model of AD, respectively. This is evidenced by a significant decline in behavioral tests, a significant increase of MDA and Aβ1-42 levels, and a significant increase in the mean number of pyknotic nuclei of neurons in the pyramidal layer of the hippocampus. Despite the fact that pre-clinical and clinical studies of the sacubitril/valsartan combination showed promising safety results, these results were criticized due to the short follow-up period and the inclusion of healthy, young volunteers. Therefore, additional research is required to determine the safety of chronic use of the sacubitril/valsartan combination, particularly in high-risk patients like those with mild cognitive impairment. Due to the duration of time required for the formation of amyloid deposits, post-marketing phase IV clinical evaluation and prolonged follow-up of large trials may be required for a comprehensive analysis.
Supplemental Material
Supplemental Material - Study of the possible effect of sacubitril/valsartan combination versus valsartan on the cognitive function in Alzheimer’s disease model in rats
Supplementary Material for Study of the possible effect of sacubitril/valsartan combination versus valsartan on the cognitive function in Alzheimer’s disease model in rats by Abdallah Salah El-din Hussein, Rahma K Abou El-Nour, Omayma A Khorshid, and Afaf Sayed Osman in International Journal of Immunopathology and Pharmacology.
Supplemental Material
Supplemental Material - Study of the possible effect of sacubitril/valsartan combination versus valsartan on the cognitive function in Alzheimer’s disease model in rats
Supplementary Material for Study of the possible effect of sacubitril/valsartan combination versus valsartan on the cognitive function in Alzheimer’s disease model in rats by Abdallah Salah El-din Hussein, Rahma K Abou El-Nour, Omayma A Khorshid, and Afaf Sayed Osman in International Journal of Immunopathology and Pharmacology.
Footnotes
Acknowledgements
The authors acknowledge the support of the Medical Pharmacology Department research team members for their help throughout this work.
Author contributions
All authors contributed to the design and interpretation of the data; AS, AO performed analysis, AO wrote the paper, OK revised it, and RK performed the histopathology. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Cairo university’s Institutional Animal Care and use committee (IACUC), medical sciences sector. The experiments were carried out in accordance with the ethical guidelines of the animal welfare application number (CU/III/F/28/19). Animals were handled according to the International Guidelines for Care and Handling of Experimental Animals.
Data availability
All data and materials are available and can be submitted when needed.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

