Abstract
Objective
The haematopoietic cytopenia (HC) of the cyclin-dependent kinase (CDK)4/6 inhibitors was evaluated using the Food and Drug Administration Adverse Event Reporting System (FAERS).
Method
Data from 1 January 2015 to 31 December 2021 has been retrieved from the FAERS database. Disproportionality analysis and Bayesian analysis were utilized in the data mining. The reporting odds ratio (ROR) with 95% confidence interval (CI) for HC was calculated for each CDK 4/6 inhibitor agent. Clinical features of the patients were collected and compared between death outcome and non-death outcome groups. Time to onset (TTO), proportion of deaths, life-threatening and hospitalizations of CDK 4/6 inhibitors-associated HC were also studied.
Results
A total of 17,235 cases of HC associated with CDK 4/6 inhibitors were identified with a median age of 65 years (interquartile range [IQR] 57–73). Palbociclib appeared the strongest signal, with the highest (ROR 9.64, 95% CI 9.46–9.83), followed by ribociclib (ROR 6.38, 95% CI 6.04–6.73) and then abemaciclib (ROR 2.72, 95% CI 2.49–2.97). Patients aged 18–64 had a higher proportion of deaths than those aged 65–84 (12.21% vs. 9.91%, p = 0.001). In Africa and Asia, the proportions of deaths were higher (31.65% and 26.13%, respectively). The median TTO was 26 days (IQR 14–65) for abemaciclib, 33 days (IQR 15–134) for palbociclib and 23 days (IQR 14–69) for ribociclib, respectively. The highest proportion of deaths, life-threatening and hospitalizations all occurred in abemaciclib (13.00%, 5.42% and 44.04%, respectively).
Conclusions
Greater proportions of deaths occurred in Africa and Asia. HC may occur early in any CDK 4/6 inhibitor regimen. Abemaciclib had the highest proportion of deaths, life-threatening and hospitalizations. Health care workers should be more concerned about CDK 4/6 inhibitors. The higher proportions of serious events, including deaths, from Africa and Asia, as well as for abemaciclib, deserve further investigations through additional pharmacoepidemiological approaches.
Keywords
Introduction
Breast cancer is the most common cancer and the leading cause of death associated with cancer in women worldwide. 1 The majority of patients, about 70%, are positive for hormone receptor (HR) and negative for human epidermal growth factor receptor (HER)2. 2 Cyclin-dependent kinase (CDK)4/6 inhibitors – palbociclib, abemaciclib and ribociclib – were approved for patients with advanced HR-positive, HER 2-negative breast cancer, based on positive findings from several randomized controlled trials (RCTs).3–5 From a pharmacological perspective, there are similarities and differences in pharmacodynamics and pharmacokinetics. 6 The most important impact on the clinical management of the disease is haematological toxicity. Grade 3 to 4 neutropenia levels associated with CDK 4/6 inhibitors reported in RCTs were highest with palbociclib (54–66%, PALOMA-2 and 3 trials) and ribociclib (27–59%, MONALEESA-2 trial) and lowest with abemaciclib (21–27%, MONARCH 2 and 3).3,4,7–9 Severe grade 4 neutropenia can cause bacterial infections and become febrile neutropenia, which can lead to death.
For treatment decisions for both clinicians and patients, comparative toxicity profiles can help guide the choice of one drug over another. Although there are several studies concerning the CDK 4/6 inhibitor-associated haematopoietic cytopenia (HC), some evidence was obtained from clinical trials3,4,7–11 and the other evidence was the meta-analysis of these trials,12–15 through which the detailed safety profile regarding HC associated with CDK 4/6 inhibitors was not fully investigated. For example, only two clinical trials (PALOMA 1 and PALOMA 3)7,16 reported the time to onset (TTO) of neutropenia and only two trials (MONARCH 2 and MONARCH 3)4,8 indicated the TTO of grade ≥ 3 neutropenia. In addition, clinical trial data with strict inclusion criteria and cohorts with restricted sample sizes may not properly reflect the real world. It is therefore important to update our understanding and provide an overview of the risks and characteristics of HC after CDK 4/6 inhibitor treatment for further prevention and management.
As a result, we sought to assess and compare the relationship between different CDK 4/6 inhibitors and HC in a large population through investigation of the Food and Drug Administration’s Adverse Event Reporting System (FAERS) so far. We also examined the onset time and the proportion of deaths, life-threatening and hospitalizations for HC of various CDK 4/6 inhibitor regimens.
Methods
Data source
We performed a retrospective pharmacovigilance study using the database of the FAERS. 17 The FAERS database collects adverse event reports from health professionals, patients and manufacturers not only in the United States, but in other areas as well. The data are publicly accessible. For this study, CDK 4/6 inhibitor data were collected in the FAERS database between the first quarter of 2015 (Q1) and the fourth quarter of 2021 (Q4).
Procedures
Haematopoietic cytopenia was obtained from the REAC files according to Medical Dictionary for Regulatory Activities (MedDRA, version 23.0) at the Preferred Term (PT) level based on HCs for Standardized MedDRA Query (SMQ) [20,000,027]. 18 Haematopoietic cytopenias (SMQ) are composed of four subgroups: HC affecting more than one type of blood cell [20,000,028], Haematopoietic erythropenia [20,000,029], Haematopoietic leukopenia [20,000,030] and Haematopoietic thrombocytopenia [20,000,031].
Cyclin-dependent kinase 4/6 inhibitors (abemaciclib, palbociclib and ribociclib) were defined as generic names in the DRUG file and selected role_cod as ‘PS’ (primary suspect). As directed by the FDA, the deduplication procedure selects the last FDA_DT when the CASEID is identical and selects PRIMARY_ID with a greater value When CASEID and FDA_DT are identical (Figure 1).
17
The HC associated with CDK 4/6 inhibitors plus endocrine agents, aromatase inhibitors (AI, including Anastrozole, Letrozole and Exemestane)or fulvestrant, were compared to single endocrine agents. The inclusion and exclusion criteria were provided in Table S1 of the supplemental file. A spreadsheet was used for data collection and the format was presented in Table S2. Descriptive analyses were used to synthesize the clinical features of the patients with CDK 4/6 inhibitors-associated HC collected from the FAERS database. We also estimated the TTO and HC outcomes for various CDK 4/6 inhibitors. Process of selecting cases of CDK 4/6 inhibitors-associated HC from the FAERS database. DEMO: demographic and administrative information; FAERS: Food and Drug Administration Adverse Event Reporting System; HC: Haematopoietic cytopenia.
Statistical analysis
Summary of major algorithms used for signal detection.

BCPNN, Bayesian confidence propagation neural network; CI, confidence interval; IC, information component; IC025, the lower limit of the 95% two-sided CI of the IC; N, the number of co-occurrences; ROR, reporting odds ratio; ROR025, the lower limit of the 95% two-sided CI of the ROR; *a: number of reports containing both the suspect drug and the suspect adverse drug reaction; b: number of reports containing both the suspect drug and other adverse drug reactions (except the event of interest); c: number of reports containing both other medications (except the drug of interest) and the suspect adverse drug reaction; d: number of reports containing other medications and other adverse drug reactions.
The chi-square test was used for comparisons among groups of categorical variables and the Kruskal–Wallis test to compare several independent samples, respectively. 19 Statistical significance was found to be p < 0.05 with 95% confidence ranges. All analyses were carried out using the SPSS statistical software (version 22.0).
Results
Descriptive analysis
Characteristics of patients with adverse events of haematopoietic cytopenia associated with CDK 4/6 inhibitors in the FAERS database (1 January 2015 to 31 December 2021).
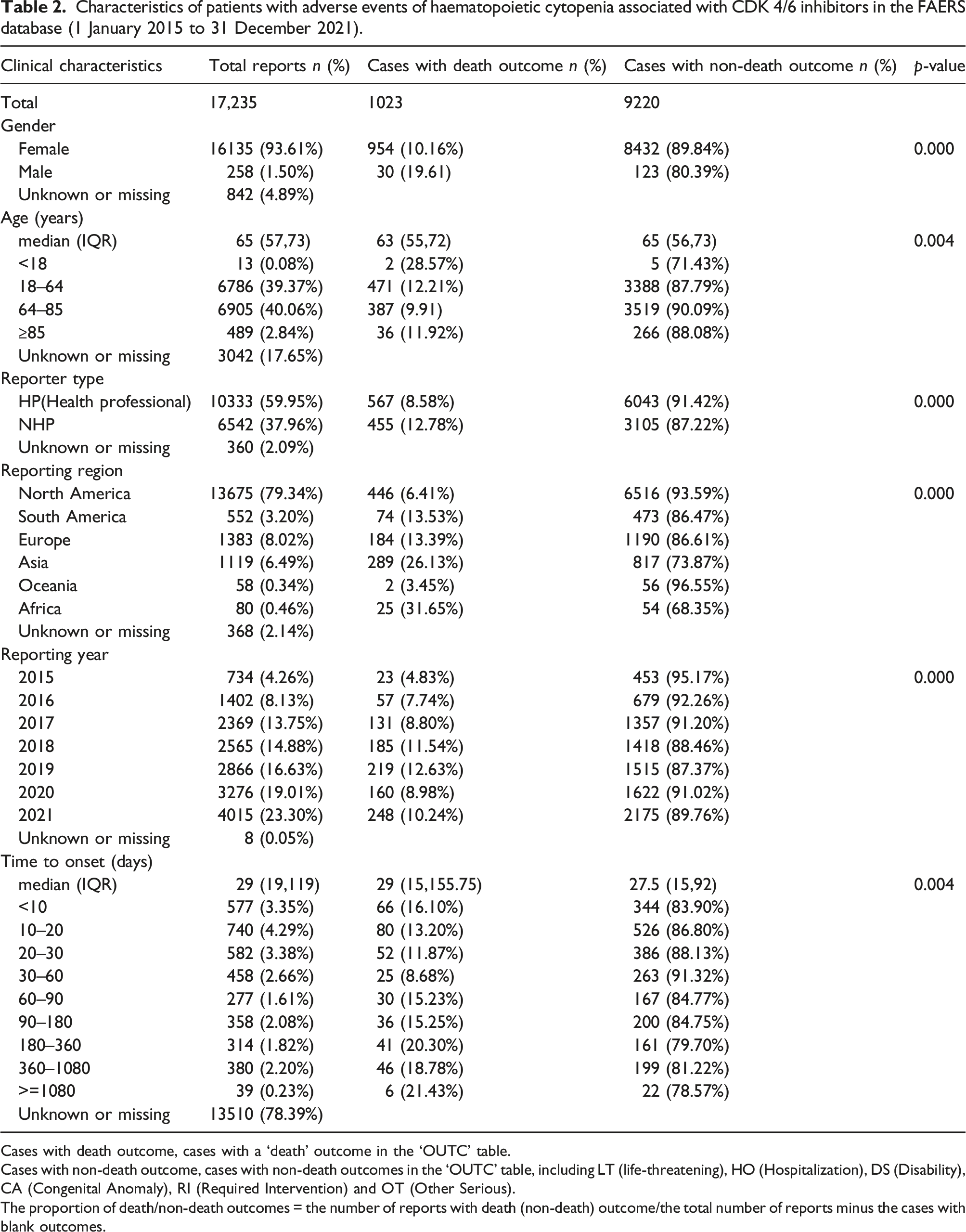
Cases with death outcome, cases with a ‘death’ outcome in the ‘OUTC’ table.
Cases with non-death outcome, cases with non-death outcomes in the ‘OUTC’ table, including LT (life-threatening), HO (Hospitalization), DS (Disability), CA (Congenital Anomaly), RI (Required Intervention) and OT (Other Serious).
The proportion of death/non-death outcomes = the number of reports with death (non-death) outcome/the total number of reports minus the cases with blank outcomes.
Signal values associated with total CDK 4/6 inhibitors and combined therapy (4/6 CDK inhibitors plus endocrines)
Associations of HC with different CDK 4/6 inhibitors and combined therapy (CDK 4/6 inhibitors plus endocrine agents).

Polytherapy 1, CDK 4/6 inhibitors plus AI.
Polytherapy 2, CDK 4/6 inhibitors plus fulvestrant.
AI, including Anastrozole, Letrozole and Exemestane.
a, number of reports containing both the suspect drug and HC; b, number of reports containing both the suspect drug and all other adverse events (except HC); c: number of reports containing both other medications (except the drug of interest) and HC; d: number of reports containing other medications (except the drug of interest) and all other adverse events (except HC).
HC, haematopoietic cytopenia; ROR, reporting odds ratio; IC, information component; 95% CI, the 95% two-sided confidence interval.
Association between CDK 4/6 inhibitors and four subgroups of HC
IC values between CDK 4/6 inhibitors and four subgroups of HC.

Analysis of cases with death and non-death outcome
As shown in Table 2, significant differences were noted with respect to gender, age, reporter type and year of reporting for cases with death outcome versus non-death outcome. Men had a higher proportion of deaths than women (19.61% against 10.16%). The age group 18–64 had a higher proportion of deaths than the age group 65–84 (12.21% vs. 9.91%, Pearson’s chi-square test, p = 0.001). The median time to event onset was 29 days (IQR 15–155.75) and 27.5 days (IQR 15–92) for cases with death outcome and non-death outcome, respectively. Time to onset of 30–60 days (8.68%) had fewer than 180–360 (20.30%) and 360–1080 (18.78%) proportion of deaths, respectively (Pearson’s chi-square test, p = 0.000 and p = 0.001, respectively). Furthermore, there was a statistical difference between cases with death outcome and non-death outcome in the reporting region (p < 0.001), with the highest percentage of deaths (31.65%, 25/79) recorded in Africa.
Time to onset of CDK 4/6 inhibitors-associated HC
Overall, the median time to the onset of 4/6 CDK inhibitors was 29 days (interquartile range [IQR] 19–119). We described the TTOs of HC for each CDK 4/6 inhibitors in Figure 2. The median TTO was 26 days (IQR 14–65) for abemaciclib, 33 days (IQR 15–134) for palbociclib and 23 days (IQR 14–69) for ribociclib, respectively. Interestingly, we found that HC could occur early in all CDK 4/6 inhibitor regimens. For palbociclib and ribociclib, 18.30% and 25.89% of cases developed HC 10–20 days after treatment, respectively. However, 21.17% of cases developed HC within 10 days after taking abemaciclib. Time to onset of adverse effects of haematopoietic cytopenia from CDK 4/6 inhibitors.
Death, life-threatening and hospitalization due to CDK 4/6 inhibitors-associated HC
To analyse the prognosis of CDK 4/6 inhibitor-associated HC, we assessed the proportion of deaths, life-threatening and hospitalizations. Due to HC following different CDK 4/6 inhibitor treatments and showed the results in Figure 3. Haematopoietic cytopenia associated with 4/6 CDK inhibitors generally gave results with the proportion of deaths, life-threatening and hospitalizations of 9.96%, 1.58% and 22.31%, respectively. The proportion of hospitalizations for abmeciclib-associated HC patients was higher (44.04%), compared with that of palbociclib and ribociclib (Pearson chi-square test for group comparison, p < 0.0167 for group comparison) Abmeciclib and ribocicllib all obtained higher proportion of life-threatening than palbociclib (Pearson Chi Square Test, p < 0.0167 for group comparison). Number of reports, proportion of deaths, life-threatening and hospitalizations for CDK 4/6 inhibitors-associated HC.
Discussion
To our knowledge, this study is the first and largest collection so far to compare the occurrences, clinical features and prognosis of HC after the therapy of different CDK 4/6 inhibitors in the real world according to the FAERS pharmacovigilance database. The 3 CDK 4/6 inhibitors in this study showed an association with HC, and the various features of the different regimens were identified.
We included 17,235 cases of CDK 4/6 inhibitors associated with HC, the largest number of such cases so far. Our study found a significant signal between all CDK 4/6 inhibitors treatment and HC. A meta-analysis of six RCT showed an increased risk of all grade haematological adverse effects in patients receiving CDK 4/6 inhibitors, while no significant difference was found in subgroup (palbociclib vs. ribociclib vs. abemaciclib). 14 Another meta-analysis of eight RCTs found higher haematologic toxicity in the palbociclib and ribociclib groups compared to the abemaciclib group. 15 The Grade 3–4 neutropenia frequencies associated with CDK 4/6 inhibitors reported in RCTs were highest with palbociclib and lowest with abemaciclib. 20 The results of the subgroup analysis conducted in this analysis were interesting and revealed that CDK 4/6 inhibitors-associated HC were most relevant to palbociclib, followed by ribociclib and then abemaciclib (palbociclib vs. ribociclib ROR: 1.47 [1.39–1.55] and ribociclib vs. abemaciclib 2.34 [2.11–2.59], respectively). In addition, all CDK 4/6 inhibitors were associated with HC in all four subgroups. Palbociclib obtained the significant signal in the haemotopoietic leukopopenia subgroup (IC: 3.52 [3.45–3.60]), followed by ribociclib (ROR: 3.08 [2.90–3.27]) and abemaciclib (ROR: 1.68 [1.50–1.89]). Palbociclib and ribociclib both got more signals in all subgroups compared to abemaciclib. Moreover, the haematopoietic leukopenia subgroup got the strongest signal than the other three subgroups. Due to the influence of bone marrow, the main toxicities associated with CDK 4/6 inhibitor are neutropenia and leucopenia, the most commonly found grade 3/4 in clinical trials. Anaemia or thrombocytocytopenia is less common.3,9,16,21
From the perspective of pharmacodynamics, each compound exhibits a different potency in the activity against CDK4 or CDK6 in enzyme testing. Palbociclib has similar activity for CDK4 and CDK6 (CDK4 IC50 = 11 nmol/L vs. CDK6 IC50 = 16 nmol/L), while ribociclib power is greater for CDK4 than for CDK6 (CDK4 IC50 = 10 nmol/L vs. CDK6 IC50 = 39 nmol/L). Abemaciclib is the strongest inhibitor, in particular to CDK4 (CDK4 IC50 = 2 nmol/L vs. CDK6 IC50 = 10 nmol/L). 22 Abemaciclib shows a greatest selectivity for CDK4 compared to CDK6. Cyclin-dependent kinase 4 is particularly important for breast tumours, whereas CDK6 plays a crucial role in differentiating haematopoietic stem cells. 23
Our study showed that CDK 4/6 inhibitors combined with endocrine therapy (ET) got significant signals compared to AI or fulvestrant. This conclusion is consistent with earlier studies.9,11,24,25 The network meta-analysis indicated that CDK 4/6s plus AI represented a better option compared with fulvestrant. 26 Our study showed CDK 4/6 inhibitors plus AI got an insignificant signal compared with fulvestrant (ROR: 1.03 [0.98–1.08]). The meta-analysis also showed similar results that no significant differences in subgroup were found between AI and fulvestrant backbones amongst haematological comparing ribociclib and abemaciclib with palbociclib. 12
The reports in age group 18–64 and group 65–84 were quite similar (39.37% vs 40.06%), while the former got a higher proportion of death outcome than the latter (12.21% vs 9.91%, p = 0.001), which may be due to febrile neutropenia was rare in older patients. 27 The proportions of death outcome are high in Africa and Asia (31.65% and 26.13%, respectively). On the other hand, North America has the highest number of reports (79.34%) with low proportion of death outcome (6.41%). All haematological adverse events can usually be adequately managed through standard supportive care. The timing of sufficient monitoring and clear communication among patients is important, just as a therapist is required to minimize misunderstandings. 23 For Asia, despite the lack of evidence of a difference in toxicity based on race or ethnicity in all Phase III tests involving the three CDK 4/6 inhibitors. 23 A race-defined clinical prediction tool was able to distinguish subgroups with significantly different risks of grade 3 neutropenia after abemaciclib initiation. 28
We found that HC could potentially occur at the initial stage of all CDK 4/6 inhibitor regimens. The median TTO was 26 days (IQR 14–65) for abemaciclib, 33 days (IQR 15–134) for palbociclib and 23 days (IQR 14–69) for ribociclib, respectively. The median time of neutropenia, regardless of grade, is from 15 to 20 days. 14 For palbociclib and ribociclib, 18.30% and 25.89% of patients developed HC 10–20 days after treatment, respectively, which is consistent with the prior researchs that neutropenia usually emerged 15 days after the first dose of palbociclib and ribociclib.7,21 For abemaciclib, 21.17% of patients developed HC within 10 days after taking drug. This was very different from the data from the abemaciclib clinical trial, which showed that all levels of neutropenia were generally observed at cycle two (28-day cycle). 4 The results suggest that clinicians should monitor the blood count of abemaciclib more quickly and earlier.
To our great surprise, abemaciclib got the highest proportion of hospitalizations (significant difference compared with ribociclib or palbociclib, p < 0.0167), proportion of life-threatening (significant difference compared with palbociclib, p < 0.0167) and proportion of deaths (unsignificant difference compared with ribociclib or palbociclib) in all CDK 4/6 inhibitors. As mentioned above, because of the higher CDK4 selectivity of abemaciclib, it has a 50% lower neutropenia rate (all grades) than palbociclib and ribociclib. 4 While abemaciclib was associated with a three-fold statistically significant probability of treatment discontinuation due to an adverse event when compared to palbociclib and no differences was observed in treatment discontinuation between ribociclib and palbociclib. 12 The outcome of HC for abemaciclib is severe, although it had the weakest signal.
Neutropenia associated with CDK 4/6 inhibitor is less serious than that of chemotherapy patients: there is no related pancytopenia and infection rates are low.3,7,21 In previous studies, less than 10% of patients developed Grade 4 neutropenia, and low levels of febrile neutropenia were reported for palbociclib (in 2.5%, 0.9% of patients in the PALOMA-2, PALOMA-3 trials, respectively) and ribociclib (in 1.5% of patients in the MONALEESA-2 trial).3,7,9 Only one patient experienced nonserious febrile neutropenia in the abemaciclib arm in the Monarch 3 trial. 4 Our study showed similar results in that significant signals appeared for palbociclib and ribociclib associated with febrile neutropenia, with an insignificant signal of abemaciclib. Overall, most cases treated with palbociclib, ribociclib and abemaciclib were quickly resolved through interruptions or reductions.3,7,9 In previous studies, we believe that although the incidence of HC in CDK 4/6 inhibitor treatment is high, the risk is low, it is not necessary to use the granulocyte-colony stimulating factor (GCSF). 23 But the data from our real-world analysis showed the situation is not so optimistic, all the health professionals should strengthen the monitoring of HC for CDK 4/6 inhibitors.
Special attention should be given to these toxicities, which can be fatal. Precise patient monitoring and managing side effects are crucial. 15 All haematological adverse events can generally be handled properly by standard supportive care. Every breast cancer center has to find its own strategy to take care of the patient and a correct treatment adjustment, such as informational flyers, patient follow-up calls and visits in the center. 23 Patients who develop grade 2 neutropenia while on treatment are more likely to develop hyperenergetic neutropenia and febrile neutropenia. They therefore need to be carefully monitored complete blood count (CBC) throughout treatment with CDK 4/6 inhibitors. 17
There are several limitations to our research. First, FAERS is a spontaneous reporting system (SRS) which is used for qualitative research and does not allow for comparing drug safety, assessing associations, calculating incidence because measures of disproportionality do not include incidence denominators. The data collected cannot be used to quantify the adverse reaction signals based on the total number of adverse reactions. 29 Signal intensity between a given drug and reaction was used only as a qualitative indicator. Second, the data available from the SRS are not as reliable as those from clinical trials and cohort studies and the identification and reporting of adverse events are not strictly controlled. It is difficult to identify significant risk factors between CDK 4/6 inhibitors and HC, since the deficiency of baseline CBC, pre-existing haematological diseases and comorbidities that may have impacts on CBC. Third, data deleted from the deleted file, followed by a question, exists because the reports may contain different but overlapping CASEID data. Only a limited number of reports were identified duplicated. However, when we attempted to remove overlaps in three or four out of four fields (event_dt, age, sex, reporter_country), a large portion of the reports would be lost. The deduplication method deserves further study. Fourth, North America accounted for the majority of the reported data (79.34%) and only 80 cases were reported in Africa. There may be considerable bias since reporting is limited and statements are undermined. Fifth, the calculation, justification and power analysis of the sample size selected in this study were not performed because all eligible adverse drug reactions will be included. Finally, during the data mining process, we noted the imperfection of the reporting of information, such as incorrect entries and incomplete reports, which can result in bias in the analysis. Although there is some hereditary limitation in the FAERS database, it points out some critical aspects of CDK 4/6 inhibitors-associated HC, providing clues for further research well designed to validate the results.
Conclusion
Based on the FAERS database, we profiled HC related to various CDK 4/6 inhibitors with more details on occurrences, clinical characteristics and prognosis. The proportions of death outcome were higher in Africa and Asia. HC could occur early in all CDK 4/6 inhibitor regimens. Abemaciclib got the highest proportion of death, life-threatening and hospitalization outcomes. More concerns should be paid for CDK 4/6 inhibitors when applied to patients with a tendency for HC. The higher proportions of serious events, including deaths, from Africa and Asia, as well as for abemaciclib, deserve further investigations through additional pharmacoepidemiological approaches.
Supplemental Material
Supplemental Material - Haematopoietic cytopenia associated with cyclin-dependent kinase 4/6 inhibitors: A real-world study of data from the food and drug administration adverse event reporting system database
Supplemental Material for Haematopoietic cytopenia associated with cyclin-dependent kinase 4/6 inhibitors: A real-world study of data from the food and drug administration adverse event reporting system database by Xiayang Ren, Cilin Yan, Le Tian and Xiangli Cui in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgements
I would particularly like to acknowledge my research partner, Cilin Yan, was instrumental in defining the path of my research. I also thank Ye Xu for her suggestion on the chart format.
Authors’ contributions
Xiayang Ren drafted the manuscript. All authors participated in data analysis and interpretation, manuscript revision, and final approval of the submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and material
The data that support the findings of this study are available upon reasonable request.
Ethics approval
Our data came from the FAERS database, which was free and publicly accessible. All published articles using FAERS have not yet been approved by the Ethics Committee.
Informed consent
Written informed consent for this study was not required as we used the publicly available FAERS database. Currently, all published articles using FAERS are not required to have informed consent.
Trial registration
Not applicable.
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


