Abstract
Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection may present with some systemic lupus erythematosus (SLE) manifestations intermingled with Kawasaki disease features. These emerging presentations were dubbed under the umbrella term ‘multisystem inflammatory syndrome in children (MIS-C)’. A one and half-year-old girl, admitted to Mansoura University Children’s Hospital (MUCH) with fever, bad general condition, vomiting, widespread maculopapular, vasculitic rash, hands and feet oedema, oral ulceration, arthralgia and lymphadenopathy. Moreover, bicytopenia, positive antinuclear, anti–double-stranded DNA antibodies and low C3 qualified her as a case of juvenile SLE. Despite the child received the initial therapy of immunosuppressive medication, her general condition deteriorated with fever persistence and rash exacerbation. At that time, the skin of her hands and feet started to peel. Thus, an expanded study for other alternatives was obligatory; SARS-CoV-2 infection testing revealed positive IgG serology, and retesting for lupus autoantibodies turned negative. HRCT chest showed bilateral basal consolidation with ground-glass appearance. Furthermore, Echo exhibited coronary artery dilation with thrombus inside. This evolution raised the concern for COVID-related MIS-C syndrome. This report provides a model of COVID-19 heterogeneity with protean immune-related manifestations. This case has a unique presentation that necessities its description, in order to provide a nidus for future studies in this new entity.
Keywords
Introduction
According to the centre for disease control and prevention (CDC), multisystem inflammatory syndrome in children (MIS-C) is defined as an individual < 21 years with fever, laboratory evidence of inflammation, with multisystem (>2) organ involvement (cardiac, mucocutaneous, respiratory and haematologic) and no alternative diagnoses. In addition to positive tests for severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection by polymerase chain reactant (PCR), serology, antigen test or COVID-19 exposure within the 4 weeks prior to the onset of symptoms. 1
MIS-C is an immune activation syndrome but not an acute infectious process, and patients do not currently have active infection. Despite, the SARS-CoV-2 infection is predominantly associated with respiratory tract inflammation; some patients can develop an abnormal inflammatory or autoimmune response involving extra-pulmonary tissues. The manifestations associated with such immune reactions are heterogeneous and can resemble some systemic autoimmune diseases. 2
Some patients may exhibit features of hyperinflammatory state mimicking Kawasaki disease (KD), macrophage activation syndrome (MAS), antiphospholipid syndrome and shock. The release of large amounts of cytokines results in systemic inflammation, which is characterized by fever, tachycardia, tachypnoea and hypotension. In addition to elevated C-reactive protein, hypercoagulation and hyperferritinaemia. 3 The coronary arteries involvement in MIS-C implies a close link with KD, and the frequency of coronary aneurysms reported with MIS-C is 8–23%. 4
SARS-CoV-2 infection may present with some systemic lupus erythematosus (SLE) manifestations including, arthralgia, serositis, chilblain lesions or antiphospholipid antibodies. Haematological involvement that is frequent dyscrasia with SLE was also documented with COVID-19 infection as lymphopenia, thrombocytopaenia or haemolytic anaemia. Furthermore, they can present with immune thrombocytopaenia (ITP) and thrombotic thrombocytopenic purpura as well. 2
Case presentation
Herein, we present a tragic case of SARS-CoV-2 infection and new-onset SLE manifestations concomitant with severe systemic hyperinflammation (MIS-C) and coronary artery dilation with thrombus formation. All data is displayed, after obtaining her parent’s written consent.
A one and half-year-old girl who is one of twin pregnancy. At the age of 9 months, she was diagnosed with secundum ASD and an oral diuretic was prescribed for her. In August 2021, she was admitted to Mansoura University Children’s Hospital (MUCH) with a history of fever and bad general condition associated with acute skin eruption of erythematous rash all over the body and her face, joint pain, vomiting and poor appetite. Her mother denied any respiratory, cardiovascular, urinary complaints or any history of contact with a COVID-19 patient.
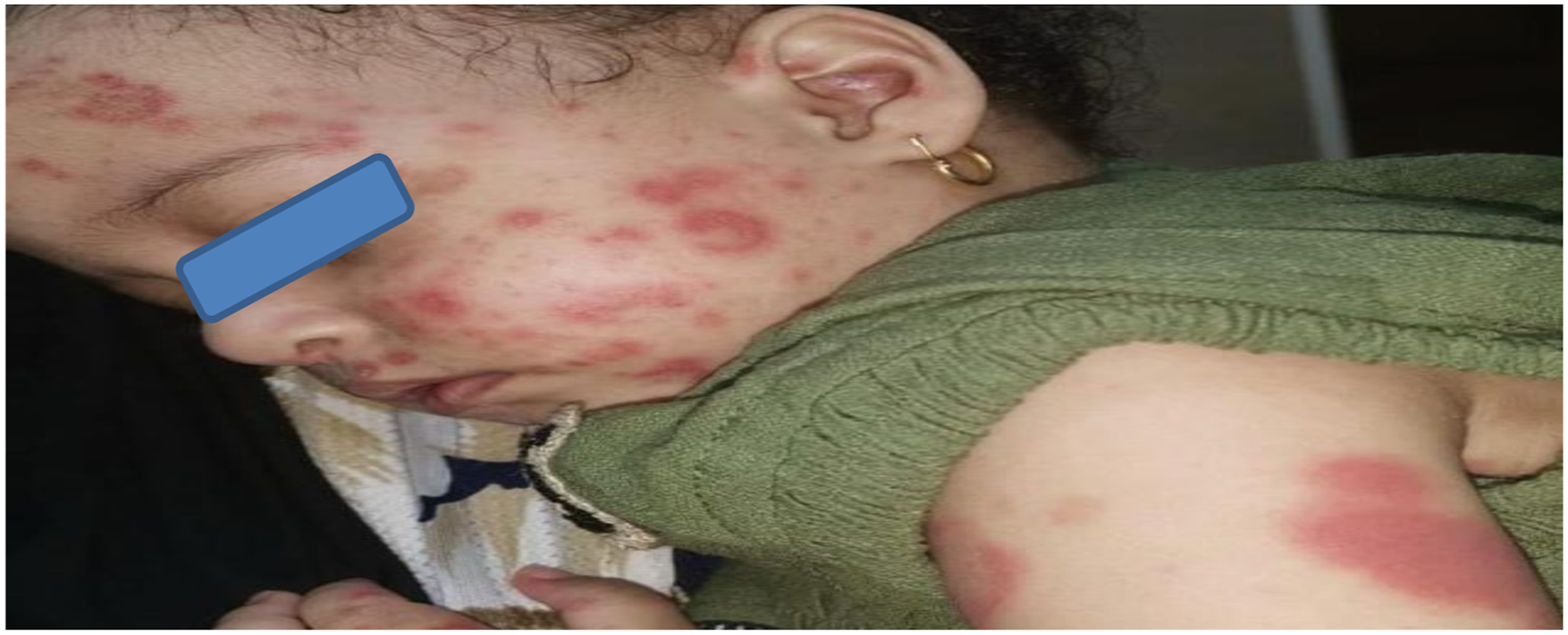
Thorough clinical examination revealed a confused girl with a GCS of 13, oxygen saturation was 98% at room temperature. Her heart and respiratory rates were 100 bpm and 20 breaths per minute, respectively, the temperature was 39°C, and her blood pressure was 110/70 mmHg. The weight and height were within normal percentiles. However, there was pallor and lymphadenopathy without jaundice or cyanosis. Normal systemic examination including cardiac and respiratory auscultation. Mucocutaneous examination showed oral ulceration, an erythematous-maculopapular rash over her face and body with areas of hyperpigmentation, vasculitic rash and oedema of both hands and feet (Figures 1 and 2). Vasculitic rash at toes tips. Erythematous-maculopapular rash over the face and left upper.
Laboratory finding of reported case.

aabnormal values.
CBC: Complete blood count, RBCs: red blood cells, HBG: haemoglobin, HCT: haematocrit, MCV: mean corpuscular volume, MCHC: mean corpuscular volume concentration, WBCs: white blood cells, PLT: platelets, ESR: erythrocyte sedimentation rate, CRP: c reactive protein, AST: aspartate aminotransferase, ALT: alanine aminotransferase, ANA: antinuclear antibody, Anti-dsDNA: Anti-double strand DNA, C3: complement3, CK: creatine kinase, EBV: Epstein-Barr virus, CMV: cytomegalovirus, HCV: hepatitis c virus, CD: cluster differentiation, Ig: immunoglobulin
Her imaging detected multiple enlarged bilateral cervical and axillary lymph nodes by superficial ultrasound. Abdominal ultrasound depicted average sized liver, gallbladder, spleen without any focal lesions, patent portal vein, normal size, echo-pattern, parenchymal thickness of both kidneys, no enlarged para-aortic lymph nodes, adnexal masses and no pelvic free fluid. Additionally, her MRI brain was normal.
Fever, diffuse maculopapular, vasculitic rash, arthralgia, oral ulceration, bicytopenia (thrombocytopaenia, leukopenia and neutropenia), positive antinuclear (ANA), anti-double stranded deoxyribonucleic acid (anti-dsDNA) antibodies, low complement (C3) level. Moreover, negative urine, blood cultures, negative viral serology and normocelluler bone marrow aspirate favour her as a case of juvenile SLE according to the SLE European League Against Rheumatism/American College of Rheumatology (Euler/ACR) criteria by 27 points. 5
Despite of receiving oral steroids, hydroxychloroquine and azathioprine, her general condition deteriorated with fever persistence and rash exacerbation. At that time, the skin of her hands and feet started to peel. Thus, more expanded research for an alternative diagnosis was obligatory especially with C reactive protein shooting to 180 mg/L.
Testing for SARS-CoV-2 infection revealed positive IgG serology with negative PCR, and the girl was retested for ANA, anti-dsDNA, which came back negative. Non-contrast CT chest showed bilateral basal patches of consolidation with ground-glass appearance and bilateral minimal pleural effusion (Figure 3). Additionally, echocardiography exhibited an ejection fraction of 76%, minimal pericardial effusion, and left main coronary artery dilation (5 mm with a Z score of 7.30) with thrombus inside. Measurements of both Troponin T and interleukin (IL)-6 levels were normal. This evolution raised the concern for new emerging SARS-CoV-2-related MIS-C syndrome. Non-contrast CT chest showed bilateral basal consolidation with faint patches of ground-glass appearance.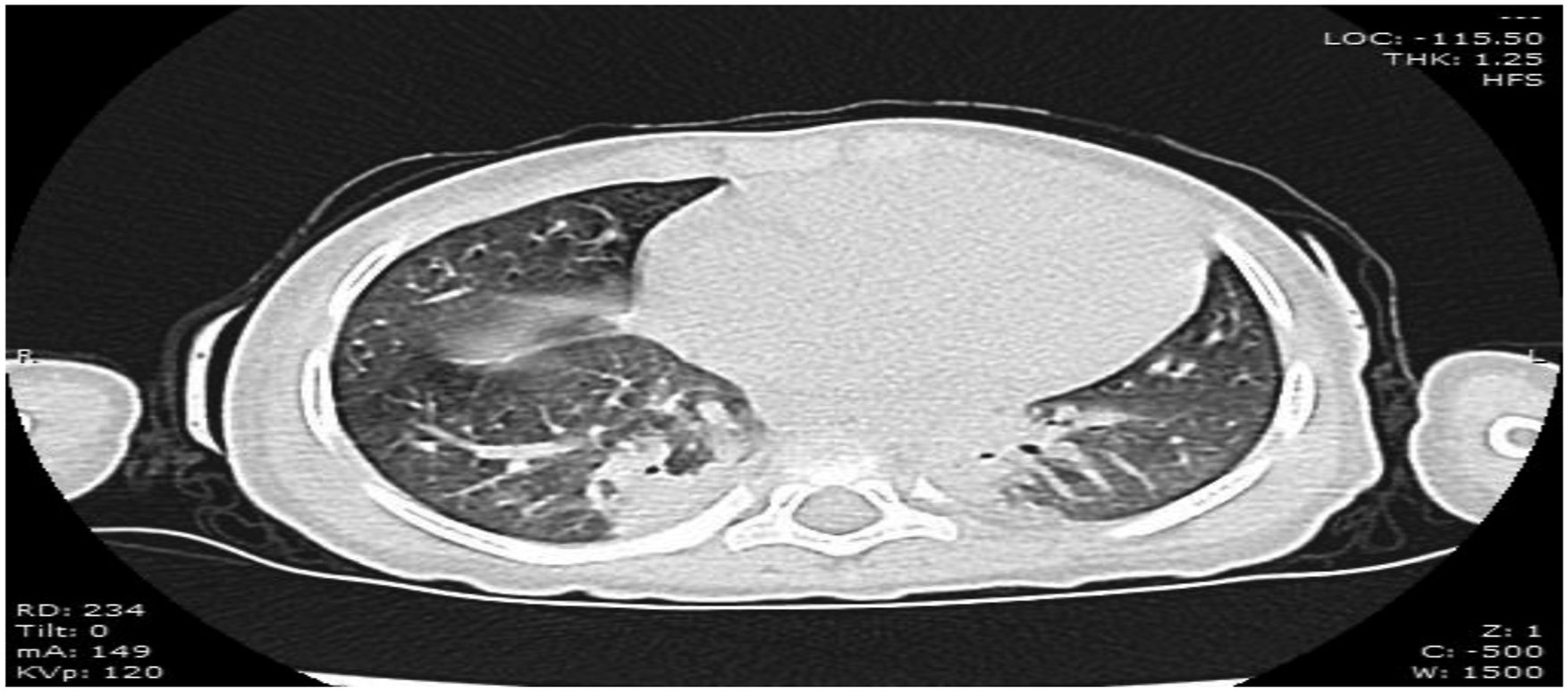
Unfortunately, despite therapy intensification with pulse steroid, IVIG, therapeutic anticoagulant and IV antibiotics, the patient had a severe pulmonary haemorrhage, bleeding from orifices, with coagulation profile prolongation and severe pancytopenia with a high probability of end-stage MAS development before she underwent cardiac arrest and died.
Discussion
SLE is an autoimmune, multisystem disease characterized by systemic inflammation and tissue damage. Viruses have been linked with SLE pathogenesis as Epstein-Barr virus, parvovirus B19, cytomegalovirus, and recently SARS-CoV-2 may work as a trigger of self-tolerance breakdown and lupus development. 6
Little is known about this link, it may be attributed to the involvement of specific acquired immune responses or interferon-related pathways. SARS-CoV-2 can induce a dysregulated cytokine release that leads to high expression of pro-inflammatory cytokines, such as IL-1, IL-6, TNF alpha and T cells shift from Th1 to Th2 similar to autoimmune diseases. 7
In addition, molecular mimicry, polyclonal B cells activation owing to long-term viral antigens persistence, bystander activation, or formation of neutrophil extracellular traps may be underlying smouldering factors for autoimmunity. Autoantibodies positivity may be a transient association with COVID-19 infection as reported previously, antinuclear and antiphospholipid antibodies were detected temporary in 35.6% and 11.1% of COVID-19 cases, respectively. 8
Only eight cases of new-onset SLE triggered by SARS-CoV-2 have been reported, their age ranged between 18 and 85 years, and the interval between COVID-19 diagnosis and SLE was 13–60 days. Lung and kidney involvement was the most common SLE manifestation among those patients. Others presented with jaundice, elevation of liver enzymes, lymphopenia, thrombocytopaenia, positive ANA, anti-dsDNA, anti-Ro, anti-La, P-ANCA and low complement levels. 9
This girl developed some SLE manifestations, like fever, cutaneous disease (diffuse maculopapular rash, purpuric eruptions and vasculitic rashes), haematological abnormalities (thrombocytopaenia, leukopenia and neutropenia), oral ulceration, arthralgia, positive ANA and anti-dsDNA antibodies. With more insult to the injury, there was an overlap with KD in the form of persistent fever, widespread exanthema, hand and feet oedema, fingers desquamation and the disease hallmark of coronary artery aneurysm.
Regardless of the patient initially fulfilled the new Euler/ACR criteria for SLE, the fever persistence, print of coronary artery dilation, gastrointestinal manifestations, negative autoantibodies on repeated testing and poor response to the initial therapy alarmed more expanded tests for the pervasive SARS-CoV-2 infection associated MIS-C. Noteworthy, the presence of these mucocutaneous, cardiac, gastrointestinal, haematological disorders were reported as significant predictors of MIS-C discrimination from other multi-system inflammatory conditions. 10
MIS-C should be managed by a multi-disciplinary team including, paediatric rheumatologists, cardiologists and infectious disease specialists. According to the ACR guidelines for the management of MIS-C, there are various therapeutic approaches that can be used as IVIG, high dose of IL-1 inhibitors, corticosteroids and anticoagulant therapy. 11
Conclusion
This case report provides a model of COVID-19 clinical heterogeneity with protean immune-related manifestations to arouse the medical committee about these emerging features. Further and solid studies are vital to explore the pathogenic pathways, which will help in the prompt detection and proper management of various COVID-19 manifestations.
Footnotes
Acknowledgement
We are thankful to child’s parents for their cooperation.
Author contributions
The authors shared in data collection, the manuscript writing, and all have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent for publication and participation
The patient’s parent provided written consent to participate in this study.
Availability of data and material
All data generated during this study are in this published article.
