Abstract
In the diagnostic of systemic sclerosis (SSc), both anti-centromere (ACA) and anti-topoisomerase I (ATA) antibodies are considered mutually exclusive, though their coexistence has been also reported in some patients. Notably, nintedanib has been approved for the treatment of interstitial lung disease associated to SSc. Herein, we present the clinical case of a 41-year-old woman with SSc who shows an immunological seroconversion (from ACA positivity to a coexistence of ACA and ATA antibodies) together with changes in her clinical phenotype. Besides, the patient responds positively to the treatment of her lung involvement with a combination of immunomodulators and antifibrotic agents.
Keywords
Introduction
Anti-centromere (ACA) and anti-topoisomerase I (ATA) antibodies are diagnostic markers of systemic sclerosis (SSc). 1 Although these autoantibodies are considered to be mutually exclusive, their coexistence has been described in 0.05–5.6% of patients.1–5 On the other hand, nintedanib, an inhibitor of tyrosine kinases, has emerged as a new drug for the treatment of SSc-associated interstitial lung disease (SSc-ILD). 6 This work is intended to present the clinical case of a 41-year-old woman that displays a limited cutaneous SSc (LcSSc) clinical phenotype and ACA antibodies but that undergoes immunological seroconversion, with coexistence of ACA and ATA together with a change in her clinical phenotype. But also, during the course of the disease and due to lung involvement, she responds favourably to the combination use of immunomodulators (rituximab and mycophenolate) and antifibrotic agents (nintedanib).
Case presentation
In 2011, a 41-year-old woman was referred to our centre because of a 1-year history of Raynaud’s phenomenon. The capillaroscopy pattern was compatible with early SSc, and she was positive for antinuclear antibodies (ANA) by Indirect Immunofluorescence (IIF) with a centromere pattern at a titre of 1:320. ACA antibodies were confirmed by fluorescent enzyme immunoassay (FEIA) and immunoblotting (IB). Three years later, the patient developed oesophageal and cutaneous manifestations, compatible with telangiectasias and a modified Rodnan skin score (mRSS) of 2. Consequently, the patient was diagnosed with LcSSc. No other organ affections were found in the initial screening (echocardiogram, lung function tests and high-resolution computed tomography (HRCT) of the lungs were normal). In June 2015, the patient developed dyspnoea on moderate exertion (mMRC 2). An HRCT of the lungs showed changes compatible with non-specific interstitial pneumonia (NSIP). Lung function tests, previously normal, had progressed to a restrictive pattern (forced vital capacity (FVC) 75%, forced expiratory volume in 1 second (FEV1) 73%, diffusing capacity of the lungs for carbon monoxide (DLCO) 45%, and diffusion coefficient (KCO) 71%), with a six-minute’s walk distance (6MWT) of 274 metres, without desaturation. Hence, the patient was prescribed prednisone (initial dose 15 mg/24h, followed by tapering to 5 mg/24h) and mycophenolate (2 g/12h). Given the unexpected fast development and progression of NSIP, further immunological analysis was carried out. The IIF performed revealed that ACA pattern was overlapping with a nucleolar pattern compatible with ATA (Figure 1). Interestingly, the presence of ATA (45 U/ml) was confirmed by FEIA and IB despite the fact that ATA had been previously tested by following the same method in the former samples and it had been negative. In the following months, there was a 13% decrease of DLCO together with a worsening of skin affection (mRSS: 5) and the appearance of digital ischaemic ulcers and flexor tenosynovitis, that until that moment the patient had not presented. Hence, in June 2016, ‘off-label’ rituximab was added, due to worsening pulmonary involvement despite mycophenolate and flexor tenosynovitis. In November 2019, O2 therapy was needed, and ‘off-label’ nintedanib was added, given the worsening of the dyspnoea (mMRC 3) and the progression of areas of fibrosis to a honeycomb pattern on HRCT. Figure 2 shows the sequential plot. Ten months later, the patient reported subjective improvement of dyspnoea (mMRC 2) and, even though she was still under O2 therapy, her HRCT findings were stable and DLCO had increased by 14%. Triple therapy has been maintained until now. On the last visit, in February 2022, the patient had dyspnoea mMRC 2 (lung function tests: FVC 79%, FEV1 78%, DLCO 48% and KCO 77%), persistent cyanosis without digital ulcers and no synovitis. Figure 3 shows a graph of the lung function parameters.

Indirect immunofluorescence image of serum from the patient. The following can be observed: nuclear dots in interphase with typical mitoses in a centromere pattern (arrows) overlapping with a finely speckled pattern and nucleolar dots with weak cytoplasmic fluorescence due to the presence of ATA (asterisks).
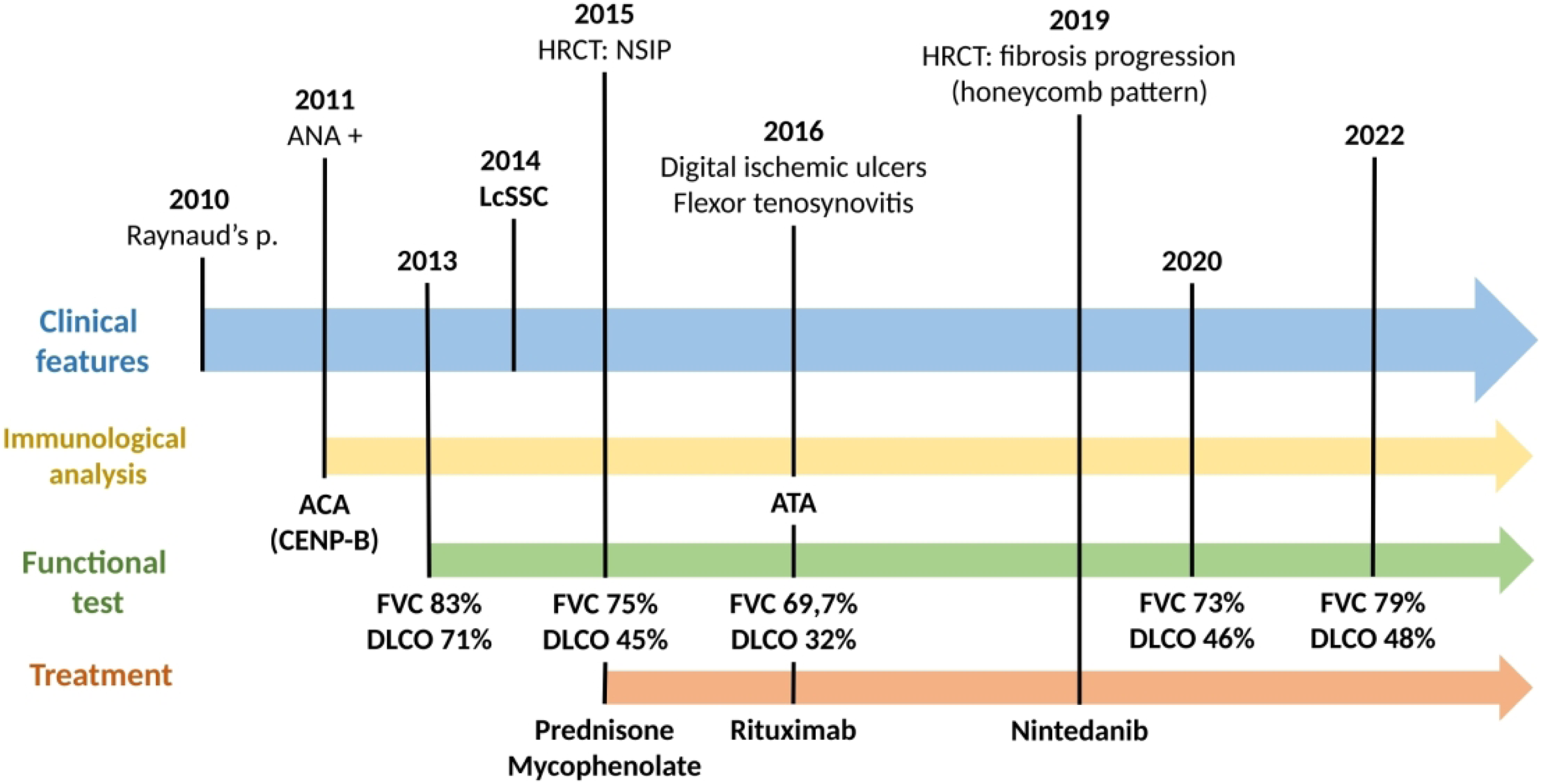
Sequential plot of the clinical and immunological results, the functional lung tests and the treatment of the patient. ANA; antinuclear antibodies, ACA; anti-centromere antibodies, ATA; anti-topoisomerase I antibodies, HRCT; high-resolution computed tomography, LcSSc: limited cutaneous systemic sclerosis, FVC; forced vital capacity, DLCO; diffusing capacity of the lungs for carbon monoxide.

Graph of the lung function parameters. FVC: forced vital capacity, DLCO: diffusing capacity of the lungs for carbon monoxide.
Discussion
In this article, we present an uncommon clinical case report of a patient with LcSSc who shows an immunological seroconversion (from ACA positivity to a coexistence of ACA and ATA antibodies) together with a clinical phenotype change. In addition, the patient displayed a good response to the combination use of immunomodulators (rituximab and mycophenolate) and antifibrotic agents (nintedanib).
The coexistence of ACA and ATA autoantibodies has been described with a prevalence of 0.05–5.6%.1–5,8 These differences depend on the technique used. 1 Mutual masking may occur with IIF when there are high titres of ACA or ATA.2,7,8 In our patient, this phenomenon was ruled out, the results being confirmed using FEIA and IB.
Some authors (Kikuchi M et al.) uphold that the coexistence of ACA and ATA antibodies is associated with severe Raynaud’s phenomenon and oesophageal dysfunction. 2 By contrast, others authors among them (Heijnen IA et al.; Srikamud S. et al.) have reported, as we did, a phenotype that is clinically similar to ATA-positive patients with more severe ILD and musculoskeletal involvement.8,9 In our case, the patient’s clinical phenotype changed coinciding in time with the appearance of ATA. In fact, this immunological seroconversion that we observed has been also described by other authors.1,2,8,9
Finally, it is worth to remark the long time effectiveness and safety of combining mycophenolate and nintedanib with rituximab, data beyond those from the SENSCIS trial. 4 In our case, after triple therapy, the patient experienced an improvement in dyspnoea, as well as in DLCO. In this sense, it is necessary to emphasize that there are a number of sources of variability in DLCO, for example, differences in equipment, testing conditions, patient factors and reference equations. 10 We did not consider repeating the test because the lung function test was going to be repeated over time from then on. At this moment, 26 months after the addition of nintedanib to mycophenolate and rituximab, lung function test still showed a stabilization. Another significant aspect is that the patient received treatment with 15 mg de prednisone, followed by tapering to 5 mg/24h. We need to be acknowledged, that glucocorticoids at dose >15 mg per day are not an appropriate treatment for SSc, due to the risk of triggering a scleroderma renal crisis. 11
Conclusions
Altogether, this clinic case evinces the uncommon coexistence of ACA and ATA in a patient diagnosed with SSc. It is important to consider repeating the assessment of a patient’s immunological status in the event of a change in clinical phenotype. Moreover, antifibrotic drugs are an effective and safe option in patients with SSc-ILD despite treatment with immunosuppressants and glucocorticoids.
Footnotes
Acknowledgements
We would like to thank the Spanish Rheumatology Society for their support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics and consent
This study complies with the Declaration of Helsinki and written informed consent has been obtained from the patient for publication.
