Abstract
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic resulted in more than five hundred million infected cases worldwide. The current study aimed to screen the correlation of different laboratory findings with disease severity and clinical outcomes of coronavirus disease (COVID-19) among Egyptian patients to obtain prognostic indicators of disease severity and outcome.
A total of 112 laboratory-confirmed COVID-19 patients were examined. According to the severity of the disease, these patients were divided into three main groups: mild, moderate and severe cases. In addition, clinical characteristics and laboratory findings, including Hb, platelet count, white blood cell count, lymphocyte percentage, neutrophil percentage, neutrophil lymphocyte ratio (NLR), D-dimer, highly sensitive C-reactive protein (HS-CRP), alanine aminotransferase (ALT), lactate dehydrogenase (LDH) and creatinine, were measured.
The presence of hypertension and/or diabetes was found to be a significant risk factor for disease severity and poor outcome. Increased respiratory rate, levels of SpO2, HS-CRP, D-dimer, NLR, ALT, LDH, lymphopenia and neutrophilia, as well as changes in chest computed tomography (CT), were associated with increased disease severity and fatal consequences. Highly sensitive C-reactive protein, D-dimer, NLR and LDH constituted excellent predictors for both disease severity and death.
Laboratory biomarkers, such as HS-CRP, D-dimer, NLR and LDH, are excellent predictors for both disease severity and death. They can predict mortality in patients at the time of admission secondary to SARS-CoV-2 infection and can help physicians identify high-risk patients before clinical deterioration.
Keywords
Introduction
Coronavirus disease (COVID-19), which is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), was first identified in Wuhan, China, in December 2019 1 . In March 2020, the World Health Organization (WHO) declared a worldwide pandemic. 2 This disease usually starts with flu-like symptoms, and about two-thirds of infected subjects remain asymptomatic3–4 The classical symptoms of the disease include fever, fatigue and cough. 4 In many cases, the disease progresses to severe pneumonia or acute respiratory distress syndrome (SARS), up to multi-organ failure with fatal consequences. 5 On the other hand, some patients might develop severe respiratory distress with fatal consequences, especially elderly patients with more comorbidities, such as hypertension, 6 diabetes, 7 dementia8, 9 and Parkinson disease10, 11 as well as those with immunocompromised disorders 12 and cancer patients. 13
The levels of many biomarkers are increased during the disease and are highly suggestive of the infection, including D-dimer, C-reactive protein (CRP), highly sensitive CRP (HS-CRP) and high-density lipoprotein.3,12,14–17 In addition, the pathological findings of chest computed tomography (CT) exhibit good consistency, and their combination can reflect the disease severity and progression, as well as therapeutic effects.3, 18 A haemogram derived marker, NLR, has been studied in various conditions and found to be related to inflammation in type 2 diabetes mellitus, 19 Hashimoto’s disease, 20 ulcerative colitis 21 and COVID-19 infection. 22 Moreover, it is correlated with plasma glucose and glycated haemoglobin (HbA1c) levels in diabetic patients. Therefore, it can be assumed that NLR could be related to the prognosis of COVID-19 subjects. Accordingly, there is a need to determine prognostic parameters, including laboratory biomarkers, clinical manifestations and factors affecting patient survival, for better disease management to predict the disease severity in a trial to reduce mortality among COVID-19 patients.17,23,24 Therefore, in the current study, we aim to determine biomarkers that can be used as prognostic indicators of the clinical outcomes of the disease.
Materials and methods
Patients
This is a case–control study included 112 hospitalised patients whose infection with the SARS-CoV-2 virus was confirmed by real-time polymerase chain reaction through throat and/or nasal swabs. The control group included 45 age-matched normal subjects. Eligibility criteria were all COVID-19 patients who were admitted to Cairo University Hospitals between April and October 2020 with complete baseline clinical and laboratory data and were on treatment and follow up. Exclusion criteria were patients with incomplete medical records or those refused to sign the informed consent.
The patients were classified into mild, moderate and severe/critical cases according to the procedure described by WHO 40 and the outcomes were recorded. Mild cases were defined by the presence of clinical symptoms and no changes observed in chest CT scans, and moderate cases included all those with respiratory symptoms associated with changes observed in CT scans. Severe cases were defined by the presence of the following three criteria: respiratory distress, with a respiratory rate ≥ 30/min, resting blood oxygen saturation ≤ 93% or partial pressure of arterial blood oxygen (PaO2)/oxygen concentration (FiO2) ≤ 300 mmHg. Critically ill cases included all severe cases that deteriorated due to respiratory failure and required mechanical ventilation, cases that involved shock and cases in which other organ failure required treatment with monitoring in intensive care units (ICUs).
The data were carefully collected from medical records, including personal data, history of comorbidities, general examination findings, oxygen saturation at admission, laboratory test reports (i.e. complete blood count (CBC), lactate dehydrogenase (LDH), HS-CRP, D-dimer and liver and kidney functions) and chest CT findings at admission. The outcome indicators of interest of this study were disease severity and mortality.
Statistical analysis
Statistical analysis was performed using SPSS version 16.0 (IBM, NY, USA). The differences in the levels of laboratory and clinical findings were analysed using a chi-square test and analysis of variance (ANOVA). In addition, Spearman’s rho correlation of clinical and radiological findings and biochemical and haematological parameters with disease severity and outcome was evaluated. An ANOVA M analysis was used to identify independent prognostic factors. Receiver operating characteristic (ROC) curves were used to specify possible parameters that could be used as indicators of disease severity and clinical outcomes (clinical improvement, cure and death).
Results
Patients’ demography and clinical data
Patients’ demographic data and clinical findings in different groups of patients based on the disease severity and disease outcome.

Data expressed as the mean ± standard deviation, *p-values were obtained using Chi Square.
Disease severity and outcome were significantly affected by radiological findings. Patients who showed normal radiological findings suffered from mild disease with no mortality. Patients who developed pneumonia or ground-glass opacity showed a high rate of severe disease (45/63) (71.4%), with high fatal consequences (25/63) (39.7%) (Table 1).
Laboratory findings and disease severity and disease outcome
Laboratory findings in different groups of patients based on the disease severity and disease outcome.

ALT, alanine aminotransferase; LDH, lactic dehydrogenase. Data expressed as the mean ± standard deviation, P-values were obtained using ANOVA analysis.
Correlation of clinical and laboratory findings with the disease severity and outcome
Spearman’s rho correlation of clinical and radiological findings with the disease severity and outcome.
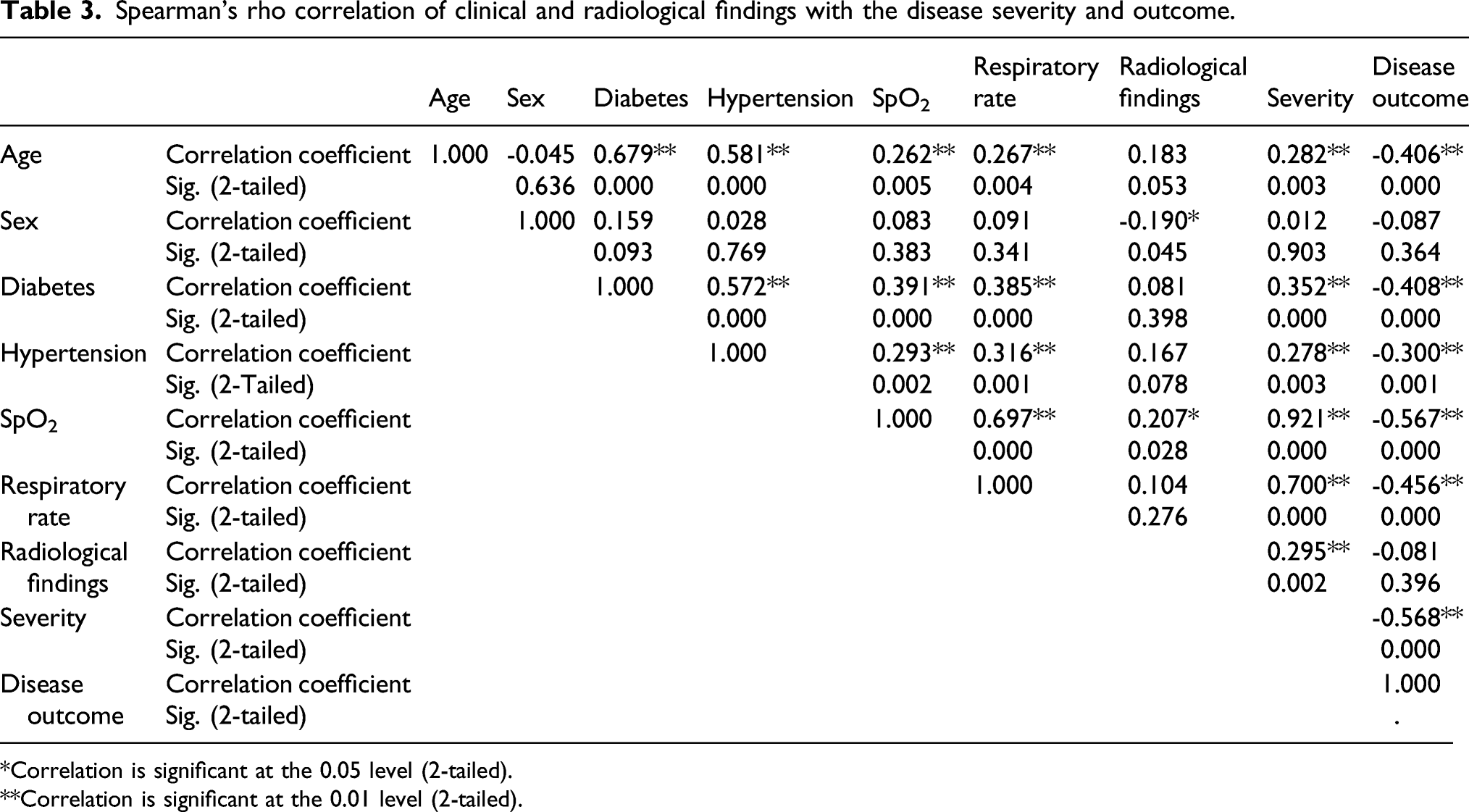
*Correlation is significant at the 0.05 level (2-tailed).
**Correlation is significant at the 0.01 level (2-tailed).
Spearman’s rho correlation of haematological parameters with the disease severity and outcome.

*Correlation is significant at the 0.05 level (2-tailed).
**Correlation is significant at the 0.01 level (2-tailed).
Spearman’s rho correlation of biochemical findings with the disease severity and outcome.

**Correlation is significant at the 0.01 level (2-tailed).
Prognostic parameters of disease severity and disease outcome
The results of the multivariate analysis revealed that HS-CRP and D-dimer are independent prognostic factors that can differentiate among mild, moderate and severe cases. The respiratory rate, SpO2, lymphocyte percentage, NLR, ALT and LDH are other independent prognostic factors which can denote severe forms of the disease. Creatinine, Hb, platelet count and TLC showed no significant differences among the groups (Figure 1). ANOVA multivariate analysis of different parameters in relation to the disease severity estimated as marginal means. (a) SpO2, (b) Respiratory rate (c) Platelets (d) Hb, (e) Segmented cell percentage, (f) Lymphocyte percentage, (g) Total leucocyte count (TLC), (h) ALT, (i) D-dimer, (j) HS-CRP, (k) LDH, (l) Creatinine.
Receiver operating characteristic curves were analysed to reach the best cut-off values for predicting disease severity and outcome. Upon evaluating the ROC curve of fatal consequences and different parameters, D-dimer, HS-CRP and LDH showed excellent test values of 0.951, 0.961 and 0.900, respectively. Creatinine showed a good value of 0.88, and ALT showed a fair value of 0.797; however, the remaining parameters did not show promising predictive values.
The ROC curve of HS-CRP versus disease severity and outcome showed excellent test values with area values of 0.978 and 0.96 (p = .00), respectively. At 167.5, which was the cut-off for HS-CRP, 95.7% of the cases were correctly identified as a severe disease and only 6.1% were incorrectly classified. At 252.5 cut-off, 82.8% of the cases were correctly classified as fatal and 8.4% were incorrectly classified.
The ROC curve of D-dimer versus disease severity and outcome showed excellent test values with 0.964 and 0.96 area values (p > .01), respectively. At 5.3 cut-off of HS-CRP, 82.6% of the cases were correctly identified and only 7.6% were incorrectly classified. At 10.2 cut-off, 82.8% of the cases were correctly classified as fatal and 4.8% were incorrectly classified. The ROC curve of LDH versus disease severity and outcome also showed excellent test values with area values of 0.906 and 0.900, respectively. At 317.5 cut-off, 73.2% of the cases were correctly classified as severe and 10.2% were incorrectly classified. At 400.5, 76.9% of the cases were correctly classified as fatal and 9.4% were incorrectly classified.
Roc curves results of different laboratory and clinical parameters in relation to the disease severity and disease outcome.

aUnder the nonparametric assumption.
bNull hypothesis: true area = 0.5.
Discussion
The severity of COVID-19 is a crucial problem in patient treatment and outcome. Many studies and meta-analysis studies investigated the possible role of different laboratory biomarkers for predicting COVID-19.11,25 The current study demonstrates the relationship between the different demographics and laboratory data, chest CT findings and disease severity and outcome. There was no significant effect of gender or age among the studied subjects. Some studies have also reported no gender variation among COVID-19 patients.18,26 However, only 26/112 patients over 60 years old were included in the current study, which can explain the contrast between our results and those obtained by previous studies that reported a significant effect of both variables on disease severity.23,27–31
Similar to previous results, the presence of hypertension and/or diabetes is considered an associated risk factor for disease severity and poor outcome.1,6,7,16,28,32–34 Low oxygen saturation and high respiratory rate increased the liability of ICU disease severity and fatal consequences, which agrees with other previous findings.27,32,34,35 Abnormal chest CT findings of the patients were significantly correlated with disease severity and outcome. Patients who presented with normal radiological findings suffered from mild disease with no mortalities, while those who developed pneumonia or had ground-glass opacity showed severe disease with highly fatal consequences. This finding is also in agreement with those obtained by previous studies.3,18
Our study reported highly significant lymphopenia and neutrophilia associated with severe and fatal cases, which was also evident in other studies.17,28,36,37 However, the ROC curve analysis did not find any of them sensitive and specific enough to be good predictors of disease severity or fatal consequences. Although a significant increase in WBC count in fatal cases is well-documented, 17 we did not find a significant increase in WBC count among severe or fatal cases. This finding can be explained by the fact that the increase in neutrophils compensates for the relative decrease in lymphocytes. A better assessment method is NLR. We found that NLR is an independent prognostic factor that correlates with disease severity and outcome, which agrees with other previous findings.28,29
In the current study, an increase in both ALT and LDH was detected in severely affected subjects as well as fatal COVID-19 patients, and both showed a significant correlation to disease severity. An increase in the ALT level was reported in 50% of fatal cases and 20% of COVID-19 survivors. 3 Meanwhile, an increase in both ALT and AST was reported in approximately 20% of COVID-19 patients. 3 Increased levels of serum LDH have also been reported in fatal SARS-CoV-2 cases.12, 38 In the current study, we found LDH to be an excellent predictor of both disease severity and death. Interestingly, it was also found to be a death predictor due to sepsis. 39
In the current study, HS-CRP and D-dimer were found to be strong independent prognostic factors for predicting disease severity and outcome. For example, the values of 167.5 and 5.8 could predict a severe disease, and those of 252.5 and 8.3 could predict fatal cases for HS-CRP and D-dimer, respectively. These findings agree with other previous findings that found a significant correlation between high levels of CRP and D-dimer with increased disease severity and poor prognosis.18,27,32 In contrast, Maddani et al. 28 reported a strong association between them and the COVID-19 rather than being an independent prognostic factor. This might be because our study dealt with mild, moderate and severe cases, while Maddani et al. studied only severe cases and compared them to mild cases. 28
Limitation
The main limitation of this study was the unavailability of involving patients from centres other than Cairo University hospitals. In addition, we did not conduct any power analysis to calculate the sample size selected for this study.
Conclusion
The presence of hypertension and/or diabetes was considered an associated risk factor for disease severity and poor outcome. Increased levels of HS-CRP, D-dimer, NLR, ALT, LDH, lymphopenia and neutrophilia, as well as changes in the chest CT, were associated with increased disease severity and fatal consequences. The ROC curves of HS-CRP, D-dimer, NLR and LDH suggested that they constitute excellent predictors for both disease severity and death.
Footnotes
Author contributions
Conceptualization, M. M. K., A.S.A., and Y. M. E.; methodology, L. A.F., L. M. K., O. S., M. A. K., M. M. K., and H. H.F.; formal analysis, L. A. F., Y. A. S. A., J. A. A..; data curation, J. A. A., L. A. F., and Y. A. S. A.; writing—original draft preparation, L. M. K., O.S. and; writing—review and editing, A. S. A., L. A. F., M. M. K. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Taif University Researchers Supporting Project Number (TURSP-2020/11), Taif University, Taif, Saudi Arabia.
Ethics approval
The protocol for this study was approved by the Institutional Review Board of National Cancer Institute, Cairo University, EGYPT. The ethical approval number: CP1937-30783 on 10 March 2020; all patients & control group gave written informed consent.
Informed consent
Each participant of this study provided informed written consent before the study.
Trial registration
Not applicable.
