Abstract
This study aimed to exploit the potential therapeutic value of palmatine in treatment of cardiac hypertrophy and the underlying molecular mechanism. Rat hypertrophy model was established by intraperitoneal isoproterenol (ISO) injection. The hypertrophy was evaluated with cardiac hypertrophic parameters, hemodynamic parameters, lipid profile, and non-specific cardiac markers. The animals were intraperitoneally administrated with either palmatine or vehicle. The relative expressions of ANP, BNP, HDAC2, HDAC5, KLF4, and INPP5F transcripts were determined by real-time polymerase chain reaction (PCR). The relative protein levels of HDAC2, HDAC5, KLF4, and INPP5F were analyzed by immunoblotting. Palmatine treatment significantly attenuated ISO-induced hypertrophy in rats and elicited remarkable repressions in ANP, BNP, and HDAC2 transcriptions but not HDAC5. The downstream effector genes KLF4 and INPP5F were greatly restored in a dose-dependent manner in response to palmatine treatment. Our data demonstrated that palmatine possessed promising therapeutic potential against hypertrophy, which was mediated by modulation of HDAC2-KLF4/INPP5F pathway.
Introduction
Cardiac hypertrophy is the abnormal enlargement of the heart muscle, which frequently associates with increased afterload. 1 Currently, the clinical management options for this complication are still very limited, and extensive investigations into the molecular mechanism underlying the etiology and progression of hypertrophy are still in urgent need. 2 Assembling evidences indicated that epigenetic modulation, especially chromatin remodeling, was critically involved in gene expression profile alterations in cardiac hypertrophy. 3 Most recently, the distinct roles of class I and class II histone deacetylases (HDACs) in the regulation of hypertrophic pathways have been elucidated. The potential therapeutic values of HDAC inhibitors or agonists in cardiac hypertrophy have been under extensive investigation, which might hold great promise for clinical applications.
Palmatine is a protoberberine alkaloid, extracted from the traditional Chinese medicine fibraureae recisae, with diverse pharmaceutical and biological activities. 4 Clinically, palmatine has been mainly used for the treatment of bacterial dysentery caused by gastrointestinal tract infection, as well as respiratory tract, urinary system, surgical-related infection, and gynecological inflammation. 5 However, the potential therapeutic value of palmatine in cardiac hypertrophy has not been exploited yet. Notably, in a recent endeavor to screen natural compounds with histone, deacetylase regulatory activity has characterized that palmatine was capable of specifically inhibiting class I HDACs, 6 which immediately prompted us to speculate whether palmatine could ameliorate cardiac hypertrophy via attenuating the pro-hypertrophic effect of class I HDACs. Our results for the first time revealed the anti-hypertrophic activity of palmatine, which warranted further investigations for its clinical applications.
Methods and materials
Animals
Male Wistar rats with average body weight of 180–220 g were purchased from Vital River Laboratories (China). All animals were housed in pathogen-free environment with appropriate temperature and humidity. Animal experiments were strictly complied with protocol approved by the Institutional Committee of Animal Care and Use of Cangzhou Central Hospital. Totally, 50 rats were randomly divided into five groups: (1) control group received saline (1.0 mL/kg body weight) subcutaneously and dimethyl sulfoxide (DMSO; 0.25 mL) intraperitoneally for 14 consecutive days; (2) cardiac hypertrophy group received ISO (10 mg/kg body weight) subcutaneously and DMSO (0.25 mL) intraperitoneally for 14 consecutive days; (3) hypertrophy group dosed with 25 mg/kg palmatine (prepared in 50 mg/mL DMSO stock solution) received both ISO subcutaneously and palmatine (25 mg/kg body weight) intraperitoneally for 14 consecutive days; (4) 50 mg dosage group received ISO and 50 mg/kg body weight palmatine for 14 consecutive days; and (5) treatment control group received saline subcutaneously and 50 mg/kg body weight of palmatine intraperitoneally for 14 consecutive days.
Real-time polymerase chain reaction
Hearts were extracted immediately after the experimental rats were sacrificed at the endpoint. Fresh tissue was homogenized and total RNA was isolated with TRIzol reagent (Invitrogen, Carlsbad, CA, USA). The quality and quantity of RNA were determined with BioAnalyzer 2100 (Agilent, Santa Clara, CA, USA) prior to any further processing. An amount of 1 μg RNA was reversely transcribed into complementary DNA (cDNA) with High-Capacity cDNA Reverse Transcription Kit (Thermo, Waltham, MA, USA). All the primers used in this study were synthesized by Shanghai Sangon (Shanghai, China) and listed in Table S1. The relative expression was calculated by 2-∆∆Ct method and normalized to GAPDH.
Western blotting
The indicated tissue samples were ultrasonicated on ice and total protein was extracted with radio-immunoprecipitation assay (RIPA) lysis buffer. The relative content of protein was determined by BCA Protein Assay Kit (Pierce, Madison, WI, USA). Equal amount of protein was resolved by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene difluoride (PVDF) membrane. The membrane was blocked with 5% skim milk and hybridized with indicated primary antibody at 4°C overnight (HDAC2 cst5133, HDAC5 cst2082, and β-actin cst12620 from Cell Signaling Technology, Danvers, MA, USA; INPP5F sc-514657 and KLF4 sc-166190 from Santa Cruz Biotechnology, Dallas, TX, USA). The unbound antibody was completely removed by rigorous wash with Tris-buffered saline with Tween 20 (TBST) at day 2. Then, the membrane was incubated with corresponding horseradish peroxidase–conjugated secondary antibody at room temperature for 1 h. After second round of washing with TBST, the blots were visualized by Enhanced Chemiluminescent Kit (Millipore, Billerica, MA, USA). The quantification was performed with densitometry scanning.
Serum analysis
Blood samples were collected from experimental rats under anesthesia at the endpoint of indicated treatments. Serum was isolated and the relative contents of triglycerides, total cholesterol, very low-density lipoprotein (VLDL), low-density lipoprotein (LDL), high-density lipoprotein (HDL), C-reactive protein (CRP), lactate dehydrogenase (LDH), and creatinine kinase-MB (CK-MB) were determined with biochemical analyzer (MSLBA06 plus Semi auto; Guangzhou Medsinglong Medical Equipment Co. Ltd., Guangzhou, China) using available biochemical diagnostic kits.
Measurement of hemodynamic parameters
The impact of palmatine on hemodynamic parameters was evaluated by invasive cannulation method. Briefly, rats were anesthetized by intramuscular administration with ketamine (20 mg/kg) and xylazine (10 mg/kg). The optimal body temperature was maintained at 37°C via external heating. The abdomen was opened to locate the trachea, behind which the carotid artery was cannulated. The hemodynamics was measured with BP 100 transducer (LabScribe Systems).
Measurement of hypertrophic parameters
The hypertrophic parameters were measured immediately after hemodynamics. Rats were euthanized by overdose of ketamine (80 mg/kg, IM). The hearts were extracted and weighed. The femur length was measured for calculation of variety of hypertrophic indices such as cardiac hypertrophic index (CHI) and left ventricular hypertrophy index (LVHI). LV wall thickness was measured using screw gauge micrometer. Cardiomyocyte diameter measurement was performed using ImageJ Analyzer Software on H&E-stained left ventricular tissue sections.
Statistical analysis
All data were obtained from at least three independent experiments and expressed as mean ± standard deviation. Pair-wise comparison was performed by one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test using SPSS 23.0 software. Difference was considered as statistically significant when P-value <0.05.
Results
Palmatine treatment decreased expression of ANP and BNP messenger RNA
We first established cardiac hypertrophy rat model by intraperitoneal administration of ISO. The successful establishment of animal model was further validated by anatomic analysis after sacrifice. The essential hypertrophic indices were observed in our model including CHI, LVHI, heart weight/body weight (HW/BW), left ventricular weight/right ventricular weight (LVW/RVW), LV wall thickness, cardiomyocyte diameter and LV collagen level, which were all listed in Table S2. ISO injection induced significant hypertrophy in our system, which was indicated by increase in all the above indexes. The dosage of palmatine was determined in view of effective pharmaceutical concentration and minimum toxicity. For treatment group, either 25 or 50 mg/kg body weight of palmatine was intraperitoneally injected with ISO simultaneously for 14 consecutive days. The hypertrophic parameters were determined as described in “Methods and materials” section. Administration with palmatine in saline-controlled rat showed no effect on hypertrophic parameters in comparison with untreated counterparts. However, in hypertrophic rats, palmatine significantly decreased related parameters in a dose-dependent manner (Table S2). The relative expressions of ANP and BNP extracted from heart tissue were measured by quantitative real-time polymerase chain reaction (PCR). As shown in Figure 1, both ANP and BNP were significantly induced in hypertrophic rats, and this upregulation was slightly but significantly reversed by coadministration of 25 mg/kg palmatine. In our 50 mg/kg treatment group, both transcriptional levels of ANP and BNP were dramatically suppressed and decreased to levels comparable to the control group. We further estimated any potential influence of palmatine on physiological expressions of ANP and BNP in the heart. Even the maximum dose of palmatine used in our study elicited little changes in both ANP and BNP. Our results unambiguously demonstrated that palmatine administration significantly suppressed the upregulation of ANP and BNP in cardiac hypertrophy.
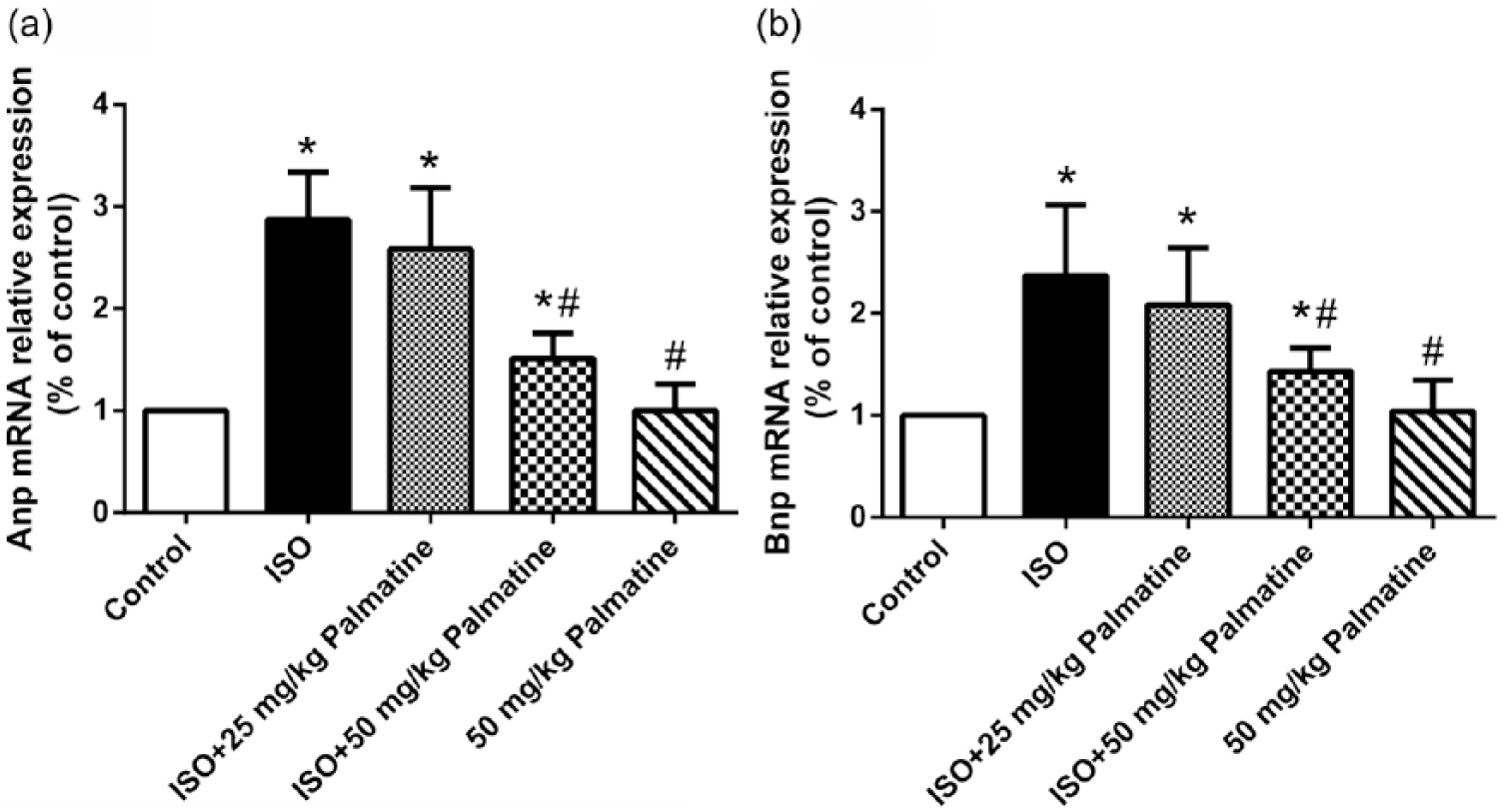
Effect of palmatine on the expression of ANP and BNP mRNAs in rat hearts. (a) The mRNA levels of hypertrophic markers ANP were analyzed by qRT-PCR. (b) The mRNA levels of hypertrophic markers BNP were analyzed by qRT-PCR. RT-PCR was performed utilizing gene-specific primers. GAPDH was used as an internal control. Values are expressed as mean ± SEM (N = 10 animals in each group). Values are expressed as mean ± SEM (N = 10 animals in each group). *P < 0.05 compared to the control group. #P < 0.05 compared to the ISO group.
Palmatine treatment inhibited HDAC2 expression
Epigenetic modulation especially chromatin remodeling was crucial for the transcriptome shift to cardiac hypertrophy. Assembling evidences suggested that opposite roles of HDAC2 and HDAC5 were involved in hypertrophy progression. Here, we set out to determine the alteration of these two histone modifiers in our animal model. ISO-induced cardiac hypertrophy showed remarkable elevations of both HDAC2 and HDAC5 messenger RNA (mRNA) expressions. Palmatine challenge imposed inhibitory effect on HDAC2 expression in a dose-dependent manner (Figure 2(a)). However, palmatine treatment exhibited no effect on HDAC5 (Figure 2(b)). Consistent with previous data, administration with palmatine did not altered base line expressions of either HDAC2 or HDAC5 in saline-injected control rats. We further validated the inhibitory effects at the translational level, where the immunoblotting results showed similar changes in proteins as their transcripts (Figure 2(c) and (d)). In agreement with previous reports, our data supported the fundamental roles of both HDAC2 and HDAC5 in prompted cardiac hypertrophy progression. More importantly, palmatine specifically suppressed expression of HDAC2 at both transcriptional and translational levels, which might underlie its therapeutic effect against hypertrophy in our rat model.

Effect of palmatine on the expression of HDAC2 and HDAC5 mRNAs and protein in rat hearts. (a and b) The mRNA levels of HDAC2 and HDAC5 were analyzed by qRT-PCR. RT-PCR was performed utilizing gene-specific primers. GAPDH was used as an internal control. (c and d) The protein levels of HDAC2 and HDAC5 were analyzed by western blot. Values are expressed as mean ± SEM (N = 10 animals in each group). Values are expressed as mean ± SEM (N = 10 animals in each group). *P < 0.05 compared to the control group. #P < 0.05 compared to the ISO group.
Palmatine restored expression of INPP5F and KLF4
Previous study suggested that HDAC2 potentially inhibited expression of KLF4 and INPP5F, which might eventually contribute to its pro-hypertrophic effect. Here, we further investigated the regulation of KLF4 and INPP5F by palmatine in our cardiac hypertrophy model. As shown in Figure 3(a) and (b), the transcriptional expressions of both KLF4 and INPP5F were dramatically suppressed in ISO-induced hypertrophic hearts. Palmatine treatment induced expressions of both factors in a dose-dependent manner. Notably in our system, 50 mg/kg palmatine was sufficient to restore the physiological expressions of both KLF4 and INPP5F. Our immunoblotting results further consolidated this stimulatory effect of palmatine at protein levels (Figure 3(c) and (d)). In line with previous report, our data demonstrated that the expressions of both KLF4 and INPP5F were significantly suppressed in hypertrophic tissues and palmatine efficiently restored physiological expressions of these effector proteins.

Effect of palmatine on the expression of INPP5F and KLF4 mRNAs and protein in rat hearts. (a and b) The mRNA levels of INPP5F and KLF4 were analyzed by qRT-PCR. RT-PCR was performed utilizing gene-specific primers. GAPDH was used as an internal control. (c and d) The protein levels of INPP5F and KLF4 were analyzed by western blot. Values are expressed as mean ± SEM (N = 10 animals in each group). Values are expressed as mean ± SEM (N = 10 animals in each group). *P < 0.05 compared to the control group. #P < 0.05 compared to the ISO group.
Effect of palmatine on hemodynamic parameters and lipid profile
Hemodynamics was heavily altered during cardiac hypertrophy progression. Thus, we first measured this parameter upon successful hypertrophic model establishment and subsequent change in response to palmatine treatment. The ventricular performance was evaluated by invasive method as described by Patel et al. As expected, the blood pressure and heart rate were significantly increased in ISO-induced hypertrophic rats, which was accompanied with decreased pressure development and decay rate (Table S3). These hemodynamic parameters were markedly improved by palmatine treatment in a dose-dependent manner. In addition, we closely examined the serum lipid profile, as dyslipidemia was an important pathological indicator of cardiac hypertrophy. As shown in Table S3, total cholesterol, LDL, VLDL and triglyceride increased, while HDL decreased in ISO-induced hypertrophic model, which was readily reversed by palmatine administration.
Effect of palmatine on non-specific cardiac markers
We further evaluated the beneficial effect of palmatine in hypertrophy by measurement of several non-specific cardiac markers such as CK-MB, LDH, and CRP (Table S4). CK-MB has been demonstrated to be significantly stimulated in coronary artery disease, aortic stenosis, and heart failure. LDH was elevated in hypertrophy-induced myocardial damage. And CRP was a systematic inflammatory factor associated with increased risk of cardiovascular disease. Our results demonstrated remarkable inductions of these three markers in hypertrophic heart, and palmatine treatment significantly decreased serous contents of LDH, CK-MB, and CRP, which suggested significant improvement with respect to myocardial function and hypertrophy.
Discussion
Recently, palmatine has been identified as candidate class I HDACs inhibitor, 6 and in view of the well-recognized pro-hypertrophy effect of class I HDACs, here we sought to exploit the therapeutic application of palmatine in hypertrophic rat model via intraperitoneal injection of ISO. Palmatine significantly attenuated ISO-induced hypertrophy in a dose-dependent manner. Consistent with previous reports, 7 the pro-hypertrophic HDAC2 stimulated by ISO was markedly abrogated by palmatine. Previous study suggested that HDAC2 might contribute to pathogenesis of hypertrophy via suppression of KLF4 and INPP5F. 8 In line with this notion, palmatine treatment restored the expressions of KLF4 and INPP5F at both transcriptional and translational levels. Therefore, our study demonstrated that palmatine modulated the HDAC2-KLF4/INPP5F pathway, which consequently contributed to its anti-hypertrophy effect.
Although palmatine was first screened with inhibitory effect against HDAC deacetylase activity, 6 our data surprisingly demonstrated that palmatine suppressed HDAC2 expression instead. The detailed molecular pathway underlying this observation still needs to be fully elucidated. Noteworthily, Hambright et al. 9 proposed that palmatine treatment specifically inhibited the Akt/mTOR/p70S6K/rpS6 axis and selectively inhibited cell growth and invasion via directly targeting ribosomal protein S6. This study, together with our current results, implicated that palmatine-mediated HDAC2 inhibition might involve regulation of Akt/mTOR pathway, which is frequently over-activated in cardiac hypertrophy.10–12 In view of the intrinsic advantage of medicinal component from the traditional Chinese medicine, the potential clinical benefits of palmatine against cardiac hypertrophy deserve further investigations.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
