Abstract
Dendritic cells (DC) are potential tools for therapeutic applications and several strategies to generate tolerogenic DCs are under investigation. When activated by cytokines and microbial products, DCs express mediators that modulate immune responses. In this regard, the metabolites generated by the activities of inducible nitric oxide synthase (iNOS) and arginase in DCs seem to play important roles. Here, we evaluated the effects of adoptive transfer of DCs generated in vitro from bone marrow precursors (BMDC) modulated with L-NAME (Nω-nitro-L-arginine methyl ester) and NOHA (NG-Hydroxy-L-arginine), inhibitors of iNOS and arginase, respectively, upon the immune response of the wild type (BALB/c) and OVA-TCR transgenic (DO11.10) mice. The modulation with L-NAME increased CD86 expression in BMDC, whereas treatment with NOHA increased both CD80 and CD86 expression. Adoptive transfer of either L-NAME- or NOHA-modulated BMDCs to BALB/c mice reduced the plasma levels of ovalbumin-specific antibody as well as proliferation and cytokine secretion in cultures of spleen cells in comparison adoptive transfer of non-modulated DCs. Conversely, transfer of both modulated and non-modulated BMDCs had no effect on immune response of DO11.10 mice. Together, these results show that the treatment with iNOS and Arg inhibitors leads to increased expression of co-stimulatory molecules in DCs, and provides evidences that L-arginine metabolism may be an important therapeutic target for modulating immune responses in inflammatory disorders.
Introduction
Several strategies to generate tolerogenic dendritic cells (DCs) are under evaluation in the attempt to modulate the immune response in several diseases. Studies,1–4 including those performed in our laboratory5–7 show that administration of oral protein leads to the generation of tolerogenic dendritic cells (DC) not only in the mucosal, but also in secondary lymphoid organs such as spleen. We previously showed that, in the presence of ovalbumin (OVA), spleen DCs from tolerant BALB/c mice co-cultured with naïve OVA-specific T cells polarize T cells preferentially to TGF-β producing Th3 cells. 5 The DO11.10 mice, which bear 80% of transgenic T-cell receptor (TCR)-α and -β chains that recognize the OVA peptide OVA323–339 that binds to I-Ad MHC class II protein, have been used in studies of oral tolerance to OVA, particularly in relation to T-cell proliferation and cytokine secretion.8,9 However, we have previously shown that, opposite of BALB/c mice, DO11.10 mice were not susceptible to tolerance after OVA ingestion. In this transgenic model, the continuous feeding with OVA did not modulate the antibody production and proliferative response.5,10,11
Another line of research has shown that nitric oxide (NO), a free radical produced by hydrolysis of L-arginine by the action of induced nitric oxide synthase (iNOS), can downregulate the immune response mediated by CD4+ T lymphocytes. Nitric oxide is involved in several physiological events such as vascular relaxation, neurotransmission, defense against pathogen, and regulation of in vivo adaptive immune response.12–16 However, overproduction of NO by activated phagocytes has been implicated in the destruction of healthy tissue in the adjacent area of inflammation, thereby contributing to the pathogenesis of several diseases.14,17–20
On the other side of the metabolic pathway of L-arginine, the enzyme arginase (Arg) converts L-arginine into L-ornithine and urea. Mammalian cells express the two isoforms of Arg, named Arg I and Arg II. 21 Arg I controls the availability of arginine in activated macrophages and downregulates NO synthesis. 22 iNOS and arginase use the same substrate and compete in conditions of limited availability of L-arginine. 21 Although the affinity of L-arginine is higher for NOS than for arginase, a maximum activity of arginase is 1000-fold higher than NOS. 23
Although the involvement of iNOS and Arg on macrophage response is well-known,12,17,22,24–29 few studies report the effects of the modulation of these enzymes on DC functions, and they only address the in vitro effects of such modulation.24,30–33 In this regard, small amount of NO production by in vitro-generated bone marrow DCs (BMDCs) seems to be able to inhibit T-cell proliferation. Literature shows that CD4+T cells are able to proliferate in the presence of DCs expressing iNOS or not. The addition of IFN-γ to co-cultures potentiated the activity of iNOS and subsequent release of NO in the supernatant reduced the proliferation of CD4+ T lymphocytes and induced T-cell apoptosis. 13 However, conflicting data showed that inhibition or absence of iNOS induces tolerance to dietary antigen. 34
The activity of both iNOS and Arg1 has also been correlated with the capacity of DCs to expand and differentiate regulatory T cells in the periphery.31,33,35–37 Cytokines secreted by Th1 and Th2 cells can competitively regulate the activity of iNOS and arginase15,24 in macrophages and DCs. Th1 immune responses lead to the induction of iNOS, resulting in production of NO, whereas Th2 responses upregulate arginase expression.24,38,39 Specifically, it was demonstrated that interleukin (IL)-4 and IL-10 induce the expression of arginase, whereas IFN-γ induces iNOS and inhibits Arg1 on macrophages and DC, in a clear Th1 and Th2 opposition.14,40 Specific inhibitors of NOS (Nω-nitro-L-arginine methyl ester;L-NAME) 41 and arginase (Nω-hydroxy-L-arginine; NOHA) 42 are currently used to investigate NO activity in adjuvant-induced arthritis, experimental colitis, and pulmonary allergy.17–20,43,44 The non-selective inhibitor L-NAME was able to block activity of constitutive NOS (cNOS). On the other hand, NOHA is an intermediate in the biosynthesis of NO by NOS and a physiological competitive inhibitor of arginase activity.23,45
In the present study, we evaluated the effects of the treatment with NOHA and L-NAME on phenotype and activity of BMDCs. We also analyzed the effects of adoptive transfer of modulated BMDCs on immune response to OVA of BALB/c and DO11.10 mice.
Our hypothesis is that the inhibition of iNOS and arginase can drive BMDCs into a tolerogenic profile. Our results indicate that modulation of BMDCs with arginase and iNOS inhibitors may be an important strategy to induce tolerance to antigens involved in the pathogenesis of inflammatory diseases.
Material and methods
Mice. Mice carrying the MHC class II restricted rearranged T cell receptor transgene, Tg (DO11.10) 10Dlo, here named DO11.10 mice, 46 and wild-type BALB/c mice were supplied by CEMIB (Centro Multinstitucional de Investigações Biológicas), UNICAMP and housed in pathogen-free conditions and fed a diet of autoclaved food and water. The study was approved by the institutional Committee for Ethics in Animal Experimentation (protocol no. 911-1).
Immunization. Mice were intraperitoneally challenged with 10 mg of OVA mixed with 1 mg of aluminum hydroxide in saline solution. After 14 days, the mice received a reinforcement dose of 10 mg of the antigen in saline solution. The mice were bled by the retro-orbital plexus after 7 days and sera were separated for antibody assays.
Antibody assays. Antibody titers to OVA were determined by a standard enzyme linked immunosorbent assay (ELISA) using an automatic reader (Multiskan II, MS, Labsystem, Helsinki, Finland) as described elsewhere. 5 Briefly, each well of a microtiter plate (Falcon, Becton-Dickinson, Franklin Lakes, NJ, USA) was coated with 1 µg of OVA in carbonate/bicarbonate buffer pH 9.5 and incubated overnight at 4°C. The wells were washed and blocked with 5% skimmed milk in phosphate buffered saline (PBS) pH 7.4. After new washing cycle, the plates were incubated for 1 h with mouse serum samples diluted with 2% skimmed milk in PBS pH 7.4 from 1:100 to 1:12,800. The washed plates were incubated with a rabbit anti-mouse Ig–HRPO conjugate (275 ng/mL) in 2% skimmed milk in PBS pH 7.4 for 1 h. The reaction was revealed by adding freshly prepared substrate (0.04% orthophenylenediamine and 0.03% H2O2 in 50 mM citric acid/di-sodium hydrogen phosphate buffer, pH 5.5). The reaction was stopped after 30 min and the absorbance was read at 492 nm. The ELISA scores were computed by running sums of the optical densities for the serum dilutions (1:100–1:12,800) for individual mice, and were expressed as the mean ± SEM of five mice from each group.
BMDCs. BMDCs were generated from bone marrow precursors as described elsewhere.47,48 Briefly, bone marrow cells were flushed from femurs and tibia of naïve BALB/c mice. Cells were seeded in six-well plates (Corning) at a density of 2 × 106 cells/well in RPMI 1640 medium (Sigma) containing 10% fetal bovine serum (FSB, Cultilab), 20 μg/mL gentamicin (USB), and 20 ng/mL of mouse recombinant granulocyte macrophage colony-stimulating factor (mrGM-CSF) (Biosource) and cultured at 37°C. On days 3 and 6, the culture medium was replaced. After 8 days of culture, BMDCs were harvested and re-cultured in 24-well plates at a density of 2.5 × 106 cells per well in the presence of either L-NAME at 1 mM or NOHA at 750 μM. BMDCs cultured in the absence of stimuli were used as control. After 48 h of the onset of the cultures, 100 mg/mL OVA (Conalbumin, Sigma) were added to monolayers that were incubated for additional 18 h.
Measurement of nitric oxide. Dosages of NO were used as described previously.49–51 Briefly, 50 μL of the culture supernatants of BMDCs cultured in the presence or absence of LPS + TNF-α were collected and transferred to a 96-well plate (Nunc, Nunclon) in triplicates. Then, 50 μL of Griess reagent were added to supernatants. After stirring, spectrophotometric readings were carried out on an ELISA reader (Multiskan MS, Labsystems, USA) at 540 nm. The concentration of nitrite (mM/1 × 106 cells) was calculated based on a standard curve of a sodium nitrite solution at 320, 160, 80, 40, 20, 10, and 5 mM. The results were expressed as mean ± SEM.
Measurement of urea. BMDC cultures were used for detection of arginase activity according to the methodology described elsewhere50,52 with some changes. Briefly, after removing the supernatant, the cells in monolayers were lysed by adding 50 μL/well of lysis solution (0.1% Triton X-100, 5 mg aprotinin and trypsin inhibitor, and 5 mg of antipain or PMSF) and the plates were kept under mild stirring for 30 min at room temperature. Then, 50 μL Tris-HCl 50 mM, pH 7.5 containing 10 mM MnCl2 was added to each well and the plates were incubated at 55°C for 10 min to activate the enzyme. Aliquots of 25 μL of activated lysates were transferred to Eppendorf tubes and 25 μL of 0.5 M L-arginine (Sigma, St. Louis, MO, USA), pH 9.7 was added. After 60 min incubation at 37°C, the L-arginine hydrolysis reaction was stopped by adding 400 μL of a mixture of H2SO4, H3PO4, and H2O (1:3:7). Then, 25 μL of a solution ISPF 9% in absolute ethanol was added to each reaction tube and the mixtures were heated for 45 min at 100°C. After resting for 10 min at room temperature in the dark, absorbance was measured in microplate reader (Multiskan MS, Labsystems, USA) at 540 nm. The quantity of urea in the reaction was determined by comparison with the absorbance of urea standard curve (1.5–1200 mg/mL). For spectrophotometric measurements of urea standard solutions, aliquots of 100 μL of each of the curve points were distributed in the microtiter plate in quadruplicate and then each well was filled with 400 μL of the mixture of acids and 25 μL of ISPF in absolute ethanol. The results were expressed as mean ± SEM. The urea concentration in the sample in μg/106 cells/mL.
Flow cytometry analysis. The DC generation was accompanied by flow cytometry (FACSAria, Becton-Dickinson), using anti-mouse CD11c- allophycocyanin (APC) conjugate (clone HL-3; BD PharMingen, San Diego, CA, USA); anti-class II MHC-PE conjugate (clone M5/114.15.2, Multinyi Biotec); anti-CD80-FITC (clone 14-10A1), anti-CD86-FITC (clone GL1, BD PharMingen), and anti-CD40 FITC (clone 3/23, BD-Pharmingen) as described previously. 53 The cells were also stained with irrelevant isotype controls (BD PharMingen) for each cell marker.
Amplification and compensation were determined by the examination of non-marked cells and analyses were carried out using the FCS-Express software. Data were represented as percentage of positive cells (%) and geometric mean fluorescence intensity (MFI).
BMDC transfer. BMDCs generated as described above were collected, pelleted at 200 g for 5 min to remove stimulus, and cell concentration was adjusted to 5 × 106 cells/mL in 0.15M NaCl. Aliquots of 100 μL of the cell suspension were injected into the retro-orbital venous sinus of each mouse of BALB/c or DO11.10 lines.
Antigen-specific proliferation. T-cell proliferation assays were carried out as described elsewhere.10,53 Briefly, spleen cells or mesenteric lymph nodes (MLN) (5 × 105 cells/well) were cultured in the presence of the antigen (0.1 mg/mL OVA) for 96 h and supernatants were collected for cytokine determination. 3-[4,5-dimethyltiazol-2-yl] diphenyltetrazolium bromide solution (MTT, Sigma, St. Louis, MO, USA) was added to the cultures (5 mg/mL; 10 μl/well) and incubated for an additional 4 h. After dissolving of formazan crystals, the optical densities were measured at 540 nm. TCD4+ cells were enriched from spleens of 8-week-old naïve BALB/c mice using CD4 microbeads, as indicated by manufacturer (Miltenyi Biotec, Germany). Purification (higher of 95%) was accompanied by flow cytometry with antibodies anti-CD3-PE and anti-CD4-FITC (BD PharMingen, San Diego, CA, USA).5,10
Co-cultures of DC-Tcells and CD28+, CTLA4+, and CD25+Foxp3+ cell staining. DCs and T-cell co-culture assays were carried out as described elsewhere. 7 T cells (5 × 105 cells/well) and DCs (2 × 104 cells/well) were co-cultured in the presence of the antigen (1 mg/mL OVA) for 96 h. Co-cultures of CD3+CD4+ cells were gated and populations marked to determinate the CD28+, CTLA4+, and CD25+Foxp3+ cells as described. Briefly, single cell suspensions were washed and suspended in staining buffer (PBS plus 0.1% bovine serum albumin plus 0.09% sodium azide [BD Becton Dickinson, San Jose, CA, USA]) and stained with surface the markers anti-CD3-PerCP-Cy5.5, anti-CD4-PECy7, anti-CD25 FITC, anti-CD28+ PE, and anti-CTLA4+ PE and their respective isotype controls for 30 min at 4°C. The samples that were used to analyze CD25+Foxp3+ Tregs were additionally incubated with fixation/permeabilization buffers (Mouse Foxp3 Buffer Set – BD Pharmingen) for 30 min, according to the manufacturer’s instructions, and stained with anti-mouse-Foxp3 PE, antibody, and the respective isotype control. All FACS reagents were purchased from BD Bioscience (San Jose, CA, USA) and used following the manufacturer’s recommendation. Amplification and compensation were determined by the examination of non-marked cells and analyses were carried out using the FCS-Express software. Data were represented as percentage of positive cells (%) and geometric MFI.
Cytokine measurement. The levels of IL-2, IL-4, IL-10, IFN-γ, and TGF-β were measured in co-culture supernatants using BD–OptEIATM ELISA sets, according to the manufacturer’s instructions. Absorbances were read at 450 nm, with the wavelength of 540 nm used for correction.
Statistical analysis. Differences were statistically analyzed using a one-way ANOVA with a Bonferroni post-test. The software Prism (version 5.0, Graphpad Software, Inc., San Diego, CA, USA) was used to prepare graphics and for statistical analysis. P values <0.05 were considered statistically significant. Data are presented as the mean ± SEM.
Results
In this work, we aim to evaluate the effects of adoptive transfer of BMDCs modulated with iNOS and arginase inhibitors upon the development of immune response in BALB/c and DO11.10 transgenic mice. To our knowledge, this is the first work showing the modulatory effects of the inhibition of arginase and iNOS in DCs on the immune response to an exogenous antigen.
First of all, we determined the effective minimal doses of L-NAME and NOHA used in the treatments of BMDCs and we found that these drugs were able to significantly inhibit iNOS and Arg activities in doses as low as 1 mM and 750 μM, respectively (data not shown). As expected, the pretreatment of BMDCs with 1 mM L-NAME led to a significant decrease in nitrite production in BMDCs stimulated with LPS + TNF-α (Figure 1a). With respect to treatment with NOHA, it was observed that the dose of 750 μM led to a significant inhibition of urea production in BMDCs treated and not treated with IL-4 (Figure 1b).

INOS and arginase activities in BMDCs upon treatment with inhibitors of L-arginine metabolism. BMDCs were generated in vitro and cultured in the presence of either L-NAME (1 mM) or NOHA (750 μM) for 24 h. BMDCs cultured in the absence of inhibitors were used as control group. (a, b) LPS (1 µg/mL) + mrTNF-α (20 ng/mL) or rmIL-4 (40 ng/mL) were added to the culture wells, to induce iNOS and Arg-1 activities, respectively, and plates were incubated for an additional 24 h. In all groups, BMDCs cultured in the absence of stimuli were used as controls. Culture supernatants were collected to NO detection (a) and cells were lysed to evaluated arginase activity by urea assay (b). (*) means P <0.05. The non-treated group did not receive any stimuli.
The BMDCs treated with the inhibitory drugs L-NAME and NOHA were also evaluated for expression of co-stimulatory molecules CD80, CD86, and CD40 in the CD11c+ MHCII+ subsets. Analysis by flow cytometry showed no change in CD40 expression after treatment with L-NAME or NOHA, compared to untreated cells (data not shown). When CD86 expression was analyzed, we noticed significant increase in the expression of this molecule in BMDCs treated with either L-NAME or NOHA, whereas the expression of CD80 was significantly increased only after the treatment with NOHA (Figure 2a).

Effects of L-NAME or NOHA treatment on surface markers expression and frequency of BMDCs and T cells. (a) DCs were generated in vitro by culture of bone marrow precursors with GM-CSF for 8 days and cultured with L-NAME (1 mM), NOHA (750 μM), or RPMI for 48 h. BMDCs were labeled with anti-CD11c-APC, anti-MHC class II-PE, anti-CD86-FITC, and anti-CD80-FITC antibodies and analyzed by flow cytometry. Data represents the percentage of positive cells (%) and geometric MFI of cells over background staining with an isotype-matched control (hatched line). Data are representative of two separate experiments. (b) DCs were generated in vitro by culture of bone marrow precursors with GM-CSF for 8 days and cultured with L-NAME (1 mM), NOHA (750 μM), or saline for 48 h and co-cultured with T CD4+ cells obtained from OVA immunized BALB/c mice, as described on M. CD25+Foxp3+ markers were stained on TCD4+ population and represented as percentages. CTLA-4 and CD28 molecule expression on TCD4+ population were expressed on MFI. Data represent the percentage of positive cells (%) and geometric MFI over background staining with an isotype-matched control (hatched line). Dashed line represents non-stimulated TCD4+ cells. Data are representative of two separate experiments.
The activity of both iNOS and Arg1 can also been correlated with the capacity of DCs to expand regulatory T cells in the periphery, as demonstrated here. In this regard, we observed that BMDCs treated with saline or L-NAME were able to expand in vitro the population of CD4+CD25+Foxp3+ cells in co-cultures with TCD4+ cells (Figure 2b).
Effects of administration of NOHA or L-NAME modulated BMDCs on immune response of BALB/c mice
To evaluate the effects of modulation of DCs on the immune response, BMDCs were treated with the iNOS (L-NAME) and arginase (NOHA) inhibitors and adoptively transferred to naïve BALB/c or DO11.10 (OVA-TCR) mice. NOHA or L-NAME-modulated BMDC, as well as non-modulated BMDC, were adoptively transferred to naïve wild-type BALB/c and transgenic DO11.10 mice. All mice were i.p. immunized with OVA. After antigenic challenge, the levels of antibodies in serum, antigen specific-proliferative response, and cytokine production were evaluated in the cultures of spleen and mesenteric lymph nodes (MLN).
As depicted in Figure 3a, the adoptive transfer of BMDCs modulated with either L-NAME or NOHA was able to reduce the levels of OVA-specific antibody in sera of immunized BALB/c mice, and NOHA-modulated BMDCs apparently seems to be more effective in this regard.
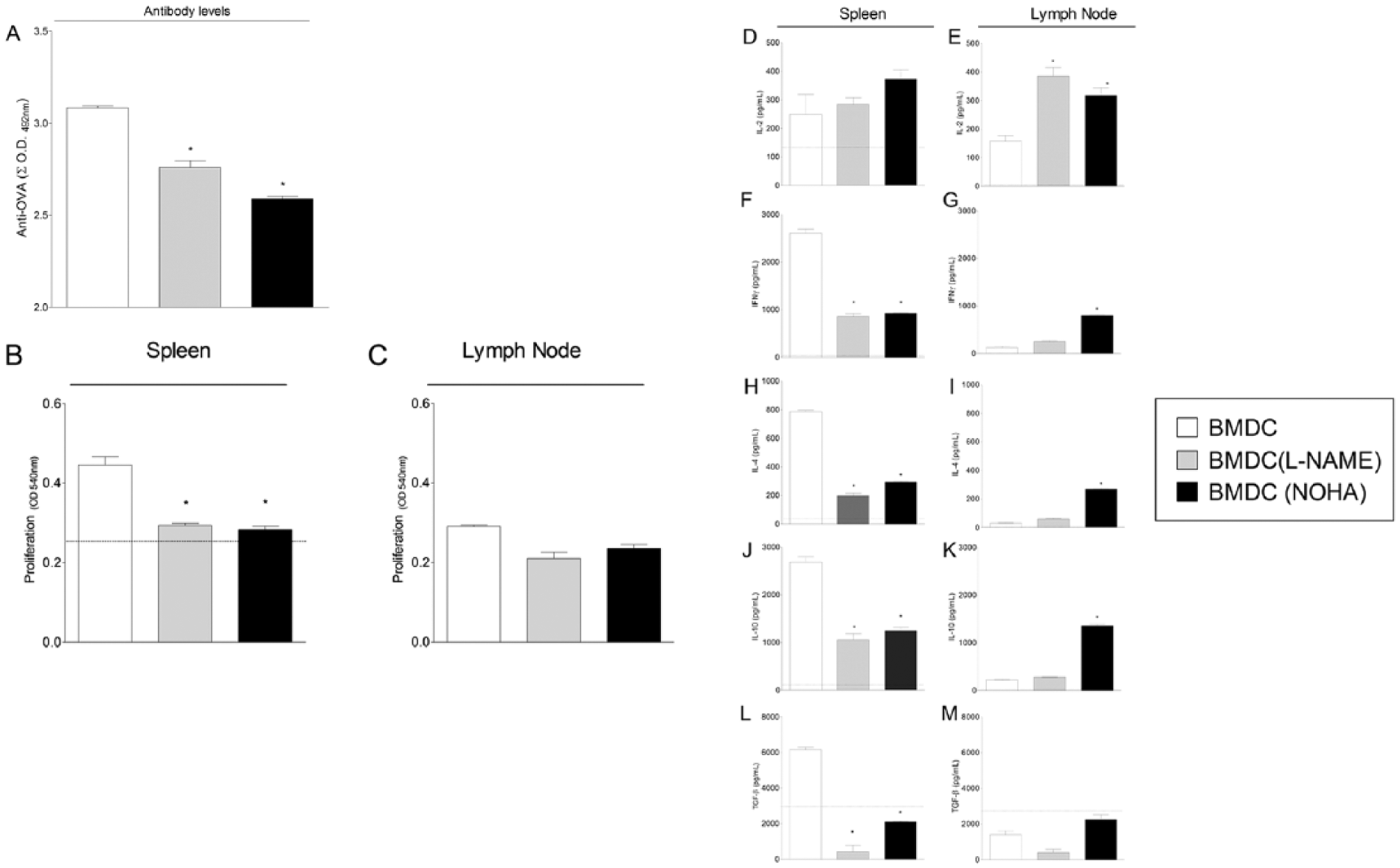
Adoptive transfer of BMDCs modulated with L-NAME or NOHA reduces antibody levels and antigen specific proliferation of immune cells in BALB/c mice. To perform the adoptive transfer, BALB/c mice (n = 6 per group) were injected by intravenous route with 5 × 105 BMDCs cultured in the presence of L-NAME (1 mM) or NOHA (750 μM) for 48 h. A group of mice that received BMDCs cultured in the absence of inhibitors was included as control. One day later, mice of all groups were challenged with 10 mg OVA + 1 mg Al(OH)3 by intraperitoneal route. At day 14, mice were boosted with 10 μg OVA in saline solution. Seven days after the last antigenic dose, mice were killed for evaluations. Sera were separated for measuring anti-OVA antibody levels in ELISA tests, and spleens and lymph nodes were removed aseptically for measuring antigen specific proliferation by MTT method. (a) The bars represent the mean ± SEM of the sum of absorbance values of individual serum dilutions from 1:100 to 1:12,800 in ELISA assays. (b, c) The bars represent the mean of absorbance ± SEM measured in the cultures subtracting naïve basal proliferation of spleen (b) and lymph node (c) cells. Cultures were performed in six replicates. Data are representative of two separate experiments. *P <0.05 compared with control group. Comparisons were carried out using one-way ANOVA followed by Bonferroni’s test.
Our results show that the adoptive transfer of BMDCs modulated with either L-NAME or NOHA led to the reduction of in vitro antigen specific proliferation of both spleen cells and MLN cells from immunized BALB/c mice in comparison to the transfer of non-modulated BMDCs. Interestingly, an increase in antigen-specific proliferation was observed in cultures of the spleen and MLN cells of immunized BALB/c mice adoptively transferred with non-modulated BMDCs compared with that observed in cell cultures of untreated mice (Figure 3b, c).
The secretion of IFN-γ, IL-4, IL-10, and TGF-β was more reduced in cultures of spleen cells obtained from BALB/c mice adoptively transferred with BMDCs modulated with either L-NAME or NOHA in comparison with cultures of cells obtained from mice treated with non-modulated BMDCs (Figure 3, D-M). However, compared to the control group (mice that did not receive BMDCs), secretion of IL-4, IL-10, and IFN-γ was upregulated in the cultures of spleen cells from BALB/c mice transferred with BMDCs modulated with the inhibitors or not.
Effects of administration of NOHA or L-NAME modulated BMDCs on immune response of DO11.10 mice
To evaluate the effects of L-arginine metabolism on the immune response of an OVA-TCR-specific model, unmodulated BMDCs or BMDCs modulated with L-NAME or NOHA were adoptively transferred to naïve DO11.10 transgenic mice. Antibody levels, antigen-specific proliferative response, and cytokine production were evaluated as previously described.
As depicted in Figure 4a, transfer of either non-modulated BMDCs or BMDCs modulated with iNOS or arginase inhibitors equally increase the specific humoral immune response of DO11.10 mice, in comparison to untreated immunized mice (dashed line). Antigen-specific proliferation of spleen T cells was not affected by adoptive transfer of either modulated or non-modulated BMDCs (Figure 4b). A slightly increased proliferation of antigen-specific cells was observed only in cultures of MLN cells from DO11.10 mice adoptively transferred with NOHA-modulated BMDCs (Figure 4c).

Adoptive transfer of BMDCs modulated with L-NAME or NOHA does not affect antibody levels and T-cell proliferation but increases cytokine levels in DO11.10 mice. To perform the adoptive transfer, DO11.10 mice (n = 6 per group) were injected by intravenous route with 5 × 105 BMDCs cultured in the presence of L-NAME (1 mM) or NOHA (750 μM) for 48 h. A group of transgenic mice that received BMDCs cultured in the absence of inhibitors was included as the control. One day later, mice from all groups were challenged with 10 mg OVA + 1 mg Al(OH)3 by the intraperitoneal route. At day 14, transgenic mice were boosted with 10 mg OVA in saline solution. Seven days after the last antigenic dose, mice were killed for evaluations. Sera were separated for measuring anti-OVA antibody levels in indirect ELISA, and spleens and lymph nodes were removed aseptically for measuring antigen specific proliferation and cytokine production in supernatants. Cells were cultured in the presence of 100 μg/mL OVA for 96 h. Supernatants were collected, complete mediums containing MTT were added to the wells, and plates were incubated for an additional 4 h. (a) Antibody levels: the bars represent the mean ± SEM of the sum of absorbance values of individual serum dilutions from 1:100 to 1:12,800 in ELISA assays. (b, c) Cell proliferation: the bars represent the mean of absorbance ± SEM measured in the cultures subtracting naïve basal proliferation of spleen (b) and lymph node (c) cells. Cultures were performed in six replicates. (d–l) Cytokine levels: the bars represent the mean ± SEM of cytokine concentrations in culture supernatants. Data are representative of two separate experiments. *P <0.05 compared with control group. Comparisons were carried out using one-way ANOVA followed by Bonferroni’s test.
As illustrated in Figure 4, IL-2 was more elevated in supernatants of spleen cells and MLN cells from DO11.10 mice treated with either non-modulated or modulated BMDCs in comparison to untreated mice (dashed line), but cells of both compartments collected from mice adoptively transferred with L-NAME-modulated BMDCs was less effective in producing this cytokine (Figure 4d, e). IFN-γ levels rise in cultures of MLN cells from DO11.10 mice transferred with L-NAME-modulated BMDCs in comparison to untreated mice, but were lower in supernatants of MLN cells from transgenic mice transferred with NOHA-modulated BMDCs (Figure 4g). IL-4 levels also rise in the cultures of spleen cells of all groups in comparison to untreated mice, except in cell cultures from mice that received NOHA-modulated BMDCs (Figure 4h, i). Although statistically non-significant (due to high dispersion), an augment of IL-10 was observed in supernatants of spleen cell cultures from DO11.10 mice treated with BMDCs modulated with L-NAME or NOHA (Figure 4j, k). TGF-β was not detected in spleen cell cultures and was below the control group in cultures of MLN (Figure 4l, m).
Discussion
Mononuclear phagocytic cells such as macrophages and DCs use two types of enzymes to metabolize L-arginine: arginase (Arg) and inducible nitric oxide synthase (iNOS). The Arg I and II are able to metabolize L-arginine into L-ornithine and urea, while the iNOS convert L-arginine to L-citrulline and NO.26,54 Although iNOS and Arg are well studied in macrophages, there are few reports related to the role of these enzymes on DC functions.24,30
Although there are some studies on the consequences of inhibition of iNOS and arginase in DCs, literature has already shown that the blockage of NOS by Manassantin A (MSA), an inhibitor of the nuclear transcription factor-kappa B (NF-κB) led to a reduction of the expression of MHC I, MHC II, CD40, CD80, and CD86 molecules in BMDC, even in the presence of LPS. 55 As a consequence of treatment with MSA, the authors report that the maturation of DCs is compromised as well as their ability to induce allogeneic T-cell activation. Myeloid-derived suppressor cells (MDSC), which include immature macrophages, granulocytes, DCs, and myeloid cells, from a CD80-deficient mouse, showed reduced Arg1 expression and activity that correlates with accentuated tumor progression. 56 The phenotypic changes in DCs observed in the present study arising from the use of iNOS and arginase inhibitors suggest that the APC function of these cells may be compromised. To test this hypothesis, adoptive transfer experiments BMDCs modulated with L-NAME and NOHA were conducted in wild-type BALB/c mice and in DO11.10 transgenic mice.
There are few data in the literature correlating alterations in antibody production and the catabolism of L-arginine, most of them coming from a transgenic mouse deficient in one of the two enzymes. In this regard, it has been shown that the NOS2-deficient mouse infected with Influenza A virus presents virus titers in the lungs significantly lower than those found in the wild-type mouse. This enhanced viral clearance seems to correlate with the production of higher levels of virus-specific IgG2a antibody and of IFN-gamma by spleen cells from deficient mice. 57 On the other hand, Noda et al. showed that iNOS-deficient mice were more susceptible to lethal infection with cytomegalovirus than iNOS-expressing mice, and generated a much higher peak virus titers in the salivary gland after acute infection. 58 However, macrophage-Arg1-deficient mice infected with the gastrointestinal helminthes Trichuris muris showed antibody levels comparable to wild-type animals in both acute and chronic infection. 59 In relation to iNOS and arginase inhibitors, the literature data have associate treatments with L-NAME to increase in IgG2a isotype (from Th1 responses), whereas treatments with NOHA have been correlated with increased levels of IgG1 (from Th2 responses).14,17–20,40–42
Although there are no specific data relating to the effects of inhibition of iNOS and arginase on adoptively transfer model of BMDC, literature data show conflicting results regarding the effects of the absence of iNOS in APCs upon T-cell proliferation. In this regard, Hoffmann et al. showed that CD8 T-cell proliferation can be inhibited by lesser amounts of APC-derived NO than is necessary to inhibit CD4 T-cell proliferation. 13 In an attempt to identify the underlying molecular mediators of immunosuppression, Ren et al. examined how apoptotic cells induce immunoregulation by DCs. 60 They found that normal DCs co-cultured with apoptotic DCs completely lose their ability to support T-cell activation. This effect, however, can be reversed by inhibitors of iNOS. The authors suggested that the exposure to the apoptotic cells rendered normal DCs capable of producing much more NO, such as in a tolerogenic prolife. 60
Interestingly, all measured cytokines (IL-2, IFN-γ, IL-4, IL-10, and TGF-β) were significantly increased in MLN cultures obtained from BALB/c mice that received BMDCs modulated with NOHA. These findings appear to indicate that modulation of arginase and iNOS leads to different results depending on the immune compartment to which the DCs migrate. On the other hand, non-modulated BMDCs seem to increase the immune response as a whole. A possible explanation is that after transfer to immunized mice, non-modulated immature BMDCs probably undergo maturation in vivo leading to the activation of naïve T cells toward effector populations Th1 and Th2. 3 Although there are some reports showing that L-NAME improve Th1 response while NOHA reduces this kind of immunity,14,17–20,40–42 our work differs from those since it shows that the transfer of BMDCs modulated with these inhibitors reduces both Th1 and Th2 responses in the spleen, but increases the two types of responses in lymph nodes associated with the intestine.
The activity of both iNOS and Arg1 can also be correlated with the capacity of DCs to expand and differentiate populations of regulatory T cells in the periphery.35,36 In this regard, we observed that BMDCs treated with saline or L-NAME were able to expand in vitro the population of CD4+CD25+Foxp3+ cells in co-cultures with TCD4+ cells. Besides, BMDCs modulated with L-NAME and NOHA were also able to increase the expression of CTLA-4 and CD28 molecules on T cells. Since the CTLA-4 molecule has a higher affinity to DC receptors (CD80/CD86) than CD28, it can be an explanation to the downregulation of proliferation and cytokine secretion.6,61
Since adoptive transfer of non-modulated BMDCs increases immune response of BALB/c mice by augmenting antibody levels, proliferative response, and cytokine secretion, a possible explanation is that these cells undergo in vivo maturation leading to the high activation of naïve T cells toward both effector populations Th1 and Th2. 3
In agreement with our data, it has been shown that depletion of L-arginine through arginase in cells of myeloid origin is an important mechanism of immunosuppression.62,63 The depletion of L-arginine in tumor cells affected the activation of T cells by inhibiting re-expression of ζ chain CD3 molecule after the internalization of Ag-TCR complex, which seems to be an efficient mechanism of tumor escape. 28 Also, “classically activated” or “killer” murine macrophages (M1) can be differentiated from “alternatively activated” phenotype or “healer” macrophages (M2) by how they metabolize L-arginine. M1 macrophages produce iNOS in response to LPS, which uses L-arginine as a substrate to produce NO and higher levels of pro-inflammatory cytokines. On the other hand, M2 macrophages constitutively produce the enzyme arginase I, which sequesters L-arginine and results in the production of ornithine and downstream polyamines and L-proline.64,65 Recent data showed that oral supplementation with an inhibitor of arginase named nor-NOHA was able to improve adipose tissue inflammation in obese subjects. The authors observed that this treatment altered macrophage phenotype from M1 to M2 in adipose tissue by upregulation of IL-10 messenger RNA. 66
The NOS isoforms have received considerable interest since these molecules are potential targets of drugs designed for several pathological conditions. 67 Inducible NOS-derived NO contributes to pathological processes, in particular neurodegenerative disorders, inflammation, and pain.67–70 It has been shown that iNOS inhibition with L-NAME restores antigen-specific T-cell proliferation and reduces the percentage of apoptotic cells in cultures of spleen cells in experimental autoimmune encephalomyelitis (EAE). 71
Increased expression of Arg1 in inflammatory infiltrates has been demonstrated in several murine models of disease as experimental glomerulonephritis, trypanosomiasis, schistosomiasis, leishmaniosis, viral and bacterial diseases, asthma and pulmonary fibrosis, and experimental autoimmune encephalomyelitis.72,73 In relation to asthma, it has been shown that the administration of nor-NOHA significantly reduced the inflammatory process induced by treatment with OVA in a model of antigen-induced airway inflammation.74,75
It has been shown that decreased arginine availability by myeloid-derived suppressor cells led to T-cell dysfunction and increased susceptibility to infection after physical injury. 76 In agreement with our data, these effects were reversed by arginase inhibitor nor-NOHA, but reproduced by adoptively transferring myeloid-derived suppressor cells or injecting arginase 1 into control mice. 76 Furthermore, suppressed CD8+ T-cell proliferation was attenuated by the iNOS inhibitor aminoguanidine hydrochloride and arginase inhibitor N-hydroxy-nor-l-arginine (nor-NOHA). 77 It has also been shown that MDSC play important immunosuppressive role in several types of cancers, including head and neck squamous cell carcinoma (HNSCC). 56 In this regard, Vasquez-Dunddel et al. 78 showed that MDSC collected from tumor, draining lymph nodes, and peripheral blood from HNSCC patients inhibit autologous antigen-independent T-cell proliferation. MDSC from all three compartments expressed high phosphorylated STAT3 levels that correlate with arginase-I expression levels and activity. The authors also showed that Stattic, a STAT3-specific inhibitor, as well as repletion of L-arginine or treatment with nor-NOHA, abolished MDSC’s suppressive function.
Literature showed that T cells obtained from the spleens of DO11.10 mice are 80% of CD4+ T cells that bore the DO11.10 transgenic TCR (OVA-specific). 79 It has been shown that i.p. injection of OVA leads to central and peripheral depletion of OVA-specific T CD4+ cells in DO11.10 mice (MURPHY; HEIMBERGER; LOH, 1990). MacConmara et al. 81 confirm that DO-11 mice have a significant population of DO-11 TCR-Tregs, which could influence the OVA323–339 peptide immune response when co transferred into Treg-depleted mice. 81 Data obtained in our lab, however, shows that even in the presence of extensive reduction of mature anti-OVA T cells, DO11.10 mice are able to produce a significant humoral immune response following immunization with OVA. 5 Since anti-OVA specific T cells represent the major population of TCD4+ cells in peripheral blood of DO11.10 mice, 80 transfer of modulated BMDCs would not be enough to modify the immune response in these transgenic mice. In fact, as we previously report, DO11.10 mice were not susceptible to the induction of oral tolerance or adoptive transfer of DCs from tolerant mice, as measured by its antibody levels, proliferation of T cells, and levels of CD86 molecules. 5 Data obtained here show that adoptive transferring of iNOS- or arginase-blocked BMDCs to the transgenic mice had the ability of modify the cytokine production in spleen and MLN cell cultures, but had few reflex in its in vivo immune response. The literature shows that T cells obtained from the spleens of DO11.10 mice are 80% of CD4+ T cells that bore the OVA transgenic TCR (OVA-specific).8,9,82 As we already described, DO11.10 mice are highly responsive to OVA, including orally.5,10,83 So, we can presume that discrepancies among BALB/c and DO11.10 response can be due to the elevated number of OVA-specific TCR found in DO11.10 mice.
Hultkrantz and collaborators 84 adoptively transferred cells from DO11.10 transgenic mice and evaluated the response in different lymphoid organs. Antigen-specific CD4+ T cells were activated in the liver-draining celiac lymph node (CLN) after ovalbumin feeding. The significantly higher proportion of the T cells in the CLN developed into the putative regulatory phenotype than in Peyer’s patches, the mesenteric and peripheral lymph nodes, and the spleen. The expression of FoxP3 was not upregulated in any of the lymph node compartments. The phenotypic and functional findings suggest that the induction of regulatory T cells in the CLN may be relevant in the control of the immune response to dietary antigens. Also, Zinselmeyer et al. 85 tracked antigen-specific CD4+ T lymphocytes in real time, in situ during the induction of oral priming versus oral tolerance. They observed a marked contrast with respect to rate and type of movement and clustering between naïve T cells and those exposed to immunogenic or tolerogenic antigen. Egan et al. 86 adoptively transferred CD4+, KJ1-26+ T lymphocytes from transgenic mice to BALB/c mice. These authors suggested that repeated and systemic exposure of T cells to their cognate Ag in the absence of “danger” signals induces initial T-cell priming and clonal expansion, but results in an anergic phenotype, corroborating our data.
Also, our data showed that iNOS and Arg modulation inhibit the function of in vitro generated DCs. The treatment with iNOS and Arg inhibitors leads to augmented expression of co-stimulatory molecules that are important for antigen presentation by DCs. Adoptive transfer of BMDCs treated with L-NAME or NOHA was able to modulate the immune response in immunized BALB/c mice, mimicking the functions of tolerogenic DCs. However, none of the treatments with BMDCs was able to modulate the immune response of DO11.10 transgenic mice.
The present study indicates that modulation of BMDCs with pharmacological inhibitors of NOS and Arg provides a mechanism of inhibition of antigen-specific responses, possibly by increasing co-stimulatory molecules on effector T cells and stimulating Treg cells in BALB/c mice. Although the modulatory mechanism may be related to co-stimulatory molecules and Treg cells, further studies on the mechanisms of action are still necessary. This study provides evidence that L-arginine metabolism in DCs may be an important therapeutic target for controlling immune responses in inflammatory disorders.
Footnotes
Acknowledgements
The authors thank Marcos Cesar Meneghetti for helping with animal care.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by São Paulo Research Foundation (FAPESP) (grant nos. 05/51520-8, 06/60128-7, 2016/21424-1, and 08/05466-0 and fellowship no. 2005/03507 [PUS]), Fundo de Apoio ao Ensino, a Pesquisa e a Extensão - FAEPEX/UNICAMP (grant no. 132/08), and National Counsel of Technological and Scientific Development-CNPq (fellowship no. 142333/2005-0 [LGRF]).
