Abstract
The purpose of this investigation was to determine the lymphocyte subset response to 30 min of moderate treadmill exercise during caffeine supplemented (6.0 mg.kg−1) and placebo conditions in caffeine-naïve and -familiar individuals. Seventeen individuals participated (caffeine-familiar = 8, caffeine-naïve = 9) completing two exercise bouts (caffeine supplemented and placebo control) 48 h apart in a counterbalanced and double-blinded fashion. Individuals were classified as follows: caffeine-naive <50 mg.d−1 and caffeine-familiar >200 mg.d−1. Whole blood samples were obtained at rest, 30 min after caffeine or placebo ingestion, immediately following exercise, and 1 h post exercise. Blood was used to analyze apoptosis (annexin V) and cellular migration (CX3CR1) responses in lymphocyte subsets (CD4+, CD8+, CD19+). Absolute changes from rest values were calculated and differences between conditions were determined through Chi-squared analysis with significance accepted at P <0.05. With regard to CD4+ and CD19+ lymphocytes, the interaction of caffeine and exercise did not affect naïve individuals to a greater extent immediately post exercise when compared to familiar, as similar apoptotic and migratory responses were observed (P >0.05). However, CD8+ lymphocyte cell death and migration responses were observed to be significantly greater at each sampling point in caffeine-familiar individuals (P <0.05). It is possible that chronic caffeine supplementation may prime CD8+ cell receptors for responsiveness to apoptosis and migration and the consequence of this form of immunosuppression in the post-exercise period should be determined.
Keywords
Introduction
The immune system is charged with maintaining overall health, and is one of the most dynamic of the body’s systems. When the physiological stress of exercise is undertaken, the immune system displays a characteristic increase in cell volume during activity followed by a decrease upon cessation.1,2 Recently, our laboratory group has focused on the lymphocyte response that occurs due to various modes of exercise, in an attempt to define the mechanism responsible for the decrease of these cells in the recovery period, termed exercise-induced lymphocytopenia.3–6 Several of our papers have provided evidence that the interplay between cellular migration and programmed cell death is responsible for the observed lymphocytopenia response.7–9
Various factors affect the dramatic decrease in lymphocyte volume following exercise. Exercise intensity appears to have the greatest influence on the post-exercise response, 8 but frequency and duration also have important effects. 9 One factor that has received little attention to date is the effect of nutritional supplements or ergogenic aids. Caffeine is one of the most widely utilized ergogenic supplements 10 because it increases endurance performance, 11 perhaps through a direct effect on muscle activation or neural processes such as adrenergic activation. 12 To our knowledge, only two investigations have assessed the effect of caffeine supplementation during exercise on the lymphocyte response.13,14 Bishop et al. found that caffeine ingestion of 6.0 mg.kg−1 1 h before a bout increased the resting concentration of both CD4+ and CD8+ lymphocytes prior to exercise, and while 90 min of cycle exercise at 70% VO2max did not have any effects on the overall count, activated subsets of both t-cell type (CD4+/CD69+, CD8+/CD69+) were significantly greater than baseline. 14 Utilizing a similar caffeine loading and exercise scheme, Fletcher and Bishop found an immunosuppressive effect on trained cyclists 1 h following the exercise bout, as antigen-stimulated CD4+/CD69+ lymphocytes were significantly decreased compared to a placebo condition. 13
It is apparent that a caffeine and exercise interaction has the potential to affect the lymphocyte response during activity. What is less apparent is the consequence with regard to lymphocyte subsets and the apoptotic or migratory response during the post-exercise period in caffeine-familiar and caffeine-naïve individuals. We hypothesized that the combination of caffeine and treadmill exercise would have a greater effect on caffeine-naïve individuals, and that the CD8+ subset would display the greatest modulation. Therefore, the purpose of this investigation was to determine the lymphocyte subset response to 30 min of treadmill exercise at an intensity between 60–80% heart rate reserve in caffeine supplemented and placebo conditions.
Materials and methods
Participants
Seventeen healthy college-age individuals (6 men, 11 women) volunteered to participate in this study. Based on our previous research, no differences for the lymphocyte apoptotic response between men and women were expected, thus participants were grouped into caffeine-familiar or caffeine-naïve as described below. 15 All participants completed a medical history questionnaire and a caffeine consumption questionnaire which included all common sources of caffeine. The general characteristics of the individuals are presented in Table 1. Using the responses from their caffeine consumption questionnaire, nine participants were labeled as caffeine-naive (<50 mg.d−1) and eight labeled as caffeine-familiar (> 200 mg.d−1). Only participants with a “low-risk” status according to the American College of Sports Medicine risk stratification algorithm 16 were allowed to participate. Prior to participation, each participant signed an informed consent form that was approved by the University Institutional Review Board for the ethical treatment of human participants in accordance with the ethical standards as found in the 1964 Declaration of Helsinki and its later amendments.
Participant characteristics.

Participants refrained from caffeine intake for 24 h prior to the study (i.e. no coffee, tea, nicotine, chocolate, soft drinks, energy drinks, etc.) and were instructed to eliminate all extra sources of caffeine from their diet during the study period.
Caffeine administration
The experimental caffeine dose (6.0 mg.kg−1) was administered to all participants according to their respective body weight (mg.kg−1) to assure blood caffeine saturation.11,17 The caffeine dose, as well as the placebo (Crystal Light alone) was given in a 10-oz water bottle with lemonade flavor on the first trial and raspberry lemonade flavor before the second trial. Participants completed two testing sessions (6.0 mg.kg−1 or placebo) 48 h apart using a double-blinded and counterbalanced design.
Maximal exercise testing
All participants completed a VO2max treadmill test that began with a walking warm up at 80.5 m.min−1 (3 mph) for 3 min. The running protocol began at 150.2 m.min−1 (5.6 mph) and increased by 16.1 m.min−1 (0.6 mph) every 2 min until volitional fatigue to determine an appropriate workload corresponding to 60–80% heart rate reserve (HRR). 18 Heart rate (HR), rate of perceived exertion (RPE), VO2, and speed were recorded.
Experimental exercise protocol
On a separate day at least 72 h after the test for VO2max, a submaximal treadmill run was performed. Participants were instructed to report to the laboratory well hydrated, not to consume any meal at least 3 h prior to testing, and to not exercise prior to the testing session. A resting blood sample was obtained and then caffeine or placebo was administered as described above, and a post-ingestion resting blood sample was obtained 30 min later. Participants warmed up for 3 min at 80.5 m.min−1 (3 mph), and then jogged at the speed that corresponded to 60–80% of the HRR. Speed was adjusted to allow participants to maintain heart rate within 60–80% of the heart rate reserve. HR was continuously measured (Polar, Oy, Finland). Blood samples were taken immediately after and 1 h post exercise. On a separate day at least 72 h following this test, the final trial (either caffeine or placebo) was completed as described. Experimental trials were completed in a counterbalanced order.
Blood analysis
All flow cytometry antibodies and buffers were obtained from e-Bioscience (San Diego, CA, USA) and Bio Vision (Bio Vision, Mountain View, CA, USA). The investigated lymphocyte subsets were: CD4+, CD8+, and CD19+ with cell surface markers annexin V+ for cell apoptosis and CX3CR1 for cell migration. Blood samples were taken through a finger stick and 200 μL whole blood was collected into tubes (Sarstedt, Newton, NC, USA). A total of 20 μL blood was placed in each of the six tubes, three contained Annexin-V-FITC and three contained CX3CR1 which were prepared with standing buffer (eBioscience, San Diego, CA, USA) and respective antibodies. After incubation (30 min), samples were centrifuged for 5 min, decanted, and thoroughly vortex before the addition of red blood cell lysis buffer (15 min). Phosphate buffered saline was added, and samples were centrifuged, decanted, and vortexed before analysis by flow cytometry (C6, Accuri, Ann Arbor, MI, USA) to determine cell counts, apoptosis, and migration. At least 10,000 events were counted in the lymphocyte gate, initially determined from front and side-scatter characteristics. Further gating to distinguish CD4+ and CD8+ populations was determined in the FL-2 channel via the PE fluorochrome. The markers for apoptosis (annexin V) or migration (CX3CR1) were further noted for cells in each subfraction via the FITC fluorochrome and FL-1 channel.
Statistical analysis
Absolute changes from rest (Δ baseline) values were calculated according to the following formula: ((measure-baseline)·baseline-1)·100. As we expected the absolute change from rest with regard to apoptotic and migratory markers to be similar to the change in cell volume, the Chi-squared test (χ2) was utilized. A large effect size was anticipated based on a previous investigation from our laboratory, 19 but to use a conservative approach sample size was calculated using a medium effect size corresponding to a 25% relative increase in post-exercise apoptosis. To detect this increase in lymphocyte apoptosis, it was determined that a minimum of eight participants per group was necessary. The statistical analyses were performed using SPSS 20.0 (SPSS, Chicago, IL, USA).
Results
Trial intensity
The average HR throughout the first trial was 157.53 ± 2.41 beats·min−1, and was not significantly different when compared to the second trial (156.82 ± 2.58 beats·min−1, P = 0.57). Additionally, no differences were noted between groups with respect to average HR for the first trial (caffeine-familiar HR = 158.38 ± 4.20 beats·min−1, caffeine-naïve HR = 156.78 ± 2.84 beats·min−1, P = 0.76) or during the second trial (caffeine-familiar HR = 157.88 ± 4.08 beats·min−1, caffeine-naïve HR = 154.78 ± 3.30 beats·min−1, P = 0.57).
CD4+ helper T lymphocytes
Caffeine supplementation affected the CD4+ lymphocyte response in naïve individuals differently than familiar individuals. Whereas caffeine ingestion had no pre-exercise effect on familiar individuals, it significantly increased the percentage of apoptotic helper T lymphocytes prior to the treadmill run (P = 0.0001; Figure 1). Caffeine consumption significantly increased CD4+ apoptotic cells in both familiar and naïve participants post exercise (P = 0.0001), and the response persisted to at least 1 h post exercise in the naïve group (P = 0.0001). Cellular migration was also differentially affected by caffeine ingestion with the familiar group showing significantly reduced CD4+/CX3CR1+ percentages at 1 h following the exercise bout (P = 0.03).
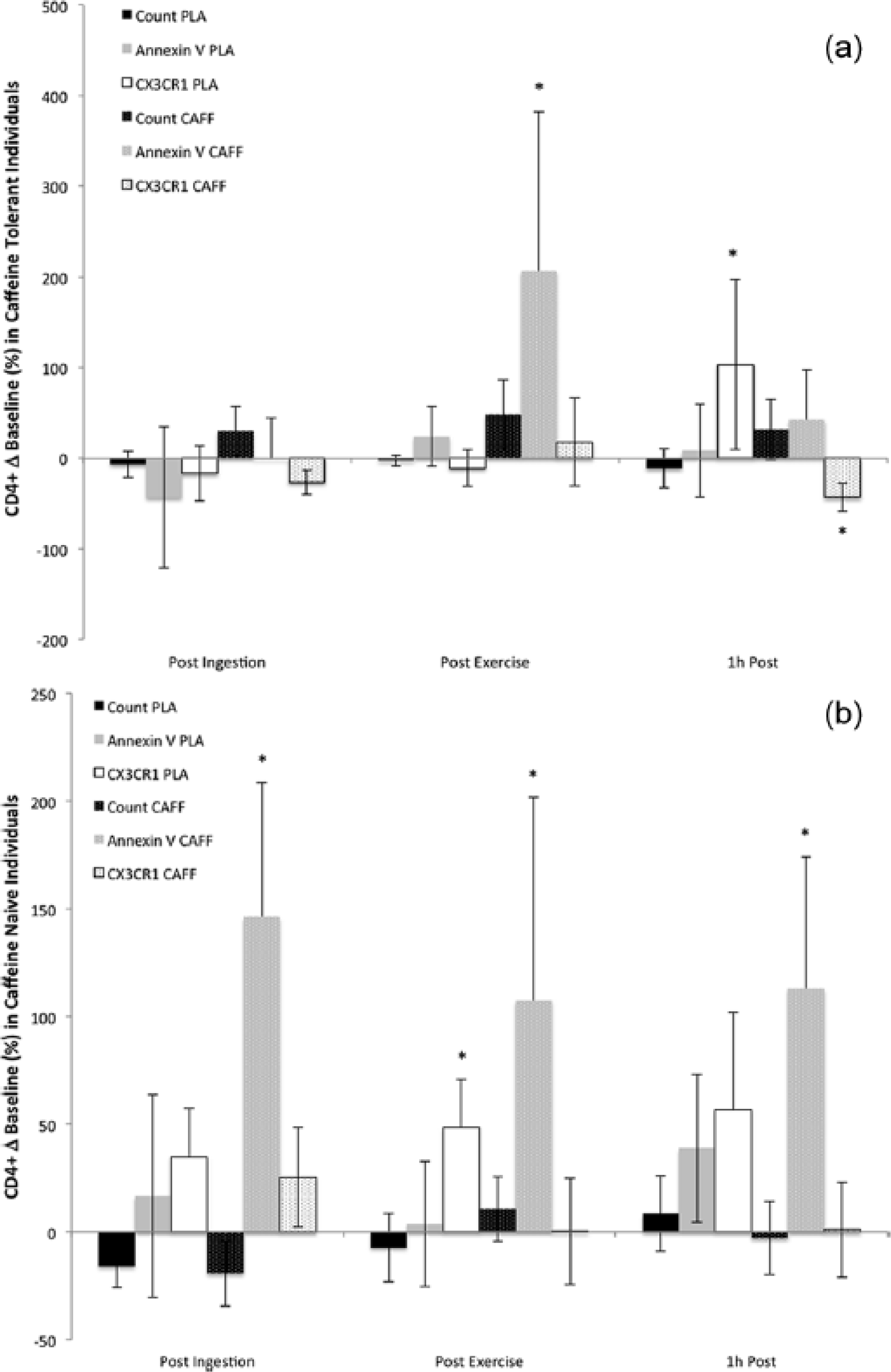
(a) Change (Δ) in helper T lymphocyte (CD4+) count, apoptosis (Annexin V), and migration (CX3CR1) compared to baseline values in caffeine familiar participants under conditions of caffeine supplementation (CAFF) and placebo (PLA) following 30 min ingestion (Post Ingestion), after 30 min moderate intensity treadmill running (Post Exercise), and at 1 h after the bout (1h Post). (b) Change (Δ) in helper T lymphocyte (CD4+) count, apoptosis, and migration compared to baseline values in caffeine-naïve participants that received of caffeine supplementation and placebo following 30 min ingestion, after 30 min moderate intensity treadmill running, and at 1 h after the bout.
CD8+ cytotoxic T lymphocytes
Caffeine supplementation differentially affected the CD8+ lymphocyte response in naïve and familiar participants. Caffeine ingestion significantly increased both apoptosis and migration markers in familiar participants at each sampling point (P = 0.0001), and had no effect at any time point in naïve individuals (Figure 2). However, in the placebo condition naïve participants displayed significantly increased apoptotic CD8+ lymphocytes post ingestion (P = 0.001) and at 1 h post exercise (P = 0.001; Figure 2b).

(a) Change (Δ) in cytotoxic T lymphocyte (CD8+) count, apoptosis (Annexin V), and migration (CX3CR1) compared to rest in caffeine-familiar participants under caffeine supplementation (CAFF) and placebo (PLA) conditions following 30 min ingestion (Post Ingestion), after 30 min moderate intensity exercise (Post Exercise), and at 1 h after the bout (1h Post). (b) Change (Δ) in cytotoxic T lymphocyte (CD8+) count, apoptosis, and migration compared to baseline values in caffeine-naïve participants that received of caffeine supplementation and placebo following 30 min ingestion, after 30 min moderate intensity treadmill running, and at 1 h after the bout.
CD19+ B lymphocytes
Compared to the placebo condition in familiar participants, caffeine supplementation significantly increased B lymphocyte migration post ingestion (P = 0.03) as well as post exercise (P = 0.001; Figure 3a). Additionally, B lymphocyte apoptosis was significantly greater at the post ingestion (P = 0.0001) and post exercise sampling points (P = 0.03) and remained significantly elevated 1 h post exercise (P = 0.001). In naïve participants caffeine supplementation significantly increased migration at all time points (pre ingestion P = 0.0001, post exercise P = 0.0001, 1 h post P = 0.03), and had a greater effect on the post exercise apoptotic response (P = 0.0001) than the placebo condition (P = 0.03; Figure 3b).
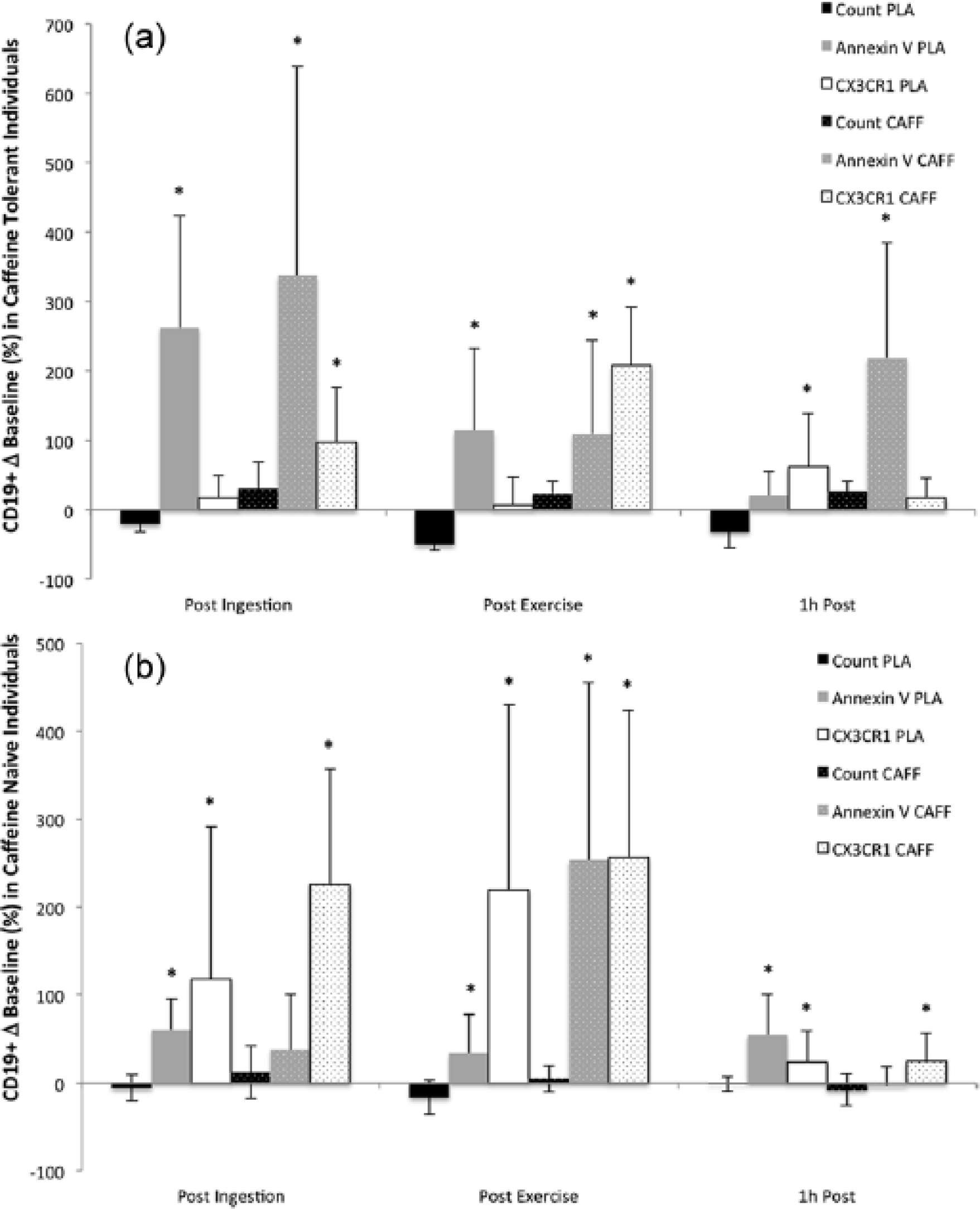
(a) Change (Δ) in B lymphocyte (CD19+) count, apoptosis (Annexin V), and migration (CX3CR1) compared to baseline in caffeine familiar participants following caffeine supplementation (CAFF) and placebo (PLA) after 30 min ingestion (Post Ingestion), 30 min moderate intensity exercise (Post Exercise), and at 1 h after the bout (1h Post). (b) Change (Δ) in B lymphocyte (CD19+) count, apoptosis, and migration compared to baseline values in caffeine-naïve participants that received caffeine supplementation and placebo following 30 min ingestion, 30 min moderate intensity treadmill running, and at 1 h after exercise.
Discussion
The aim of the present investigation was to determine the effects of caffeine supplementation on lymphocyte subset apoptotic and migratory responses following acute moderate exercise. It was hypothesized that the interaction between caffeine supplementation and treadmill exercise would have a greater influence on naïve participants, and that the CD8+ subset would display the greatest modulation. Overall, the combination of caffeine and exercise did not affect naïve participants to a greater extent immediately post exercise when compared to familiar participants, as similar apoptotic and migratory responses were observed for CD4+ and CD19+ lymphocytes. In addition, the cytotoxic lymphocyte cell death and migration responses were observed to be much greater at each sampling point in caffeine familiar participants which was counter to our hypotheses.
The present results correspond well with our previous findings utilizing increasing intensities of treadmill running, 8 resistance training, 7 and a simulated tennis match. 6 Exercise intensity in the present investigation was between 60–80% of the HRR, and includes the 76% of VO2max used previously which showed increases in CD4+ apoptosis, and CD8+ and CD19+ migration. 8 At the end of the caffeine-supplemented exercise period in the current study we found increases in naïve and familiar CD4+ apoptosis, increased CD8+ cellular migration in familiar individuals, and increased CD19+ cellular migration in naïve and familiar groups. Immediately following a resistance training bout designed for hypertrophy (3 sets of 9 exercises) significant increases were observed in CD4+ and CD8+ apoptosis and cell migration markers. 5 Additionally, a simulated tennis match (580 total serves and ground strokes) produced significant increases in CD4+ apoptosis, and in CD8+ apoptosis and cell migration. 6
Previous research into the interaction between caffeine supplementation and exercise has reported an immunosuppressive effect in the helper and cytotoxic T lymphocytes of trained cyclists following an exercise bout. 13 The results of the current investigation support and extend knowledge regarding the potential underlying mechanisms associated with this phenomenon. We found that caffeine supplementation significantly increased apoptosis markers in helper T lymphocytes immediately post exercise in both caffeine-naïve and -familiar individuals, with little effect on migration. Additionally, caffeine increased both cell death and migratory markers on cytotoxic T lymphocytes of the familiar group, but had no significant impact on naïve individuals. While not measured in the current study, it is possible that caffeine has an effect by altering the intracellular calcium concentration. 20 Caffeine has been shown to inhibit lymphocyte production of TNF-α and IL-2 in vitro, 21 as well as TNF-α expression from peripheral blood mononuclear cells obtained from healthy individuals. 22
Previous investigations utilizing caffeine supplementation and cycling exercise have found a greater response in cytotoxic T lymphocytes.13,14 It was proposed that these responses are mediated through adenosine/adenylate cyclase 23 and β2-adrenergic receptors/cAMP. 24 One interesting finding of the present study is that caffeine administration increased cell death and migration in cytotoxic T lymphocytes in the caffeine-familiar group only, suggesting that chronic supplementation may somehow prime receptors for responsiveness in this cell subset. Further investigation into this response and the underlying mechanism/consequences is warranted.
This study has a number of limitations, such as the reduced number of participants (although the achieved power was acceptable), absence of measurements of blood caffeine metabolites, and the control of dietary habits several days prior to testing sessions to reduce any residual caffeine tolerance which may have interfered with test results. While we failed to account for dietary habits that may have affected the exercise response, acute glucose supplementation has been reported to have no effect on T lymphocyte cell death, 25 and we are unaware of any investigations utilizing acute fat feeding and the exercise-induced lymphocyte apoptotic response. Another facet that can be considered a limitation is that we only provide data on numbers of cells expressing a particular surface marker that is associated with apoptosis or migration/cell adhesion and whether these cells effectively went into cell death or migrated into, for example, the vascular endothelium was not investigated. Another limitation is that we did not collect information regarding participants who may have utilized caffeine as an ergogenic aid prior to their normal exercise routines. As this practice has the potential to alter the immune responses reported in the present investigation, future studies should take this issue into account during their initial study design. Additionally, in attempt to control the study design we required the participants in the caffeine-familiar group to refrain from consumption for a 24 h period. It is possible that this influence could have affected the exercise interaction and response of lymphocyte subfractions, and that we were actually measuring a withdrawal response in these individuals. Future investigations utilizing caffeine-familiar individuals should take this particular issue into consideration.
In conclusion, our results indicate that cytotoxic T lymphocyte responses to cell death and migration are different to an exercise stimulus between naïve and familiar individuals. This finding should be taken into consideration in future exercise immunology-related investigations. As a magnified post-exercise lymphocytopenia response was observed in the CD8+ lymphocytes of caffeine familiar individuals, future studies should be directed toward determining the consequence of this form of immunosuppression and identifying the time course of this transient state. On the other hand, we found that caffeine status has no effect on the response of helper T- and B-lymphocytes as similar post-exercise changes in apoptosis and migration were observed between groups.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sector.
