Abstract
Small B-cell lymphocytic lymphoma/chronic lymphocytic leukemia, which typically affects elderly people, is a group of conditions that are not clinically uniform. It has been suggested that using the combined activity of the monoclonal antibody anti-CD20 (rituximab) and
Introduction
Small B-cell lymphocytic lymphoma/chronic lymphocytic leukemia (SLL/CLL), which typically affects the elderly, is a group of illnesses that are not clinically uniform. 1 In some SLL/CLL patients, the leukemia is slowly progressing and characterized by several years of survival without intensive therapy. In other patients, however, after an initially indolent period, serious progression of the leukemia occurs, most often correlated with the B-cell IgV mutated or unmutated status of leukemia cells. 2 Typically, chemotherapy with the anti-CD20 antibody rituximab (RTX) is used for treatment, depending on the type of the B-leukemia or lymphoma.
RTX (MabThera®) is a chimeric monoclonal antibody specific to the CD20-epitope on leukemia/lymphoma cells. It has been applied in clinical therapy for several years, and its therapeutic value is well described.3,4 Preliminary work carried out in our laboratory suggests that RTX cytotoxicity may be increased in the presence of the
LLO belongs to the bacterial hemolysin family of proteins with a molecular weight of 58 kDa. LLO molecules bind the cholesterol-containing domains of membranes, polymerize there, and form pores.5,6 The cytotoxic activity of LLO depends on its concentration. 7 A low concentration induces pores in the cell membrane, which allows for intracellular import of Ca2+ ions and activation of several cytoplasmic systems, finally inducing apoptosis. A higher concentration of LLO induces formation of larger pores, and the cell content effluxes, resulting in cell necrosis.7,8
The aim of the present work was cytometric analysis of B-lymphocytic leukemia cells following treatment with both LLO and RTX. The Raji cell line is especially useful as a model of human B-lymphocyte leukemia/lymphoma and RTX treatment because it expresses CD20; as a control, we used the Jurkat cell line, which does not express CD20. Our intention was to evaluate the combined effect of LLO and RTX activity because both induce cell membrane damage at higher cell cytotoxicity activity levels.9,10 It has been suggested that more effective B-cell killing by the combined activity of both components could possibly induce a better therapeutic effect.
Materials and methods
Cell lines and preparation of leukemia cells and cultures
The Raji (ATTC, CCL-86) human Burkitt lymphoma B-cell line is CD20, CD19 positive and CD3 negative. Jurkat (ATCC, CRL-2063) human leukemia T-cells are CD3, CD4 positive in 28% of cells and CD19, CD20 negative. The Jurkat cell line thus serves as a negative control for RTX activity. The cell lines were cultured in RPMI 1640 (Gibco®) medium supplemented with L-glutamine, streptomycin (TZF Polfa), penicillin (TZF Polfa) (100 units/mL penicillin with 0.1 mg streptomycin/mL), and 10% inactivated fetal bovine serum (FBS, Sigma). Experiments with cell lines were performed in plastic 48- and 96-cell culture cluster wells (Costar) at 37°C under 5% CO2 in a humidified incubator. Before each experiment, the medium was changed to fresh, and then the number of cells per mL of medium was determined before the experiment by dilution. The study protocol was approved by the Bioethical Commission at The Centre of Postgraduate Medical Education in Warsaw (17 July 2014).
Listeriolysin O
Modified LLO was produced by gen
Anti-CD20 antibody
RTX is a mouse–human chimeric IgG1 monoclonal antibody that has been well characterized (MabThera®, Rituximab, Roche). It was obtained at 10 mg/mL concentration (Hoffmann-La Roche AG).
Cytotoxicity test
The
Human peripheral blood plasma
Human peripheral blood plasma was obtained from the whole blood of healthy volunteers who have agreed to take part in the experiment. Peripheral blood taken in EDTA (final concentration, 0.5 mM) was centrifuged for 10 min RCF 440 ×
LLO cytotoxicity
The dilutions of LLO preparation in 10 μL saline were prepared in test tubes containing 0, 0.5, 1, 2, 3, 4, 5, 6, 8, or 10 μL LLO. Then 50 μL of the cell suspension was added as 1×106 cells/mL (Raji or Jurkat) in the culture medium with 10% inactive FBS, but without active human mixed plasma. After 30 min of incubation time, cytotoxicity was assayed with the PI and FDA test by cytometry.
Preparation of RTX mixtures with LLO
The protocol was optimized based on the preliminary experiments. A mixture was prepared containing 1 μL RTX (10 mg/mL) and 9 μL LLO (0.1 mg/mL) at an activity of 55–60 HU. In the control samples the volume of the second component was replaced by physiological saline. The reaction mixture was left at room temperature for 15 min (pH 7) with stirring to give time for interaction between sample components. Then the reacted mixture was briefly stored at 4°C until used in the assay.
Flow cytometry
In a FACS Canto II cytometer, at least 25,000 events were collected at 488 nm from each sample, and data were analyzed using the FACS Diva application (Becton Dickinson). Experiments were repeated three times. The negative control tests were performed in parallel. In some tests, the total number of events was compared using Peak Flow TM Green flow cytometry reference beads (Life Technologies).
Calculation of median lethal dose, LD50
The activity of LLO and RTX follows a sigmoid cytotoxicity course. The median lethal cell dose (LD50) was read from data of four tests for each analyzed cell line with different doses of LLO, RTX, and their mixture. The best fitted linear equation was obtained for points with a significant decrease in viability. Average results were transferred to a chart, and the best fitted linear equation of the received points was elaborated using the Cricket Graph program. In the brackets for each LD50 result, the coefficient of determination, R2, is presented to indicate how well data fit a statistical model.
Statistics
The cytometric results are presented as median values and percentile values, P25 and P75. Statistical analysis of cytometric experiments was performed using the global linear model after logit transformation of values. Results of hemolytic tests are presented as mean ± SD. Analysis of variance was performed, and differences were considered to be significant when
Results
LLO cytotoxicity
The cytotoxicity of LLO at 55–60 HU on Raji and Jurkat cell lines assayed without active human plasma was different (Figures 1 and 2). To assess differences between cell lines, the results of viability for each dose of LLO were averaged, and the best fitted linear equations were calculated. The method of calculation is presented as a graph (Figure 3). The Raji and Jurkat lines had different LD50 values and sensitivity at each analyzed LLO dose. We found that LLO cytotoxicity was higher for Jurkat than for Raji cells in all tested concentrations. The Jurkat cell line had lower viability for each dose of LLO, and its median viability of cells (LD50) was achieved at 2.25 µL while the Raji cell line had a higher viability and LD50 at 3.89 µL of LLO. The sensitivity to cytotoxic activity of LLO in Jurkat cells was 1.7 times that of Raji cells.

The cytotoxicity of LLO (0.1 mg/mL, activity 55 HU) on Raji and Jurkat cells. In the test, the cytotoxicity on the Jurkat cells after 30 min of incubation was higher (direct reading LD50 = 2.2) than that on Raji cells (direct reading LD50 = 4.1). Incubation time, 30 min at 37°C in the presence of 10% FBS. Cell concentration, 1×106 cells/mL. n = 4; error bars = SD.

Cytometric comparison of LLO cytotoxicity for Raji and Jurkat cells. Dot plot for selected doses of LLO 0 and 5 μL. The cytotoxicity after an incubation time of 30 min at 37°C was assayed with the PI and FDA tests. In gate P2 beads labelled with FITC were collected. For gate P1 events were analyzed on the right dot plot of FDA fluorescence versus PI fluorescence. Percent of living cells was read in the Q4 quadrant and is presented next to the dot plot. On the right side of the dot plot is the mean value of living cells as compared with the control. In the brackets are directly read mean values of living cells. n = 4; cell concentration 1×106 cells/mL.

LD50 calculation method for LLO (0.1 mg/mL) with an activity 55 HU on Raji and Jurkat cells. The linear equation is presented with the coefficient of determination (R2). The calculated reading of LD50 = 2.25 for Jurkat cells and LD50 = 3.89 for Raji cells was found.
RTX cytotoxicity assay with human plasma from different healthy volunteers and its optimal concentration
The cytotoxic activity of human plasma from different volunteers on Raji cell lines in the presence of 0.5 μg and 2 μg RTX differed significantly. The test suggested that the human peripheral blood plasma exerted individual complement activity at each concentration of RTX and is an individual feature. Results of cytotoxic activity of RTX observed with human plasma complement proteins from different individuals showed differences in activity with the Raji cells after 30 min of testing (Figure 4). Therefore, for further experiments, we used a mixture of an equal volume of human blood plasma from three healthy volunteers.
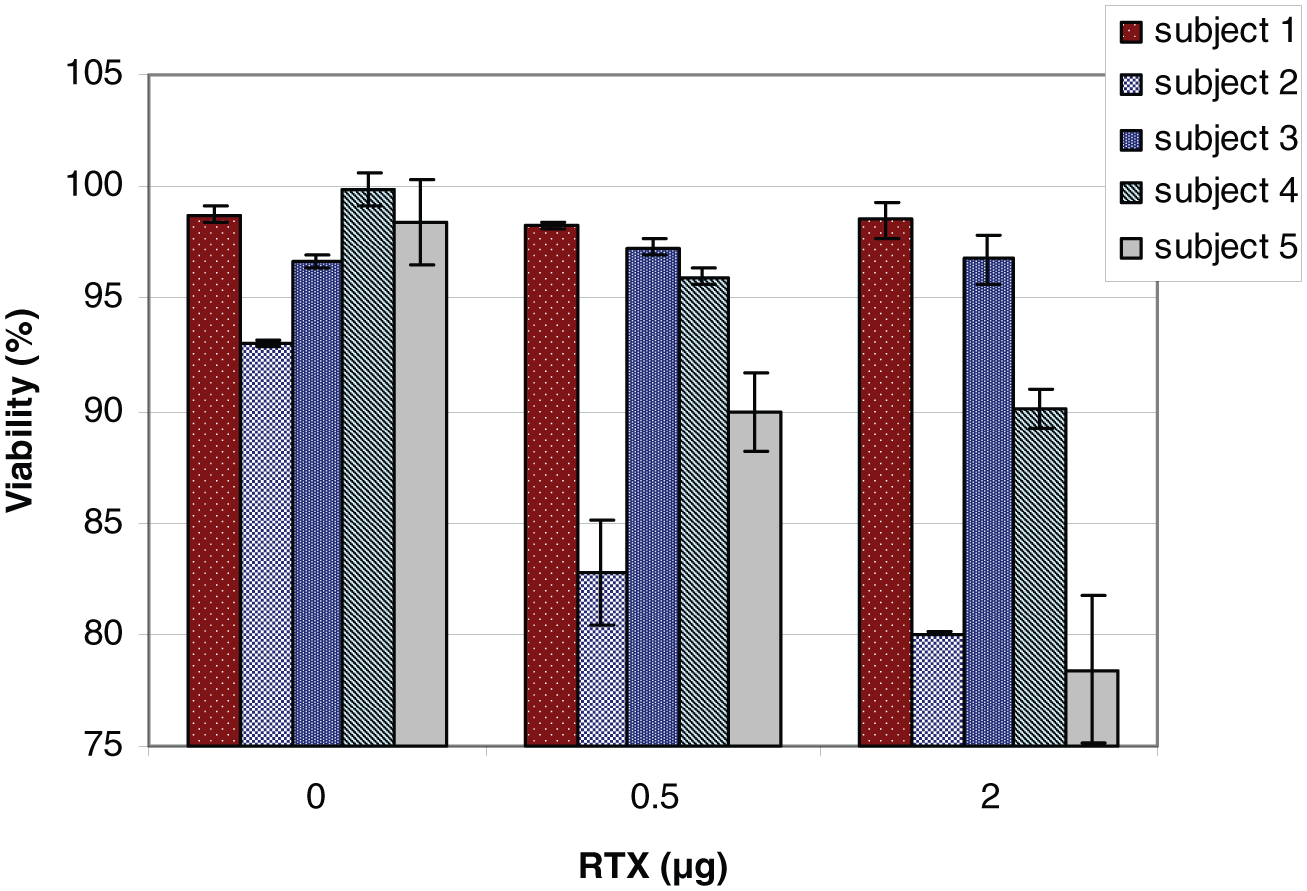
The activity of human plasma from five healthy volunteers on the Raji cell line in the presence of 0.5 μg or 2 μg RTX. Human plasma was obtained from different volunteers and applied at concentration 5%. Incubation time, 30 min; medium 10% FBS; n = 3; cell concentration, 1×106 cells/mL; error bars = SD.
To determine the activity of the mixed plasma from three healthy volunteers, we incubated Raji and Jurkat cells at a concentration of 1×106 cells/mL in standard conditions for 30 min in reaction medium in the presence of 10% FBS with the addition of different amounts of active mixed human plasma (1%, 2%, 3%, 5%, 10%) and with different doses of RTX (2 μg and 10 μg). With the increase in the mixed plasma concentration, a decrease in Raji cell viability was observed, greatest at the 5% concentration of mixed plasma. At the 10% concentration of mixed plasma for the 2 μg and 10 μg doses of RTX, gels/clots were observed in the test with Raji cells, which prevented proper execution of flow cytometry measurement. Under the same conditions, Jurkat cells at each plasma concentration for both doses of RTX showed no changes in viability. Based on these observations, we concluded that 5% concentration of the peripheral blood plasma mixture will be optimal for activity and repeatability of the applied method.
Human plasma cytotoxicity assay in the presence of RTX: Concentration dependence
To further distinguish the complement-dependent cytotoxicity (CDC) effect in the presence of RTX in Raji and Jurkat cells and to determine the optimal timing of experiments, measurements at different time points were conducted. After 30 min of incubation of the cell lines, the mean viability for the RTX doses of 0.5 μg and 2 μg was close to that of the control sample (Figure 5). Significant statistical differences (

Impact of RTX concentrations on Raji and Jurkat cells at 30 min and 120 min. Final RTX dose in the cell reaction medium, with 10% FBS and 5% human mixed plasma. n = 3;
RTX concentration dependence at 24 h is presented in Figure 6. In the cytotoxic test with Raji cells, RTX treatment at 0.05 μg/mL and 0.5 μg/mL in the presence of 5% human plasma resulted in 62% and 3% living cells on the PI test, respectively, and 50% and about 18% on the Trypan blue test (Figure 6; Table 1). With the same concentration and conditions, the impact on Jurkat cells was minimal, and close to 98–100% of living cells remained after treatment (Figure 6; Table 1). Active human plasma without antibody does not kill Raji or Jurkat cells. Figure 6 shows that the minimal inhibitory concentration of RTX assayed with both methods for Raji cells was in the range of 0.1–0.05 μg RTX in 1 mL at a concentration of 1–2×106 cell/mL. In analyzed range of dose RTX significant statistical differences for control cell line Jurkat were observed.

RTX impact on Raji and Jurkat cells at 24 h incubation. Final RTX dose in the cell reaction medium containing 10% FBS and 5% human mixed plasma. n = 3; error bars = SD.
Results of Trypan-blue viability test with Raji and Jurkat cells incubated 24 h with RTX in the presence of 5% of human plasma mixture.

Eukaryotic cell viability was evaluated microscopically with Trypan-blue and with the cytometric test with PI, and we considered this as the model for testing CDC in the presence of RTX for B lymphocytes. We found that the highest RTX activity was observed after 24 h, but at higher (>10 μg) concentrations, a nonspecific effect on Jurkat cells was observed. Consistent with previous findings, we found that the most optimal time frame was 120 min for evaluating LLO and antibody effects.
Cytotoxic activity of RTX and LLO combination on Raji (B-cell) or Jurkat (T-cell) cells
The activities of the components present in the suspension of Raji cells for 120 min depended on their concentration. Solutions of RTX, LLO, and their combination in the proportion 1 × 9 were prepared before application to cell suspensions. LLO applied at 0.5–5 µL (0.1 mg/mL) had low cytotoxicity for Raji or Jurkat cells. RTX treatment of Raji cells at 5 μL (10 mg/mL, 5 μg/cell sample) resulted in a decrease in living cells to 55–60% (Figure 7). The cytotoxic effect on Raji cells of the RTX combination with LLO was greater at each tested dose. The highest decrease in viability, 45%, occurred at the dose 5 μL of mixture RTX (5 μg/cell sample) and LLO (0.5 μg/cell sample). At this dose the significant statistical differences for

Result of survival analysis of Raji cells treated with gradually increasing concentrations of RTX (0.3 mg/mL), LLO (0.1 mg/mL), and a mixture of RTX and LLO. Mean ± SD;
A similar experiment was performed on Jurkat cells treated as the control cell line, and the results are presented in Figure 8. LLO or RTX had no effect on Jurkat cells within concentrations as tested before. For the combination of RTX 1 μL (10 mg/mL) and 9 μL LLO (0.1 mg/mL), cytotoxicity was detected at a dose of 2 μL (RTX 2 μg/cell sample LLO 0.2 μg/cell sample), and the viability was 90%; at 5 μL, viability was 65% and both values were statistically significant (

Result of survival analysis of Jurkat cells treated with gradually increasing concentrations of RTX (0.3 mg/mL) or LLO (0.1 mg/mL) or a mixture of RTX and LLO. Mean ± SD;
The data obtained from different tested substances allowed for calculation of the LD50 (Table 2). This step was helpful in planning further analyses.
The LD50 calculated from the results of the cytotoxic effect on Raji or Jurkat cells of LLO, RTX, or a mixture of RTX and LLO. The coefficient of determination, R2, is presented in brackets; n = 4.

The results may be summarized as follows: Addition of LLO at the tested concentration to the suspension of Raji cells or Jurkat cells did not result in significant differences in cytotoxic effects in either cell line. Under the tested conditions, the LLO cytotoxicity was similar for both cell lines and close to the dose of 0.95 µg. RTX at the tested concentration range had a cytotoxic effect on Raji cells, but not on Jurkat cells. No cytotoxicity was observed for the control Jurkat cells when the dose of RTX mixture was 5.6 μg. The LD50 value of the RTX cytotoxic effect on Raji cells increased about 1.5 times when LLO was present in the medium. The impact of the mixture of RTX and LLO on Jurkat cells was higher than LLO alone, and the LD50 value was achieved at the 6.89 μg dose of the mixture. RTX in the presence of LLO had a nonspecific cytotoxic effect on Jurkat cells but in comparison to Raji cells at lower cytotoxicity. The additive effect on Raji cells was observed for lower concentrations of the LLO and RTX mixture.
Discussion
Chemotherapy is a traditional treatment for cancer patients; however, the activity of the drugs applied is not selective and often at suboptimal doses. More selective targeting of tumors by cytotoxic drugs may be applied as chemoimmunotherapy with joint chemo- and antibody treatment. When RTX is applied alone in the treatment of B-cell leukemia or lymphoma, it is effective in about 50% of patients at 375 mg/m2 weekly for 4 weeks. 4 Leukemia cell resistance to therapy may be associated with expression of complement regulatory proteins such as CD59 on the surface of the target cells. 11 Because of these limitations, there is an ongoing effort for improvement of leukemia treatment by a combination of RTX with chemotherapy.12–14 It seems possible that some cell membrane toxins, such as LLO, might be helpful in achieving this goal. Here, we tested the effect of the combined activity of RTX and LLO in model human leukemia cell lines.
The mechanisms of action of RTX have been extensively investigated, but the intracellular events leading to apoptosis of target cells remain to be elucidated. The CD20 epitope binding RTX is localized on B-lymphocytes, at the large loop of the CD20 cell membrane surface protein. 15 RTX induces apoptosis in binding cells by a mechanism of the complement system (CDC) and/or cell antibody-dependent cytotoxicity (ADCC),3,16–18 as well as by the induction of apoptosis.19–22
RTX is chimeric antibody, with human IgG1 and κ constant regions, providing the Fc portion necessary for CDC or ADCC induction of apoptosis.18,20 Moreover, it has been hypothesized that the number of CD20 receptors on lymphatic cells determines sensitivity to RTX 23 and that consequently, certain cytokines may increase the efficacy of RTX by up-regulating the expression of CD20. 24
CD20 is a B lymphocyte specific membrane protein that has been suggested to function as a calcium channel. 25 Like most cell membrane proteins, it is localized within membrane domains called rafts and is directed there after antibody binding. 26 Moreover, the membrane rafts are known as cholesterol-enriched membrane domains and may be preferable sites for the cholesterol-binding toxin LLO. 6 In addition, RTX activity directly depends on cell membrane cholesterol content. 27 The antibody binding site and toxin binding site appear at the same region of the cell membrane, suggesting a potentially effective interaction of both factors (Figure 9). Binding of antibody to the cell membrane in the presence of the complement proteins induces formation of pores composed of the C9 protein complex, and almost at the same site, LLO binds to cholesterol and results in the formation of toxin pores. The hypothesized joint interaction of both components in the cell membrane pore formation implies the possibility of a more effective cytotoxic activity, as observed here.

Scheme of RTX and LLO activity on a CD20-positive cell. (a) LLO interacts with cholesterol mostly within lipid domain rafts, leading to the formation of pores in the lipid membrane. The created pores induce inflow of Na+ and Ca2+ ions to the cells and outflow of the cell components. (b) RTX binding induces activation of complement (CDC) and translocation of the CD20–RTX complex into the cell membrane rafts. Activation of this enzymatic cascade leads to the formation of pores in the lipid membrane and lysis of the target cell. (c) Both processes (a and b) occur within the same region of the cell membrane, inducing a higher cytotoxicity response than they do separately. (d) Control sample that does not present any factors that can trigger apoptosis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the European Union structural funds, Innovative Economy Operational Program POIG.01.01.02-00-109/09-00.
