Abstract
The global incidence of human papillomavirus (HPV)-positive head and neck squamous cell carcinoma (HNSCC) has increased markedly over recent decades, leading to a fundamental shift in the epidemiology and clinical management of head and neck malignancies. HPV-positive HNSCC represents a biologically and clinically distinct disease entity, characterized by unique molecular features, improved treatment responsiveness, and a more favorable prognosis compared with HPV-negative tumors. However, the optimal integration of molecular insights with evolving therapeutic strategies remains an ongoing clinical and translational challenge. In this review, we summarize the etiological role of HPV in the pathogenesis of HNSCC and delineate key differences in epidemiology, molecular biology, and clinical presentation between HPV-positive and HPV-negative disease. We further examine current standard-of-care treatments, including surgery, radiotherapy, and chemotherapy, with particular emphasis on de-escalation strategies aimed at reducing long-term toxicity while maintaining oncologic efficacy in HPV-positive patients. Emerging immunotherapeutic approaches have demonstrated encouraging activity in recurrent or metastatic disease. Moreover, therapeutic vaccine development, including DNA-, mRNA-, peptide-, and viral vector-based platforms targeting HPV E6/E7 oncoproteins, represents a rapidly evolving area with the potential to enhance antitumor immunity. This review also highlights ongoing clinical trials that may reshape the future landscape of treatment for HPV-positive HNSCC. Collectively, these advances emphasize the importance of HPV status in guiding personalized management strategies and improving patient outcomes.
Keywords
Introduction
Head and neck squamous cell carcinoma (HNSCC) is a group of malignancies arising in the oral cavity, pharynx, hypopharynx, larynx, and nasal cavity, ranking as the seventh most common malignancy worldwide.1,2 Human papillomavirus (HPV) was first linked to oral mucosal malignant squamous cell tumors in 1983.3–5 Over the past few decades, persistent infection with high-risk types of HPV has become a leading cause of oropharyngeal squamous cell carcinomas (OPSCC). 6 A retrospective analysis by Ang et al. identified tumor HPV status as a strong and independent prognostic factor for survival in patients with oropharyngeal cancer. 7 The role of HPV in non-OPSCCs is less well defined, but some reports suggest a possible association between HPV infection and non-OPSCC. Rooper et al. 8 reported that among 52 cases of HNSCC other than non-OPSCC, 65% exhibited classic histologic features of HPV-positive OPSCC, including endophytic growth, minimal keratinization, and hyperchromatic nuclei without koilocytic changes. Zhu et al. 9 found 131 cases (8.51%) of HPV 16 positivity among 1539 patients with non-OPSCCs. Therefore, a comprehensive evaluation of the epidemiological, diagnostic, and prognostic roles of HPV-positive HNSCC across different anatomical sites is essential for optimizing risk stratification and individualized treatment.
HPV is a non-enveloped virus with a circular double-stranded DNA genome that targets the basal cells of stratified epithelium. 10 While the immune system typically clears most HPV infections naturally, persistent infection with high-risk HPV types (e.g. HPV 16, HPV 18) can result in anogenital cancers (e.g. cervical, anal, vulvar, penile) or HNSCC.11,12 More than 200 HPV types have been identified. Among the 14 high-risk types, HPV-16 accounts for 90% of HPV positive HNSCC cases. 13 The oncogenic potential of high-risk HPV types is primarily mediated by the viral oncoproteins E6 and E7, which promotes malignant transformation, and tumorigenesis through integration into the host genome. 14
HPV-positive HNSCC often occurs in younger, non-smoking individuals and is associated with significantly improved prognosis. 15 However, conventional treatment regimens, especially high-dose chemoradiotherapy, often lead to severe long-term toxicities such as dysphagia, xerostomia, and neurocognitive decline. 16 The heightened sensitivity of HPV-positive HNSCC to chemoradiotherapy has sparked growing interest in emerging treatment de-escalation strategies aimed at minimizing long-term toxicity without compromising therapeutic efficacy. As a result, treatment de-escalation strategies have gained increasing attention, aiming to preserve excellent oncologic outcomes while minimizing morbidity. Concurrently, immunotherapeutic approaches and therapeutic vaccines targeting viral antigens such as E6 and E7 are being actively explored, given the intrinsic immunogenicity of HPV-positive tumors.
Despite these advances, several critical challenges remain. First, the optimal criteria for patient selection in de-intensification trials are still under investigation. Second, long-term data on oncologic outcomes and quality-of-life metrics following reduced-intensity treatments are limited. Third, while immunotherapy has shown potential in various cancer types, its role in HPV-positive HNSCC remains incompletely understood, with variable clinical responses and unclear biomarkers of efficacy. This review focuses on the evolving therapeutic landscape of HPV-positive HNSCC. We first outline its molecular pathogenesis, emphasizing the roles of viral oncoproteins and associated signaling pathways. Subsequently, we discuss current standard treatments, ongoing de-intensification efforts, and the therapeutic potential of immunotherapy and vaccine-based interventions.
Search methodology
This narrative review was based on a literature search of using PubMed, Web of Science, and Google Scholar. Keywords included “Human papillomavirus (HPV),” “head and neck squamous cell carcinoma (HNSCC),” “HPV-positive,” “HPV-negative,” “oropharyngeal carcinoma,” “non-oropharyngeal squamous cell carcinomas,” “pathogenesis,” “immunotherapy,” “PD-1/PD-L1,” “diagnosis,” “therapeutic vaccines,” and “E6/E7 oncoproteins.” To ensure comprehensive and up-to-date coverage, the literature search was restricted to studies published between 2000 and 2025, with the inclusion of landmark publications of major relevance. Inclusion criteria of studies are as follows: (a) Recently published systematic reviews and meta-analyses to summarize high-quality evidence; (b) for the diagnostic section, we selected the most recent diagnostic criteria and guidelines for HNSCC, while also incorporating relevant recently systematic reviews and meta-analyses, as well as representative original studies; and (c) for therapeutic strategies, we preferentially included completed and formally published clinical studies exclusively involving patients with HNSCC, focusing on therapies that have reached advanced phases of clinical development (e.g. late phase II/III trials).
Epidemiology
Global incidence and trends
HPV-positive HNSCC has shown a significant upward trend worldwide in recent years.17,18 It is estimated that HPV infection may replace tobacco as the leading etiologic factor for HNSCC globally, a trend driven primarily by the rising incidence of OPSCC, in which HPV-associated cases now exceed tobacco-related oral cancers in several regions. 19 Although a modest increase in HPV-positive tumors has also been reported at non-oropharyngeal sites, carcinogenic HPV infection remains a relatively uncommon contributor to non-OPSCC.20,21 According to data from the National Cancer Institute and the SEER database, the incidence of HPV-positive OPSCC in the United States is 4.62 cases per 100,000 individuals, approximately 2.5-fold higher than that of HPV-negative OPSCC, whereas the incidence of HPV-positive non-oropharyngeal HNSCC is substantially lower at 0.62 cases per 100,000. 22 Epidemiological evidence continues to show an increasing incidence of HPV-related OPSCC in several countries, particularly in high-income countries, accounting for approximately 72% of all HNSCC cases. 23 In contrast, the prevalence remains significantly lower in low- and middle-income countries. 19 This geographic disparity may be attributed to differences in sexual behavior, public health infrastructure, and the implementation of HPV vaccination programs.
Risk factor
HPV infection is the most direct pathogenic factor of HPV-positive HNSCC; oral sex, especially, is one of the main transmission routes. Many studies have pointed out that the number of sexual partners, oral sex partners, and the early age of first sexual intercourse are all positively correlated with the risk of oral HPV infection.24,25 A recent multicenter case-control study conducted in the United States found that individuals with more than 10 lifetime oral sex partners had a 4.3-fold increased risk of developing HPV-related oropharyngeal cancer (OPC). Notably, those who initiated oral sex before the age of 18 had a significantly higher risk (adjusted odds ratio (OR) = 1.8), even after accounting for the number of oral sex partners and smoking history. Moreover, a higher intensity of exposure, measured as more than five oral sex partners per decade of sexual activity (“oral sex-years”), was associated with nearly a threefold increased risk. 26
Tobacco and alcohol remain important cofactors. While they are not primary drivers of HPV-related carcinogenesis, their presence may accelerate progression or worsen prognosis. A meta-analysis showed a significantly better 5-year overall survival (OS) for HPV positive non-smokers. Smoking is a negative prognostic factor for OS in patients with hr-HPV-positive HNSCC. 27 A retrospective study by Lai et al. 28 identified a significant interaction between alcohol consumption and HPV positivity (hazard ratio (HR) = 6.00; 95% confidence interval (CI): 1.03–35.01), indicating that alcohol use was associated with an average 26.1% increased risk of disease recurrence in patients with HPV-positive OPSCC. Additionally, a Mendelian randomization study revealed that smoking and alcohol independently increase the risk of both HPV-positive HNSCC. 29 Immunosuppressive states are also considered high-risk factors for persistent HPV infection. In these populations, the incidence of HPV-related tumors is significantly higher than in the general population.30,31 The cohort study by Beachler et al. 32 confirmed that the standardized incidence rate of HPV positive HNSCC is elevated in HIV-infected individuals, indicating that immunosuppression plays a role in its development.
Molecular mechanisms of HPV-positive HNSCC
Roles of HPV viral oncoproteins E5, E6 and E7
The carcinogenicity of HPV16 and HPV18 is primarily driven by the viral oncoproteins E6 and E7, which promote malignant transformation by interfering with host cell cycle regulation and DNA repair pathways. 33 The E6 protein exerts its oncogenic effects primarily by targeting the tumor suppressor protein p53 for degradation via the ubiquitin-proteasome pathway. High-risk HPV E6 inactivates p53 through two distinct mechanisms; association with E6AP to degrade p53 and association with p300 to block p300-mediated p53 acetylation and activation. 34 Loss of p53 function disrupts DNA damage response, apoptosis, and genomic stability. 35 Additionally, E6 activates telomerase reverse transcriptase (TERT), promoting cell immortalization. 36 These combined effects create a permissive environment for uncontrolled proliferation and accumulation of mutations (Figure 1).
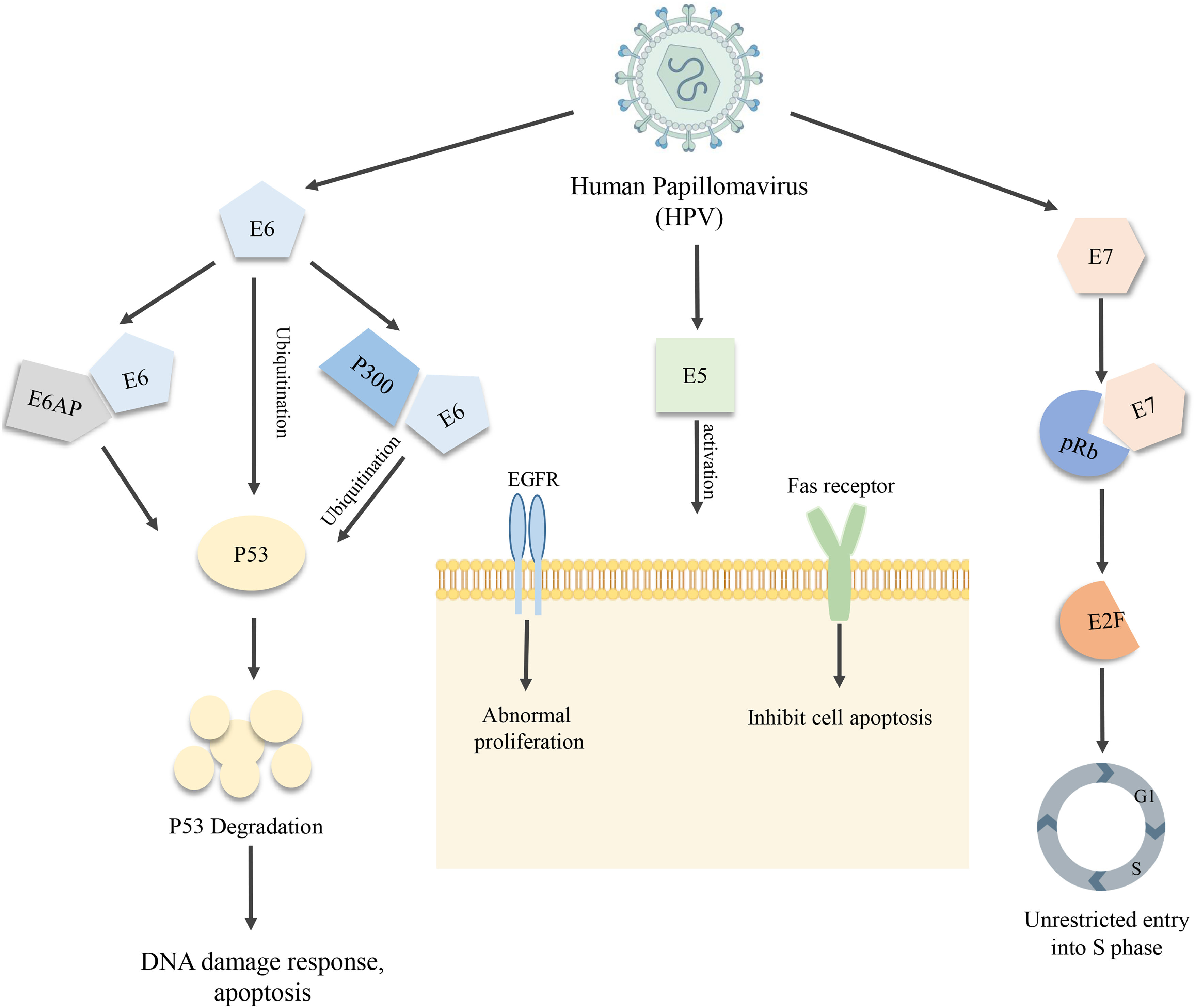
Mechanisms of oncogenic HPV proteins E5, E6, and E7. E6 promotes p53 degradation and activates hTERT, leading to impaired apoptosis and cellular immortalization. E7 inactivates pRb, driving cell cycle progression. E5 enhances EGFR signaling, inhibits apoptosis, and supports immune evasion.
The E7 protein promotes the degradation of retinoblastoma protein, releasing the transcription factor E2F and driving the abnormal cell cycle progression from the G1 phase to the S phase. 35 It also contributes to epigenetic alterations by inhibiting histone deacetylases. These effects enable unchecked cellular proliferation, a hallmark of cancer.
In addition to E6 and E7, the E5 oncoprotein also plays a role in oncogenic transformation. 37 E5 can induce aberrant cellular proliferation through ligand-mediated activation of epidermal growth factor receptor (EGFR). Also, E5 inhibits apoptosis by interfering with Fas receptor–mediated death signaling and contributes to immune evasion through modulation of host immune recognition, collectively promoting malignant transformation. 38
Key molecular pathways
Numerous studies have demonstrated aberrant activation of signaling pathways in HNSCC, among which phosphatidyl inositol 3-kinase (PI3K)/AKT/mTOR pathway is the most frequently altered oncogenic pathway. 39 Studies have shown that E6 and E7 in HPV 16 regulate PI3K/Akt/mTOR signaling by interacting with host proteins.40,41 Liu et al. 42 found that PIK3CA or PIK3R1 were the only oncogenes mutated in HPV-positive tumors, and patients harboring multiple mutations in PI3K pathway-related genes were more likely to present with advanced-stage disease, suggesting that dysregulation of the PI3K pathway is associated with HNSCC progression. Verhees et al. 43 reported that PI3K inhibitor reduced the activity of PI3K/AKT/mTOR pathway in HPV-positive HNSCC, leading to increased apoptosis and decreased oxidative and glycolytic metabolism. This indicates that the PI3K/AKT/mTOR pathway plays an important role in the development of HPV-positive HNSCC. Carracedo et al. found that activation of mTORC1 was positively correlated with a low-invasive phenotype, and was associated with significantly improved survival in HNSCC subgroup. 44 Katase et al. 45 demonstrated that DKK3 enhanced the malignant properties of HNSCC through activation of the PI3K/AKT/mTOR and mitogen-activated protein kinase (MAPK) pathways.
In addition to the PI3K/AKT/mTOR pathway, other signaling pathways also play important roles in the development and progression of HNSCC. STAT family proteins mediate cellular responses to cytokines and growth factors. Aberrant activation of the
Tumor microenvironment of HPV-positive HNSCC
HPV infection exerts a significant influence on the tumor microenvironment (TME) in patients with HNSCC (Figure 2). The composition and metabolic activity of immune infiltrates within the TME are critical determinants of anti-tumor immune responses and are closely associated with therapeutic efficacy and long-term prognosis. 51 HPV-positive HNSCC exhibits heightened immunogenicity due to the sustained expression of viral tumor-associated antigens, particularly the E6 and E7 oncoproteins, which promote the recruitment and activation of immune cells involved in both innate and adaptive immunity. 52 This enhanced antigenic landscape drives robust infiltration of immune effector cells into the tumor milieu, including CD8+ cytotoxic T lymphocytes, CD4+ helper T cells, PD-1+ exhausted T cells, CD19/CD20+ B cells and natural killer (NK) cells.53,54 Such an “inflamed” immune phenotype is generally associated with improved treatment responses and prolonged OS. Despite this immunologically active environment, HPV viral proteins also possess immunosuppressive properties, contributing to a complex immunomodulatory balance within TME. For instance, HPV16 E6 has been shown to downregulate the expression of Toll-like receptor 3 (TLR3), thereby impairing innate immune sensing. 55 Moreover, both E6 and E7 oncoproteins disrupt key cytosolic nucleic acid sensing pathways, including the RIG-I/MDA5-MAVS and cGAS-STING signaling cascades. Specifically, E6 suppresses RIG-I ubiquitination and subsequent downstream signaling, while E7 attenuates STING function, facilitating immune evasion and a more immunosuppressive TME.56,57 This interplay between immune activation and suppression gives rise to a distinctive immunological landscape in HPV-associated tumors. The dual effects of HPV, simultaneously enhancing antigen presentation while subverting innate immune surveillance, establish a unique immune balance within the TME. From the perspective of immune cell composition, HPV-positive tumors consistently demonstrate higher densities of CD8+ and CD4+ tumor-infiltrating lymphocytes (TILs), as well as increased NK cell infiltration, compared with HPV-negative counterparts. 58 This distinctive immune profile provides a strong biological rationale for the observed sensitivity of HPV-positive HNSCC to immunomodulatory therapies and supports ongoing efforts to integrate immune-based strategies into treatment paradigms.
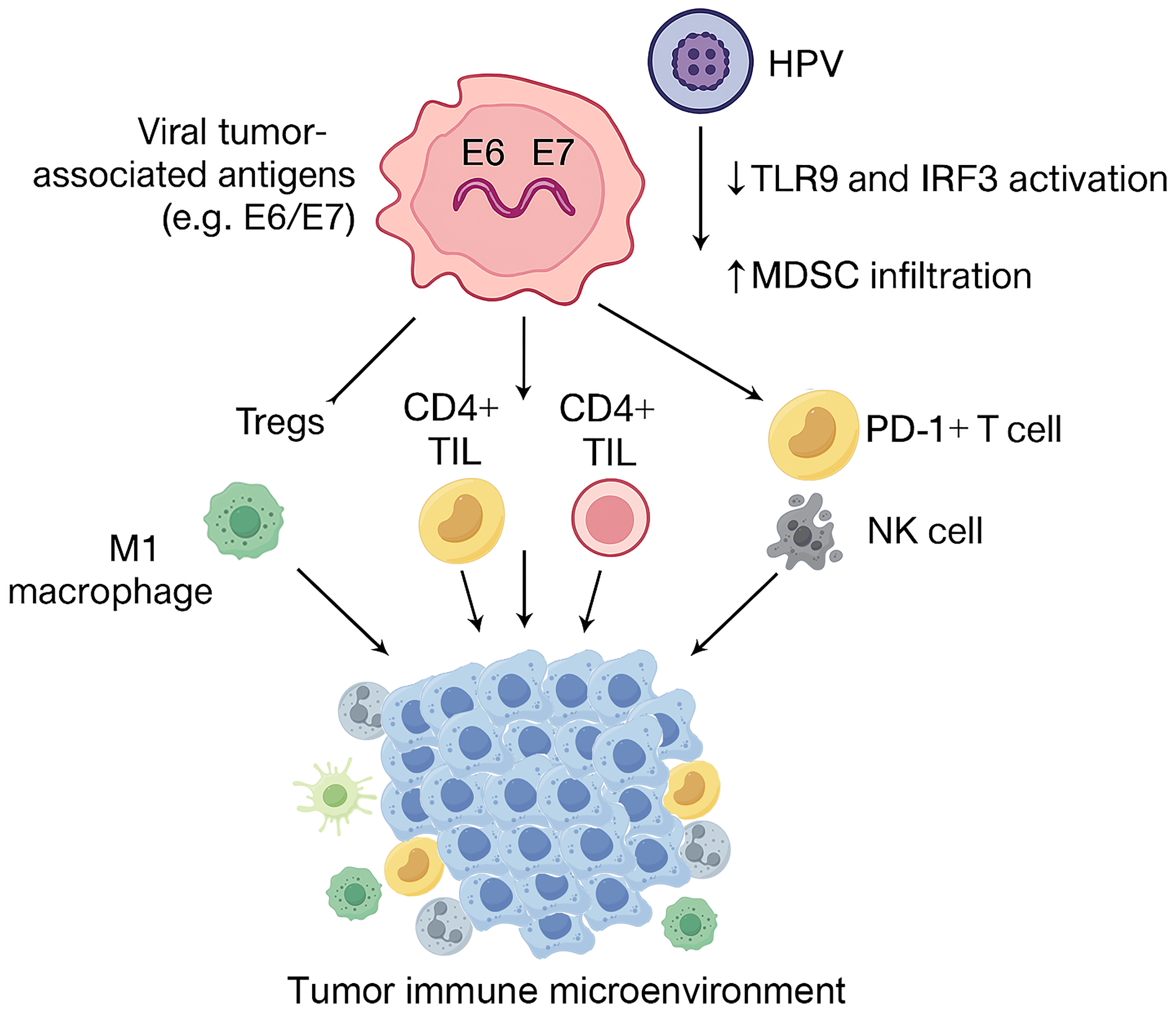
Tumor microenvironment (TME) of HPV-positive HNSCC. CD8+ cytotoxic T lymphocytes, CD4+ helper T cells, PD-1+ exhausted T cells, CD19/CD20 + B cells, and natural killer (NK) cells infiltrate the tumor environment in HNSCC.
Diagnosis and biomarkers of HPV-positive HNSCC
With the increasing understanding of the role of HPV in the pathogenesis of HNSCC, diagnosis of HPV-positive HNSCC is critical for prognostic assessment and the development of individualized treatment strategies. This section systematically reviews the histological features, imaging characteristics, molecular diagnostic methods, and emerging biomarkers of HPV-positive HNSCC, aiming to provide a basis for clinical diagnosis and management.
Pathological diagnosis and histological characteristics
Typical HPV-positive HNSCC arises predominantly in the oropharynx, particularly within the palatine tonsils and base of the tongue, a predilection attributable to the unique susceptibility of the reticulated crypt epithelium. Histopathologically, HPV-positive tumors are characterized by marked cellular atypia and variable degrees of squamous differentiation. However, hallmark features of conventional squamous differentiation, such as intercellular bridges and keratin pearls, are frequently absent. 59 Morphologically, HPV-positive HNSCC exhibits a spectrum of differentiation: well-differentiated tumors may resemble stratified squamous epithelium, exhibiting layered cell architecture and irregular keratinization, with keratin pearl formation being a defining feature (Figure 3(a)). In contrast, poorly differentiated tumors typically comprise immature-appearing cells with marked nuclear pleomorphism and atypical mitotic figures, often lacking organized stratification or keratin production (Figure 3(b)). HPV-negative HNSCCs are more often moderately to well differentiated, whereas HPV-positive tumors frequently display poor differentiation and basaloid morphology, which serves as a useful histologic clue in distinguishing HPV-related disease. 60
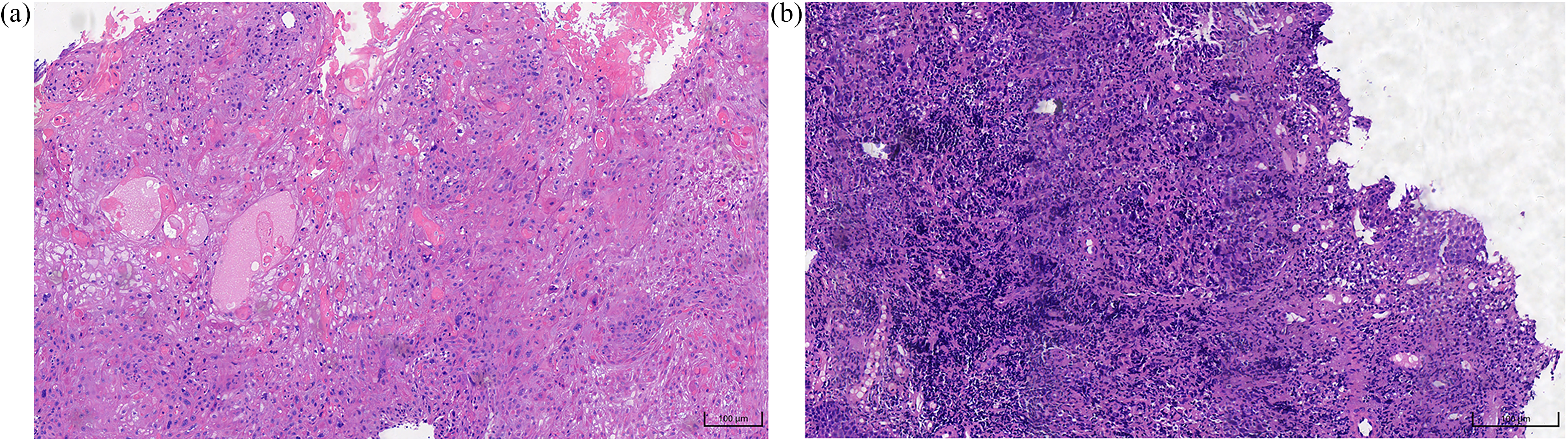
Histopathology of head and neck squamous cell carcinoma (HNSCC). (a) Well-differentiated squamous cell carcinoma showing intercellular bridges, keratinization, and keratin pearls (×20). (b) Poorly differentiated squamous cell carcinoma. It has nuclear pleomorphism and atypical mitoses, without obvious stratification or keratinization (×20).
Recent advances in artificial intelligence (AI) have further expanded diagnostic capabilities. Klein et al. 61 developed a deep learning algorithm (OPSCCnet) that analyzed standard gigapixel hematoxylin and eosin whole-slide images, which demonstrated superior performance compared with conventional p16 immunohistochemistry (IHC) and HPV DNA testing in selected clinical settings. AI-based histopathology foundation models have also identified distinct HPV-associated histologic patterns and delineated overlapping HPV-related subtypes with potential prognostic relevance. 62 However, most current AI models rely on single-institution datasets, and the lack of large, multi-center, and ethnically diverse cohorts remains a major limitation to their generalizability.
Imaging diagnostic basis and characteristics
Although imaging modalities cannot definitively determine HPV status, several radiologic features have been associated with HPV-positive tumors. Computed tomography (CT) enables visualization of tumor morphology, size, and anatomical boundaries. Yan et al. 63 summarized the diagnostic performance of CT for identifying HPV-positive HNSCC through a meta-analysis. The pooled sensitivity, specificity, and accuracy were 77.7%, 72.2%, and 63.8%, respectively, with an overall positive predictive value of 68.6%. These findings suggest that reliance on conventional CT imaging features alone is insufficient to achieve optimal diagnostic performance, highlighting the need to further refine imaging characteristics of HPV-positive HNSCC to improve diagnostic efficacy.
18F-fluorodeoxyglucose positron emission tomography-computed tomography (18F-FDG PET-CT) is widely used to assess the tumor metabolic activity. However, its ability to distinguish HPV-positive from HPV-negative HNSCC remains controversial. While Tahari et al. 64 reported higher glycolytic parameters, including maximum standardized uptake value (SUVmax) and SUVmean, in HPV-positive tumors. Clark et al. 65 showed no significant differences in SUVmax between HPV-positive and HPV-negative groups. Nevertheless, metastatic lymph nodes in HPV-positive OPSCC often demonstrate higher FDG uptake, which may aid in predicting p16-positive disease. 66
Diffusion-weighted imaging, which reflects cellular density through the apparent diffusion coefficient (ADC), has also shown potential in differentiating tumor biology. A meta-analysis by Payabvash et al. 67 revealed that the mean ADC values of primary HPV-positive HNSCC lesions were significantly lower than those of HPV-negative lesions, supporting the potential use of ADC as a quantitative imaging biomarker.
Molecular testing methods and diagnostic criteria
According to the 8th edition of the AJCC TNM staging system, p16 IHC is used as a surrogate marker for HPV-driven OPSCC, with positivity defined as diffuse nuclear and cytoplasmic staining in more than 70% of tumor cells (Figure 4). However, the applicability of p16 IHC across different anatomical subsites remains controversial. Wang et al. 68 reviewed 30 studies published between 2007 and 2019 and reported pooled sensitivity and specificity values of 0.94 (95% CI: 0.92–0.95) and 0.90 (95% CI: 0.89–0.91), with strong concordance between p16 IHC and in situ hybridization (ISH). Despite its high sensitivity, p16 IHC may yield false-positive results, particularly in non-oropharyngeal tumors. Gallus et al. 69 therefore suggested that p16 IHC should be used as a standalone test only in populations with high HPV prevalence and proposed a prevalence threshold above which the false-positive rates were acceptable. In line with this recommendation, public health agencies in Sweden and Denmark advocate confirmatory high-risk HPV nucleic acid testing following p16 positivity. A subsequent meta-analysis by Wang et al. 70 systematically analyzed whether the diagnostic efficacy of p16 positivity for HPV infection remained consistent across all sites of squamous cell carcinoma. The results demonstrated that p16 showed higher sensitivity and overall diagnostic accuracy in OPSCC (area under the curve (AUC) ≈ 0.96) than in non-OPSCC (AUC ≈ 0.95). Overall, p16 positivity exhibited superior diagnostic efficacy for HPV infection in oropharyngeal cancer compared with non-oropharyngeal sites, indicating that the interpretation of p16 status in non-oropharyngeal head and neck cancers should be approached with caution.
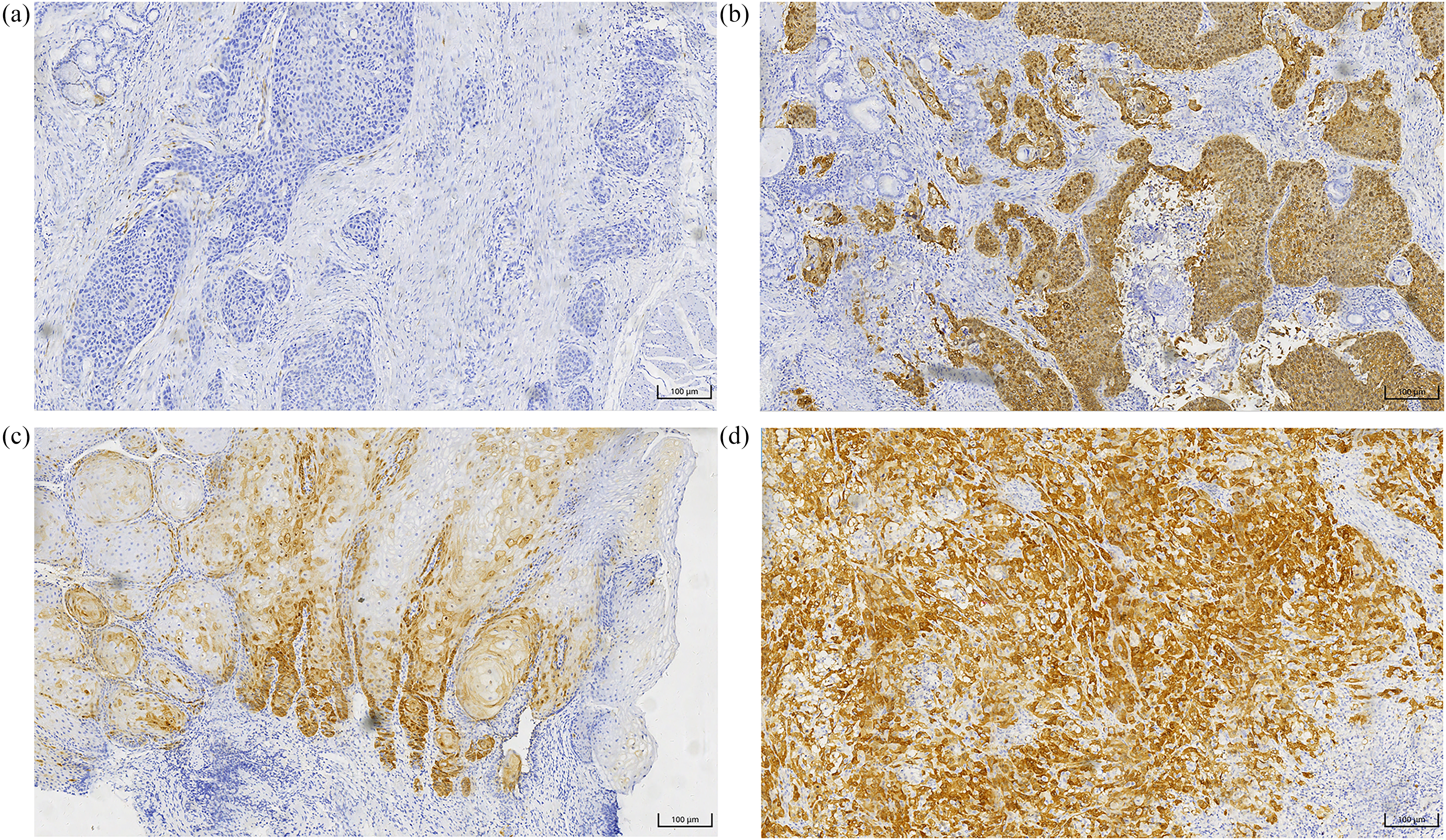
p16 immunohistochemistry in head and neck squamous cell carcinoma (HNSCC). (a) p16-negative moderately differentiated HNSCC shows faint staining with the same p16 antibody, the intensity of which is suggestive of HPV-negative disease (×20). (b) p16-positive moderately differentiated HNSCC, immunohistochemical staining for the cell cycle protein p16INK4A demonstrates diffuse nuclear and cytoplasmic staining, suggestive of human papillomavirus (HPV)-positive disease (×20). (c) p16-positive well-differentiated HNSCC (×20). (d) p16-positive poorly differentiated HNSCC (×20).
Detection of the presence of HPV DNA or E6/E7 messenger RNA (mRNA) using polymerase chain reaction (PCR) or ISH provides higher specificity for biologically active HPV infection. Liquid biopsy has also emerged as a promising approach, enabling the detection of circulating nucleic acids from blood samples. Allen et al. 71 reported an in vitro diagnostic method using serum-derived miRNAs to enhance diagnostic specificity and facilitate early detection of primary or metastatic disease. Mes et al. 72 developed and validated a novel rapid molecular assay based on the combined detection of HPV DNA and E6 mRNA in formalin-fixed paraffin-embedded tumor tissue, achieving diagnostic accuracies of 97% in OPSCC and 100% in oral squamous cell carcinoma.
While p16 IHC is currently the clinical standard for identifying HPV-driven OPSCC, particularly in high-prevalence regions, its limitations outside the oropharynx should be recognized. Accordingly, confirmatory molecular testing is strongly recommended for non-oropharyngeal cancers and in settings with low HPV prevalence, reflecting ongoing controversies and evolving guidelines regarding optimal HPV testing strategies in head and neck malignancies.
Biomarkers
Several molecular markers used for HPV diagnosis, including p16, HPV DNA, and HPV E6/E7 mRNA, also serve as relevant biomarkers, providing insight into prognosis, treatment response, and potential therapeutic targets (Table S1). HPV DNA is consistently detectable in the saliva of patients with HPV-positive HNSCC, making salivary HPV DNA analysis via liquid biopsy an efficient and non-invasive diagnostic tool.73,74
HPV circulating tumor DNA (ctDNA) is an emerging biomarker capable of real-time assessment of treatment response and disease recurrence. 75 Haring et al. 76 developed and validated a droplet-digital-PCR-based assay for detecting circulating HPV16 ctDNA in plasma and demonstrated its utility in monitoring therapeutic response in metastatic HPV-positive OPSCC. Furthermore, a systematic review by Jones et al. 77 confirmed that HPV ctDNA is potential for HPV-positive HNSCC, it should be noted that this study covered patients from multiple sites, including the oropharynx, hypopharynx, larynx, and external auditory canal, demonstrating broad applicability.
Platelet-derived growth factor (PDGF) and its receptors (PDGFR) are expressed in various malignant tumor cells and contribute to cancer proliferation, invasion, metastasis, and angiogenesis through modulation of the PI3K/AKT and MAPK/ERK signaling pathways.78,79 In vitro studies have shown differential expression of vascular endothelial growth factor (VEGF), PDGF, and PDGFR-α/β between HPV-positive and HPV-negative cell lines, as well as distinct responses to chemotherapy. 80 Moreover, small-molecule tyrosine kinase inhibitors have been reported to suppress PDGF/PDGFR expression in HPV-positive models. 81 These findings suggest that PDGF/PDGFR may represent a potential biomarkers and therapeutic target in HPV-positive HNSCC. However, further clinical studies are required to validate their functional relevance.
Treatment management
Patients with HPV-positive HNSCC generally exhibit a more favorable prognosis than those with HPV-negative disease. Accordingly, increasing attention has been directed toward treatment de-escalation strategies aimed at reducing therapy-related toxicity while preserving excellent oncologic outcomes and quality of life. In parallel, immunotherapy has achieved substantial progress in the recurrent and metastatic setting, and both prophylactic and therapeutic HPV vaccines are anticipated to play an increasingly important role in future treatment paradigms. Treatment-related clinical studies are summarized in Table 1, including study populations, sample sizes, primary endpoints, key clinical outcomes, and major limitations, providing a comprehensive framework for evaluating the clinical utility of different therapeutic strategies.
Summary of therapeutic strategies for HPV-positive HNSCC.
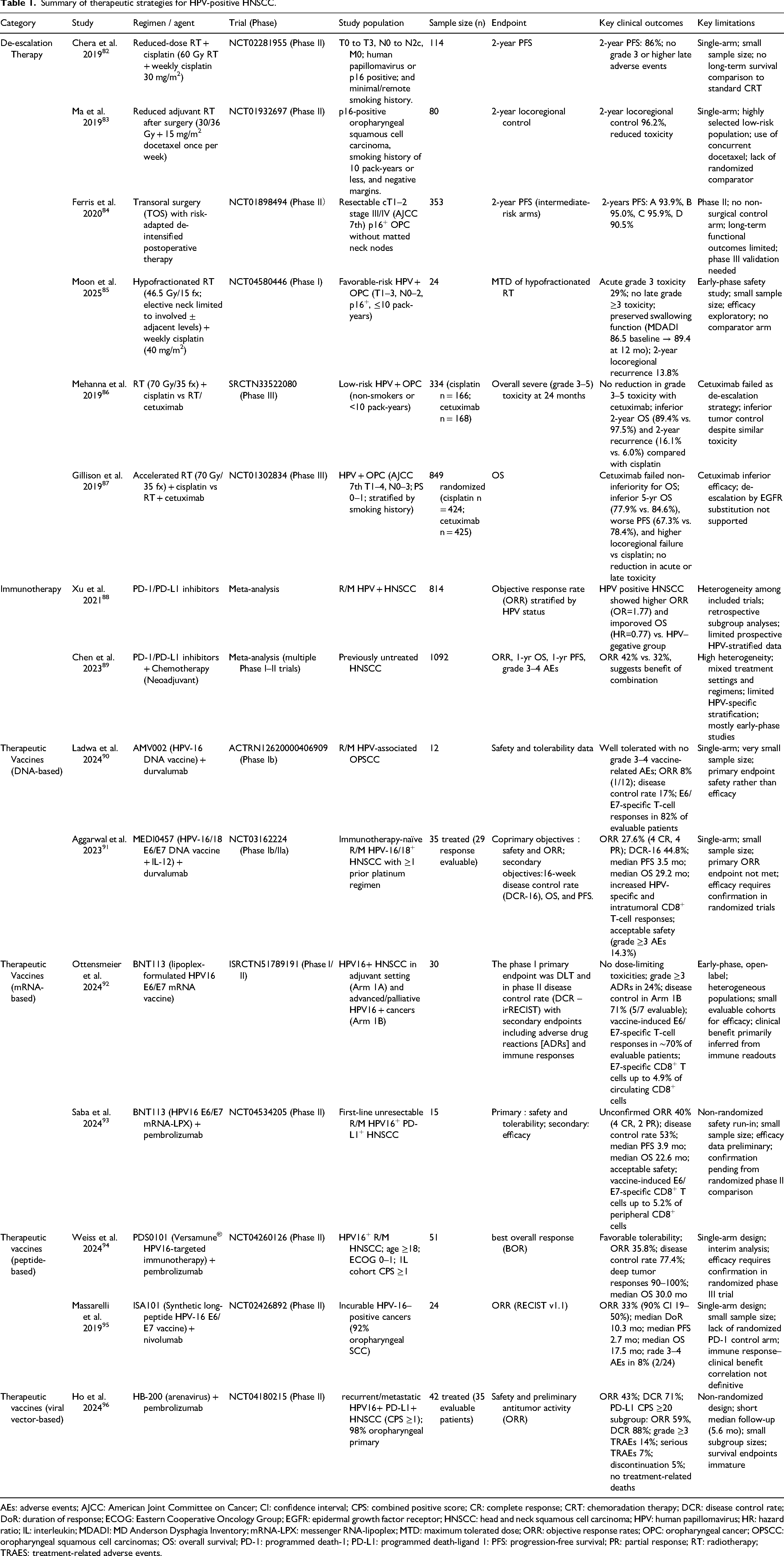
AEs: adverse events; AJCC: American Joint Committee on Cancer; CI: confidence interval; CPS: combined positive score; CR: complete response; CRT: chemoradation therapy; DCR: disease control rate; DoR: duration of response; ECOG: Eastern Cooperative Oncology Group; EGFR: epidermal growth factor receptor; HNSCC: head and neck squamous cell carcinoma; HPV: human papillomavirus; HR: hazard ratio; IL: interleukin; MDADI: MD Anderson Dysphagia Inventory; mRNA-LPX: messenger RNA-lipoplex; MTD: maximum tolerated dose; ORR: objective response rates; OPC: oropharyngeal cancer; OPSCC: oropharyngeal squamous cell carcinomas; OS: overall survival; PD-1: programmed death-1; PD-L1: programmed death-ligand 1: PFS: progression-free survival; PR: partial response; RT: radiotherapy; TRAES: treatment-related adverse events.
Traditional treatment approaches
Surgery, radiotherapy (RT), and chemotherapy, used alone or in combination, remain the cornerstone of HNSCC management, with treatment selection guided primarily by tumor node metastasis (TNM) stage and tumor site. 97 Surgery is most employed for oral cavity cancers, whereas RT is more frequently used for pharyngeal and laryngeal malignancies. According to the National Comprehensive Cancer Network 2022 guidelines, patients with HPV-positive oropharyngeal carcinoma at stage T1–2N0 may be managed with definitive RT or primary surgical resection with neck dissection. For patients with T1–2N1 disease (lymph nodes <3 cm), surgery, RT, or concurrent chemoradiotherapy are all acceptable options. 98 HPV-positive tumors are sensitive to chemoradiotherapy, resulting in excellent clinical outcomes, with reported 5-year progression-free survival (PFS) rates of 85–90% and OS rates approaching 95%. 99 However, cumulative RT and chemotherapy is associated with substantial long-term toxicities, including xerostomia, dysphagia, and temporomandibular joint dysfunction, underscoring the need for treatment optimization. 100
De-escalation strategies of (chemo)radiation
Given the favorable treatment responsiveness of HPV-positive HNSCC, appropriately reducing treatment intensity represents an ideal strategy. Standard therapy typically consists of 70Gy intensity-modulated radiation therapy (IMRT) combined with cisplatin (100 mg/m2 every 3 weeks). 101 To mitigate long-term toxicity while maintaining survival outcomes, multiple de-intensification trials have been conducted.
In a phase II trial, Chera et al. 102 evaluated 60 Gy IMRT combined with weekly cisplatin (30 mg/m2) in 43 non-smoking patients with HPV-positive disease, demonstrating high pathological complete response rates and reduced toxicity compared with standard regimens. A subsequent phase II trial enrolling 114 patients reported similar outcomes, supporting the feasibility of this approach. 82 The phase II MC1273 trial demonstrated that in carefully selected patients with HPV-positive OPSCC (low smoking history, negative surgical margins), reducing adjuvant RT to 30–36 Gy combined with chemotherapy after surgery significantly decreased toxicity while preserving swallowing function and achieving a 2-year locoregional control rate of 96.2%. 83 Similarly, the ECOG-ACRIN 3311 trial showed that intermediate-risk patients receiving 50 Gy RT alone after transoral resection achieved a 2-year PFS of 95.0%, with lower toxicity compared with higher-dose RT. 84 However, both studies selected strictly screened intermediate-risk patients (e.g. low smoking history, negative surgical margins); therefore, the results may not be applicable to all HPV-positive oropharyngeal cancer patients, especially those with higher-risk characteristics (e.g. long smoking history, positive surgical margins, or multiple lymph node metastases). Furthermore, these studies had relatively small sample sizes and short follow-up periods, requiring further long-term studies to validate their applicability to a broader patient population. Therefore, future research should include more high-risk patients, employ longer follow-up periods, and explore biomarker-guided personalized treatment strategies.
A phase I trial by Moon et al. 85 confirmed that for patients with early HPV-related oropharyngeal cancer (T1-3N0-2, p16 positive, ≤10 pack-years), the regimen of hypofractionated RT (52 Gy/20 fractions in high-risk areas and 46.5 Gy/15 fractions in low-risk areas) completed within 3 weeks combined with weekly cisplatin therapy was well tolerated, with an acute grade 3 toxicity rate of 29%, stable swallowing function (MD Anderson Dysphagia Inventory (MDADI) composite: score baseline 86.5→12 months 89.4), and a 2-year local regional control rate of 86.2%. This treatment paradigm warrants further investigation with biomarker integration to assess for efficacy.
EGFR has been identified as an oncogenic driver in squamous cell carcinoma, which can promote cell proliferation through multiple pathways. 103 Its monoclonal antibody, cetuximab, has been proposed as a de-escalation strategy in these patients with the aim of reducing the toxicity associated with standard cisplatin-based therapy. The open-label, randomized phase III De-ESCALaTE HPV trial evaluated whether cetuximab combined with RT could reduce treatment-related toxicity while maintaining efficacy compared with cisplatin plus RT. Compared with the standard cisplatin regimen (100 mg/m2 on days 1, 22, and 43 of radiotherapy) combined with RT (total 70 Gy), cetuximab (400 mg/m2 loading dose followed by seven weekly infusions of 250 mg/m2) plus RT (total 70 Gy) failed to reduce toxicity and was associated with significantly worse tumor control. 86 These findings were further corroborated by the similar phase III RTOG 1016 trial, which demonstrated that although toxicity profiles were comparable, patients in the cetuximab arm experienced significantly worse overall survival and progression-free survival. 87 This trial is pivotal in highlighting the potential risks of using cetuximab for de-escalation, and underscores the need for careful evaluation of alternative agents in treatment regimens.
Currently, many large-scale phase III randomized controlled studies investigating treatment de-escalation strategies are still ongoing, with the long-term goal of reducing treatment-related toxicity without affecting the excellent survival of HPV-positive HNSCC patients. However, any treatment de-escalation therapy is not currently recommended for HPV-related HNSCC patients outside of clinical trials. Additionally, song et al. develop and validate a prognostic and predictive radiomic image signature to inform survival and chemotherapy benefit using CT scans. They found that the patients did not benefit from chemotherapy, and could be considered for treatment de-escalation. 104 This could become an important reference method for de-escalation therapy.
Immunotherapeutic strategies
PD-(L)1 inhibitor: In recent years, immune checkpoint inhibitors targeting programmed cell death protein 1 (PD-1) and its ligand PD-L1 have gained widespread clinical application. Currently, most PD-(L)1 inhibitor studies in HNSCC have been conducted in the HPV-negative population. However, growing evidence supports their efficacy in HPV-positive subtypes as well. Nivolumab (CheckMate-141 trial) and pembrolizumab (KEYNOTE-048 trial) have received FDA approval for the treatment of platinum-refractory recurrent/metastatic HNSCC. 105 In HPV-positive patients, these agents have demonstrated objective response rates (ORR) of approximately 18%, with a median OS extended to around 8.4 months. 106 Several meta-analyses have indicated a significantly higher ORR in HPV-positive patients compared to HPV-negative individuals following PD-1/PD-L1 inhibitor treatment (pooled OR ≈ 1.77, 95% CI: 1.14–2.74), along with a reduced risk of all-cause mortality (HR ≈ 0.77). 88 Moreover, multiple ongoing phase III clinical trials are investigating combinations of immunotherapy with radiation and chemotherapy, aiming to enhance treatment efficacy. In a comprehensive meta-analysis by Chen et al., 89 which included 1092 patients across 24 studies, the combination of neoadjuvant PD-1/PD-L1 inhibitors with chemotherapy yielded a higher ORR compared to immune monotherapy in previously untreated HNSCC patients (32% vs. 42%, subgroup difference p = 0.60). These findings support the potential of integrated immunochemotherapy strategies in the management of HPV-associated HNSCC.
HPV-positive tumors, due to their inherent immunogenicity and “inflamed” tumor microenvironment, are thought to be more susceptible to immunomodulatory therapies. This is supported by clinical and meta-analytic evidence suggesting improved objective response rates and OS in HPV-positive patients compared to their HPV-negative counterparts. However, despite these encouraging signals, the absolute response rates to PD-1/PD-L1 inhibitors remain modest, and a significant proportion of patients do not benefit from monotherapy. Moreover, combination strategies, such as integrating immune checkpoint inhibitors with radiotherapy, chemotherapy, or therapeutic vaccines, offer potential synergy but require careful optimization to avoid overlapping toxicities. Although early-phase trials have demonstrated safety and enhanced efficacy signals, confirmatory phase III data are still needed before widespread adoption.
Targeted vaccines
In HPV-driven cancers, the primary tumor-associated antigens are the viral oncoproteins E6 and E7. 107 Most therapeutic vaccine platforms aim to deliver these antigens to antigen-presenting cells to stimulate antigen presentation through both MHC class I and II pathways, thereby inducing robust CD8+ cytotoxic and CD4+ helper T-cell immune responses. 106 In the context of HPV-positive HNSCC, several vaccine strategies have emerged, broadly categorized into molecular vaccines (based on DNA, RNA, or peptides) and those utilizing viral or bacterial vectors.
AMV002 is a DNA vaccine that contains two plasmids encoding fusion variants of HPV16 E6 and E7 proteins. A single-center phase Ib study investigated the safety and tolerability of AMV002 in combination with the PD-L1 inhibitor durvalumab in patients with recurrent OPSCC. The combination was well tolerated and immunogenic, with detectable T-cell responses observed in treated patients. 90 To date, AMV002 has primarily been studied in OPSCC, and no systematic studies in broader HNSCC populations have been reported. Another candidate, MEDI0457 (also known as INO-3112), is a plasmid-based DNA vaccine composed of three plasmids encoding HPV-16 and HPV-18 E6/E7 antigens, along with interleukin-12 to enhance immunogenicity. Results from phase Ib/II studies indicated that MEDI0457 induced durable HPV16/18 antigen-specific immune responses both systemically and intratumorally. 108 Furthermore, a phase Ib/IIa trial assessing the combination of MEDI0457 with durvalumab in patients with HPV-positive recurrent/metastatic HNSCC demonstrated good tolerability and immunogenicity, with increases in peripheral HPV-specific T cells and intratumoral CD8+ T-cell infiltration. 91
BNT113 is a lipoplex-formulated uridine-modified mRNA vaccine encoding the E6 and E7 oncoproteins of HPV-16. Results from phase I/II clinical trials demonstrated that BNT113 was generally well tolerated in patients with HPV16-positive HNSCC. Importantly, the vaccine induced robust antigen-specific immune responses, with E6/E7-specific T cells comprising up to 4.9% of circulating CD8+ T cells. 92 In the phase II AHEAD-MERIT trial, the combination of BNT113 with pembrolizumab was confirmed to be both safe and clinically effective (ESMO-IO 2022 #155P). Furthermore, HPV16 E7-specific CD8+ T cells were observed to constitute up to 5.2% of peripheral CD8+ T cells, suggesting strong systemic immunogenicity of the vaccine in combination with immune checkpoint blockade. 93
PDS0101 (Versamune® HPV) is a novel investigational immunotherapy targeting HPV16, designed to elicit a potent and durable T-cell–mediated immune response against HPV16-positive malignancies. In a single-arm phase II clinical trial, the combination of PDS0101 and pembrolizumab continued to demonstrate favorable tolerability in first-line treatment of patients with recurrent/metastatic HPV16-positive HNSCC. 94 ISA101 is a synthetic long-peptide vaccine composed of nine overlapping peptides derived from HPV-16 E6 (five 32-mers and four 25-mers) and four overlapping 35-mer peptides covering the full-length sequences of HPV-16 E6 and E7 oncoproteins. 109 In a phase II clinical trial, the combination of ISA101 with nivolumab achieved an overall response rate of 33% and a median OS of 17.5 months in HPV16-positive HNSCC patients, surpassing the outcomes typically observed with nivolumab monotherapy. 95
HB-200 is a novel arenavirus-based therapeutic vaccine designed to induce robust E6/E7 antigen-specific antitumor immune responses. A phase II clinical trial led by Ho et al. 110 is currently underway to evaluate the efficacy of HB-200 as an adjuvant treatment for minimal residual disease in HPV-positive HNSCC, as confirmed by NavDx testing following definitive therapy. Preliminary results from another trial investigating the combination of HB-200 with pembrolizumab in patients with recurrent/metastatic (R/M) HPV16-positive HNSCC have shown promising clinical activity and favorable tolerability. 96
Therapeutic HPV vaccines represent a promising immunologic approach in HPV-positive HNSCC, offering the potential to elicit targeted, durable, and systemic anti-tumor immune responses. By focusing on the viral oncoproteins E6 and E7, these vaccines enable precise immune targeting with minimal off-tumor effects. Across different platforms, including DNA-based, mRNA-based, peptide-based, and viral vector-based vaccines, early-phase clinical trials have demonstrated encouraging safety profiles, robust antigen-specific T cell responses, and potential synergy with immune checkpoint blockade. However, current studies have some limitations. First, most vaccine-based studies in HPV-positive HNSCC remain early-phase investigations (phase I or Ib/II), are predominantly single-arm in design, and involve relatively small and heterogeneous patient cohorts. These trials frequently enroll heavily pretreated patients with recurrent or metastatic disease, thereby limiting the generalizability of the findings and precluding definitive conclusions regarding comparative efficacy. Second, while many trials report robust immune readouts, such as increases in circulating HPV E6/E7–specific CD8+ T cells, enhanced intratumoral T-cell infiltration, or favorable cytokine profiles, these immunologic endpoints do not consistently translate into durable clinical benefit. ORRs and survival outcomes remain modest in a substantial proportion of patients, underscoring a persistent gap between immune activation and effective tumor control. This discrepancy likely reflects multiple biological barriers, including immunosuppressive tumor microenvironments, regulatory T-cell and myeloid-derived suppressor cell infiltration, antigen loss, and baseline or treatment-induced T-cell exhaustion. Third, combination strategies integrating therapeutic vaccines with PD-1/PD-L1 blockade, chemotherapy, or radiotherapy appear biologically rational and have demonstrated acceptable safety profiles and preliminary efficacy signals. However, most supporting data are derived from non-randomized studies, and the relative contribution of each therapeutic component remains unclear. The optimal sequencing, dosing, and patient selection criteria for these combinations have yet to be defined, and there is an ongoing need to balance enhanced antitumor immunity against the risk of cumulative toxicity. Finally, HPV-positive HNSCC itself represents a biologically heterogeneous disease. Differences in viral integration status, tumor mutational burden, immune infiltration patterns, and host immune competence may all influence responsiveness to immunotherapy and vaccines. Future trials will need to incorporate biomarker-driven stratification, standardized immune monitoring, and clinically meaningful endpoints to better identify patients most likely to benefit. In summary, while immune checkpoint inhibitors and therapeutic HPV vaccines represent promising components of the treatment landscape for HPV-positive HNSCC, current evidence is largely hypothesis-generating. Well-designed, randomized phase III trials with clearly defined clinical endpoints are essential to bridge the gap between immunogenicity and durable clinical benefit and to establish the precise role of these strategies in routine clinical practice.
Conclusion
HPV-positive HNSCC represents a distinct biological and clinical subtype characterized by unique molecular drivers, immune microenvironment features, and therapeutic sensitivities. From a clinical perspective, accurate identification of HPV-driven tumors is essential for prognosis and treatment decision-making. Although p16 immunohistochemistry remains the standard surrogate marker for HPV-positive OPSCC, its diagnostic specificity and reliability are substantially reduced in non-oropharyngeal sites, underscoring the importance of confirmatory molecular testing. In this context, emerging biomarkers, including HPV DNA, and HPV E6/E7 mRNA circulating HPV DNA and imaging-based radiomic signatures, show promise for noninvasive diagnosis, treatment monitoring, and early detection of recurrence. However, substantial variability in detection platforms and analytical sensitivity currently limits cross-study comparability and clinical translation, indicating a need for standardized validation in prospective, site-specific cohorts, particularly in non-OPSCC.
Therapeutically, the excellent outcomes observed in HPV-positive HNSCC have stimulated substantial interest in treatment de-escalation strategies to reduce long-term toxicity while maintaining oncologic efficacy. While early-phase studies suggest that reduced-intensity radiotherapy and risk-adapted postoperative approaches may be feasible, most de-escalation treatment studies currently focus on patients with specific disease stages, and the generalizability of their results requires further validation. Furthermore, de-escalation decisions remain primarily based on clinical and pathological risk stratification rather than tumor biological characteristics. The lack of reliable, biology-based predictive biomarkers continues to limit the implementation of biomarker-guided personalized treatment adjustments. In parallel, immune checkpoint inhibitors and therapeutic HPV vaccines targeting the E6/E7 oncoproteins represent promising immunotherapeutic approaches. Despite the encouraging immunogenicity and early efficacy signals (particularly in combination regimens), clinical benefit remains heterogeneous. And the biological determinants of response and resistance are not fully elucidated, limiting optimal patient selection and rational treatment sequencing.
It should be acknowledged that, although this review is titled HNSCC, most of the high-quality evidence currently available pertains to HPV-positive OPSCC. HPV-associated carcinomas arising in non-oropharyngeal head and neck sites remain relatively uncommon and under-investigated, and the existing literature is limited in both quantity and consistency. Nevertheless, we conducted a comprehensive and systematic search to include relevant studies on HPV-related non-oropharyngeal HNSCC whenever available. Additional limitations of this review include heterogeneity in HPV detection methods, and the predominance of retrospective or early-phase clinical studies, particularly within the immunotherapy and vaccine literature.
In summary, HPV-positive HNSCC represents a distinct clinical and molecular entity with a better prognosis and unique therapeutic opportunities compared to its HPV-negative counterpart. Advances in our understanding of HPV pathogenesis have opened the door to novel diagnostic, prognostic, and therapeutic approaches. Future research should focus on refining de-intensification protocols, developing therapeutic vaccines, personalizing immunotherapeutic strategies and AI-driven analytics to optimize outcomes for HPV-positive HNSCC patients.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155261428136 - Supplemental material for HPV-positive head and neck squamous cell carcinoma: From molecular pathogenesis to therapeutic frontiers
Supplemental material, sj-docx-1-jbm-10.1177_03936155261428136 for HPV-positive head and neck squamous cell carcinoma: From molecular pathogenesis to therapeutic frontiers by Ying Li and Shengfei Zhou in The International Journal of Biological Markers
Footnotes
Acknowledgements
Not applicable.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Authors’ contributions
Conceptualization: Ying Li; Methodology: Shengfei Zhou; Formal analysis and investigation: Ying Li and Shengfei Zhou; Writing—original draft preparation: Ying Li and Shengfei Zhou; Writing—review and editing: Ying Li, Shengfei Zhou. All authors read and approved the final manuscript.
Funding
This work was supported by the fund from Sanya Central Hospital (The Third People's Hospital of Hainan Province) (No. SYZXYY202303).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
Supplemental material
Supplemental material for this article is available online.
