Abstract
Introduction
Matrix metalloproteinases (MMPs) are enzymes participating in tumorigenesis and tumor progression through their proteolytic and cell-signaling properties. They are regulated mostly by endogenous tissue inhibitors of metalloproteinases (TIMPs). The expression of both MMPs and TIMPs is often altered in cancers. Many studies investigated their potential as circulating cancer biomarkers, with confounding results, which might be induced by preanalytical issues, particularly by using serum instead of plasma. The study aims to investigate plasma levels of selected MMPs and TIMPs in association with the diagnosis and prognosis of colorectal cancer.
Methods
The clinico-pathological data of 148 patients operated for colorectal cancer were collected from the medical records system at the University Hospital Pilsen. Sixty-eight age-matched healthy subjects were included as controls. Plasma levels of MMP-2, -7, -8, -9, -10, and TIMP-1, -2, -3, and -4 were assessed with multiplex immunoassays with the technology xMAP.
Results
MMP-8 and -9 levels were significantly elevated in patients (P= 0.0002 and 0.0009, respectively), TIMP-2 levels were significantly decreased in colorectal cancer patients (P = 0.0016). When comparing the early colorectal cancer (stage I and II) with advanced colorectal cancer (stage III and IV), MMP-8 and TIMP-1 were significantly increased in advanced colorectal cancer (P = 0.0173 and <0.0001, respectively). The area under the curve and the receiver operating characteristic were between 0.680 and 0.530 for all studied biomarkers. In univariate analysis, overall survival was significantly elevated in patients with MMP-7, -8, or TIMP-1, which was higher than cut-offs (hazard ratio = 4.57, 2.03, and 7.64, respectively).
Conclusion
These findings suggest that plasma MMP-7, MMP-8, and TIMP-1 are potential prognostic biomarkers for colorectal cancer . None of the investigated biomarkers revealed diagnostic potential.
Introduction
Circulating cancer biomarkers assume an essential role in the management of colorectal cancer (CRC). Traditional serum markers, notably carcinoembryonic antigen (CEA) and cancer antigen 19-9 (CA 19-9), are routinely employed for tumor staging, monitoring disease progression, and identifying tumor recurrence or metastasis. However, the limited sensitivity and specificity of these markers restrict their efficacy as diagnostic and prognostic tools. 1 Novel cancer biomarkers are being investigated that may complement traditional markers, with a particular emphasis on biomolecules implicated in tumorigenesis or those that may elucidate the biological activity of tumors such as markers of differentiation and proliferation.2,3
In this context, circulating matrix metalloproteinases (MMPs) and their specific tissue inhibitor metalloproteinases (TIMPs) have been explored as potential diagnostic and prognostic biomarkers, either stand-alone or in combination with traditional markers. However, the findings from these investigations have yielded inconsistent results. 4
MMPs—various functions in cancer
MMPs are a large family of zinc-dependent endopeptidases, 23 of which have been identified in humans. These proteins primarily function in the proteolytic degradation of extracellular matrix (ECM) components and the remodeling of tissue. MMPs exhibit similar structural characteristics, and the specific structure of each MMP determines the target ECM components. Based on their specificity and structural properties, MMPs are categorized into six distinct groups: gelatinases, collagenases, stromelysins, matrilysins, membrane-type MMPs, and other MMPs. 5
MMPs are integral to a wide array of biological processes, including tissue repair, embryogenesis, angiogenesis, and wound healing. In addition to degrading various ECM components, MMPs are capable of degrading non-matrix proteins such as cytokines, chemokines, growth factors, and cell receptors, thereby modulating numerous cellular signaling pathways. Specifically, MMPs facilitate cell proliferation, migration, and differentiation, and contribute to processes such as apoptosis, angiogenesis, and immune responses.6,7
The activity of MMPs is precisely regulated at multiple levels, including messenger RNA (mRNA) expression, the activation of proenzymes, and inhibition by endogenous tissue inhibitors known as TIMPs. Dysregulation of MMP activity can result in various pathologies characterized by tissue destruction, particularly cancer. MMPs have been linked to carcinogenesis through their roles in tumor progression, invasiveness, and metastasis. MMPs are produced by a diverse range of tissues and cells, including connective tissue, fibroblasts, leukocytes, and platelets. Notably, the expression of numerous members of the MMP family within cancerous tissues is induced not only in malignant cells but also in the surrounding stromal cells. 6 Their expression is frequently upregulated in cancers, including CRC, often correlating with advanced tumor stages, increased invasion, metastasis, and poorer survival outcomes. 8 Research has indicated that MMP overexpression is associated with unfavorable prognoses, suggesting their potential utility as biomarkers for assessing outcomes in CRC patients. Nevertheless, the roles of MMPs in cancer are complex and may even be protective, depending on the specific substrates involved.7,9
Given that changes at the cellular level may be detected in body fluids, elevated levels of MMPs in human blood could serve as valuable diagnostic and prognostic indicators for a variety of cancer types, including colorectal malignancies. 4 The gelatinases MMP-2 and -9 have been studied most often due their role in CRC progression and metastasis 10 ; other MMP subtypes such as MMP-7, -8, and -10 have been investigated less frequently.
TIMPs—more than proteolytic inhibitors
TIMPs are glycoproteins—endogenous inhibitors of MMPs activity—that bind to MMPs. In humans, four homologous TIMPs have been identified: TIMP-1, -2, -3, and -4. Each TIMP possesses the capability to inhibit multiple MMPs, and vice versa. 11
The expression levels of TIMPs are expected to correlate consistently with the inhibition of cancer progression. However, clinical findings reveal a degree of contradiction, indicating that both overexpression and underexpression of TIMPs may facilitate tumor progression. The upregulation and activation of MMPs have been recognized as critical mechanisms in disease processes, notably in promoting invasion and metastasis. Consequently, the observed increase in TIMP expression has often been interpreted as an indication of ineffective inhibition of ECM degradation. 12
It is acknowledged that TIMPs serve functions beyond their role as MMP inhibitors, suggesting a more intricate contribution to cancer biology than previously understood. 13 TIMPs may act as signaling molecules with cytokine-like properties, impacting various biological processes such as cell growth, apoptosis, differentiation, angiogenesis, and oncogenesis.14,15 Furthermore, TIMPs display tissue-specific expression, which is regulated at the transcriptional level by a variety of cytokines and growth factors. In conditions such as cancer, particularly in the case of TIMP-1, the enhanced expression could be attributed to the emerging signaling functions of TIMPs.14,15
Despite their common evolutionary structure, the four TIMP proteins exhibit distinct properties and regulatory mechanisms. 16 TIMP-1, the most extensively studied member of the TIMP family in relation to cancer, is consistently found to be elevated in both cancerous tissues and the bloodstream of patients. This overexpression is associated with poor prognosis. TIMP-2 presents heterogeneously with its expression varying based on the tissue of origin; for instance, in CRC, TIMP-2 is overexpressed in tissue while being underexpressed in blood. 16
Both TIMP-1 and TIMP-2 have been shown to possess not only inhibitory but also growth-promoting effects within tumors. There is evidence indicating that TIMP-3 is silenced in various human cancers, including CRC, 13 and this silencing correlates with poor prognostic outcomes. TIMP-4, the least studied member of the family, also shows a heterogeneous expression trend similar to that of TIMP-2.
The overexpression of TIMP-1 or the silencing of TIMP-3 is consistently correlated with cancer progression and poor patient prognosis. Experimental studies reveal that TIMPs contribute significantly to many cancer hallmarks, and deregulation of TIMPs is commonly observed in both tumors and stroma. 13 The expression levels of TIMPs have been linked to both favorable and unfavorable outcomes in CRC.
Additionally, akin to MMPs, the blood levels of TIMPs may serve as potential surrogate circulating biomarkers for CRC.
In our case-control study, we conducted a comprehensive analysis of blood plasma levels of MMP-2, -7, -8, -9, and -10, alongside TIMP-1, -2, -3, and -4, and we investigated their potential as biomarkers for diagnosis and prognosis.
Preanalytical considerations
When assessing circulating MMPs and TIMPs in blood, it is imperative to take preanalytical considerations into account. Serum samples may not be the most suitable option for evaluating MMP and TIMP levels due to variability in the release of these proteins from platelets and leukocytes during the processes of coagulation and platelet activation. This variability can result in artificially elevated levels of MMPs and TIMPs in serum compared to plasma. 17 It was shown that even the rapid separation of serum that minimizes the time between blood drawing and centrifugation does not prevent the falsely high MMP-9 content in serum. 18
Numerous published studies examining circulating MMP levels have relied on serum, which may have led to erroneous interpretations due to these preanalytical factors. Notably, significant discrepancies between serum and plasma levels of MMPs have been observed in healthy individuals participating in CRC screening. Research has indicated that serum levels of MMP-1, -2, -7, -8, -9, and -13 are significantly higher than those measured in citrate plasma. 19 Yet, another study reported the good correlation between serum and plasma concentrations of MMP-9 and -2, and TIMP-2 suggesting that the validity of previous studies using serum may not be affected by the differences in MMP concentrations associated with the use of serum instead of plasma. 20
Considering these publications, we opted to use blood plasma rather than serum to minimize the influence of these preanalytical variables.
Patients and methods
The cohort
The blood EDTA plasma samples from patients were provided by the Biobank of Faculty of Medicine in Pilsen Charles University and University Hospital Pilsen. A total of 148 patients (47 women and 101 men, aged 35–87 years, median 69.5 years) undergoing surgery for CRC at the Department of Surgery, University Hospital Pilsen, between 2008 and 2017 who had histologically verified malignant CRC, participated in the study. Additionally, 68 age-matched healthy subjects, with no history of past or present oncological disease, were included as controls. All subjects provided written informed consent prior to their inclusion in the study. The study protocol had received approval from the Local Ethics Committee at the University Hospital in Pilsen on March 18, 2008.
For the analysis of results based on the stage of CRC, the patient group was stratified into two subgroups: one group (Early, stage I + II) consisting of 85 patients and the other group (Advanced, stage III + IV) consisting of 60 patients. Survival data were collected from the National Oncological Registry of the Czech Republic.
Blood collection
Blood samples were collected within 3 days before surgery. Peripheral venous blood was obtained in EDTA plasma collection tubes following a standardized procedure, using the VACUETTE blood collection system (Greiner Bio-One Company, Kremsmünster, Austria). Plasma was separated by centrifugation at 1300×g for 10 min. The plasma specimens were then aliquoted and immediately frozen at −80 °C.
Plasma MMP and TIMP measurement
Multiplex panels (R&D Systems, Bio-Techne, Minneapolis, MN, USA) were used to measure the levels of MMP-2, -7, -8, -9, and -10, plus TIMP-1, -2, -3, and -4. The measurements were performed on the Luminex multiplex bead array system, according to the manufacturer’s protocol.
Statistical analysis
Statistical Analysis Software version 9.4 (SAS Institute Inc., Cary, NC, USA) was used for all statistical analyses. The Mann–Whitney U test was used to compare the groups. Inferential statistical analysis was conducted to evaluate time-to-event parameters, such as overall survival, using the Cox proportional hazards regression model. The Kaplan–Meier method was used to determine the probability of survival. The log-rank test was performed to compare survival outcomes between the two groups. Additionally, Wilcoxon Rank Sum tests were used to compare the distributions of selected parameters across groups. A P-value < 0.05 was considered statistically significant. Receiver operating characteristic (ROC) curves were generated to assess the diagnostic accuracy of each parameter, and the sensitivity and specificity at the optimal cut-off point were determined.
Results
Table 1 summarizes the clinical and pathological characteristics of the patients. The control cohort comprised 68 age- and sex-matched healthy subjects. The median patient age was 69.5 years (range 35–87 years), and 68% of patients were male (n = 101). Most patients (57%) had early-stage CRC (stages I and II). Of the cancers, 65% were localized in the colon and 35% in the rectum.
Clinico-pathological characteristics of colorectal cancer patients (n = 148).
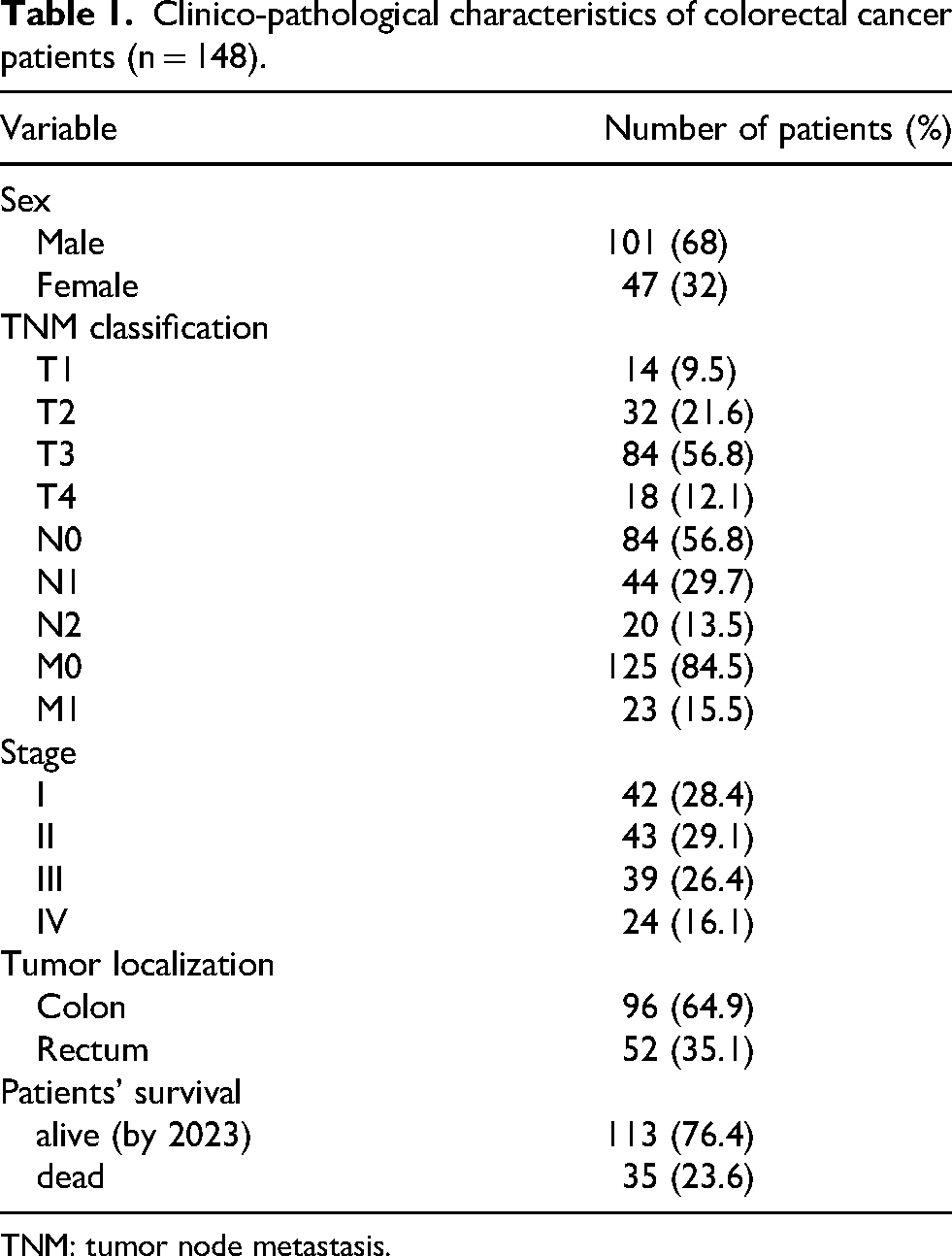
TNM: tumor node metastasis.
Analytical performance of MMP and TIMP assays
In Supplementary Table 1, several performance characteristics of MMP and TIMP assays are reported. The plasma samples were diluted 1:50 (MMP-2 and -9, and TIMP-1, -2, -3, and -4) and 1:10 (MMP-7, - 8, and -9) before analysis. The calibration was performed with 6 standards (MMPs) and 8 standards (TIMPs), and 5P logistic regression was used for the calibration curve plot. The MMP and TIMP levels in all samples were above the lower limits of quantification. Intra-assay coefficients of variations were between 4.3% and 7.9%, and inter-assay coefficients of variations were between 9.9% and 11.6%. In-house pooled serum was used as a control sample and was measured in duplicate; the results were consistent with the average value.
A comparison of plasma MMP and TIMP levels between patients and controls and their relation to tumor stage
Plasma concentrations of selected MMPs and TIMPs were measured in CRC patients (n = 148) and healthy subjects (n = 68), as shown in Table 2. The levels of MMP-8 and -9 were significantly higher in CRC patients compared to healthy subjects, with P-values of 0.0002 and 0.0009, respectively. Conversely, the levels of TIMP-2 were significantly lower in CRC patients than in healthy subjects (P = 0.0016). The plasma levels of MMP-2, -7, and -10, and TIMP-1, -3, and -4 did not show significant differences between the two groups.
Plasma levels of MMPs and TIMPs in CRC patients (n = 148) and healthy controls (n = 68).
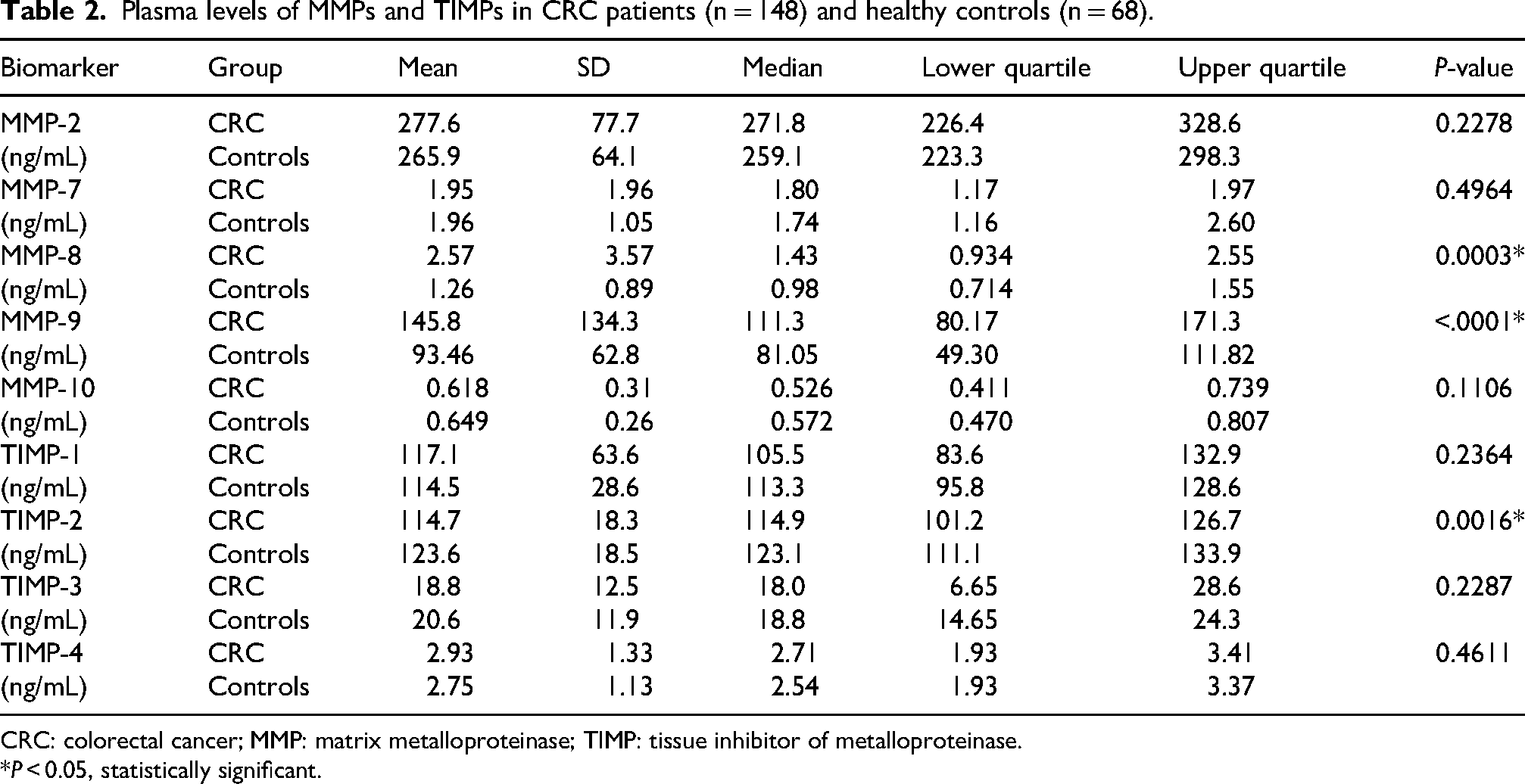
CRC: colorectal cancer; MMP: matrix metalloproteinase; TIMP: tissue inhibitor of metalloproteinase.
*P < 0.05, statistically significant.
We conducted an analysis of plasma levels of MMPs and TIMPs to determine how their levels correspond with disease stage. Patients were categorized into two groups: early stage (stages I and II) and advanced stage (stages III and IV). Our findings indicate that the concentrations of MMP-7 and -8, and TIMP-1 were significantly lower in the early stage compared to the advanced stage, with P-values of 0.0375, 0.0173, and <0.0001, respectively (see Table 3). Furthermore, plasma concentrations of MMP-10 were observed to be higher in the advanced stages, approaching statistical significance. Conversely, the levels of MMP-2, and -9, and TIMP-2, -3, and -4 did not exhibit significant differences between the early and advanced stages.
Plasma levels of MMPs in CRC patients at early and advanced stages of the disease (Early stage = stage I and II, n = 85, advanced stage = stage III and IV, n = 60).
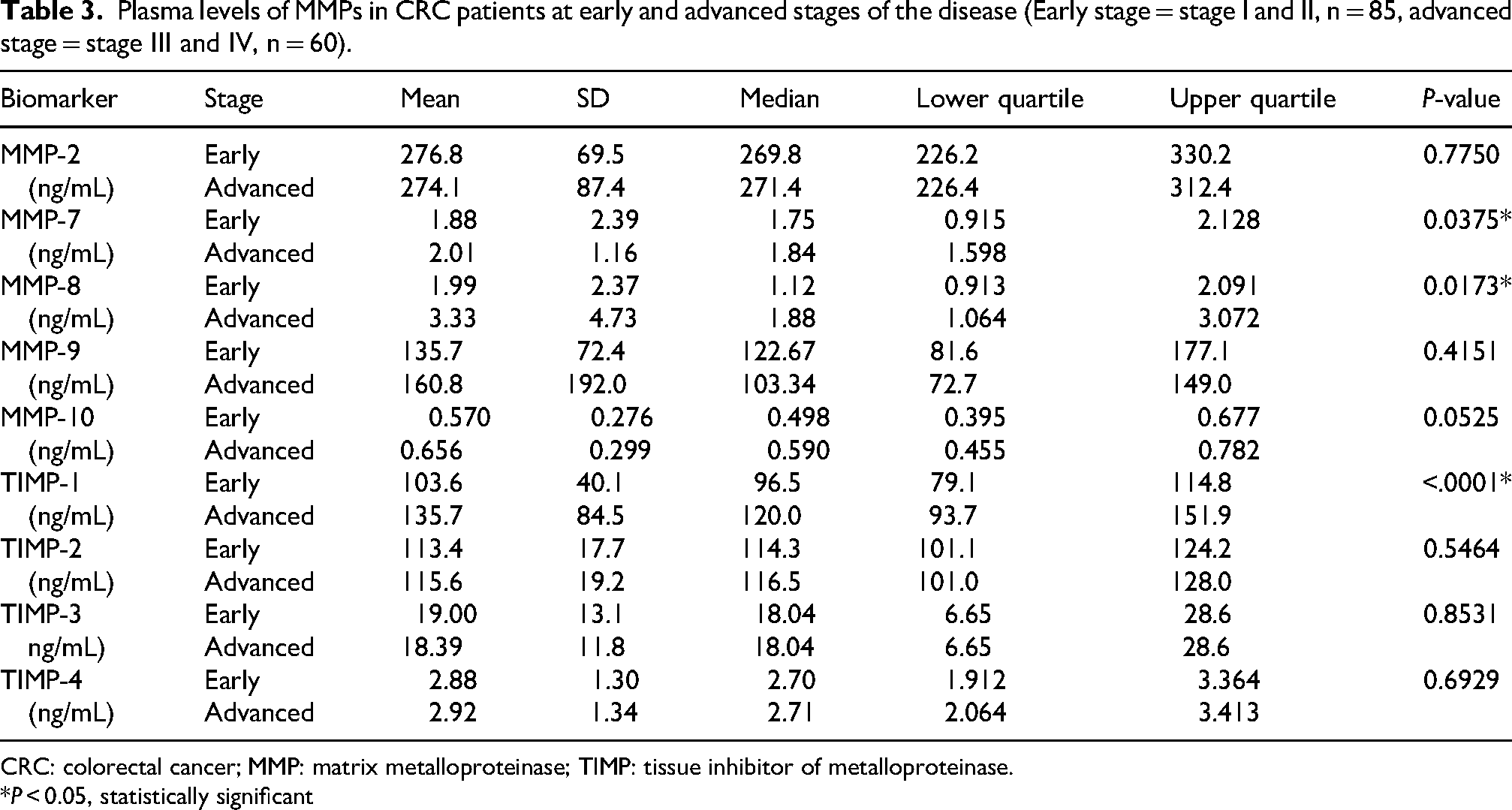
CRC: colorectal cancer; MMP: matrix metalloproteinase; TIMP: tissue inhibitor of metalloproteinase.
*P < 0.05, statistically significant
High SD values observed in Tables 2 and 3 were caused by a few outlier values for which there is no evidence to exclude them. However, non-parametric Wilcoxon two-sample test used for the evaluation, median, lower quartile, and upper quartile values are not affected by such outliers.
Evaluation of biomarker potential for diagnostics
The adjusted cut-off values for potential diagnostic biomarkers MMPs and TIMPs, specifically MMP-7, -8, and -9 and TIMP-1 were calculated to distinguish between normal and pathological levels. They are summarized in Table 4, together with other parameters. The sensitivities of MMP-8 and -9 at 95% specificity were 22.97% and 19.58%, respectively.
MMP and TIMP levels and calculated cut-off, sensitivity at 95% specificity, positive and negative predictive values and risk ratios.
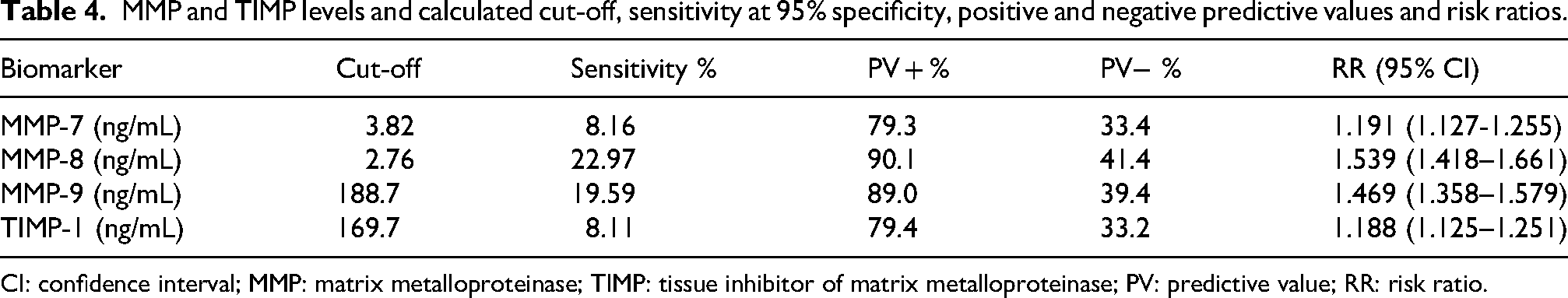
CI: confidence interval; MMP: matrix metalloproteinase; TIMP: tissue inhibitor of matrix metalloproteinase; PV: predictive value; RR: risk ratio.
The clinical performance for diagnostics was evaluated by ROC analysis. MMP-9, and -8 and TIMP-2 had the best area under curve (AUC) from all studied biomarkers, namely 0.6760, 0.6542, and 0.6357, respectively.
Analysis of survival and prognosis
Prognostic potential of studied biomarkers for overall survival (1800 days, i.e., 4.9 years) was evaluated. Univariable Cox regression analyses were conducted using logarithm base 10 of the plasma levels of biomarkers. Plasma MMP-7, and -8, and TIMP-1 were identified as independent prognostic factors (P-value <0.05), with TIMP-1 and MMP-7 demonstrating the most substantial prognostic potential (hazard ratios (HR) of 7.634 and 3.692, respectively). Kaplan–Meier graphs for MMP-7, and -8 and TIMP-1 are presented in Figure 1 (a) to (c).
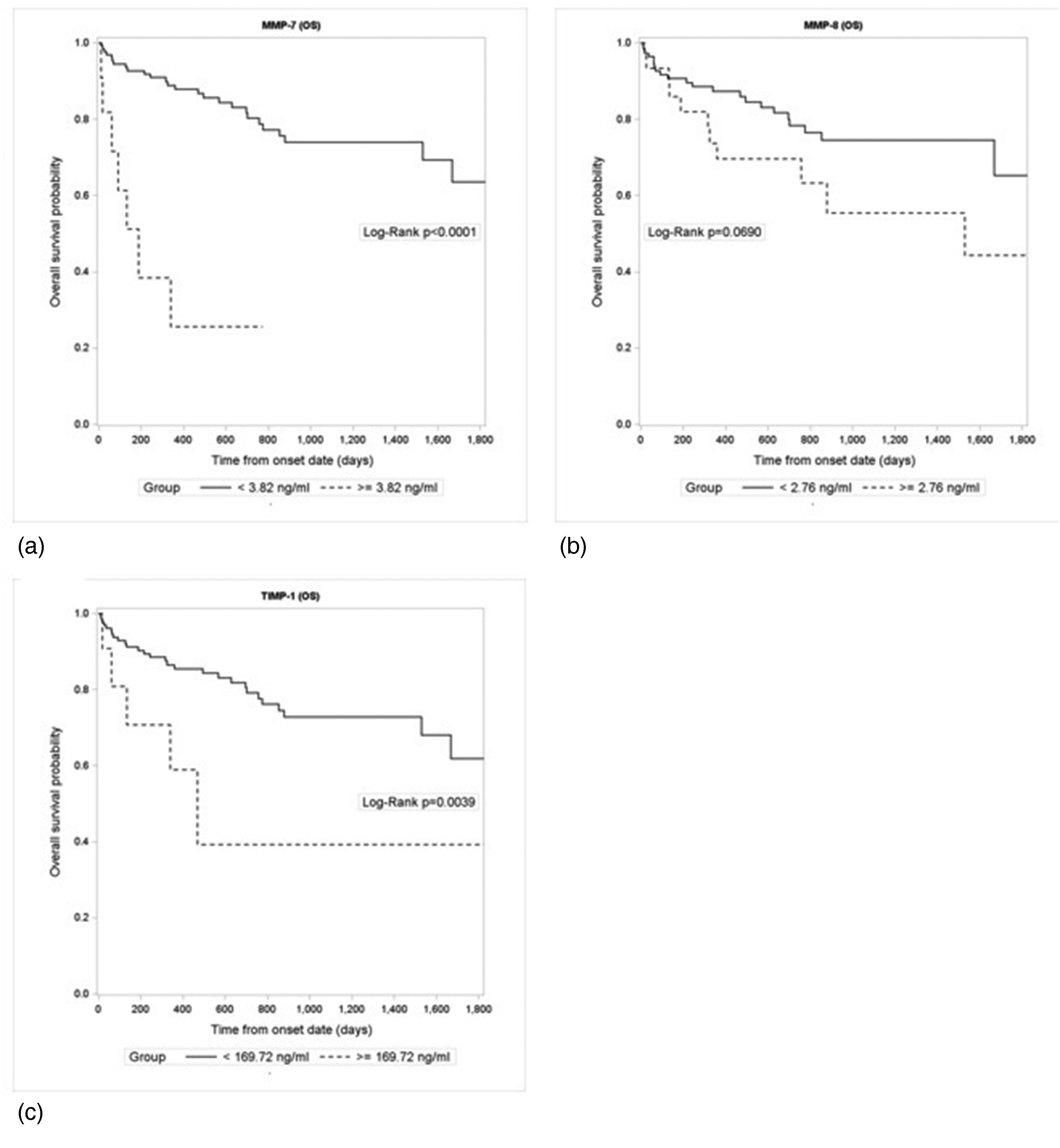
Kaplan–Meier curves of overall survival (OS) for MMP-7 (a), MMP-8 (b) and TIMP-1 (c).
Additionally, multivariate survival analysis was performed using a stepwise selection method based on all measured levels of MMPs and TIMPs. TIMP-1 was selected as most promising, showing an HR of 7.226 and a chi-square value of 19.80.
Discussion
MMPs and TIMPs are proteins that play significant roles in tumor growth, invasion, and metastasis, exhibiting both tumor-promoting and tumor-suppressive characteristics. Increased expression of MMPs and TIMPs in CRC tissues was observed. Numerous studies have explored the potential of different MMPs and TIMPs as additional circulating biomarkers for CRC; however, the findings have been inconsistent and, at times, conflicting.21–26 This variability may arise, among others, from the sensitivity of these proteins to pre-analytical issues, mainly the choice of biological material for analyses. Blood serum has been commonly used for such analyses21,22,24,25,27,28; however, it can result in false positive and inconsistent results, as MMPs and TIMPs are concentrated in leukocytes and platelets and may be released during the clotting process. Blood plasma (either EDTA or citrate) is more appropriate material for MMPs and TIMPs assays 19 and has been selected by some researchers.29–37 Probably the most consistent is the work of the Danish research group on plasma TIMP-1 as diagnostic and prognostic biomarker in CRC patients29,30,32,36: their prospective validation study confirmed plasma TIMP-1 as a stage independent prognostic marker for CRC patients. 29
In our study, we employed EDTA plasma to quantify the levels of MMP-2, -7, -8, -9, and -10, as well as TIMP-1, -2, -3, and -4 in pre-operative samples from CRC patients. Our objective was to assess their potential as diagnostic and prognostic biomarkers for CRC. With respect to consistency and to minimize a material bias, we only discussed the results obtained from plasma.
We observed that the concentrations of the measured proteins did not significantly differ in relation to clinicopathological variables, including T (tumor), N (nodal involvement), M (metastasis), tumor localization (colon vs. rectum), or the affected side of the colon (left versus right). We found that plasma levels of MMP-8, and -9, and TIMP-2 were significantly elevated in the CRC group compared with healthy controls. Conversely, the plasma levels of MMP-2, -7, and -10, and TIMP-1, -3 and -4 did not show significant differences between these two groups. The notable increase in serum levels of MMP-8 and -9 in CRC patients suggests that these MMPs may be associated with tumor progression. Similar findings were reported by Gimeno-Garcia et al. 33 regarding MMP-2 and -9. Gehrstein et al. 37 reported a significant increase in plasma MMP-9 but not MMP-7, TIMP-1 and TIMP-4 in CRC patients compared with healthy controls. The study of Tutton et al. 38 demonstrated that both plasma MMP-2 and -9 were increased in CRC patients and in relation to advanced cancer stage compared with healthy controls. In our study, plasma concentrations of MMP-7 and -8, and TIMP-1 were significantly higher in patients with advanced cancer (stages III and IV) relative to those with early-stage disease (stages I and II); other studied biomarkers did not show a difference. Nonetheless, ROC curve analyses found that the diagnostic value of these biomarkers was limited. Notably, plasma MMP-9 exhibited the highest performance among the proteins studied, achieving an AUC of 0.676.
We assessed the prognostic potential by analyzing survival and HR for each protein studied. Our results indicate that plasma MMP-7, MMP-8, and TIMP-1 could be independent prognostic factors (P-value <0.05); TIMP-1 and MMP-7 demonstrated the most substantial prognostic potential (HRs of 7.634 and 3.692, respectively), as determined by univariate regression analysis. In the multivariate analysis of all proteins examined, TIMP-1 was selected as the most promising independent prognostic factor, showing an HR value of 7.226. Similarly, high preoperative plasma levels of MMP-7 and TIMP-1 were independent unfavorable prognostic factors for CRC patients in the study of Kushlinskii et al. 39 The prospective validation study published by Birgisson et al. 29 confirmed plasma TIMP-1 as a stage independent prognostic marker for CRC patients.
MMP-7, or matrilysin, plays a role in cell proliferation and apoptosis, angiogenesis, invasion, and metastasis. According to immunohistochemical studies, MMP-7 expression correlates significantly with nodal or distant metastasis in colorectal carcinoma and seems to be useful in identifying high-risk groups for adverse outcomes in patients with early invasive colorectal carcinomas. 40 A significant difference in the level of mRNA MMP-7 expressions between tumor colorectal and control colorectal tissues was demonstrated and might be helpful for the prognosis of CRC. 41
MMP-8, also known as neutrophil collagenase, plays a significant role in mediating inflammation and therefore possesses antitumor activity. 42 A review of the literature indicates that in cancers such as breast, skin, and oral tongue, MMP-8 appears to inhibit cancer cell invasion and proliferation; conversely, elevated levels of MMP-8 in liver and gastric cancers have been associated with a poorer prognosis. 42 These findings indicate that some of the actions of MMP-8 might be tissue dependent. In the context of CRC, increased MMP-8 levels correlated with greater malignancy, reduced survival rates, and enhanced systemic inflammation. 28
TIMP-1 exhibits both MMPs inhibitory activities (e.g., inhibition of the tissue remodeling process) and MMP-independent functions (e.g., stimulation of cell proliferation, anti-apoptotic effect, pro- and anti-angiogenic effects, and so on). 43 Both retrospective and prospective studies demonstrated that elevated plasma TIMP-1 in combination with remodeling elevated CEA may be valuable diagnostics30,36,43 and prognostic 29 biomarkers of CRC. Our findings have indicated the prognostic but not the diagnostic role of plasma TIMP-1.
We are aware of some limitations concerning our study. The study was retrospective, using previously collected plasma samples from patients with verified CRC and age-matched healthy subjects as controls; this approach may introduce bias since samples from the patient cohort and the control subjects have not been collected simultaneously and therefore probably not according to similar standard operating procedures. Moreover, the control group was relatively small compared with the patients’ group. Finally, the results were not validated on an independent cohort. A prospective, multicentric study is needed to confirm our results.
Conclusion
Our retrospective study selected plasma MMP-7 and -8 and TIMP-1 as independent prognostic biomarkers of CRC with TIMP-1 having the highest potential. High plasma MMP-7, -8 or TIMP-1 may be associated with poor prognosis.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155251388492 - Supplemental material for Plasma matrix metalloproteinases 7 and 8 and tissue inhibitor of metalloproteinases 1 as prognostic biomarkers in colorectal cancer
Supplemental material, sj-docx-1-jbm-10.1177_03936155251388492 for Plasma matrix metalloproteinases 7 and 8 and tissue inhibitor of metalloproteinases 1 as prognostic biomarkers in colorectal cancer by Marie Karlíková, Magda Čurillová, Ladislav Pecen, Václav Karnos, Václav Karnos and Ondřej Topolčan in The International Journal of Biological Markers
Footnotes
Ethical considerations
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional review Board and Local Ethics Committee of the University Hospital in Pilsen, decision from March 18, 2008. All data were collected anonymously and securely, ensuring participant confidentiality.
Consent to participate
Informed consent was obtained from all participants prior to their inclusion in the study.
Consent for publication
Not applicable.
Author contributions
Marie Karlíková: formal analysis, investigation, methodology, validation, project administration, writing—original draft, writing—review and editing. Magda Čurillová: investigation. Ladislav Pecen: statistical analyses. Václav Karnos Jr.: investigation. Václav Karnos: conceptualization. Ondřej Topolčan: conceptualization, methodology, writing—review and editing. All authors have read and approved this final version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Health of the Czech Republic - conceptual development of research organisation (University Hospital in Pilsen - FNPl 00669806) and the project BBMRI.cz, reg. no. LM2023033.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
