Abstract
Background:
Several studies have suggested an association between tumor-associated autoantibodies (TAAbs) and breast cancer. However, most research has focused on imaging techniques, with few studies examining the combined use of TAAbs and imaging to distinguish benign and malignant breast nodules.
Methods:
Our study included 197 women with breast nodules. We collected clinical data, preoperative breast ultrasonography, and mammography results. Serum TAAbs were detected using enzyme-linked immunosorbent assay. Logistic regression analysis assessed the ability of factors to distinguish benign from malignant nodules. Receiver operating characteristic curves were plotted, and predictive models were constructed.
Results:
Five TAAbs (BRCA2, TP53, ATAD2, NY-ESO-1, CAGE) exhibited significant differences (p < 0.001) between benign and malignant breast nodules, with specificity of 95.83% (area under the curve = 0.722). Pearson's χ2 results revealed a close association between the levels of 5-TAAbs and breast cancer stages. Additionally, preoperative 5-TAAbs combined with ultrasonography and mammography increased diagnostic accuracy of benign nodules by 10.42% and reduced misdiagnosis of malignant nodules by 14.85%. Multivariate logistic analysis identified age (p < 0.001), preoperative 5-TAAbs (p = 0.004), breast ultrasonography (p = 0.01), and mammography (p < 0.001) as independent factors for malignant nodules. Combining 5-TAAbs with age and imaging improved differentiation, achieving sensitivity of 85.42% and specificity of 94.06%.
Conclusion:
Tumor-associated autoantibodies, especially the 5-TAAbs, have certain clinical value in the early diagnosis of breast cancer and can serve as an effective auxiliary tool for preoperative breast ultrasonography and mammography in patients with breast nodules.
Introduction
Breast cancer is the most common malignancy worldwide and the leading cause of cancer-related deaths in women. 1 According to data from the International Agency for Research on Cancer of the World Health Organization, breast cancer ranks first in terms of newly diagnosed cases of cancer. 2 In China, breast cancer is also the most common malignancy in women, with approximately 416,000 new cases reported annually, accounting for 18.4% of global cases. Breast cancer has age-standardized incidence and mortality rates of 36.1 and 8.8 per 100,000 population, respectively. 3 Early detection, diagnosis, and treatment are key factors in reducing the mortality of breast cancer. Screening in the population helps to detect breast cancer at an early stage, promote early treatment, and thereby reduce mortality. 4 Ultrasonography and mammography are commonly used screening methods for breast cancer, with ultrasonography being more widely used in China. 5 However, breast ultrasonography has limited sensitivity and specificity in identifying lesions and differentiating between malignant and benign nodules, especially in cases of dense breast tissue. 6 Furthermore, due to its lack of sensitivity, breast mammography provides reduced benefits for young women. 7 Importantly, breast mammography is not suitable for detecting early primary lymph node-negative breast cancer and ductal carcinoma in situ.8,9 Moreover, breast mammography can lead to false-positive results, especially in women—particularly those undergoing screening at a younger age (e.g., before 40) as they undergo more frequent examinations and subsequent follow-ups. 10 Statistics show that 1–10% of patients experience overdiagnosis through breast mammography. Although mammography and ultrasonography are commonly used for breast cancer screening, challenges still exist in differentiating between the benign and malignant conditions through imaging examinations. In addition, mammography involves a certain level of radiation exposure, and due to the limitations of imaging examinations, many patients with benign nodules also need to undergo painful and costly biopsy procedures. Thus, more effective diagnostic methods are urgently needed for distinguishing between the benign and malignant breast nodules.
The tumor microenvironment plays a decisive role in the occurrence, development, and treatment of tumors. 11 Due to somatic cell mutations and genomic instability, the proteome of tumor cells undergoes phosphorylation, acetylation, and glycosylation modifications, resulting in the generation of tumor-associated antigens (TAAs). When TAAs are recognized by the immune system, the body produces tumor-associated autoantibodies (TAAbs). 12 Through the amplification effect of humoral immunity, autoantibodies in the peripheral blood of patients are much more abundant than tumor-associated antigens, and their half-life is longer. 6 TAAbs have been studied as biomarkers or tools for early cancer detection because they can recognize antigens produced by human cells at low concentrations in a conformational manner. 12 In addition, their involvement in the pathogenesis and progression of cancer, including breast cancer, has been extensively studied. 13 Therefore, by detecting TAAbs in peripheral blood and combining them with imaging examinations is expected to improve the sensitivity and specificity of breast cancer screening, thereby reducing misdiagnosis and unnecessary biopsies. Based on the published literature and the public database GEPIA (http://gepia2.cancer-pku.cn/), eight TAAbs (BRCA2, 14 TP53, 15 NY-ESO-1, 16 PARP1, 17 ATAD2, 18 CAGE, 19 Annexin A11 20 and TRIM21 21 ) were closely related to the occurrence and development of breast cancer, which indicated their potential as biomarkers for the early diagnosis of breast cancer. Therefore, in this study, eight TAAbs were investigated in the serum of patients with breast nodules, to determine whether the diagnostic performance of the breast cancer can be improved with TAAbs involvement.
Materials and methods
Study subjects
This retrospective study included 197 female patients with breast nodules who received treatment at the Fudan University Shanghai Cancer Center from May 2019 to January 2022. The diagram for the flow of participants through the study was summarized in Figure 1. All patients were found to have breast nodules by breast mammography and ultrasonography, and were pathologically confirmed through surgery. According to the pathological results, patients with breast nodules were divided into the benign group (96 cases) and the malignant group (101 cases). Data were collected retrospectively from the medical records database, including results of breast mammography and ultrasonography, gender, age, menstrual status, tumor-node-metastasis (TNM) staging, etc. Inclusion criteria: (i) patients who underwent breast mammography and ultrasonography at Fudan University Shanghai Cancer Center and had complete clinical, pathological, and physiological data; (ii) patients who received breast-conserving surgery, total mastectomy, radical surgery, etc., for breast tumor treatment. Exclusion criteria: (i) pregnant and lactating women; (ii) patients with a history of breast surgery on the same or contralateral side; (iii) patients who received new adjuvant therapies such as chemotherapy or endocrine therapy before surgery; (iv) patients with other malignant tumor history; (v) patients with severe liver, kidney, or heart dysfunction or other systemic diseases; and (vi) patients with incomplete case data and missing surgical pathological results.
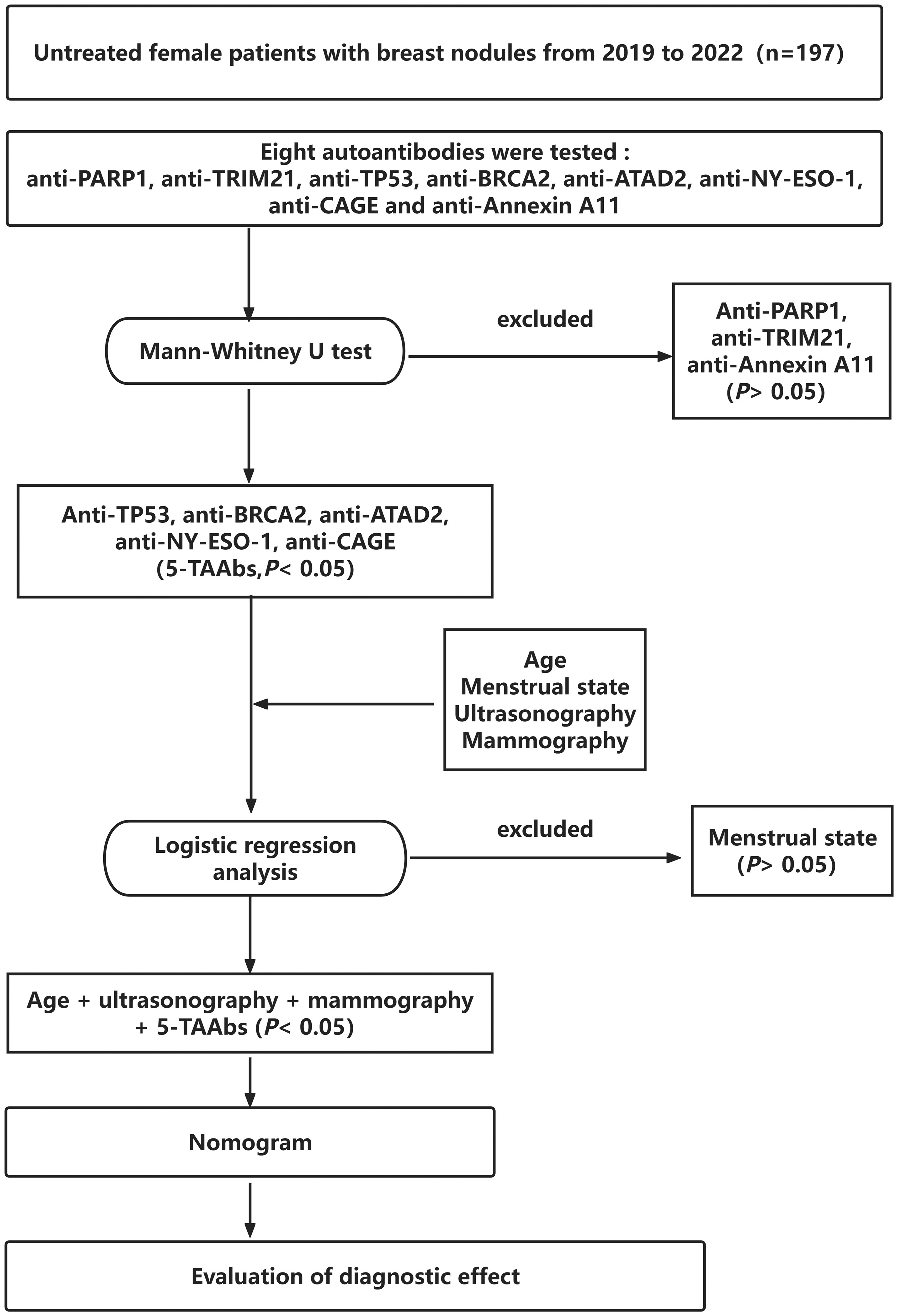
The flow chart of this study. From 197 untreated female breast nodule patients (2019–2022), eight autoantibodies were screened. Anti-PARP1 (p = 0.62), anti-TRIM21 (p = 0.18), and anti-Annexin A11 (p = 0.27) were excluded by Mann–Whitney U test (significance threshold p < 0.05). Five tumor-associated autoantibodies (5-TAAbs: anti-TP53, -BRCA2, -ATAD2, -NY-ESO-1, -CAGE; all p < 0.001) were retained. Subsequent logistic regression excluded menstrual state (p = 0.41), incorporating age, ultrasonography, mammography, and 5-TAAbs for nomogram development.
Methods
Serum samples were collected from patients’ pre-surgery and stored at −80 °C before testing. Serum levels of eight types of TAAbs were detected by enzyme-linked immunosorbent assay (ELISA). This study used commercial serum TAAbs detection kits purchased from Hangzhou Cancer Probe Biotechnology Co. Ltd to quantitatively detect eight types of TAAbs (anti-PARP1, anti-TRIM21, anti-TP53, anti-BRCA2, anti-ATAD2, anti-NY-ESO-1, anti-CAGE and Annexin A11). In addition to conducting a thorough review of the published literature, we also utilized the GEPIA 2 public database (http://gepia2.cancer-pku.cn/) for data analysis. First, we extracted gene expression data for breast cancer and normal tissues from the GEPIA 2 database. Second, we selected the relevant TAAbs based on their high expression levels and significant biological relevance in breast cancer, as identified through comparative analysis in the database. Finally, we assessed the expression levels of these markers and their correlation with breast cancer to support their potential as early diagnostic biomarkers. Optical density was measured using a BioTek ELx800 microplate reader (wavelength: 450 nm, software v2.01.14). Specifically, the serum samples were mixed and centrifuged at 5000 rpm for 5 min before detection to ensure consistency of results. The experimental procedure was conducted according to the manufacturer's instructions. First, 50 μL of diluted serum samples were added to wells coated with the corresponding antigens and incubated at room temperature for 1 h. After three washes, 50 μL of diluted secondary antibody (HRP-conjugated goat anti-human IgG) was added. Following the addition of the substrate, the color development reaction was terminated after 15 min by adding 50 μL of stop solution. The optical density at a wavelength of 450 nm was measured within 30 min after the reaction ended using the enzyme label analyzer, and determine the concentration of target TAAbs in reference units. All samples were tested simultaneously, and each sample was tested twice. During and after the TAAbs panel testing, the characteristics of the samples were blinded to the test performers.
Definitions
BI-RADS (Breast Imaging-Reporting and Data System), which is a tool used to grade the results of breast imaging examinations. According to the National Comprehensive Cancer Network breast cancer screening guidelines, the BI-RADS classification system published by the American College of Radiology is used as the standard for describing and assessing mammography and ultrasonography imaging features. BI-RADS lesions can be divided into six categories. 22 Among them, BI-RADS 0 indicates an incomplete evaluation that requires further imaging examinations (such as breast X-ray or magnetic resonance imaging) for further assessment. BI-RADS 1: the likelihood of malignancy is 0%. BI-RADS 2: benign findings with no malignant X-ray features, and the likelihood of malignancy is 0%. BI-RADS 3: used only for almost certain benign lesions, with a malignancy likelihood of 0–2%. BI-RADS 4: the likelihood of malignancy ranges from 2% to 95% and can be further subdivided into 4A (2–10% likelihood of malignancy), 4B (10–50% likelihood of malignancy), and 4C (50–95% likelihood of malignancy). BI-RADS 5: highly suspicious for malignancy, with a likelihood of malignancy ≥ 95%. BI-RADS 6: already confirmed as malignant through biopsy and requires aggressive treatment. 22 In this study, the results of breast ultrasonography and mammography are classified into two categories: BI-RADS ≤ 3 and BI-RADS > 3. BI-RADS ≤ 3 indicates a “benign” result, while BI-RADS > 3 indicates a “malignant” result.
Eight TAAbs in the serum of patients with breast nodules were detected in this study. The selection of the suitable cut-offs for each TAAbs was adjusted by combining the manufacturer's recommendations with the distribution of the specific antibody population level. The eight TAAbs “positive” reference value were as follows: anti-PARP1 (0–25 U/mL), anti-TRIM21 (0–10 U/mL), anti-TP53 (0–30 U/mL), anti-BRCA2 (0–30 U/mL), anti-ATAD2 (0–25 U/mL), anti-NY-ESO-1 (0–20 U/mL), anti-CAGE (0–30 U/mL), and anti-Annexin A11 (0–25 U/mL). If one or more TAAbs was higher than the positive judgment value, it was defined as “positive”; if only all TAAbs detection results were below the positive judgment value, it was defined as “negative.”
Statistical analysis
SPSS (version 25.0, IBM, Armonk, New York, NY, USA), R software (version 3.6.3, https://www.r-project.org/), and GraphPad Prism 8.0 were used for data analysis and plotting. Counting data were expressed as frequencies (percentages), and inter-group comparisons were performed using the chi-square test or Fisher’s exact test. Quantitative data conforming to a normal distribution were compared between two groups using the independent sample t-test, and multiple group comparisons were made using one-way analysis of variance. Non-normally distributed data between two groups were compared using the Mann–Whitney U test for two independent samples. Univariate and multivariate logistic regression analyses were used to screen independent indices (p < 0.05 indicated statistical significance) predicting the probability of malignant breast nodules. Receiver operating characteristic curves (ROC) were drawn with pathological results as the gold standard, the area under the curve (AUC) was calculated, and the diagnostic value of serum breast cancer-related TAAbs detection for differentiating between benign and malignant breast nodules was evaluated by calibration curves and calibration plots.
Results
Clinicopathological characteristics
A total of 197 female patients with histopathologically confirmed breast nodules were included in this study. The demographic characteristics are shown in Table 1. It was pathologically confirmed that 96 cases were benign breast nodules (including 66 fibroadenoma, 9 intraductal papilloma, 9 galactoma, 7 adenosis of mammary gland, 5 mammary hyperplasia) and 101 cases were malignant breast nodules (including 69 Luminal B subtype, 16 HER-2 positive, 10 Luminal A, and 6 triple-negative). Key differences emerged between the groups: patients with benign nodules had a median age of 40.75 years and were predominantly premenopausal (84.38%, 81/96), while those with malignant nodules had a higher median age of 53.99 years and were more likely to be postmenopausal (57.43%, 58/101). Based on the BI-RADS classification criteria of the American College of Radiology, most benign nodules were classified as BI-RADS ≤ 3 in both ultrasonography (87.50%) and mammography (87.50%). In contrast, most malignant nodules were classified as BI-RADS > 3 (ultrasonography: 72.28%; mammography: 83.17%). Additionally, for malignant breast nodules, TNM staging revealed that 28.71% were Stage I, 40.59% Stage IIA, 18.81% Stage IIB, 7.92% Stage III, and 3.96% Stage IV.
Baseline patient characteristics (n = 197).
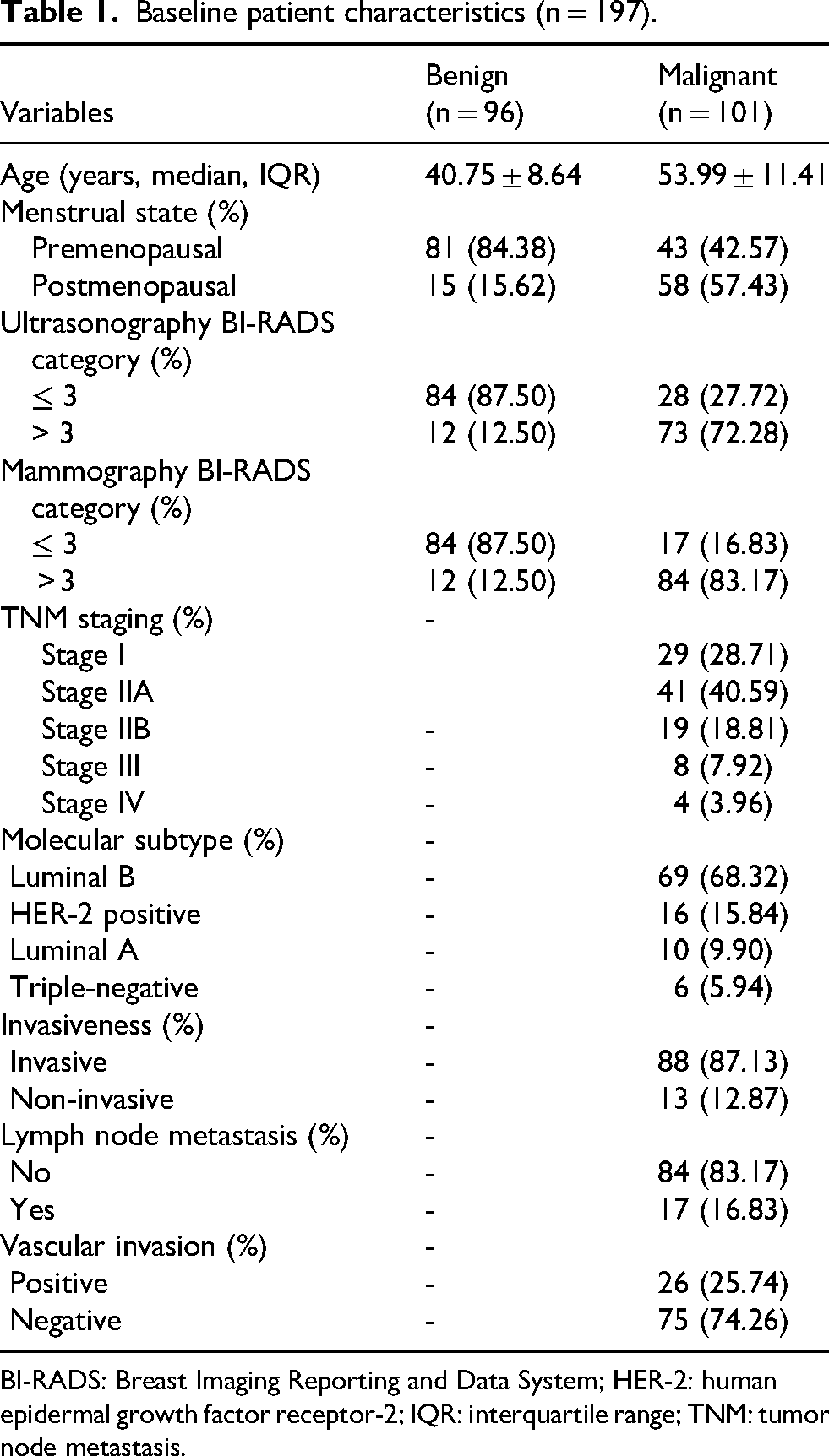
BI-RADS: Breast Imaging Reporting and Data System; HER-2: human epidermal growth factor receptor-2; IQR: interquartile range; TNM: tumor node metastasis.
Levels of TAAbs in patients with breast nodules
Serum levels of eight tumor-associated TAAbs, (BRCA2, TP53, ATAD2, NY-ESO-1, CAGE, TRIM21, Annexin A11, and PARP1) in patients with breast nodules were measured by ELISA. The results showed that five TAAbs (5-TAAbs: BRCA2, TP53, ATAD2, NY-ESO-1, and CAGE) exhibited significant differences between the benign and malignant breast nodules. In contrast, TRIM21, Annexin A11, and PARP1 showed no significant differences (p ≥ 0.05) (Figure 2(a)–(e)).
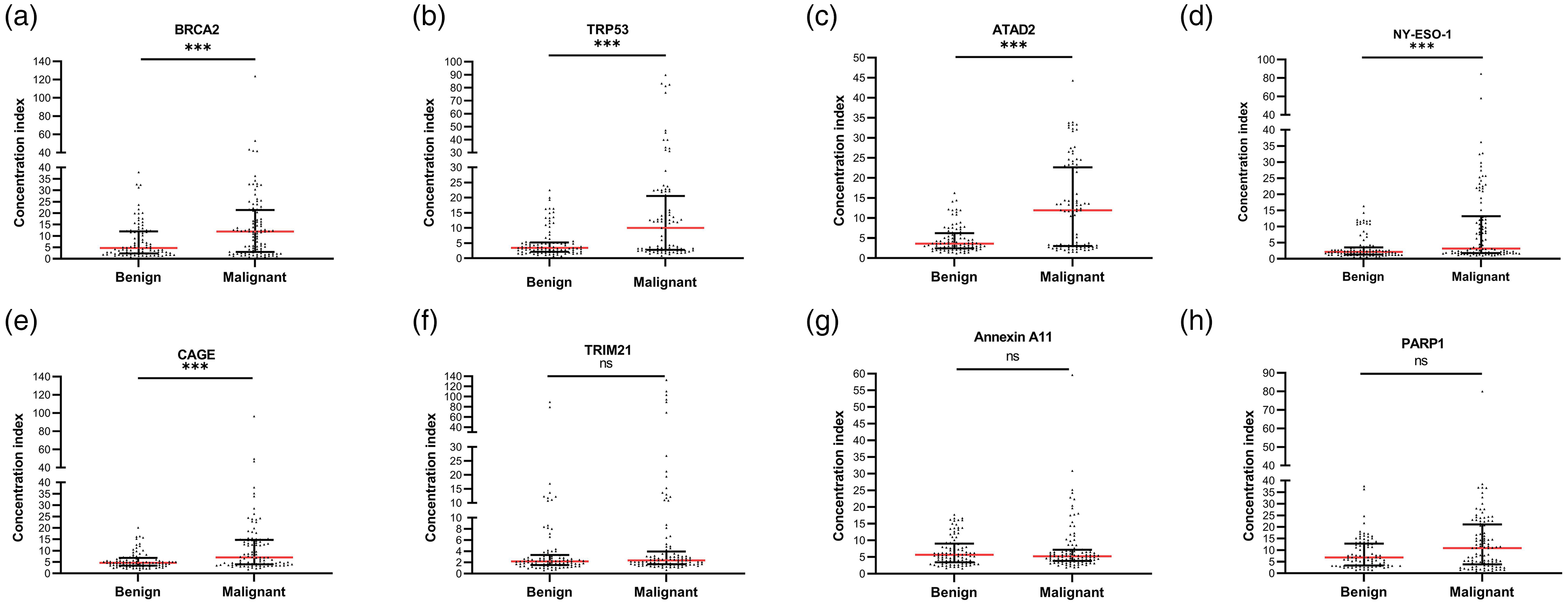
Serum TAAbs concentrations between the benign and malignant groups. Statistical significance by Mann–Whitney U test: ***p < 0.001; ns = not significant (p ≥ 0.05). Significantly elevated concentrations in malignant nodules were observed for anti-BRCA2 (a), anti-TP53 (b), anti-ATAD2 (c), anti-NY-ESO-1 (d), and anti-CAGE (e) (all p < 0.001).
Discrimination ability of the 5-TAAbs between the benign and malignant breast nodules
Non-parametric Mann–Whitney U tests revealed that the ROC curve was plotted for the TAAbs with significant differences, and the AUC was calculated. Among the five TAABs, TP53 demonstrated the highest discriminative power with an AUC of 0.681, followed by NY-ESO-1 (AUC = 0.666), CAGE (AUC = 0.664), BRCA2 (AUC = 0.630), and ATAD2 (AUC = 0.626). The standard errors of the ROC curves were 0.038 for TP53, 0.038 for NY-ESO-1, 0.039 for CAGE, 0.040 for BRCA2, and 0.037 for ATAD2, with asymptotic significance levels of p < 0.001 for TP53, NY-ESO-1, CAGE, and ATAD2, and p = 0.002 for BRCA2. Further analysis revealed that the combined detection of 5-TAAbs had a sensitivity of 46.53%, a specificity of 95.83%, a positive predictive value of 92.15%, a negative predictive value of 63.01%, and an accuracy of 70.56% in distinguishing benign from malignant breast nodules. The ROC analysis showed an AUC of 0.722 for the combined detection of 5-TAAbs (Figure 3).
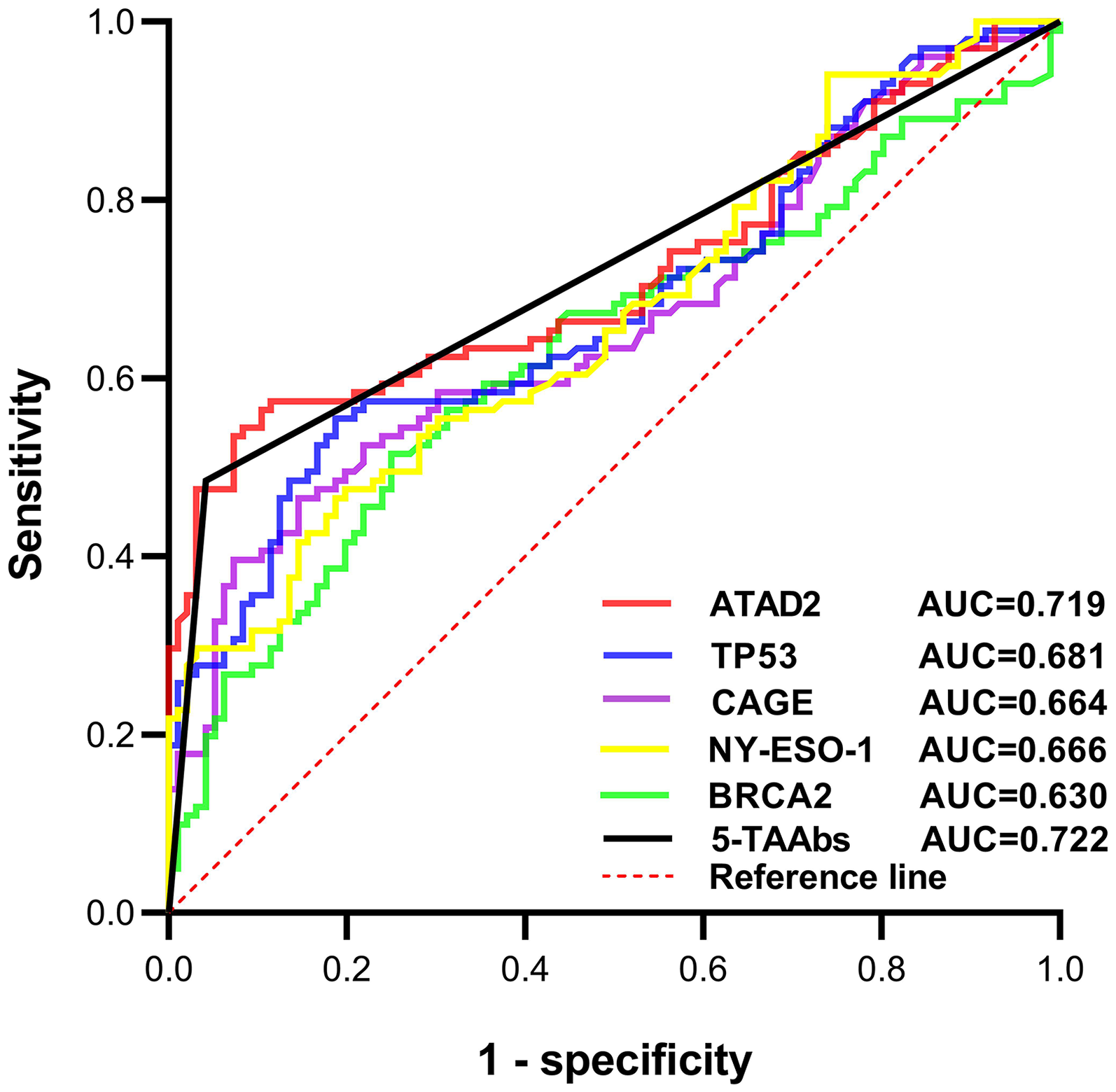
ROC curve analysis of five TAAbs in breast nodules. AUC values with 95% confidence intervals: 5-TAAbs = 0.722 (0.65–0.79), ATAD2 = 0.719 (0.65–0.79), TP53 = 0.681 (0.61–0.75). Dashed line: reference (AUC = 0.5).
Relationships between 5-TAAbs and clinicopathological parameters of breast cancer
The association of 5-TAAbs with the clinical data, including TNM staging, molecular subtype, tumor infiltration, lymph node metastasis, and vascular invasion in 101 breast cancer patients was analyzed. Results revealed a close association between the levels of 5-TAAbs and TNM staging in breast cancer (p < 0.001), indicating that higher levels of 5-TAAbs were associated with more advanced stages of breast cancer (Table 2). However, no significant correlations were observed between the levels of 5-TAAbs and molecular subtype (p = 0.175), tumor infiltration (p = 0.68), lymph node metastasis (p = 0.507), or vascular invasion (p = 0.462) (Table 2).
Association between 5-TAAbs and clinical pathological parameters of breast cancer (n = 101).
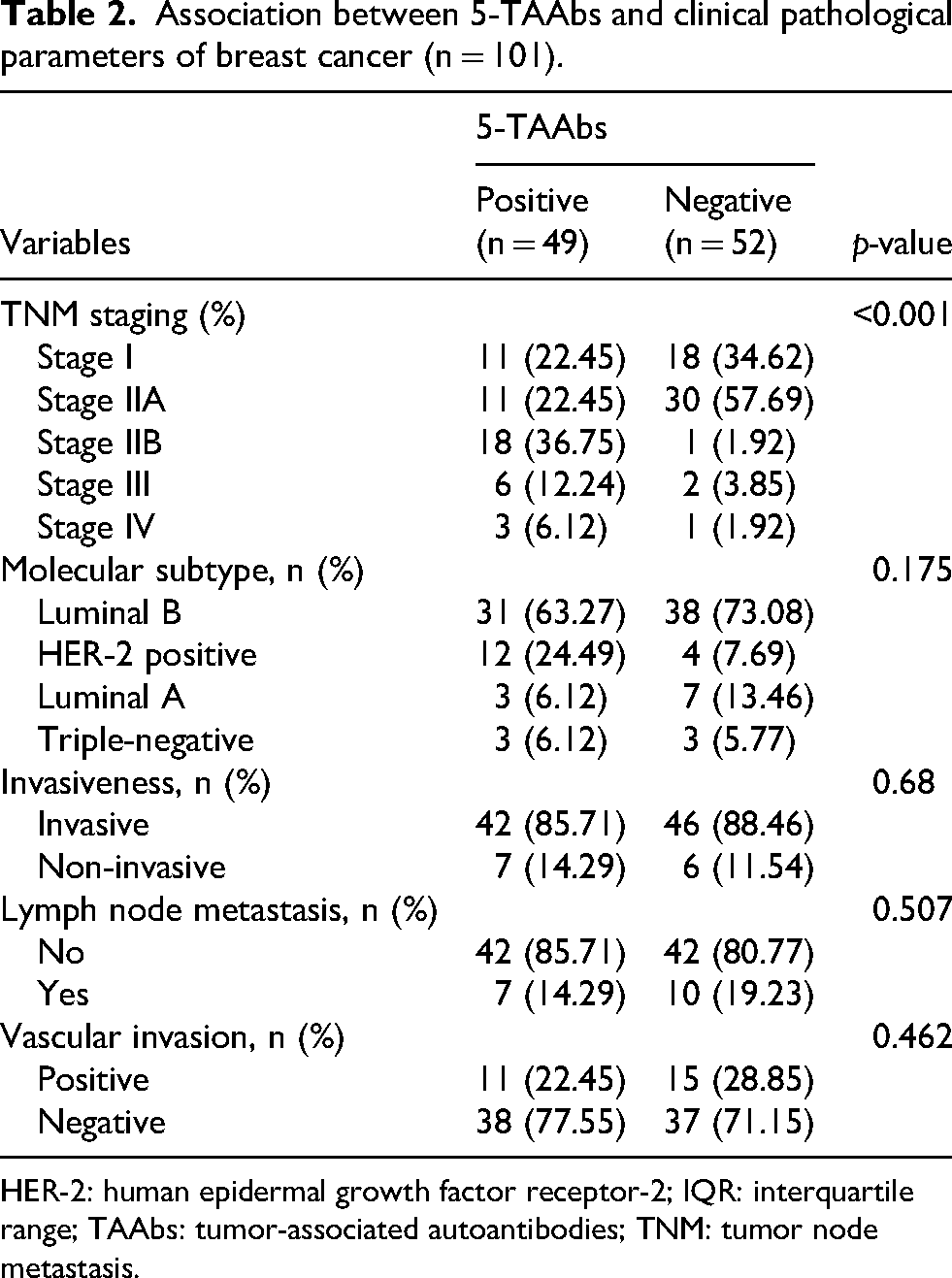
HER-2: human epidermal growth factor receptor-2; IQR: interquartile range; TAAbs: tumor-associated autoantibodies; TNM: tumor node metastasis.
Combination detection of 5-TAAbs is an effective auxiliary tool for breast ultrasonography and mammography in malignant breast nodules diagnosis
In this study, 197 cases of breast nodules underwent preoperative breast imaging examinations, including breast ultrasonography and mammography. After postoperative pathological diagnosis, 96 cases were diagnosed as benign, and 101 cases were diagnosed as malignant. Among the 96 patients with benign nodules, 84 patients were classified as BI-RADS category 3 or lower based on preoperative imaging examinations, with a diagnostic accuracy of 87.50% (84/96); however, incorporating 5-TAAbs detection improved diagnostic accuracy rate by 10.42%. Meanwhile, 14 patients were classified as BI-RADS category 3 or higher, with a false-positive (misdiagnosis) rate of 14.58% (14/96); however, incorporating 5-TAAbs detection reduced the misdiagnosis rate by 2.08% (Figure 4(a)). Among the 101 patients with malignant nodules, 39 were classified as BI-RADS category 3 or lower based on preoperative imaging examinations, with a misdiagnosis rate of 38.61% (39/101). This group was likely missed by imaging due to the subtle characteristics of malignant tumors that standard imaging techniques may fail to adequately visualize, particularly in cases of dense breast tissue. However, incorporating 5-TAAbs detection reduced misdiagnosis rate by 14.85%. Simultaneously, 95 patients were classified as BI-RADS category 3 or higher, with a diagnostic accuracy rate of 94.06% (95/101); furthermore, incorporating 5-TAAbs detection increased the diagnostic accuracy by 2.97% (Figure 4(a)).
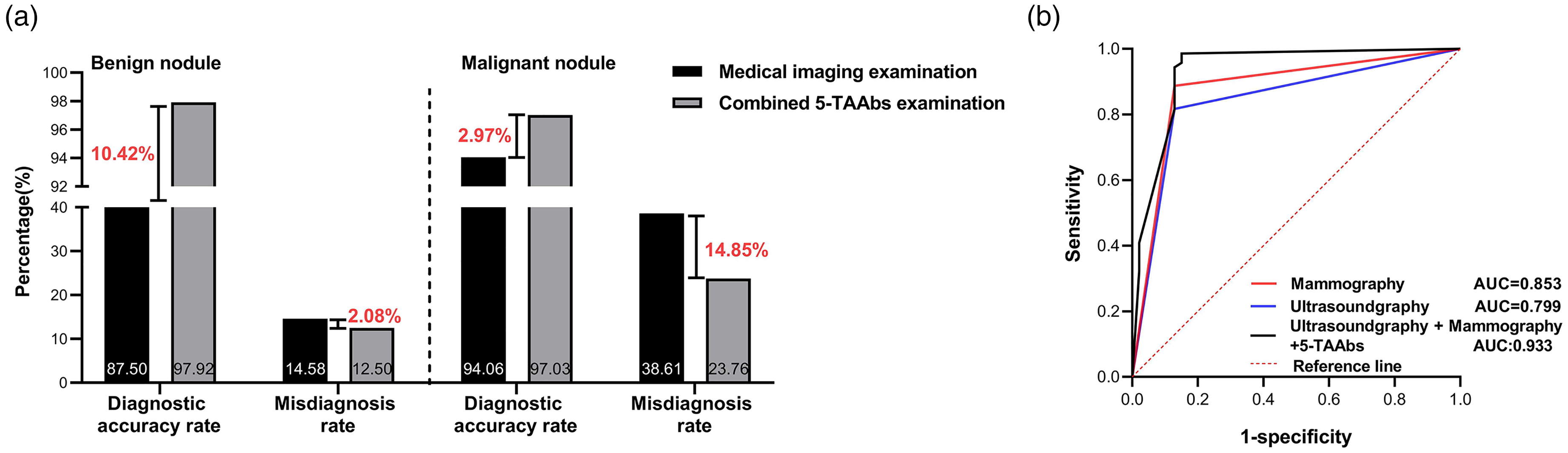
Diagnostic performance of combined 5-TAAbs detection with breast ultrasonography and mammography for breast nodules. (a) Comparison of diagnostic accuracy and misdiagnosis rates between conventional medical imaging alone and combined 5-TAAbs detection in benign and malignant breast nodules. Incorporation of 5-TAAbs increased diagnostic accuracy by 10.42% and reduced misdiagnosis rate by 2.08% for benign nodules. For malignant nodules, 5-TAAbs improved diagnostic accuracy by 2.97% and reduced misdiagnosis rate by 14.85%. (b) Receiver operating characteristic (ROC) curves for mammography (red), ultrasonography (blue), and their combination with 5-TAAbs (black) in differentiating high-risk breast lesions (B3–B4). The combination achieved superior discriminatory power (AUC = 0.933, 95% CI: 0.894–0.973), compared to mammography (AUC = 0.853) and ultrasonography (AUC = 0.799) alone. The combined approach demonstrated a sensitivity of 98.6% and specificity of 84.9%.
Furthermore, given the diagnostic challenges associated with B3–B4 lesions, where conventional imaging often shows reduced accuracy, the ROC curve analysis demonstrated that the combination of 5-TAAbs with ultrasonography and mammography provides superior discriminatory power (Figure 4(b)). Specifically, this combination achieved an AUC of 0.933 (95% confidence interval (CI): 0.894–0.973), compared to ultrasonography alone (AUC = 0.844, 95% CI: 0.778–0.910) and mammography alone (AUC = 0.879, 95% CI: 0.821–0.937). Additionally, the combined diagnostic approach achieved a sensitivity of 98.6% and a specificity of 84.9%. This enhancement confirms the critical role of 5-TAAbs in supplementing breast imaging examinations, providing a more reliable basis for clinical decision-making regarding high-risk lesions.
Univariable and multivariable logistic regression analyses of indicators related to the breast nodules
The univariate analysis demonstrated that age (p < 0.001), menopausal state (p < 0.001), ultrasonography and mammography BI-RADS categories (p < 0.001), and 5-TAAbs (p < 0.001) were significantly associated with the presence of breast nodules (Table 3). In the subsequent multivariate analysis, adjusting for potential confounders, we found that except for menopausal status, the other factors remained independent predictors of breast cancer development. Menopausal status may lack significance due to its strong collinearity with age, as well as the inherent variability in hormonal levels that can influence breast tissue. Notably, 5-TAAbs emerged as a strong independent predictor, with an adjusted odds ratio of 16.43 (95% CI: 4.11–65.74, p < 0.001), suggesting its stable association with malignancy, though the wide confidence interval reflects uncertainty due to sample size constraints (Table 3). ROC curves were plotted to independently evaluate the diagnostic performance of each indicator (age, ultrasonography, mammography and 5-TAAbs) and their combination, as well as the corresponding sensitivity and specificity. The results showed that the AUC of the combined detection was 0.957, which was significantly higher than that of the individual indicators: the AUC for ultrasonography was 0.799, for mammography was 0.853, and for 5-TAAbs was 0.722. With a sensitivity of 85.42% and a specificity of 94.06%, indicating that when the four indicators involved in this model were combined for the differentiation of benign and malignant breast nodules, optimal performance was achieved (Figure 5(a)). Based on these findings, we developed a preoperative breast nodule benign–malignant discrimination nomogram that demonstrates high predictive performance and may serve as a clinically useful tool to assist in preoperative diagnosis. The nomogram formula is as follows: Score = 1.103 + (−0.121) × [Age] + 1.759 × [Ultrasonography BI-RADS category] + 2.292 × [Mammography BI-RADS category] + 2.87 × [5-TAAbs]. When using the nomogram, the corresponding score sum is calculated by locating each variable on the axis and its corresponding point, thereby determining the predicted probability of breast nodule benign–malignant discrimination on the bottom axis (Figure 5(b)). In addition, the calibration curve for the prediction model (Figure 5(c)) demonstrated excellent agreement between the predicted values and actual observed values for the preoperative benign–malignant discrimination of breast nodules. This strong calibration is further supported by a C-index of 0.957 (95% CI: 0.918–0.987) and an average absolute error of 0.032. This integrated model provides a clinically actionable tool for individualized preoperative risk stratification, potentially reducing unnecessary biopsies while improving early malignancy detection.
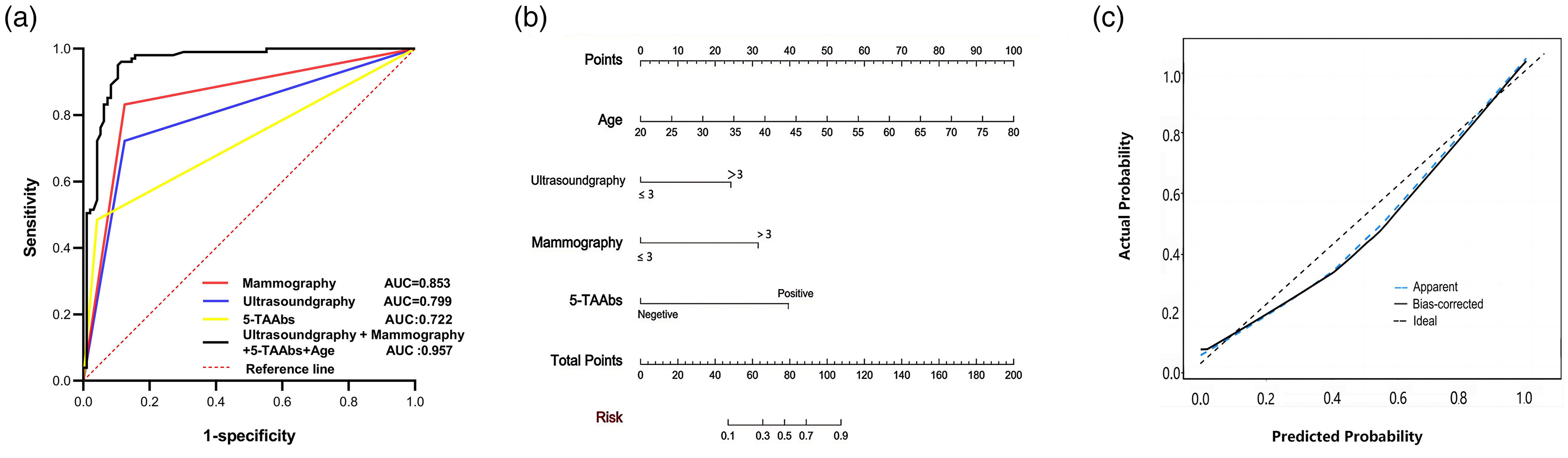
Benign–malignant breast nodule discrimination based on age, 5-TAAbs, ultrasonography, and mammography. (a) ROC curve analysis of ultrasonography, mammography and 5-TAAbs in breast nodules. (b) Nomogram model of serum TAAbs associated with malignant breast nodules. (c) Calibration curve of the model of the nomogram.
Logistic regression analysis of the factors associated with the breast nodules.
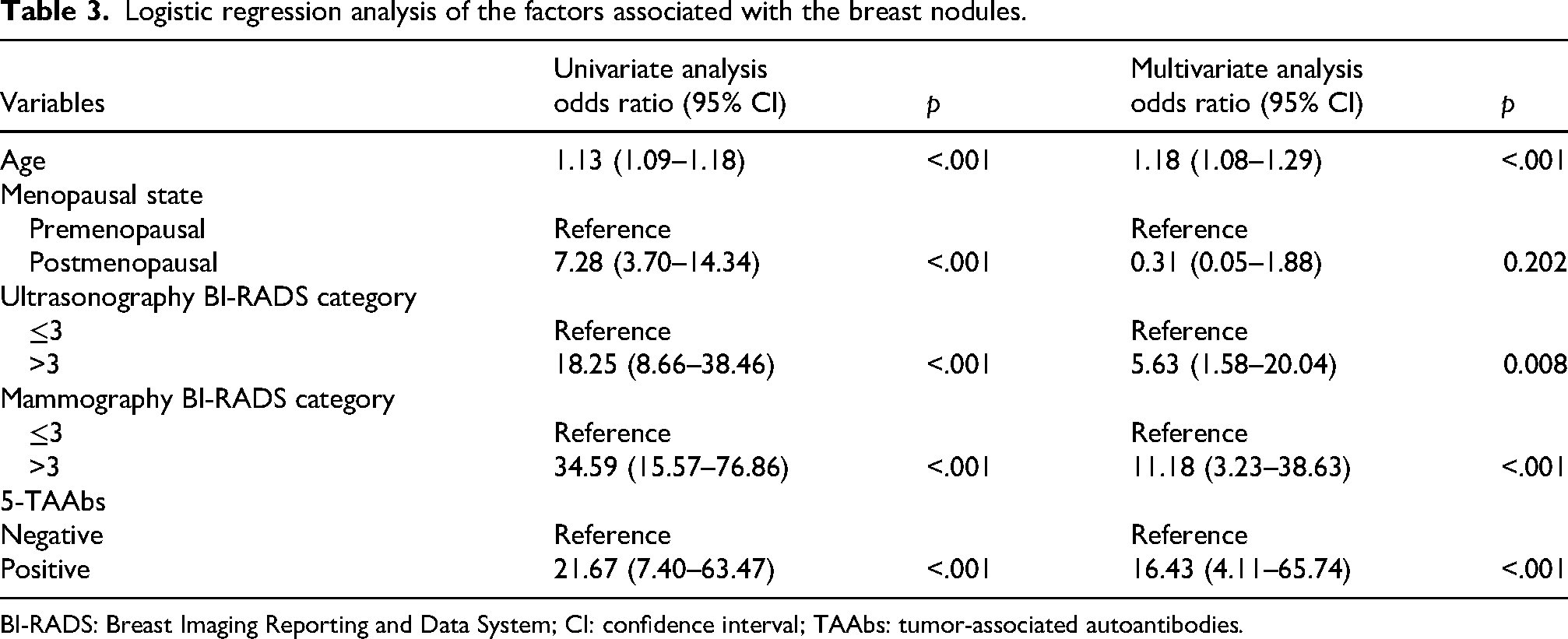
BI-RADS: Breast Imaging Reporting and Data System; CI: confidence interval; TAAbs: tumor-associated autoantibodies.
Discussion
Breast nodules are a common sign in women, often caused by an imbalance of endocrine hormones, presenting with clinical symptoms such as breast lumps, cyclical breast pain, and nipple discharge. 23 The development of breast cancer is a long-term process involving multiple genetic changes and stages of accumulation. The lack of specific clinical symptoms and efficient biomarkers complicates the early differentiation of benign and malignant breast nodules. 24 Endogenous antibodies can be amplified and measured in the early stages of cancer, making them promising biomarkers for distinguishing breast cancer from healthy tissue.6,25 Numerous studies have shown that TAAbs, including tumor-associated types, can be investigated as biomarkers for early cancer detection due to their ability to recognize low concentrations of antigens not affected by tissue density.22,26 Specifically, various TAAbs have demonstrated promise in the early detection of lung cancer, with both individual antibodies and their panels serving as effective serum biomarkers to differentiate malignant tumors from benign lung nodules. 27 Similarly, previous studies have also indicated that various endogenous TAAbs can effectively detect breast cancer, particularly holding potential value in early breast cancer screening. 26 For example, TP53 antibodies were found in breast cancer patients but absent in healthy controls. 28 Blixt et al. 29 reported significantly higher levels of MUC365 TAAbs in early breast cancer patients compared to those with benign conditions. Zhu et al. 30 discovered significantly higher TAAbs responses to PARP1 and BRCA1/BRCA2 in breast cancer sera. In this study, we evaluated eight tumor-associated endogenous antibodies, identifying five (anti-BRCA2, anti-TP53, anti-ATAD2, anti-NY-ESO-1, anti-CAGE) with potential as tumor biomarkers for early breast cancer, indicating cancer presence and an enhanced immune response. Additionally, Hong et al. 31 reported that a panel of TAAbs pane (BMI-1, HSP70, MMP-7, NY-ESO-1, and PRDX6) could differentiate early breast cancer patients from normal controls, with a sensitivity of 59.6% and a specificity of 90.2%. Hurley et al. 32 found that TRIM21 TAAbs, in combination with NY-ESO-1 and TP53, serve as biomarkers for early detection in ovarian cancer. Wang et al. 33 tested 13 TAAbs in basal-like breast cancer patients, finding that TP53, TRIM21, and others effectively differentiated basal-like breast cancer from other types with 33% sensitivity and 98% specificity. In this study, 5-TAAbs demonstrated improved discriminatory ability compared to individual TAAbs in distinguishing between benign and malignant breast nodules, with good diagnostic performance (AUC = 0.702). Moreover, 5-TAAbs was correlated with the clinical stage of breast cancer and served as an independent factor for differentiating benign and malignant breast nodules, exhibiting high specificity (95.83%).
Currently, ultrasonography and mammography are commonly used screening methods for breast cancer. 34 However, both mammography and ultrasonography have significant limitations in breast cancer diagnosis. Mammography has only 20% sensitivity for 85 mm tumors and even lower for smaller ones, leading to missed diagnoses. Additionally, some breast cancers grow along normal structures, making detection difficult, and false-positives are a common issue with both methods.35,36 Additionally, breast ultrasonography has reduced sensitivity in dense breast tissue, prevalent in younger women. 37 At the same time, breast imaging tests such as mammography and ultrasonography can also lead to false-positives or overdiagnosis, resulting in unnecessary treatments and psychological distress. 38 According to the 2021 Chinese Anti-Cancer Association guidelines, BI-RADS category 4 patients should undergo biopsies, which can lead to unnecessary trauma and false diagnosis rates. 39 Furthermore, despite advancements in medical imaging technologies, these methods rely on costly equipment and specialized training, limiting their application in large-scale screening.40–42 Therefore, there is an urgent need to find more effective diagnostic methods to improve the ability to differentiate breast nodules.
This study found that adding 5-TAAbs testing before surgery in patients with breast nodules serves as an effective auxiliary tool for breast ultrasonography and mammography. Specifically, this combination increases the diagnostic accuracy of benign breast nodules by 10.42% and reduces the misdiagnosis rate of malignant breast nodules by 14.85%. Furthermore, we conducted a comprehensive analysis of the independent predictive significance of age, breast ultrasonography and mammography BI-RADS classification, and 5-TAAbs, as well as their combined testing for benign and malignant breast nodules. The combination of these methods achieved a sensitivity of 85.42% and a specificity of 94.06%. This combined testing significantly enhanced overall diagnostic performance, with an AUC of 0.944, indicating that the combination of “Ultrasonography + Mammography + 5-TAAbs + Age” has the potential to assist in distinguishing between benign and malignant breast nodules and has certain value in the diagnosis of breast cancer. This combination allows for a more detailed understanding of breast nodules, potentially leading to improved diagnostic accuracy and reduced rates of unnecessary biopsies. By assessing the combined effects of these markers, this study offers a novel perspective on their role in enhancing breast cancer diagnosis.
Finally, it should be noted that although TAAbs testing shows potential in early breast cancer screening, it is currently in the research stage and has not been widely applied in clinical practice. Except for the limitations of retrospective studies, this study established the model based on a single center. Therefore, we will focus on expanding the sample size and conducting further external validation in diverse populations across multiple centers to determine the exact value and clinical prospects of these TAAbs as reliable biomarkers for breast cancer in future research, thereby providing more possibilities for the earlier intervention of breast cancer.
Conclusion
Tumor-associated autoantibodies not only serve as early biomarkers for breast cancer detection and prevention but also act as strong assistants in breast mammography and ultrasonography. The combination of breast mammography, ultrasonography, and TAAbs tests has greater clinical significance in distinguishing between benign and malignant breast nodules. It improves the diagnostic efficacy of breast cancer and can be used as an adjunctive diagnostic tool, providing more reference information and diagnostic evidence for clinical practice.
Footnotes
Acknowledgments
We gratefully acknowledge the Department of Clinical Laboratory, Fudan University Shanghai Cancer Center for providing clinical test data and professional analysis support.
Author contributions
Xiaorong Yang: Writing—original draft, methodology, investigation, formal analysis, data curation. Tianqing Yan: Writing—review and editing, methodology, investigation, formal analysis. Yanchun Wang: Methodology, investigation, formal analysis. Ying Tong: Funding acquisition, supervision, project administration. Xiaolu Ma: Funding acquisition, supervision. Lin Guo: Writing—review and editing, validation, supervision. Renquan Lu: Writing—review and editing, Investigation, Funding acquisition, Conceptualization. Xiaorong Yang and Tianqing Yan contributed equally to this work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number NSF-82072876, NSF-82373383, NSF-82372332, NSF-82303314).
Data availability statement
Data will be made available on request.
Informed consent statement
Written informed consent was obtained from all patients for the use of the medical records for research purposes.
Institutional review board statement
This study was approved by the ethics committee of the Fudan University Shanghai Cancer Center. Approval Number: 050432-4-2307E
