Abstract
Introduction
Breast cancer is the most common cancer among women, and metabolic syndrome (MetS) is a risk factor for breast cancer, especially postmenopausal breast cancer. We evaluated the role of the advanced glycated end products (AGEs) levels contributing to the association between MetS and breast cancer risk.
Methods
Plasma AGEs were measured in a case-control study nested within the Hormones and Diet in the Etiology of Breast Tumors (ORDET) cohort, including 40 incident postmenopausal breast cancer cases (20 with MetS and 20 without) and 40 postmenopausal controls (20 with MetS and 20 without). The association between AGEs and breast cancer was analyzed using Bayesian logistic regression models. An informative prior for the exposure coefficient, modeled as a normal distribution, centered on the natural logarithm of an odds ratio ((OR)=1.635) derived from prior evidence, was employed alongside weakly informative priors (WIPs). Bayesian linear regression with WIPs was used to examine the association between MetS and AGEs. Estimates were reported with SDs and 90% and 95% credible intervals (CI).
Results
AGEs were associated with higher breast cancer risk both with the informative prior (OR = 1.745, SD):0.362; 90% CI:1.218–2.390; 95% CI:1.137–2.548) and the WIP (OR = 1.861, SD = 0.661; 90% CI:1.026–3.082; 95% CI:0.924–3.528) specification. Although the difference in plasma AGEs in women with and without MetS was not significant, we found a suggestion of higher levels in women with MetS (mean difference in standardized AGEs between individuals with and without MetS = 0.155, SD = 0.245; 90% CI:−0.246 to 0.553; 95% CI:−0.322 to 0.625).
Conclusions
These data, although from a small sample of women, support a role of endogenous AGEs in the pathological pathways underlying the association between MetS and breast cancer development.
Introduction
Breast cancer (BC) is the most diagnosed cancer among women. 1 Older age, tobacco use, obesity, and sedentary lifestyle are risk factors for BC. 2
Metabolic syndrome (MetS), a cluster of metabolic abnormalities that develops in people with impaired insulin sensitivity, is also a major risk factor for BC. MetS is defined by the presence of any three of the following: hyperglycemia, hypertension, hypertriglyceridemia, low high-density lipoprotein (HDL) cholesterol, and large waist. 3 A recent meta-analysis found higher BC risk in postmenopausal women with MetS.4,5
Several mechanisms may explain the association of MetS with BC. Insulin resistance-pathophysiological state underlying MetS can increase risks by increasing levels of insulin-like growth factor (IGF)-1 (which has mitogenic and anti-apoptotic effects) plus estrogen and androgen.6,7 Despite these mechanisms, some people with MetS do not have insulin resistance, 8 and some obese individuals do not have MetS. 9 Thus, neither obesity nor insulin resistance can fully explain the traits associated with MetS.
Epidemiological studies mainly investigated hypothesized mediators of the MetS-BC association: expression of individual genes/proteins, levels of sex hormones, insulin and IGF-I and chronic inflammatory state. However, these studies have not identified clinical/molecular predictive risk factors.
Among the molecular mechanisms potentially involved in the MetS-BC association, the Receptor for Advanced Glycation End-products (RAGE) axis activation by AGEs is emerging as an important contributor, by fueling the oxidative stress and chronic inflammatory milieu, which are involved in the development of BC. AGEs are formed following non-enzymatic glycation of free amino acid groups on proteins, lipids, and nucleotides with reducing sugars. 10 The AGEs formation is part of normal metabolism. However, in conditions of metabolic stress their production is accelerated. 11 Thus, one could hypothesize that AGEs accumulation, and the consequent RAGE axis activation, may represent a pivotal piece in the pathways underlying the MetS-BC association.
Here, we report results of an analysis of plasma AGEs from 40 incident BC cases and 40 matched controls from the Hormones and Diet in the Etiology of Breast Tumors (ORDET) cohort study. This analysis is part of broader project aiming to elucidate the biological and molecular characteristics mediating the MetS-BC associations comparing proteomics and metabolomics profiles in MetS-positive and MetS-negative postmenopausal women who do/do not develop BC.
Methods
Study population
Women were selected from the 2008 case-control study nested in the ORDET cohort that assessed associations between MetS, sex hormones and BC. 5 Specifically, 877 postmenopausal women were included (175 cases; 702 controls). From this study, we randomly selected—among those who still had a plasma sample available—20 women with BC and MetS; 20 with BC without MetS; 20 healthy women with MetS, and 20 healthy women without MetS.
Baseline characteristics of postmenopausal case and control BC subjects.
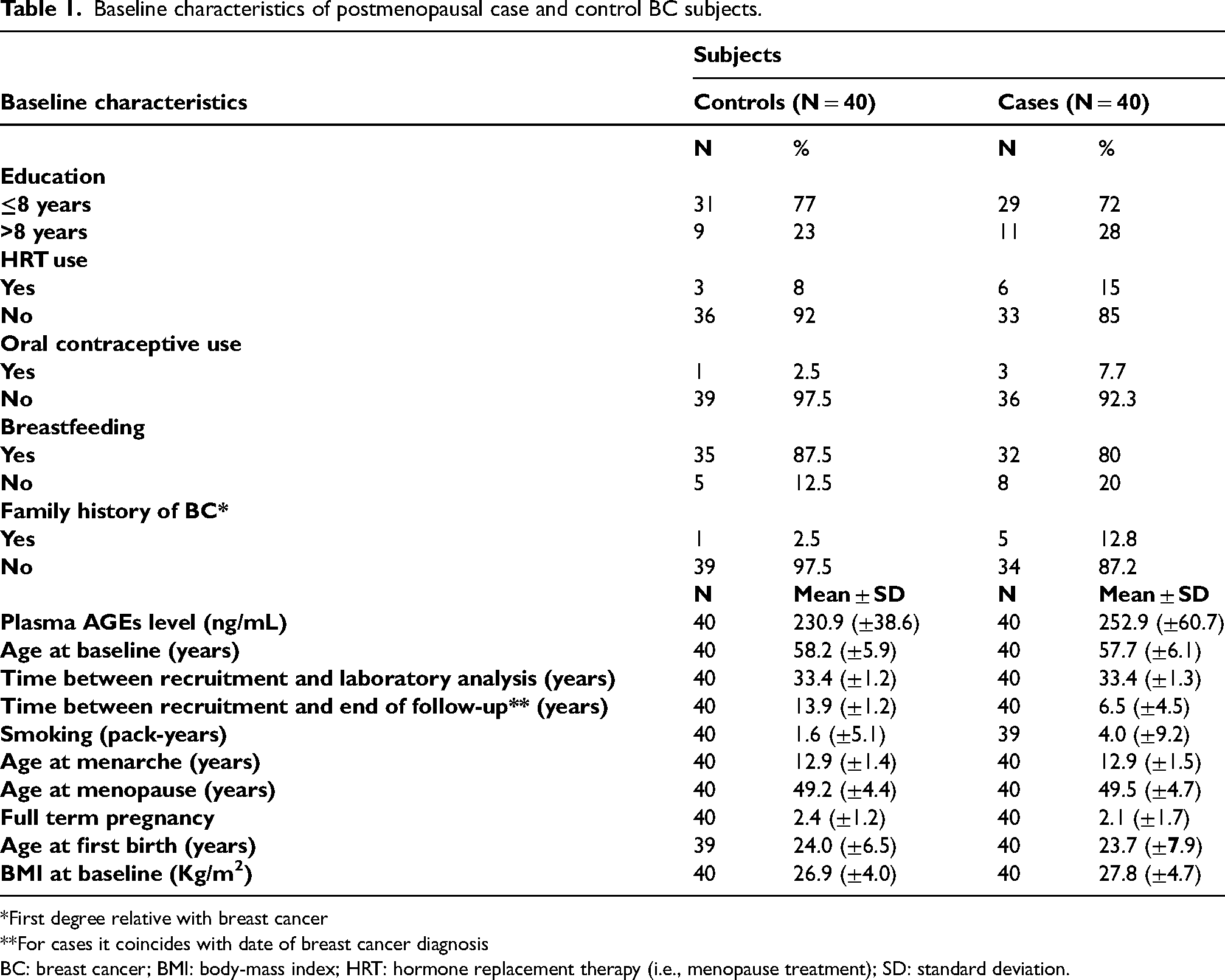
*First degree relative with breast cancer
**For cases it coincides with date of breast cancer diagnosis
BC: breast cancer; BMI: body-mass index; HRT: hormone replacement therapy (i.e., menopause treatment); SD: standard deviation.
ORDET is an Italian prospective study designed to investigate the BC etiology in 10,786 healthy women, aged 35–69 years, recruited in Varese province in 1987–1992. Women were excluded if they had undergone hormone treatment in the 3 months prior to admission, had chronic or acute liver disease, or had received bilateral ovariectomy. These women gave blood and urine samples, completed dietary and lifestyle questionnaires, and had their anthropometric measurements recorded by trained nurses. This study was approved by the Ethical Review Board of the National Cancer Institute of Milan.
Cancer incidence information, available from the local cancer registry (Varese Cancer Registry) was linked to the ORDET database to identify BC cases incident up to end of December 2008.
Measurement of plasma AGEs
AGEs were measured by the 96-well assay Human AGE ELISA Kit (Nordic Biosite). All analyses were performed according to the manufacturers’ instructions. Each sample was diluted 1:100 and measured in duplicate. Matched (for age at recruitment and recruitment date) pair case-controls were analyzed in the same analytical batch. Quality control samples were analyzed in each batch to monitor repeatability and reproducibility. The microplate reader Infinite Pro 2000 (Tecan, Männedorf, Switzerland) was used to analyze the absorbance of the samples at 450 nm. Data were processed with the data analysis software Magellan Pro 7.3 (Tecan).
Statistical analysis
Baseline characteristics of BC cases and controls were described as means ± SD (continuous variables) and counts with percentages (categorical variables).
The association between AGEs and BC cases was analyzed using Bayesian logistic regression models, adjusted for confounders, including time between recruitment and end of follow-up, duration of sample storage, and age. Continuous variables were standardized before the analysis to address differences in scale within the data. The primary analysis employed an informative prior (IP) for the exposure coefficient, based on the natural logarithm of the odds ratio ((OR) = 1.635) reported by Peng et al., 12 while weakly informative priors (WIPs) were used for all other regression coefficients. The specified priors were standard normal distributions, reflecting the standardization of continuous variables while maintaining weak information appropriate for the moderate sample size of 80 observations.
A sensitivity analysis was performed to evaluate the robustness of the findings to prior specification, using an additional logistic regression model with a WIP for the exposure coefficient instead of the IP.
Potential non-linearity in the exposure–outcome relationship was evaluated by comparing the linear logistic model to a logistic model with a penalized cubic spline function for the exposure. Model performance was assessed by examining differences in the expected log posterior density (ELPD) to determine the best fit. Prior predictive checks were conducted to ensure that the chosen priors permitted odds ratios to span a credible range of variability, capturing realistic effect sizes while avoiding extremes that could compromise inference.
To complement the primary analysis, Bayesian linear regression with WIPs was used to examine the association between metabolic syndrome and AGEs, adjusting for the same confounders.
For all models, the prior for the intercept was specified as a normal distribution with a mean of 0 and a variance of 2.5. Models were estimated using Hamiltonian Monte Carlo with the No-U-Turn Sampler, 13 which dynamically adjusts trajectory length during sampling. Four parallel chains of 2000 iterations each, including 1000 for warm-up, were used. Convergence was assessed through trace plots and diagnostic statistics, such as Rhat, bulk effective sample size, and tail effective sample size. Main estimates were reported with SDs and 90% and 95% credible intervals (CI). Throughout this study, the term “significant” is used to indicate findings based on Bayesian credible intervals, where intervals containing zero suggest a lack of strong evidence for the association.
Analyses were conducted in R (version 4.2.3, 2023-03-15) using the brms package, 14 interfacing with the Stan probabilistic programming language.
Results
Table 1 describes characteristics of cases and controls at recruitment. There were only slight differences: cases had higher use of oral contraceptives and hormone replacement therapy, a higher frequency of family history of BC and breastfed less than controls.
Compared to controls, cases also had higher body mass index and smoked more (Table 1).
Concerning the regressions, model convergence was successfully achieved, with all diagnostic measures confirming reliable parameter estimates. The comparison of ELPD between the linear and penalized spline models showed a difference of −0.4 (SD = 0.7). This indicates a slight preference for the linear model, which was selected based on its marginally better performance, the principle of parsimony, and interpretability.
Results of the logistic regression analysis and the sensitivity analysis regarding the prior are reported in Table 2. For the standardized AGEs coefficient, the IP specification yielded an OR of 1.745 (SD = 0.362; 90% CI: 1.218–2.390; 95% CI: 1.137–2.548). The WIP specification gave an estimated OR of 1.861 (SD = 0.661; 90% CI: 1.026–3.082; 95% CI: 0.924–3.528).
Estimated OR, SD, and 90% and 95% CIs for AGE in relation to BC case outcomes.

All estimates are adjusted for potential confounders, including time distance, time to sample collection, and age. The analysis incorporates two types of priors: IP and WIP.
AGE: advanced glycation end products; BC: breast cancer; CIs: credible intervals; IP: informative prior; OR: odds ratios; SD: standard deviations; WIP: weakly informative prior.
In the complementary analysis, the mean difference in standardized AGEs levels between individuals with and without MetS was 0.155 (SD = 0.245; 90% CI: −0.246 to 0.553; 95% CI: −0.322 to 0.625).
Discussion
In our small case-control study nested in the ORDET cohort on postmenopausal women, we found that increased AGEs levels were associated with higher BC risk. Although the AGEs difference in women with and without MetS was not significant, we found a suggestion of higher levels in women with MetS.
To the best of our knowledge, this is the first study that prospectively evaluated the association between blood AGEs levels and BC risk. Our findings of a direct association between blood AGEs and BC risk are in line with those suggested by previous cross-sectional studies evaluating endogenous AGEs in BC cases and controls.
Only two previous studies compared blood levels of AGEs in BC cases and controls. In 2007, Tesarová et al. 15 detected increased AGEs and decreased soluble RAGE (sRAGE) levels in serum of 120 patients with BC compared to 92 healthy controls. A case-control study on 1051 pairs of age-matched BC patients and control women, evaluating the cross-sectional association between plasma levels of AGEs and BC risk, 12 found that higher levels were associated with increased odds of BC. The findings of our study align with the evidence reported by Peng et al., 12 which identified a strong positive association between AGEs and BC. While our models suggested a positive relationship, the wide credible intervals (90% and 95%) reflected the uncertainty probably induced by the small sample size. A key strength of the Bayesian approach was its ability to incorporate prior information, as demonstrated by leveraging findings from Peng et al.'s study. 12 This approach reduced the uncertainty of our estimates, yielding more reliable insights despite limited data. Notably, even when non-informative priors—priors with minimal assumptions about the data—were used, the positive association between blood AGE levels and BC risk remained evident.
Excess endogenous AGEs exert their detrimental role on carcinogenesis both (a) directly catalyzing the formation of reactive oxygen species and inciting oxidative stress and inflammation at sites of AGEs accumulation; and (b) binding and activating the cell-surface RAGE that subsequently trigger a downstream cascade of pro-oxidant and pro-inflammatory mediators. 16 All these processes may lead to BC development through pathogenic effects on organ homeostasis, genetic integrity, and cellular signaling cascades. 17
The main mechanisms through which AGEs exert their proinflammatory and prooxidant effects is the activation of RAGE, which is highly expressed under pathological conditions. 18 RAGE has different subtypes, among which sRAGE can bind to AGEs, but fails to stimulate the RAGE-mediated signaling cascades promoting inflammation and oxidative stress. 19 Thus, sRAGE may exert a protective role against carcinogenesis15,20 by preventing AGEs to exert their pathological role. Peng et al. 12 also found that the AGEs/sRAGE ratio was associated with increased odds of BC, but sRAGE levels were negatively associated with BC. 12 Besides sRAGE, the human body has evolved other detoxification systems to reduce the burden associated with AGEs, including the glyoxalase system and the Nrf2 pathway. 16 These detoxification systems are less efficient in conditions of increased oxidative stress, 16 as it occurs in the presence of MetS. 19
Our results show a direct, although less robust (both the 90% and 95% CIs for the mean difference in standardized AGE levels between individuals with and without metabolic syndrome include zero) association between MetS and higher plasma AGEs, thus providing a potentially useful indication supporting previous experimental findings of higher endogenous AGEs accumulation and activation in the presence of MetS-enhanced inflammation and oxidative stress.16,19 However, this warrants further investigation in future studies with larger sample sizes.
The main strengths of our study include its prospective design, and the availability of the information on the presence of MetS, that enabled us to evaluate the role of endogenous AGEs as potential biomarkers involved in the association between MetS and BC. The main limitation is the small sample size, that prevented us in finding stronger and significant association. Moreover, due to the small number of included women, we could not control for some BC risk factors that may confound the association between plasma AGEs levels and disease occurrence, such as previous oral contraceptive or hormone therapy use, parity, and family history of BC.
In summary, our results confirm previous findings of a potential role of endogenous AGEs in the pathological pathway underlying the association between MetS and BC development. However, these results will have to be carefully interpreted when those coming from the proteomics and metabolomics analysis will be available, in order to gain a deeper insight in the complex mechanisms through which MetS enhances breast carcinogenesis.
Footnotes
Acknowledgements
We thank NuMe Lab - Nutrition and Metabolomics Laboratory, Fondazione IRCCS Istituto Nazionale dei Tumori, Milan, for analysis on the plasma samples. We also thank the staff off the Scientific Direction, Fondazione IRCCS Istituto Nazionale dei Tumori, Milan, for administrative support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by the Italian Ministry of Health. Grants number: RF-2019-12370396 and GR-2019-12370292. Ministero della Salute, (grants number RF-2019-12370396 and GR-2019-12370292).
Authors contributions
Conceptualization: SS, PM, CA; methodology: SS, PM, CA; laboratory analysis: GG, EV, ADP; statistical analysis: FP; writing original draft: SS, CA, FP; writing, review, and editing: CA, SS, MQ, CV, GG, EV, FP, CV; supervision: SS, PM, CA, CV, GG, EV, FP, MQ, ADP. All authors have read and agreed to the published version of the manuscript.
