Abstract
BACKGROUND:
Long non-coding RNAs (lncRNAs) were recently identified as crucial regulators of papillary thyroid cancer (PTC). However, the clinical role and regulatory functions of lncRNA cancer susceptibility candidate 2 (CASC2) in PTC remain unknown.
METHODS:
LncRNA CASC2 expression was examined in plasma samples from 68 PTC patients and 39 patients with nodular goiter (NG). Cell proliferation, migration and invasion abilities were evaluated using CCK8 assay and transwell migration and invasion assay. QRT-PCR and western blot analysis were performed to detect the expression of epithelial-to-mesenchymal (EMT) markers ZEB1, E-cadherin and vimentin in PTC cells.
RESULTS:
We demonstrated that lncRNA CASC2 expression was significantly downregulated in tumor tissues and plasma samples in patients with PTC compared with those in nodular goiters (
CONCLUSIONS:
Thus, these results indicated that lncRNA CASC2 was a predictor for LNM of PTC patients and may serve as a potential target of PTC treatment.
Introduction
Thyroid carcinoma is the most common malignant tumor of the endocrine system. Papillary thyroid carcinoma (PTC) is the major pathological type of thyroid carcinoma [1, 2]. Over the last several decades, PTC incidence has dramatically increased. Although the majority of PTCs are curable, more than 30% cases represent unfavorable prognosis due to relapse or lymphatic metastasis [2, 3]. Thus, exploring the underlying molecular mechanism of PTC is great significance.
Long non-coding RNAs (lncRNAs) are a class of transcripts longer than 200 nucleotides with limited protein coding function [4]. Recent studies have identified that lncRNAs serve as diagnostic biomarker and potential therapeutic targets in some tumors [5, 6]. LncRNAs play crucial regulators in a variety of tumor development or metastasis including thyroid carcinoma [7]. For instance, overexpression of long non-coding RNA NR_036575.1 contributes to the proliferation and migration of papillary thyroid cancer [8]. Long non-coding RNA MEG3 suppresses migration and invasion of thyroid carcinoma by targeting of Rac1 [9]. Long non-coding RNA PVT1 modulates thyroid cancer cell proliferation by recruiting EZH2 and regulating thyroid-stimulating hormone receptor (TSHR) [10]. Plasma lncRNA GAS8-AS1 as a potential biomarker of papillary thyroid carcinoma in Chinese patients [11]. In the study, we aim to investigate the clinical significance and functional role of lncRNA CASC2 in papillary thyroid carcinoma.
Here, we demonstrated that lncRNA CASC2 expression was significantly downregulated in tissues and plasma samples in patients with PTC in compared with those in nodular goiters. Decreased plasma lncRNA CASC2 expression associated with lymph node metastasis (LNM). In vitro, we demonstrated that overexpression of lncRNA CASC2 inhibited cell proliferation, invasion and EMT process. Thus, our results indicated that lncRNA CASC2 was a predictor for LNM and a potential target of PTC treatment.
Materials and methods
Plasma samples
The plasma samples from 68 PTC patients and 39 patients with nodular goiter (NG) were obtained at Department of Thyroid Surgery, the Second Affiliated Hospital of Nanchang University between January 2014 and June 2016. The plasma samples were from blood samples before operation and then were centrifuged. All samples were stored at
Cell line culture
The three human PTC cell lines (TPC-1, K1 and BCPAP) and a normal human thyroid cell line (Nthy-ori 3-1) were purchased from Cell Bank of the Chinese Academy of Sciences (Shanghai, China). Cell lines were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Gibco, Grand Island, NY) and supplemented with 10% fetal bovine serum (FBS; Gibco, Grand Island, NY) at 37
Plasmid construction and cell transfection
The full length lncRNA CASC2 sequence was subcloned into the pcDNA3.1 vector (Invitrogen, Carlsbad, CA, USA). Cell transfection were performed using lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). The pcDNA3.1-CASC2 or pcDNA3.1-vector plasmid was transfected into TPC-1 and BCPAP cells, respectively. Cells were harvested at 48 h after cell transfection.
RNA Extraction and quantitative real-time polymerase chain reaction (qRT-PCR)
Total RNA was extracted from plasma samples or cells using Trizol Reagent (Invitrogen Life Technologies, Carlsbad, CA, USA) according to the manufacturer’s instructions. Total RNA quality was detected using a NanoDrop
LncRNA CASC2 expression was lower in plasma samples in PTC patients. (A) LncRNA CASC2 expression was detected in 68 plasma PTC patients and 39 patients with nodular goiter (NG) using qRT-PCR analysis. GAPDH was used as the internal control. (B) LncRNA CASC2 expression was detected in 68 tissue samples in PTC patients and 39 patients with nodular goiter (NG) using qRT-PCR analysis. GAPDH was used as the internal control. (C) LncRNA CASC2 expression was detected in three human PTC cell lines (TPC-1, K1 and BCPAP) and a normal human thyroid cell line (Nthy-ori3-1) using qRT-PCR analysis. GAPDH was used as the internal control. (D) The expression of lncRNA CASC2 in PTC with negative or positively lymph node metastasis was shown. 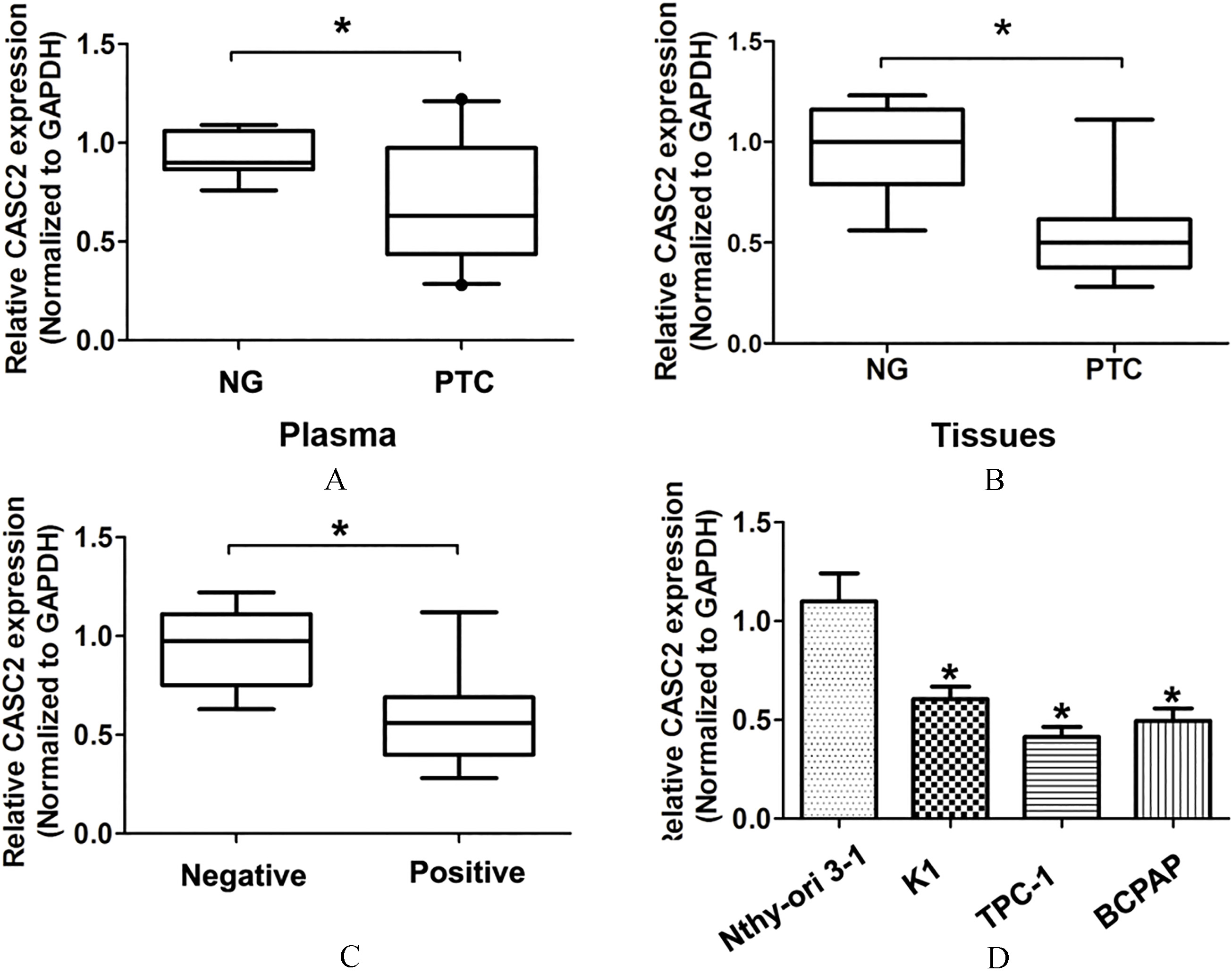
Cell migration and invasion assays were performed without or with Matrigel-coated chambers with 8-
Western blot analysis
The protein were extracted using the radioimmunoprecipitation assay (RIPA) (Sigma-Aldrich, St. Louis, MO, USA). Equal amounts of protein were separated onto sodium dodecyl sulfate polyacryl-amide gel electrophoresis gels (SDS/PAGE) and then transferred to nitrocellulose membranes (Millipore, Billerica, MA). The membranes were blocked with 5% non-fat milk and then incubated with primary antibodies including ZEB1 (Abcam, Cambridge, UK), E-cadherin (Cell Signaling Technology, Danvers, MA, USA), N-cadherin (Cell Signaling Technology, Danvers, MA, USA) and GAPDH (Cell Signaling Technology, Danvers, MA, USA) at 4
Statistical analysis
All of data in the study was analyzed using SPSS 18.0 (Version 18.0, SPSS, Inc., Chicago, IL, USA). Results are shown as the mean
The association between CASC2 expression and clinicopathological feathers of PTC patients was shown
The association between CASC2 expression and clinicopathological feathers of PTC patients was shown
Multivariate Cox regression analysis identified the risk factor for patients with lymph node metastasis in PTC patients
LncRNA CASC2 expression is lower in plasma samples in PTC patients and associates with lymph node metastasis
In the study, qRT-PCR analysis was performed to examine the expression of lncRNA CASC2 in the plasma samples from 68 PTC patients and 39 patients with nodular goiter (NG). Our results demonstrated that lncRNA CASC2 expression was significantly lower in plasma samples and tumor tissues in PTC patients compared to patients with nodular goiter (Fig. 1A and B,
Overexpression of lncRNA CASC2 expression inhibites cell proliferation, migration and invasion of PTC
To explore the potenital functional significance of lncRNA CASC2 expression in PTC carcinogenesis, we performed CCK8 cell proliferation, cell migration and invasion assays by lncRNA CASC2 overexpression. Two PTC cell lines (TPC-1 and BCPAP cells) were selected for lncRNA CASC2 overexpression for following experiments (Fig. 2A). The CCK8 cell proliferation assays showed that overexpression of lncRNA CASC2 inhibited the cell proliferation ability in TPC-1 and BCPAP cells, compared to corresponding control groups (Fig. 2B and C). Consistently, we demonstrated that cell migration and invasion abilities were suppressed after lncRNA CASC2 was overexpressed in TPC-1 and BCPAP cells, compared to corresponding control groups (Fig. 2D and E). Altogether, these results indicated that overexpression of lncRNA CASC2 expression suppressed cell proliferation and invasion of PTC.
Overexpression of lncRNA CASC2 expression inhibits cell proliferation, migration and invasion ability. (A) The lncRNA CASC2 expression was detected using qRT-PCR analysis after TPC-1 and BCPAP cells were transfected with pcDNA-vector or pcDNA-CASC2 plasmid. (B) and (C) Cell proliferation was detected using CCK8 assays after TPC-1 and BCPAP cells were transfected with pcDNA-vector or pcDNA-CASC2 plasmid. (D) and (E) Cell migration and invasion abilities were detected using transwell assays after TPC-1 and BCPAP cells were transfected with pcDNA-vector or pcDNA-CASC2 plasmid. 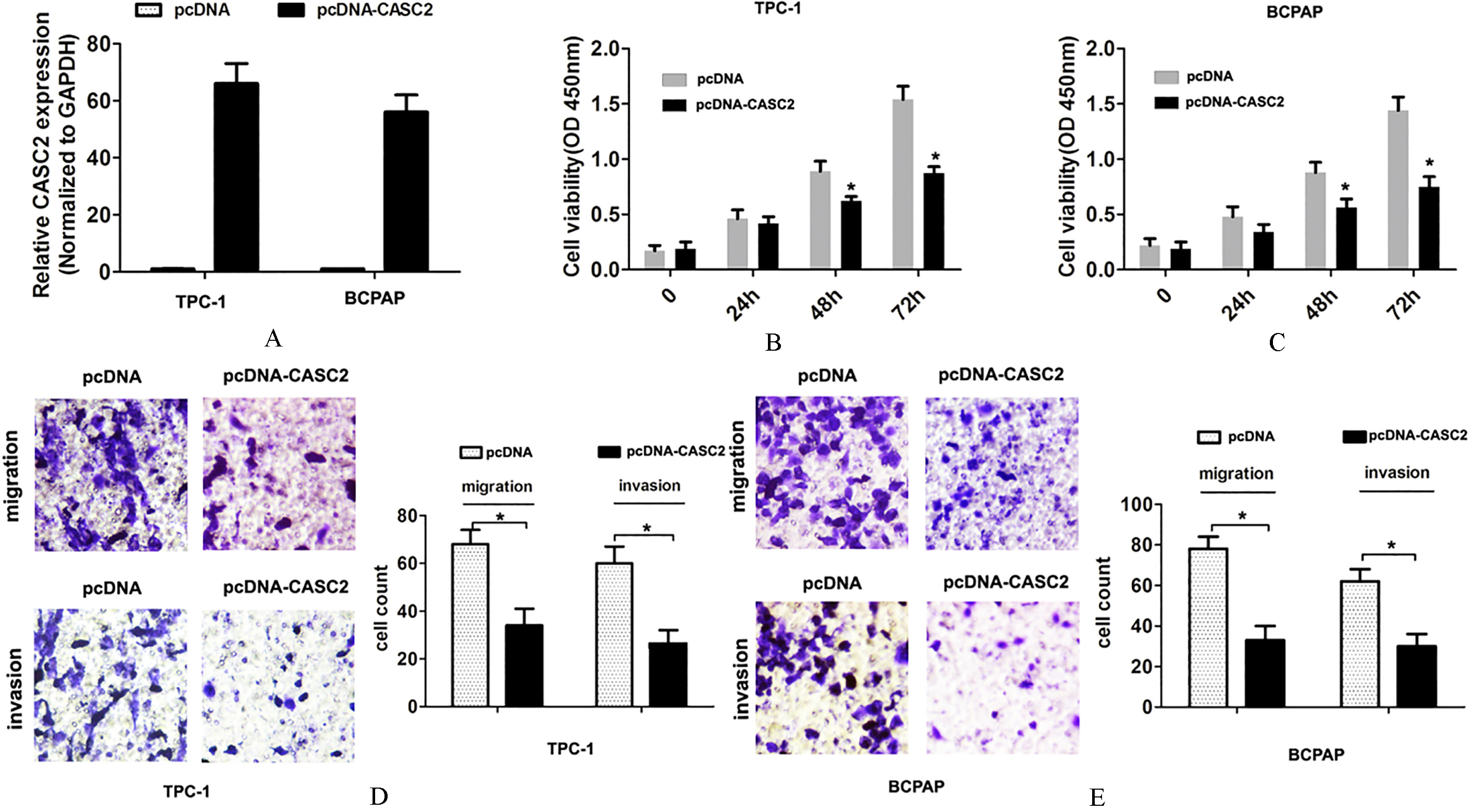
As we known, the epithelial-mesenchymal transition (EMT) process significantly associated with tumor invasion in PTC [12]. To further investigate the underlying molecular mechanisms of lncRNA CASC2 involved in tumor invasion, we detected the EMT pathway related makers ZEB1, E-cadherin and N-cadherin after lncRNA CASC2 overexpression. QRT-PCR assays results showed that overexpression of lncRNA CASC2 significantly reduced the mRNA expression of the mesenchymal cell markers ZEB1 and N-cadherin, but increased the mRNA expression of the epithelial cell marker E-cadherin in TPC-1 and BCPAP cells (Fig. 3A and B). Western blot analysis results showed that overexpression of lncRNA CASC2 significantly reduced the protein expression of the mesenchymal cell markers ZEB1 and N-cadherin, but increased the protein expression of the epithelial cell marker E-cadherin in TPC-1 and BCPAP cells (Fig. 3C and D). These results indicated that overexpression of lncRNA CASC2 expression inhibited EMT pathway.
Overexpression of lncRNA CASC2 expression inhibits epithelial-mesenchymal transition pathway. (A) The mRNA expression of ZEB1, E-cadherin or N-cadherin was detected using qRT-PCR analysis after TPC-1 and BCPAP cells were transfected with pcDNA-vector or pcDNA-CASC2 plasmid. (A) The protein expression of ZEB1, E-cadherin or N-cadherin was detected using western blot after TPC-1 and BCPAP cells were transfected with pcDNA-vector or pcDNA-CASC2 plasmid. 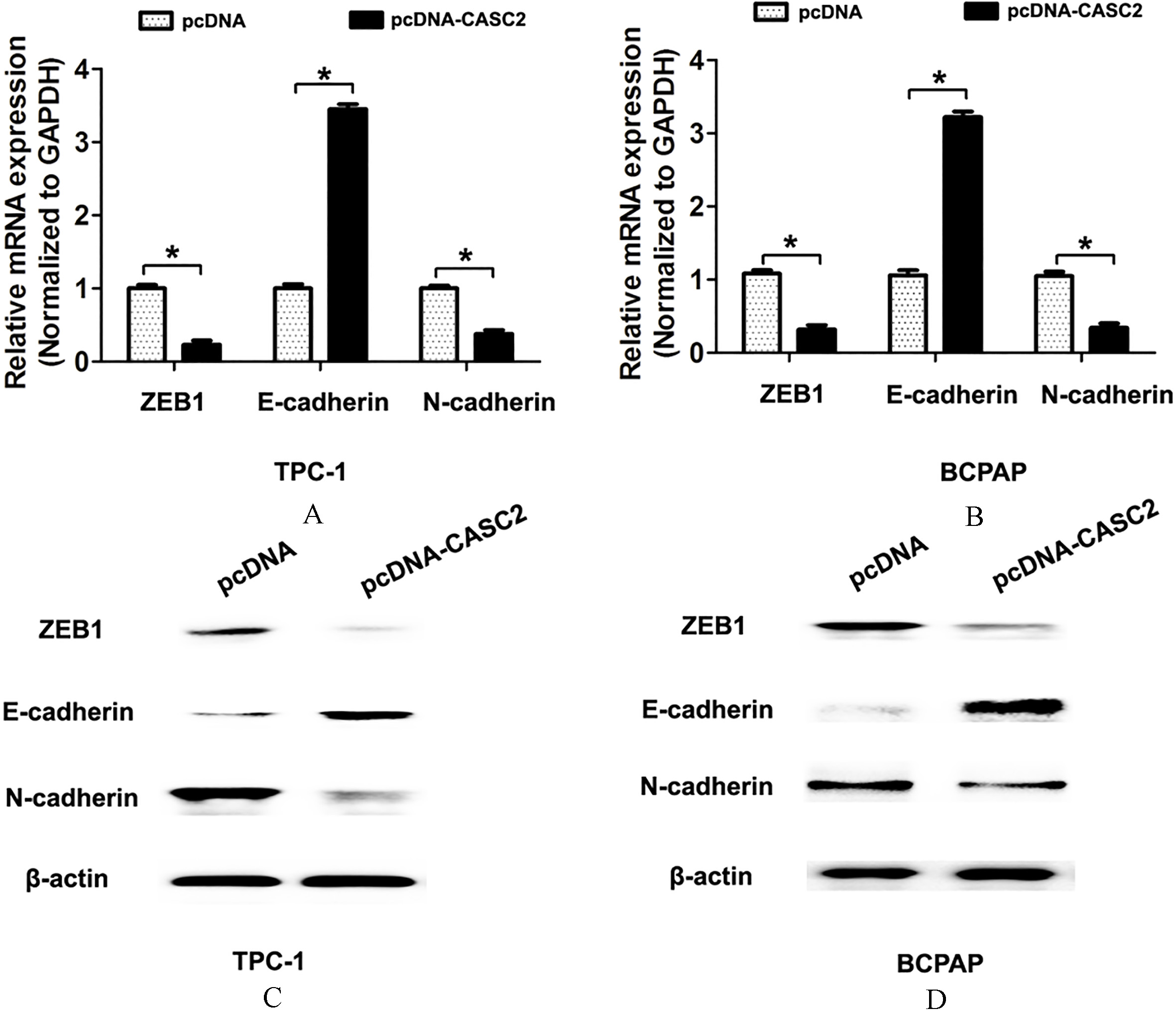
Most PTC patients have good prognosis after surgery in combination with radioiodine and levothyroxine treatment [13]. However, some of patients present unfavorable prognosis due to relapse or lymphatic metastasis. In the study, by examing the expression of lncRNA CASC2 in the plasma samples from PTC patients and nodular goiter (NG), we demonstrated that lncRNA CASC2 expression was lower in plasma samples in PTC patients compared to nodular goiter. Furthermore, patients who had lymph node metastasis showed lower lncRNA CASC2 expression, which indicated that lncRNA CASC2 had a significant clinical value for predicting lymph node metastasis of PTC patients.
In previous study, lncRNA CASC2 was found to act as a crucial regulator of tumor development. For example, lncRNA CASC2 interacts with miR-181a to modulate glioma growth and resistance to TMZ Through PTEN pathway [14]. Down-regulation of lncRNA CASC2 promotes cell prolifeCASC2c as an unfavorable prognosis factor interacts with miR-101 to mediate astrocytoma tumorigenesis [15]. Long non-coding RNA CASC2 regulates cell biological behaviour through the MAPK signalling pathway in hepatocellular carcinoma [16]. The long noncoding RNA CASC2 inhibits tumorigenesis through modulating the expression of PTEN by targeting miR-18a-5p in esophageal carcinoma [17]. Low expression of long noncoding RNA CASC2 indicates a poor prognosis and promotes tumorigenesis in thyroid carcinoma [18]. In our results, we found that lncRNA CASC2 expression was associated with lymph node metastasis of PTC patients, which is not consistent with previous reports described by Xiong et al. [18]. We speculated that due to the different from the number of patients and our results only reflect single center analysis. To explore the functional significance of lncRNA CASC2 in PTC carcinogenesis, we overexpressed the lncRNA CASC2 expression in PTC cells. After lncRNA CASC2 expression was overexpressed in PTC cells, our results indicated that overexpression of lncRNA CASC2 expression suppressed cell proliferation, migration and invasion abilities. These findings indicated that lncRNA CASC2 affected the malignant behaviors of PTC.
EMT pathway is closely related to tumor invasion and metastasis. Recent studies have demonstrated that lncRNA affects tumor invasion and metastasis by regulating EMT pathway [19]. In the study, we demonstrated that overexpression of lncRNA CASC2 significantly reduced the mRNA and protein expression of the mesenchymal cell markers ZEB1 and N-cadherin, but increased the mRNA and protein expression of the epithelial cell marker E-cadherin in PTC cells. These results indicated that overexpression of lncRNA CASC2 inhibited cell invasion by affecting EMT pathway.
In conclusion, our results found that plasma lncRNA CASC2 expression is lower in PTC and associated with lymph node metastasis. In vitro, we demonstrated that overexpression of lncRNA CASC2 expression suppressed cell proliferation and invasion ability. Furthermore, we demonstrated that overexpression of lncRNA CASC2 inhibited cell invasion by affecting EMT pathway. Thus, these results indicated that lncRNA CASC2 may be a potential target of PTC treatment.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest in this paper.
