Abstract
Background
Gastric cancer is a common cancer developed in a carcinogenesis process from precancerous lesions including chronic gastritis, intestinal metaplasia, and dysplasia. Survivin, an inhibitor-of-apoptosis protein, is associated with the initiation and progression of gastric cancer. The present study aimed to evaluate the immunohistochemical expression patterns of survivin and its relationship with early diagnosis of gastric cancer in Iranian patients.
Methods
In this retrospective case-control study, immunoexpression of survivin was investigated on sections obtained from formalin-fixed paraffin-embedded tissue blocks of 38 chronic gastritis, 32 intestinal metaplasia, 20 dysplasia, 28 gastric adenocarcinoma, and 22 controls.
Results
Survivin immunoexpression in chronic gastritis was higher than controls, but this difference was not statistically significant (P > 0.05). However, survivin immunoexpression had a steady significant increase from control and chronic gastritis to intestinal metaplasia to dysplasia to gastric adenocarcinoma (P < 0.05). Sensitivity, specificity, and area under the curve of survivin immunohistochemical test for the diagnosis of gastric cancer were 87.5%, 74.4%, and 0.85, respectively. Males had a significantly higher survivin expression than females (P < 0.001). Also, survivin expression was significantly higher in older patients than in younger ones (P < 0.001).
Conclusion
It seems that the steady increase in survivin expression from different precancerous lesions to gastric adenocarcinoma suggests that survivin can be used as a potential biomarker for the prevention and early diagnosis of gastric cancer.
Introduction
Gastric cancer (GC) is the fifth most diagnosed and the fourth leading cause of malignancy-related death worldwide, leading to approximately 700,000 deaths per year. 1 While surgical resection and advances in chemotherapy have improved the overall survival rates, prognosis of GC still remains poor, especially in patients with advanced stages of GC. 2 Thus, there is a need to identify biomarkers for early diagnosis, determining prognosis, and prediction of therapeutic resistance. Both genetic and environmental factors have an impact on its incidence and development. Helicobacter pylori infection, alcohol consumption, smoking, and family history are among the factors that increase the risk for GC development. 3
Gastric adenocarcinoma (GA) is the most common histological type of GC comprising 95% of these cancers. 4 Correa et al. showed that GA developed from precancerous lesions in a multi-step process commonly known as Correa's cascade. These precancerous lesions were reported as non-atrophic gastritis, atrophic gastritis, intestinal metaplasia (IM), dysplasia, and invasive carcinoma respectively. 5 Previous studies showed that patients with precancerous lesions had a higher risk of GC.6,7 Therefore, the prognosis of GC can be improved by early diagnosis of precancerous stages. Our previous studies showed that there was a gradual increase in the level of some biomarkers such as p53 and Ki-67 in order of Correa's cascade, which likely supports Correa's theory.8,9
Dysregulated apoptosis plays a significant role in several aspects of carcinogenesis including tumor onset, progression, and resistance to therapies such as radiotherapy and chemotherapy. 10 Survivin, the smallest member of the inhibitor of apoptosis protein family, has many roles in cell death, mitosis, angiogenesis, migration, and metastasis. 11 Polymorphisms of the survivin gene were proved to be associated with the risk of different malignancies including GC. 12 Also, it has been shown that survivin is expressed in different malignant, embryonic, and fetal tissues. 13 However, it is much less expressed in proliferative normal tissues and is approximately absent in non-proliferative normal tissues. 14 It has been shown that higher survivin expression is associated with initiation, progression, poor prognosis, metastasis, and therapeutic resistance of different cancers, and is thereby a promising biomarker. 15 A growing body of evidence has shown that higher levels of survivin expression are associated with GCs having higher tumor-node-metastasis (TNM) stages, depth of invasion, vascular invasion, distant metastasis, poorer prognosis, and low patient survival.16,17
Although there is compelling evidence for the high levels of survivin expression in GC, its levels in precancerous lesions and its ability to be used as a biomarker for early diagnosis of GC are poorly defined. Although some studies have investigated the survivin expression in precancerous lesions of GC,18–20 some precancerous lesions of Correa's cascade have not been studied and the results are controversial or dubious.
Considering the critical importance of apoptosis in carcinogenesis, we aimed to assess the immunoreactivity of survivin in normal tissues, chronic gastritis (CGS), IM, dysplasia, and GA, to investigate its diagnostic and prognostic importance as an indicator of the progression of precancerous lesions to GC.
Material and methods
This case-control study comprised 140 gastric biopsies obtained from the pathologic archive at Ali-ebne-Abitaleb Hospital, Zahedan University of Medical Sciences (ZAUMS), Zahedan, Iran. Specimens had been obtained from patients admitted from 2014 to 2019. Gastric biopsies were obtained from the parts of stomach that the gastric lesions were seen during upper gastrointestinal endoscopy. Most of them were from the antrum and a small portion were from the body of the stomach. The inclusion criteria were formalin-fixed paraffin-embedded tissue blocks with the diagnosis of CGS, IM, dysplasia, GA, and normal gastric tissue. Normal gastric tissues adjacent to the lesions served as the control group. The exclusion criteria were specimens of patients with immune disorders, inadequate and unsuitable samples, and specimens lacking the required information. The clinical data, including age, sex, and tumor site, were obtained without recording the patients’ names. To approve the diagnosis of samples, all the specimens were re-evaluated.
The study was conducted according to the Declaration of Helsinki for conducting research involving human subjects, and the study protocol was approved by the Ethical Committee of Zahedan University (IR.ZAUMS.Rec.1396.326).
Immunohistochemistry
The paraffin-embedded tissue blocks were cut into 4 µm sections using an automated microtome (Lecia, RM2255, Germany) and mounted on glass slides coated with HistoGrip (Histogrip50× CL00- 8050-Cedarla Co).
The sections were deparaffinized in xylene, then hydrated in graded ethanol solutions (pure and 96% ethanol, each one twice, in order), and rinsed in distilled water. Antigen retrieval was achieved by boiling tissue slides with 1 M sodium citrate buffer (pH: 6) for 20 min at 126°C in an autoclave (Prestige Medical, series 2100 classic, Iran). After that, the slides were put at room temperature to cool down. To quench the endogenous peroxidase activity, the slides were washed with distilled water for 5 min and then with Tris-buffered saline (TBS, pH: 7.6) three times and each time for 5 min. Then, the sections were incubated in blocking serum 1.5% diluted in phosphate-buffered saline (PBS) for 1 h. Subsequently, sections were incubated overnight at 4°C with the primary polyclonal antibody, anti-survivin antibody (abcam469, USA). Afterwards, the slides were washed with TBS twice and each time for 5 min. The sections were incubated with the secondary biotinylated antibody for 30 min and washed with TBS. AB reagent was placed on the sections for 30 min and then washed with TBS. Three drops of peroxidase substrate containing DAB Chromogen were placed on each section until the staining intensity was acceptable. After that, sections were washed with distilled water and counterstained with Hematoxylin Mayer to produce the color contrast. Then, sections were washed with distilled water, dehydrated in graded ethanol solutions, and made clear with xylene.
Positive controls (colorectal cancer) and negative controls (gastric tissue slides lacking the primary antibody) were employed for each batch of slides. Immunoreactivity was detected microscopically on masked label slides by two expert histologists. This scoring system was used to measure the survivin immunoexpression: immunohistochemistry (IHC) score (0–8) was calculated as the sum of an intensity score (0-absent, 1-weak, 2-moderate, and 3-strong) and score distribution (0: 0%, 1: < 1%, 2: 1% to 10%, 3: 10% to 33%, 4: 33% to 66%, and 5: 66% to 100%) for each tissue sample. 21
Statistical analysis
The Statistical analysis was performed using Statistical Package for Social Sciences (SPSS) software version 20.0 (IBM, Armonk, NY, USA). Mean ± SEM values were calculated for each variable in all groups, and the Mann–Whitney U-test and the Kruskal–Wallis tests were used to compare the study groups. Fisher's exact test was used for intergroup analysis. Spearman's rank correlation was used to evaluate the correlation coefficient between the variables. Differences were considered significant if P-values were <0.05.
Results
This study comprised 140 gastric biopsy specimens including 22 control specimens (50.50 ± 15.40 years, median age: 47, age range: 28–75, M/F ratio: 18/4), 38 specimens with CGS (50.03 ± 13.59 years, median age: 48, age range: 29–75, M/F ratio: 28/10), 32 specimens with IM (49.81 ± 11.01 years, median age: 48, age range: 32–68, M/F ratio: 22/10), 20 specimens with dysplasia (52.30 ± 13.83 years, median age: 48, age range: 32–76, M/F ratio: 15/5), and 28 specimens with GA (50.00 ± 13.13 years, median age: 48, age range: 32–86, M/F ratio: 21/7).
Immunohistochemistry of gastric tissue samples revealed positivity for survivin in Figure 1.
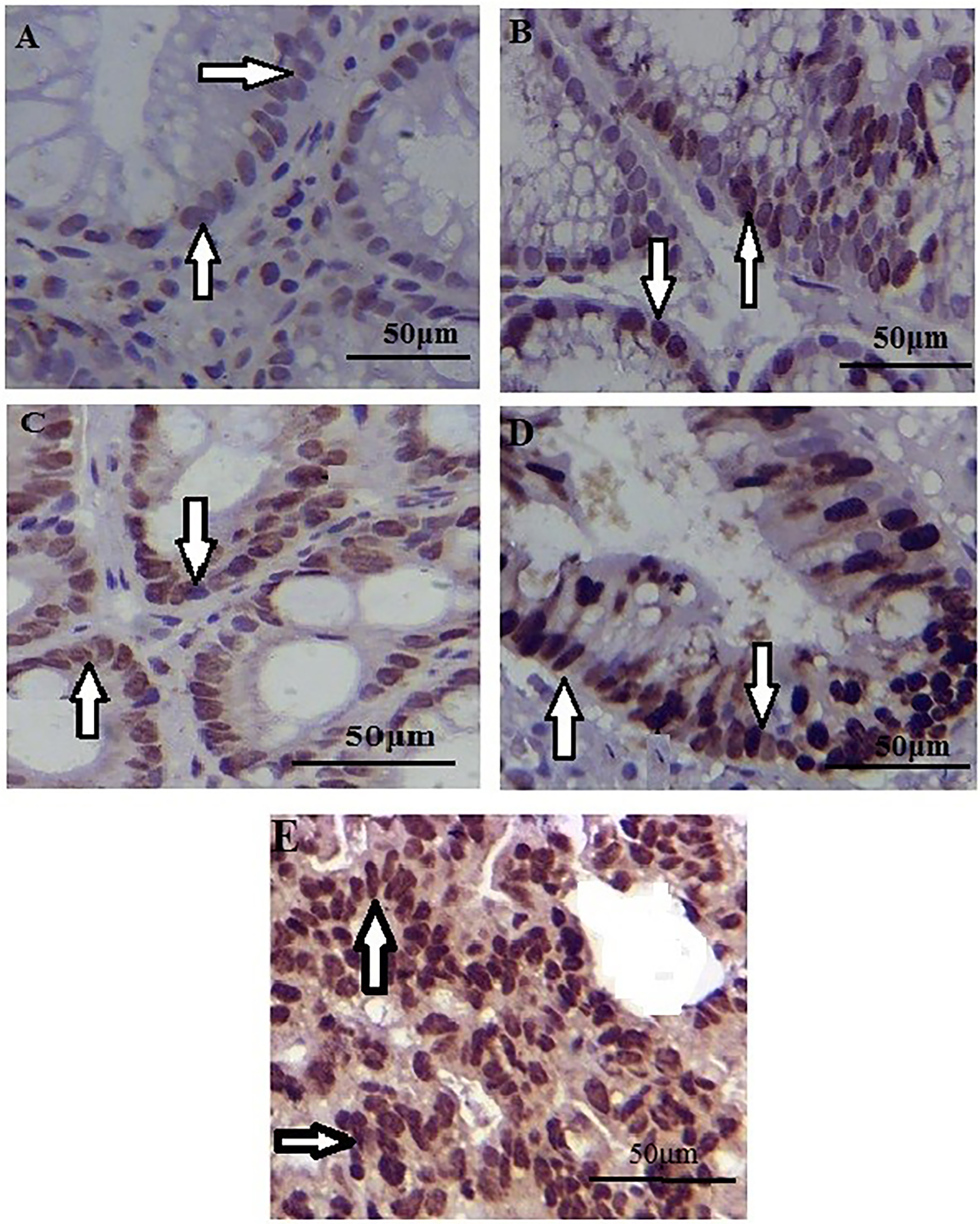
Survivin immunoexpression in control (a), chronic gastritis (b), intestinal metaplasia (c), dysplasia (d), and gastric adenocarcinoma (e) tissues (immunoperoxidase ×400). Survivin positive expressions in cells are shown with arrows.
The results of the immunohistochemical expression of survivin in tissue samples are shown in Table 1 and Figure 2. The control, CGS, IM, dysplasia, and GA groups were not significantly different in terms of age and sex (P > 0.05).
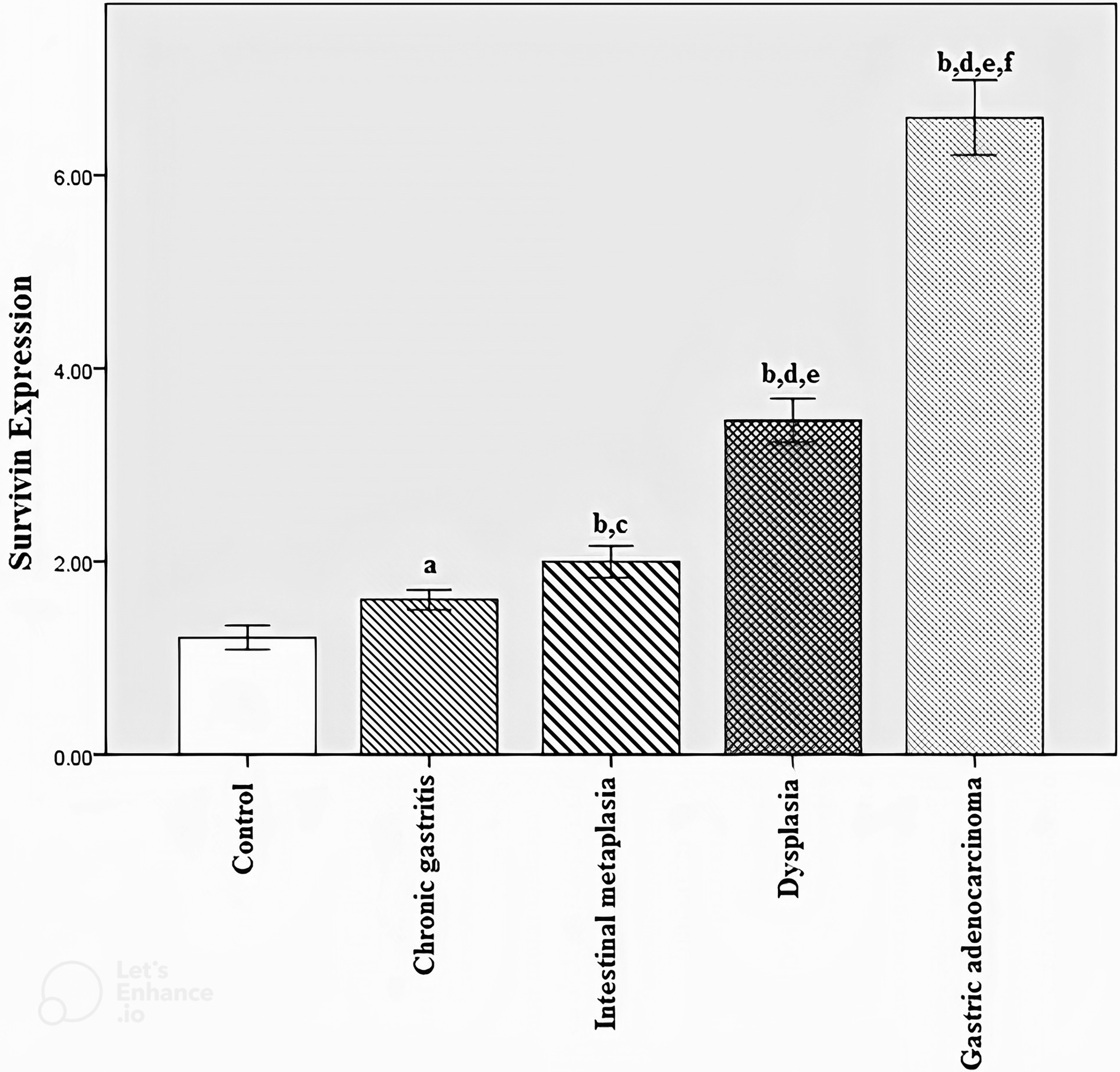
Survivin expression in control, chronic gastritis, intestinal metaplasia, dysplasia, and gastric adenocarcinoma tissue samples. (a) P = 0.080 compared to the control group. (b) P < 0.001 compared to the control group, Bonferroni correction PBC < 0.001. (c) P = 0.036 compared to the chronic gastritis group. (d) P < 0.001 compared to the chronic gastritis group, Bonferroni correction PBC < 0.001. (e) P < 0.001 compared to the intestinal metaplasia group, Bonferroni correction PBC < 0.001. (f) P < 0.001 compared to the dysplasia group, Bonferroni correction PBC < 0.001.
The immunoexpression level of survivin in control, chronic gastritis, intestinal metaplasia, dysplasia, and gastric adenocarcinoma tissue samples.

SEM: standard error of the mean.
The Kruskal–Wallis test revealed that all groups were significantly different in terms of the expression level of survivin (P < 0.001).
The Mann–Whitney U and Fisher's exact tests showed that survivin protein levels in IM, dysplasia, and GA groups were significantly increased compared to the control group (P < 0.001). However, there was no statistically significant difference between the control and CGS group (P > 0.05). There was a steady significant increase from CGS to IM to dysplasia to GA (P < 0.001) (Figure 2).
To investigate whether survivin can be an appropriate biomarker for the diagnosis of GC, area under the curve (AUC), sensitivity, and specificity of IHC tests for the diagnosis of GC using survivin were estimated and were 0.85, 87.5%, and 74.4%, respectively (Figure 3).
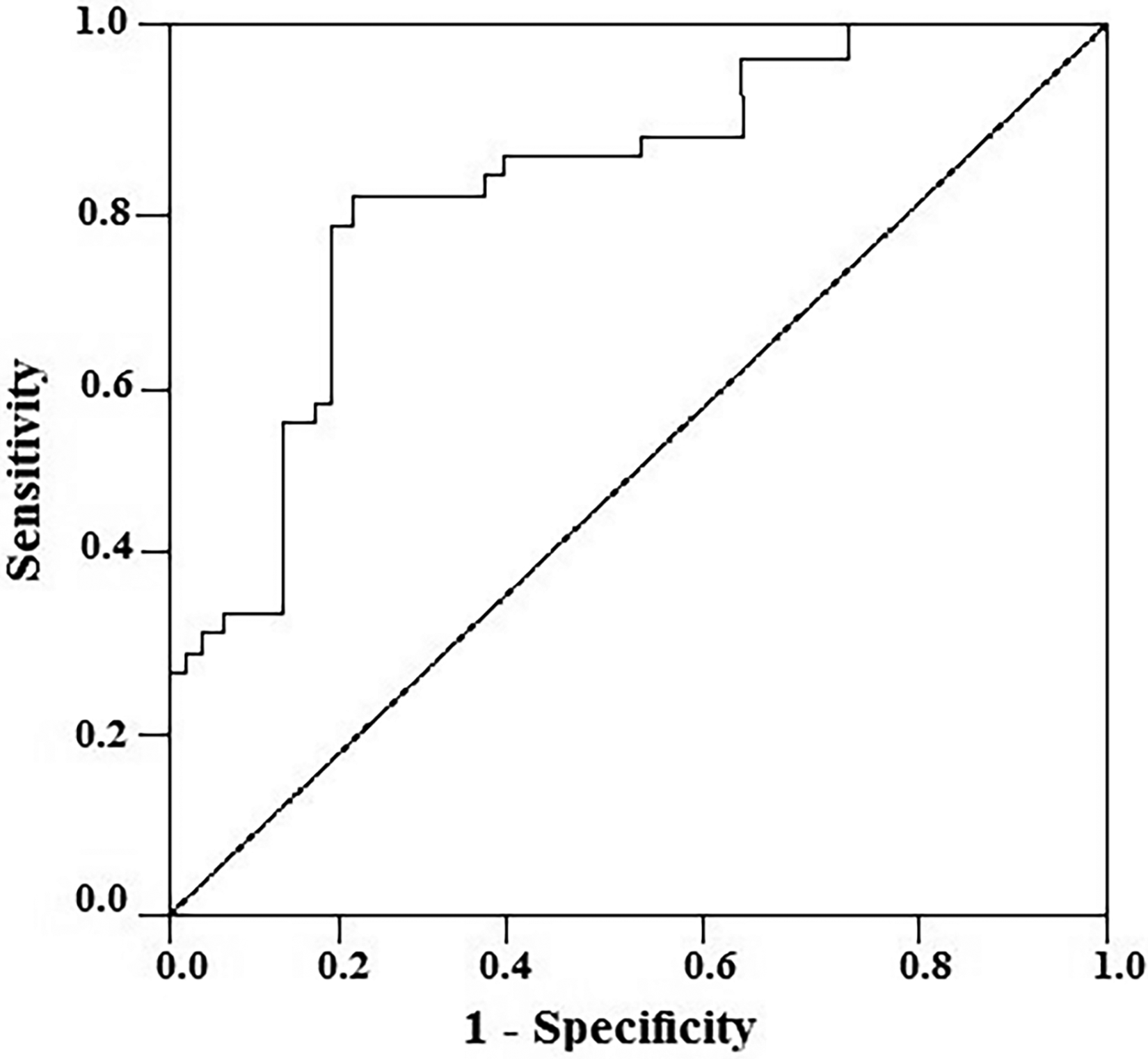
Receiver-operator characteristic (ROC) curve of the survivin for discriminating cancer patients from normal control. Area under the curve = 0.85 (95% CI: 0.76–0.94), sensitivity = 87.5%, and specificity = 74.4%.
The survivin protein levels gradually increased from normal tissue through CGS, IM, and dysplasia to GA with a significant Spearman's correlation (r = 0.898, P < 0.001).
The expression level of survivin was statistically significantly increased in male subjects compared to the female subjects (P < 0.001).
Survivin was expressed in a higher amount in older subjects (age > 40y) compared to younger subjects (age < 40y) (P < 0.001).
Discussion
The process of carcinogenesis in GA consists of a process known as Correa's cascade in which precancerous gastric lesions, including gastritis, IM, and dysplasia in that order, progress into GA. 5 Secondary prevention of GC includes screening and treating precancerous lesions. Therefore, there is a need for biomarkers that can distinguish GC progression in its early stages. 22 Survivin, a member of the inhibitor of apoptosis protein family, is differentially expressed in different stages of GCs and is effective in determining the prognosis of the patients. 16 Although the presence of survivin expression in precancerous gastric lesions has been observed,19,20 there is a need for a thorough and more accurate examination of this issue and comparison of different precancerous lesions to find whether survivin can be an appropriate biomarker for the screening of GC. Therefore, in this study, we investigated the survivin expression in precancerous and cancerous gastric lesions.
In our study, the control group expressed survivin in a low amount. This is in line with the results of Yusufu et al. 16 However, other studies including Helal et al. 23 observed that the control group was negative for survivin expression. Pandey et al. 19 explained this discrepancy by mentioning that survivin-positive normal tissues were from subjects with a history of simultaneous tobacco chewing and cagA+ Helicobacter pylori infection. Therefore, the difference can be because of the inequality in the prevalence of H. pylori and tobacco chewing between different populations. In addition, genetic and environmental variations, such as polymorphisms, in the different patient populations can contribute to the observed difference.
We observed that there was a statistically insignificant increase in survivin expression from the control to the CGS group. In line with our findings, Helal et al. 23 showed that the level of survivin expression was not significantly different in both the CGS group and the control group.
The present study demonstrated that survivin expression in IM, dysplasia, and GA was significantly higher than in the control group. This agrees with the results of Pandey et al. 19 and Da et al. 20 This may explain why precancerous gastric lesions are more prone than normal tissue to develop into cancerous tissue through bypassing apoptosis mechanisms.
Helal et al. 23 showed that survivin expression was higher in GA than in CGS. These findings concur with the results of our study. In addition, we demonstrated that survivin expression in IM and dysplasia was significantly higher than in CGS.
In the present study, we showed that GA and dysplasia have a significantly higher survivin expression than IM. However, Pandey et al. 19 and Da et al. 20 did not show this significant difference. This discrepancy can be because of the geographical and ethnic differences between the patient populations. In addition, the smaller size of their groups can be another reason for this discrepancy.
The results of the present study showed that survivin protein levels in GA were higher than in dysplasia, which is in line with the results of Pandey et al. 19 However, Da et al. 20 did not show this significant difference between these groups. The genetic and epigenetic variations as well as the presence or absence of H. pylori may be able to explain this controversy.
Based on our findings, the immunohistochemistry tests are 87.5% sensitive and 74.4% specific for the diagnosis of GA. Pandey et al. 19 showed that nuclear survivin had an AUC and positive predictive value of 0.822 and 89.06%, respectively, for distinguishing between GA and other lesions. Both studies suggest that survivin can be a potential biomarker for the diagnosis of GC.
Our study demonstrated that survivin expression is significantly higher in male subjects than female subjects. This agrees with the results of Helal et al. 23 In addition, this result is in concordance with the previous studies stating that male subjects are more prone than female subjects to GC. 24
Based on our findings, survivin expression was higher in older subjects (age > 40y) compared to younger subjects. This is in concordance with older age being a risk factor for GC. 25 However, Zhu et al. 26 did not find any significant difference between older subjects (age > 55) and younger subjects (age < 55). We chose the age of 40 as the cut-off because of the decreasing age of incidence of GC. In addition, race, nutrition, and other risk factors of GC can be involved in this discrepancy.
Our results showed a steady rise of survivin expression from CGS to IM to dysplasia to adenocarcinoma. It is well known that H. pylori is one of the main risk factors for GA. 3 Surprisingly, Valenzuela et al.27,28 demonstrated that H. pylori induces the loss of survivin and reduces its expression in gastric tissue. This discrepancy can be explained by the findings of Liu et al., 29 stating that the acute infection with H. pylori causes an increase in the pro-apoptotic factors while the chronic infection with H. pylori causes a decrease in the pro-apoptotic factors. However, their study did not investigate the survivin expression in these two different conditions. Therefore, future studies are needed to analyze the effects of acute and chronic H. pylori infection on survivin expression in Correa's cascade.
Non-steroidal anti-inflammatory drugs (NSAIDs) can cause gastric injuries such as gastritis and ulcer. 30 NSAIDs have pro-apoptotic activities and they prevent tumorigenesis partly via survivin down-regulation. 31 This is in concordance with our results that as the survivin increases, tumorigenesis progresses. Furthermore, this also implies that NSAIDs-induced gastritis probably has a lower potential to develop into GA, and future studies are needed to investigate this hypothesis.
The present study has the advantage of investigating almost all precancerous lesions of Correa's cascade, which was missing in the previous studies. The main limitation of our study is the small size of samples. Future studies are needed to examine the association between the cellular and subcellular expression of survivin and H. pylori infection in precancerous and cancerous lesions.
Conclusion
The present study demonstrates the gradual increase of survivin expression from CGS to IM to dysplasia to GA. This finding demonstrates that survivin can be utilized as a diagnostic biomarker of GC. This may open new avenues for prevention and early diagnosis of GC.
Footnotes
Acknowledgment
The authors appreciate the vice-chancellor of research and technology of Zahedan University of Medical Sciences that financially supported the research (Grant No: 8613) (IR.ZAUMS.REC.1396.326).
Author contributions
A. Mahmoudzadeh-Sagheb contributed to design and literature review and drafted the manuscript. M. Panahi and S. Jami participated in data collection and analysis. B. Moudi contributed in study design and statistical analyses and the interpretation of results and the manuscript. Z. Heidari and H. Mahmoudzadeh-Sagheb conceived and co-designed the study, supervised all the experimental designs and made a significant contribution to the revision of the manuscript. All authors read, modified, and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was supported by the vice chancellor of Research and Technology of Zahedan University of Medical Sciences (Grant No: 8613).
Ethics approval and consent to participation
The study was conducted according to the Declaration of Helsinki for conducting researches involving human subjects and the study protocol was approved by the Ethical Committee of Zahedan University (IR.ZAUMS.Rec.1396.326).
