Abstract
Hypoxia-inducible factor 1α (HIF-1α) triggers tumorigenesis and progression in hepatitis B virus (HBV)-related hepatocellular carcinoma (HCC). Inconsistent findings have been reported on the influence of HIF-1α over-expression on the clinical outcomes of HBV-related HCC. This study aims to clarify the role of HIF-1α overexpression in the tumorigenesis and prognosis of HBV-induced HCC. Systematic and comprehensive search of online papers was carried out to elucidate the contribution of HIF-1α expression to susceptibility of HBV-induced HCC. STATA 12.0 software was utilized to analyze available data extracted from all eligible literature. Publication bias and sensitivity were comprehensively analyzed. A total of 23 published studies involving 2244 subjects were finally screened. The HIF-1α expression was remarkably upregulated in HBV-induced HCC tissues than in normal liver tissues, non-tumorous tissues, paraneoplastic tissues, and non-HBV HCC tissues. The high HIF-1α expression tended to be positively related to capsular infiltration (odds ratio (OR) 1.767; 95% confidence interval (CI) 1.058, 2.950). The HIF-1α expression was relevant to lymph node metastasis (OR 3.778; 95% CI 1.666, 8.568). High levels of HIF-1α expression tended to be closely implicated in portal vein invasion (OR 6.728, 95% CI 2.191, 20.656) but were irrelevant to alpha-fetoprotein, cirrhosis, Edmondson grading, tumor size, age, gender, and histological grade. Analysis of pooled data showed that HIF-1α was not statistically relevant to poor overall survival in HBV-related HCC. Our data provides compelling evidence that HIF-1α overexpression may imply a greater probability of invasion and metastasis in patients with HBV-induced HCC.
Introduction
Hepatocellular carcinoma (HCC) is currently the sixth most prevalent malignancy owing to its rapidly increasing incidence. 1 It is one of the most fatal malignant neoplasms worldwide due to the high risk of rapid progression. Increasing lines of evidence support that more than half of HCC cases are attributed to hepatitis B virus (HBV) infection. HBV-infected patients are prone to progress to HCC, which has high mortality. 2 In spite of the rapid technique improvements of surgical intervention, radiation, chemotherapy and liver transplantation, the long-term outcome after the completely effective treatment of HBV-related HCC remains unsatisfactory, but the traditional molecular predictive markers of alpha-fetoprotein (AFP) was discovered to possess limited value in accurately predicting the clinical survival, progression, and recurrence of HCC. 3 In this regard, new biomarkers should be identified to precisely predict carcinogenesis, progression, and prognosis for improvement of the long-term survival of patients with HBV-related HCC.
Hypoxia is a commonly shared microenvironment among several human malignancies, which has crucial impact on the malignant outcome of various solid tumors. The hypoxic microenvironment contributes to cancer progression by activating tumor angiogenesis, invasion, and metastasis.4,5 Angiogenesis takes a vital part in the aggressive progression and malignant outcomes of human malignancies, hypoxia-inducible factor (HIF)-α plays a vital role in promoting the high proliferation and metastasis of cancer cells by inducing tumor-associated angiogenesis. 6 HIF-α is principally composed of the oxygen-regulated subunits of HIF-1α and HIF-2α, which is mainly activated and mediated by multiple kinds of hypoxia-induced genes under the environment of oxygen deficiency. 7 In particular, the HIF-1α-derived tumor hypoxia response is regulated by low oxygen tension. 8 HIF-1α activates the transcription of different hypoxia-induced genes to facilitate metabolic adaptation of tumor cells to hypoxic conditions. 9 Evidence indicates that aberrant HIF-1α overexpression participates in various malignancies in terms of carcinogenesis and clinical progression.10–12 HIF-1α overexpression is relevant to tumorigenesis and poor prognosis in diverse tumors. 13
HIF-1α up-regulation was reported in HBV-induced HCC in existing medical literature. 14 However, the effect of HIF-1α on the tumorigenesis and prognosis of HBV-induced HCC remains in conflict with previous reports.15,16 Hence, available literature was systematically reviewed to assess the oncogenic and prognostic effect of HIF-1α overexpression on HBV-induced HCC.
Materials and methods
Data sources
A systematic literature search was conducted on PubMed, EMBASE, Scopus, OVID, Web of Science, ELSEVIER, and CNKI databases. All eligible publications and other relevant articles available online prior to May 2022 were considered for this meta-analysis. Studies were selected and retrieved by a manual search of the following terms “HIF-1α,” “hypoxia inducible factor-1α,” and “HCC,” “hepatocellular carcinoma,” “Hepatocarcinoma,” “hepatic cancer,” “liver cancer,” “liver tumor,” “hepatic neoplasm” as well as “Hepatitis B Surface Antigen,” “HBV,” “HBsAg,” and “hepatitis B.” The literature search was restricted to publications in English or Chinese.
Criteria for inclusion and exclusion
We identified eligible papers fulfilling the following conditions: (a) studies with case-control design; (b) HCC was diagnosed by pathological examination; (c) HBV infection was detected by testing serum HBV surface antigen (HBsAg) positivity; (d) HIF-1α protein levels were detected by immunohistochemical staining (IHC); (e) the relationship between clinical characteristics and HIF-1α level was investigated and had sufficient data; and (f) the most informative studies were filtered in cases when multiple related articles were published.
We removed studies complying with the following conditions: (a) basic laboratory research about cell lines or animal trials; and (b) research without original or sufficient data.
Data extraction
Three investigators (Jinlin Peng, Jizhou Wu, and Lei Wang) independently screened publications to confirm eligible articles and extract data. All the included articles were evaluated for quality by utilizing Newcastle–Ottawa Scale (NOS) quality assessment. High-quality studies with scores ≥5 were quantitatively analyzed.
Statistical methodology
STATA version 12.0 (USA) was employed to calculate statistical data. Synthesized odds ratio (OR) together with the corresponding 95% confidence interval (CI) were applied to assess the effect of HIF-1α on HCC. Q statistic at P-value < 0.10 and I2 ≥ 50% confirmed substantial inconsistency across trials. 17 A random or fixed effects model was used to synthesize data when faced with heterogeneity or homogeneity. Sensitivity analysis was performed to evaluate the effect of single research on the pooled results. Publication bias was assessed using funnel plot symmetry as well as Begg's and Egger's tests. Significance was defined at P-value < 0.05.
Results
Study identification and characteristics
A total of 2759 potentially applicable articles were screened, and 2736 were excluded from analysis after careful examination (Figure 1). Twenty-three studies were considered eligible for quantitative analysis15,16,18–38; of which 21 were conducted in China, 1 in Japan, and 1 in Egypt. A total of 2244 specimens were involved in the analysis. All patients with HCC were evaluated to confirm HBV presence by detection of HBsAg, 1606 HBV-related HCC tissue samples, 162 adjacent tissue samples, 375 non-HBV HCC tissue specimens, and 101 normal tissue specimens. Three studies analyzed the overall survival (OS) rate. The main baseline characteristics and quality assessment of each included article is recorded in Table 1.
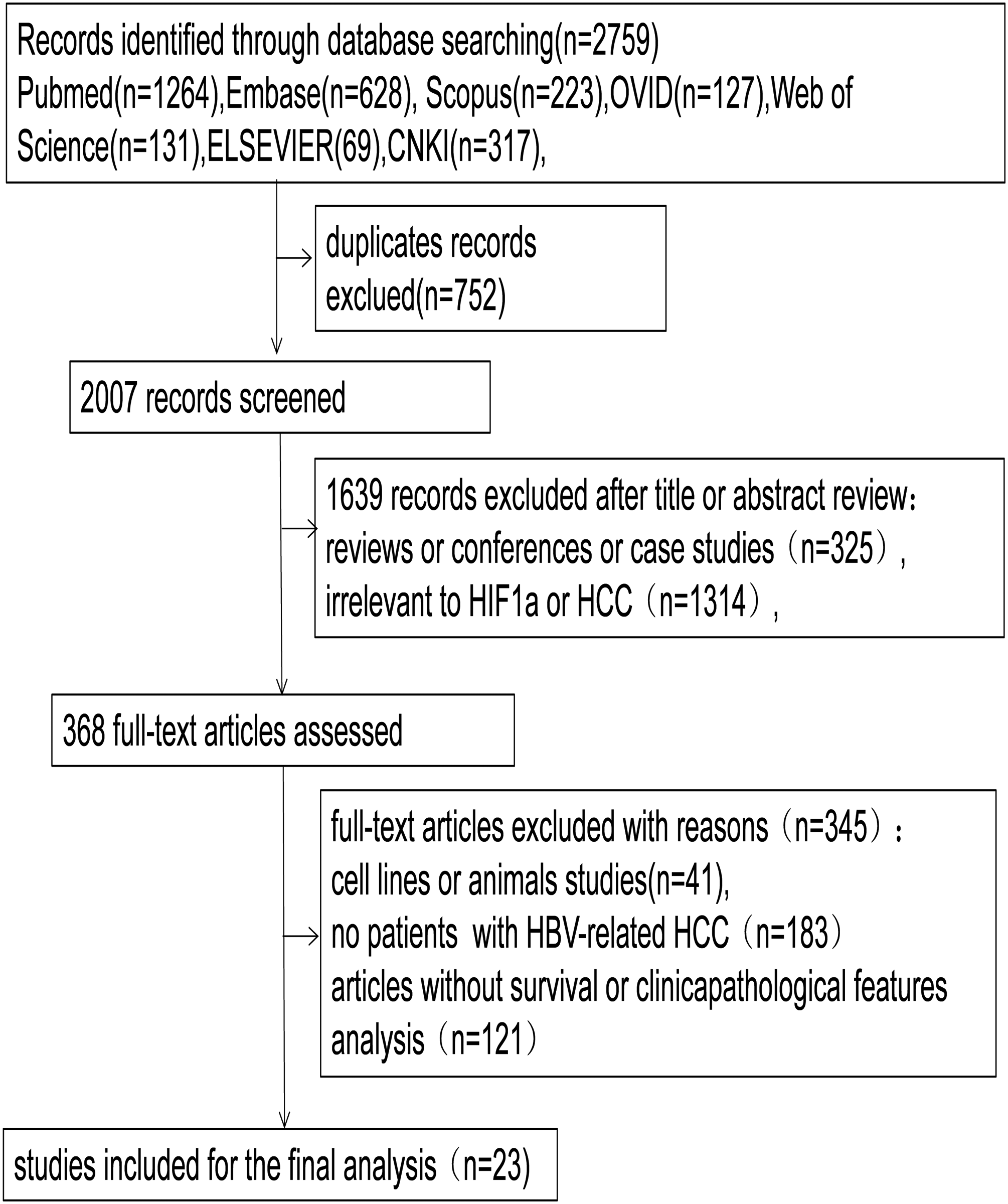
Flow diagram of selecting studies in the meta-analysis.
Basic characteristics of all included studies.
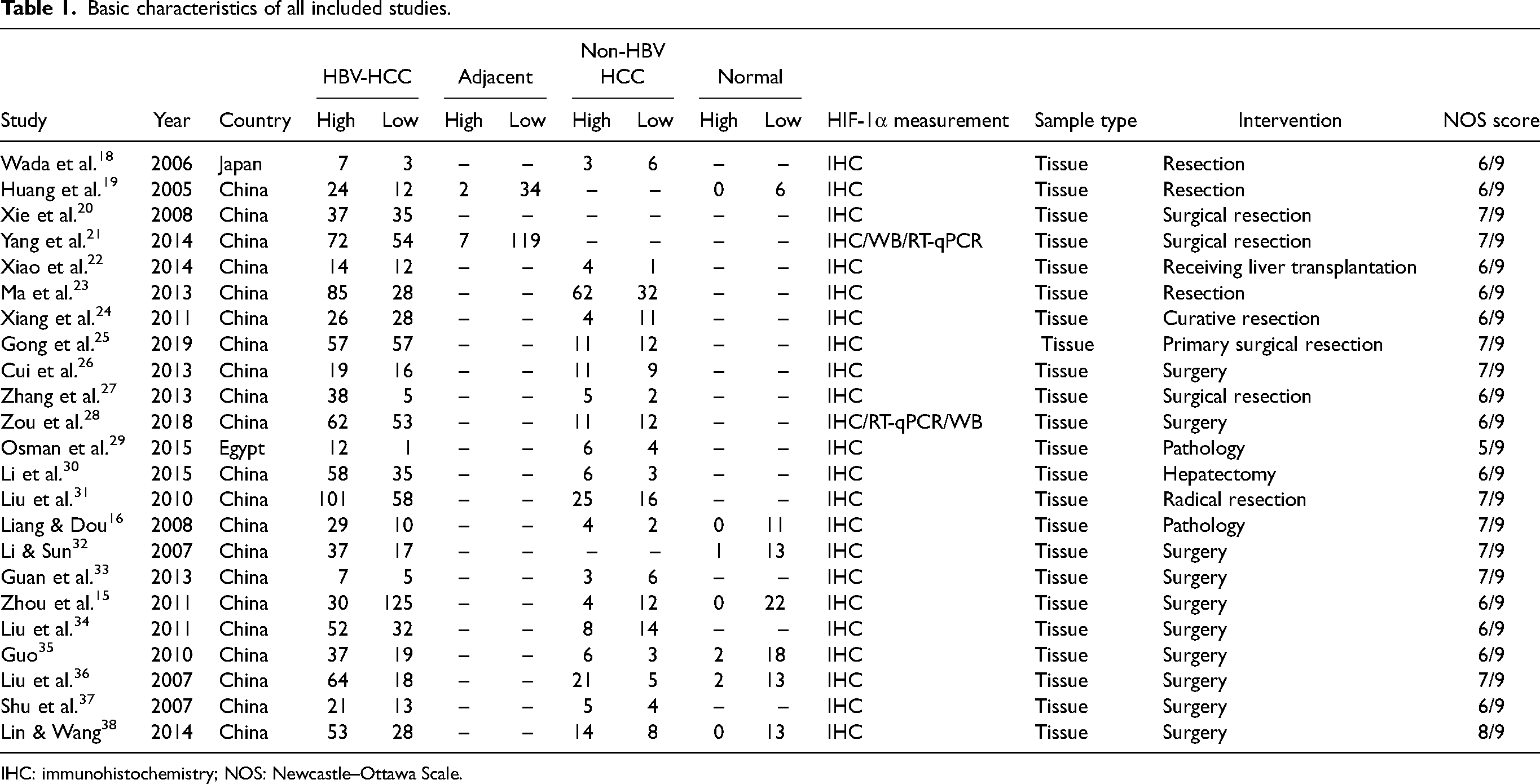
IHC: immunohistochemistry; NOS: Newcastle–Ottawa Scale.
HIF-1α expression and susceptibility to HBV-related HCC
The HIF-1α protein in HCC was more frequently overexpressed than that in non-tumor tissues, with a pooled OR of 25.109 (95% CI = 14.160, 44.522; z = 11.03; P = 0.000; I2 = 0%). The HIF-1α level was markedly increased in HCC than in normal liver tissues, with a pooled OR of 24.644 (95% CI 10.601, 57.289; z = 7.45; P= 0.000; I2 = 0.0%). The HIF-1α level in HBV-related HCC tissues was elevated compared with that in non-HBV HCC tissues, with a pooled OR of 1.353 (95% CI 1.052, 1.741; z = 2.35; P = 0.019; I2 = 0.0%). HIF-1α was distinctly overexpressed in HCC tissues than in adjacent tissues, with a pooled OR of 24.727 (95% CI 11.773, 51.937; z = 8.47; P = 0.000; I2 = 0.0%); The results are summarized in Table S1 and Figure 2.
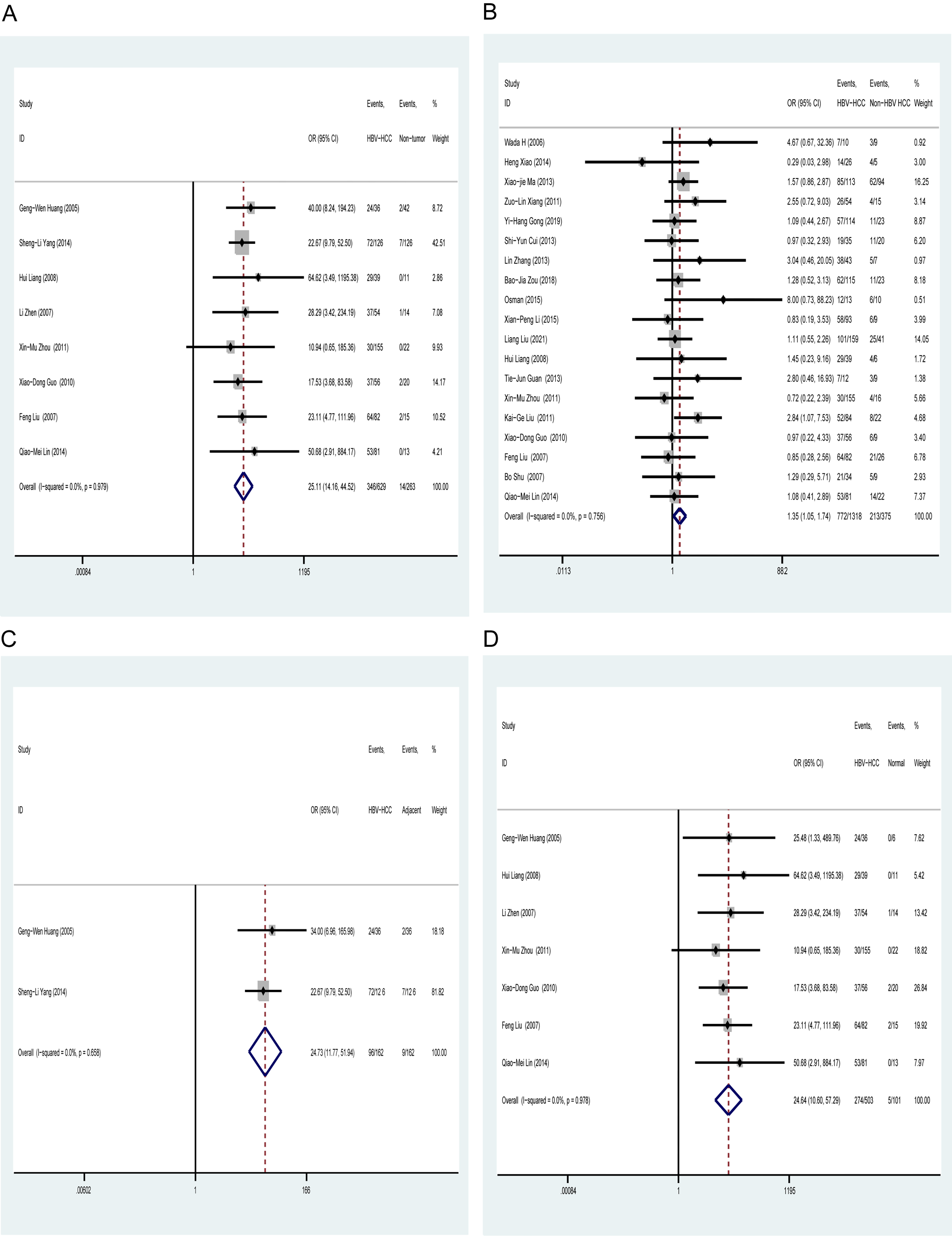
Hypoxic inducible factor-1α expression of HBV-related HCC tissue and other different liver tissues. (a) Neoplasm vs. non-tumor samples (including adjacent tissues and normal tissues). (b) Neoplasm vs. non-HBV-related HCC samples. (c) Neoplasm vs. adjacent samples. (D) Neoplasm vs. normal samples. HBV: hepatitis B virus infection; HCC: hepatocellular carcinoma.
Correlation of HIF-1α expression and clinical features in HBV-related HCC
HIF-1α overexpression was significantly positively associated with lymph node metastasis (OR = 3.778; 95% CI 1.666, 8.568), with no heterogeneity (I2 = 0.0%; P = 0.322). HIF-1α was positively correlated with portal vein invasion (OR = 6.728; 95% CI 2.191, 20.656), with acceptable heterogeneity (I2 = 56.6%; P = 0.100). HIF-1α was statistically associated with capsular infiltration (OR = 1.767; 95% CI 1.058, 2.950), with slight heterogeneity (I2 = 49.7%; P = 0.137). HIF-1α expression was not related to other tumor-related factors including histological grade, gender, age, tumor size, Edmondson grading, cirrhosis, and AFP. The results are summarized in Table S2 and Figure 3.
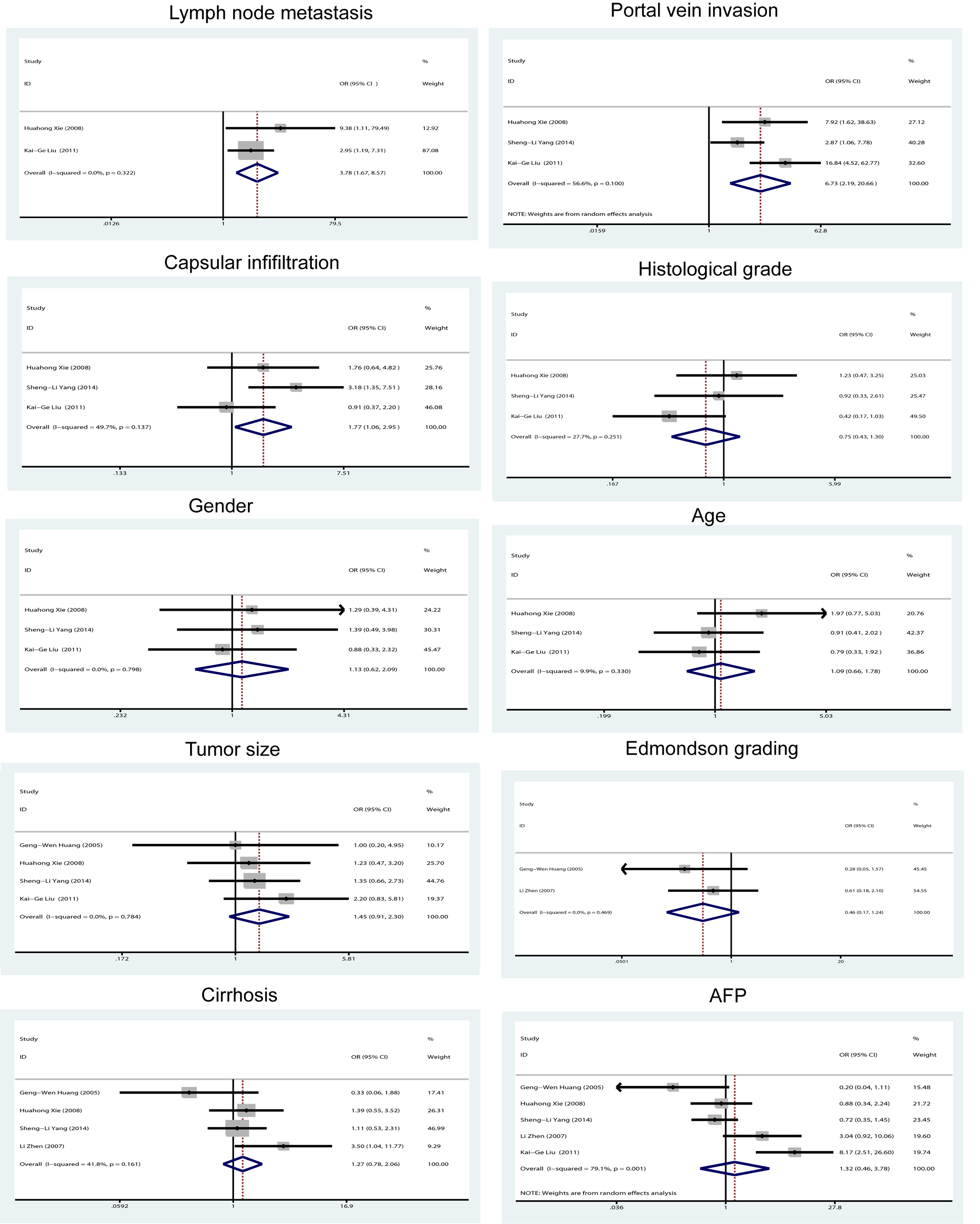
Association of hypoxic inducible factor-1α expression with clinicopathological factors in HBV-related HCC.
Prognostic value of HIF-1α protein overexpression in HBV-related HCC
Three studies19–21 were pooled to estimate the prognostic effect of the HIF-1α protein expression on HBV-related HCC (Figure 4). HIF-1α was not statistically relevant to poor OS in HBV-related HCC, with a pooled OR of 1.433 (95% CI 0.977, 2.102; z = 1.84; P = 0.065). Data were analyzed for slight heterogeneity by using a fixed-effects model among primary studies (P = 0.192; I2 = 39.5%).
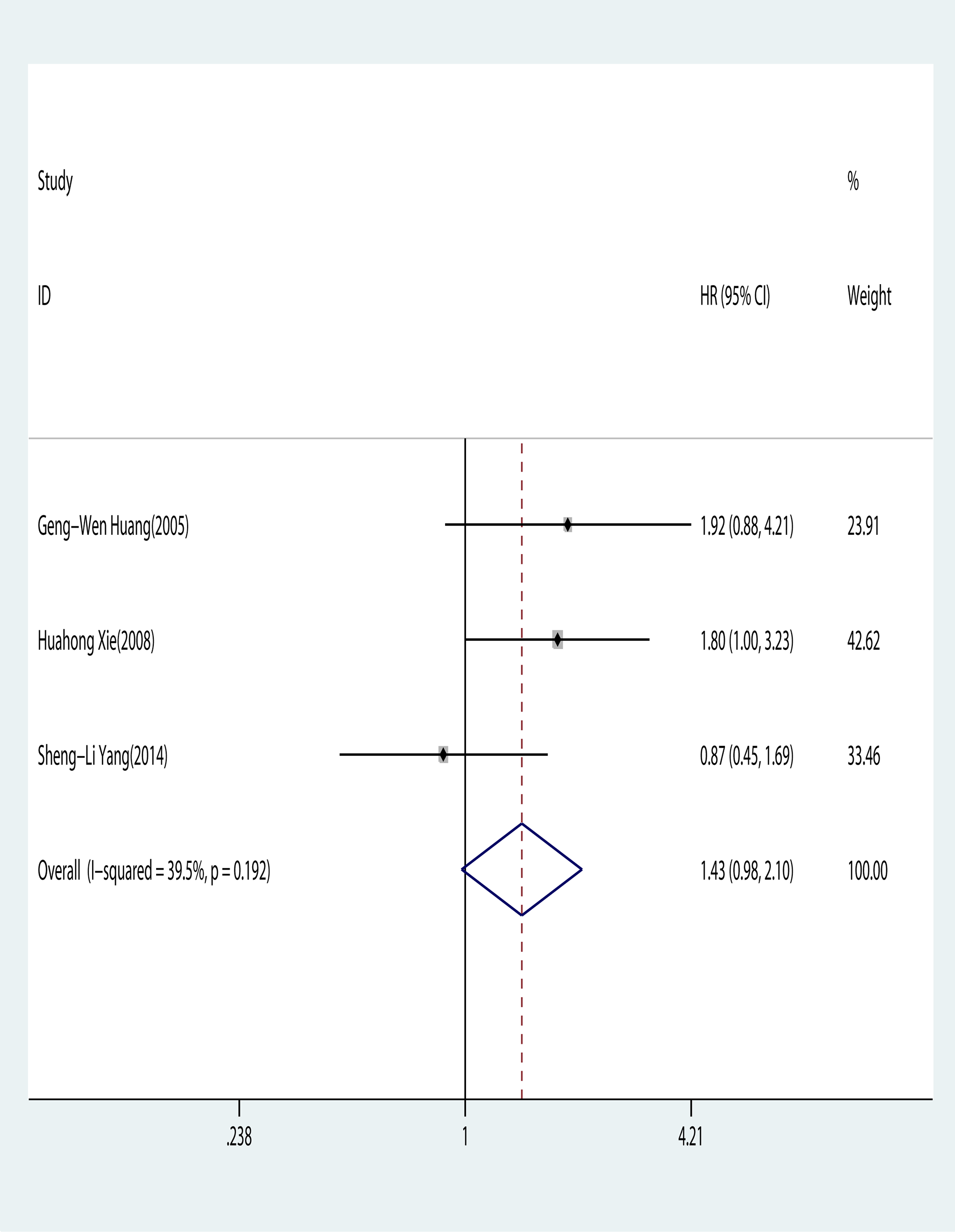
Association of hypoxic inducible factor-1α expression with overall survival in HBV-related HCC.
Publication bias and sensitivity analysis
Begg's and Egger's tests displayed no evidence of potential publication bias, and the funnel plot was presented to be symmetric. Sensitivity analysis confirmed the lack of considerable effect of a single study on the pooled results. Our findings were robust among all studies (Table S1 and Figure S1).
Discussion
HCC is a malignancy with high mortality worldwide. HBV infection can frequently stimulate human hepatocarcinogenesis. Identification of potential clinical effective biomarkers for accurate determination of disease occurrence, progression, and prognosis in HBV-related HCC plays a critical role in selecting suitable targeted therapies for prolonging patient survival. Hence, the present work was conducted to search for new biomarkers and to summarize evidence on their suitability for predicting the prognosis of HBV-related HCC.
Hypoxia serves as a frequent microenvironment characteristic in various types of solid malignancies due to intense metabolic activity of tumor cells. 39 The adaptive response to tumor hypoxic conditions for numerous tumor cells is mainly fulfilled by HIF-1α, which is a key modulator of angiogenesis in response to hypoxia and accelerates metastasis and progression in a vast array of cancer. 40 Increased clinical trials provided evidence that HIF-1α up-regulation is enrolled in carcinogenesis and tumor development, metastasis potential, failure of targeted therapies, increased mortality; hence they induce unfavorable prognosis in a battery of malignant tumors.13,40,41 Angiogenesis damage in hepatic cirrhosis and the rapid proliferation of tumor cells induces the formation of a hypoxic microenvironment, which function as a powerful trigger in stimulating HIF-1α overexpression in HCC. 42 The carcinogenic effects of HIF-1α have been detected in HBV-related HCC under hypoxic conditions. However, the results from previous literature are conflicting.15,16 The present work aimed to drive a more precise estimation on the relation of HIF-1α and HBV-related HCC.
Our final analysis combined the clinical outcomes of 2244 patients from 23 studies. The results confirmed that HIF-1α levels were elevated in HBV-related HCC tissues rather than in non-HBV HCC tissues, normal liver tissues, and adjacent tissues. HIF-1α overexpression significantly increased the susceptibility to HBV-induced HCC. HIF-1α upregulation also induced the overproduction of angiogenesis by mediating several transcriptional factors and hence contributed to the tumor metastasis and progression, 43 which might trigger HBV-related hepatocarcinogenesis under hypoxia. 20 Another study found that HIF-1 elevates the levels of HBV RNA transcript, core protein, and HBV genome replication in liver cell lines, 44 which suggests that HIF-1 may contribute to the occurrence of HCC by promoting HBV gene replication and raise transcript levels in vivo. Decreasing the transcription activity of HIF-1 prevented HIF-1-mediated effects on the HBV lifecycle. Thus, HCC cell proliferation can be blocked with HIF-1α inhibitors. 45
Our analysis provided evidence that HIF-1α expression was closely related to lymph node metastasis, portal vein invasion, and capsular infiltration. Several papers confirmed that HIF-1α overexpression was positively associated with a greater risk of lymph node metastasis in HCC, 46 and inhibition of HIF-1α has been documented to promote HCC cell apoptosis in cell lines. 47 HIF-1α positive expression was described to be closely related to portal vein tumor thrombus in previous work, and portal vein invasion served as an important risk factor for poor prognosis in HCC. 48 A recent study found that overexpression of HIF-1α is strongly linked to capsular infiltration and portal vein invasion in HCC. 21 The reason behind this may be due to the elevated levels of Notch3 expression in HCC cells caused by overexpression of both HBx and HIF-1α, ultimately leading to vascular infiltration in HCC. 49 Multiple reports provided evidence that the presence of lymph node metastasis, portal vein invasion, and capsular infiltration was the main risk factor for tumor occurrence and tumor invasiveness, which can increase the possibility of systemic diffusion and biological aggressiveness in malignant tumors. 40 As such, malignancies with high HIF-1α expression could possess a more aggressive and metastatic biological behavior, thereby increasing the risk of systemic metastasis and tumor invasion in HCC, 21 which infers that once cancer cells acquire a high expression of HIF-1α, tumor cells gain obvious propensity for tumor invasion and metastasis among HBV-related HCC patients.
The majority of eligible studies examining the correlation between HBV-related HCC and HIF-1α expression for our analysis were conducted on Chinese populations, which may be due to the fact that the many HIF-1α gene polymorphisms conferred remarkably increased the susceptibility of digestive system cancers in east Asian populations.50–52 Also, a recent study further confirmed that Chinese populations are particularly susceptible to developing HBV-related HCC as a result of certain HIF-1α gene polymorphisms. 14
We elucidated the prognostic effect of HIF-1α on HBV-induced HCC based on the available data in three eligible studies. HIF-1α expression did not affect OS in HBV-induced HCC; however, this result contradicted previous findings. 20 The conflicting results could be due to the limited data available in the original research. In this regard, the effect of HIF-1α on the survival of patients with HBV-related HCC cannot be concluded.
Analysis of publication bias provided evidence on the lack considerable discrepancy among eligible studies. Sensitivity analysis confirmed that no highly influential research could affect the overall conclusion in the present work.
This review has several limitations that should be acknowledged when interpreting the findings. First, most of the clinical studies were conducted in Asian countries because more than half of new patients diagnosed with HCC originated from China. Additional studies in other geographic regions are urgently needed. Second, the assessment of high HIF-1α expression was inconsistent among all eligible studies because of the flexibility of immunohistochemistry. Finally, the number of available included studies and the patient sample size were relatively small, which has led to the restricted value of the data obtained.
Conclusions
In summary, HIF-1α overexpression could indicate a high possibility of tumorigenesis and progression for HBV-related HCC. Additional dynamic studies with larger populations evaluating the effects of HIF-1α expression in HCC should be conducted to obtain more reliable results in the future.
Supplemental Material
sj-doc-1-jbm-10.1177_03936155231204391 - Supplemental material for Association of hypoxia-inducible factor 1α expression with susceptibility to hepatitis B virus-related hepatocellular carcinoma: A meta-analysis
Supplemental material, sj-doc-1-jbm-10.1177_03936155231204391 for Association of hypoxia-inducible factor 1α expression with susceptibility to hepatitis B virus-related hepatocellular carcinoma: A meta-analysis by Lei Wang and Jin-lin Peng in The International Journal of Biological Markers
Supplemental Material
sj-doc-2-jbm-10.1177_03936155231204391 - Supplemental material for Association of hypoxia-inducible factor 1α expression with susceptibility to hepatitis B virus-related hepatocellular carcinoma: A meta-analysis
Supplemental material, sj-doc-2-jbm-10.1177_03936155231204391 for Association of hypoxia-inducible factor 1α expression with susceptibility to hepatitis B virus-related hepatocellular carcinoma: A meta-analysis by Lei Wang and Jin-lin Peng in The International Journal of Biological Markers
Supplemental Material
sj-doc-3-jbm-10.1177_03936155231204391 - Supplemental material for Association of hypoxia-inducible factor 1α expression with susceptibility to hepatitis B virus-related hepatocellular carcinoma: A meta-analysis
Supplemental material, sj-doc-3-jbm-10.1177_03936155231204391 for Association of hypoxia-inducible factor 1α expression with susceptibility to hepatitis B virus-related hepatocellular carcinoma: A meta-analysis by Lei Wang and Jin-lin Peng in The International Journal of Biological Markers
Footnotes
Acknowledgments
None.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Lei Wang and Jin-lin Peng. The draft of the manuscript was written by Lei Wang and Jin-lin Peng. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
