Abstract
Introduction
Papillary thyroid carcinoma is the most common malignancy of the endocrine system. Most papillary thyroid carcinoma patients enjoy excellent outcomes. However, in patients with biologically aggressive features, additional prognostic and predictive data may aid disease management. Dysregulation of the endocannabinoid system including the cannabinoid receptors 1 and 2 (CB-1R and CB-2R) during carcinogenesis has been extensively studied over the last few decades. The aim of this study was to evaluate immunohistochemically the expression levels of both receptors in patients with papillary thyroid carcinoma and benign diseases, and to compare these rates and the histopathologically and clinically prognostic features.
Methods
The pathological materials and clinical data of 100 patients with papillary thyroid carcinoma and 40 with benign diseases were retrospectively re-evaluated. All tissues were immunohistochemically stained for CB-1R and CB-2R. The expression levels of CB-1R and CB-2R in papillary thyroid carcinomas, and benign lesions were recorded and compared with the pathological and clinical features.
Results
The expression levels of both receptors were significantly higher in papillary thyroid carcinoma patients than in those with benign conditions (P = 0.001). CB-1R expression correlated with both extrathyroidal extension (P = 0.022) and capsular invasion (P = 0.001). CB-2R expression was associated with the risk group of the American Thyroid Association stratification system (P = 0.004).
Conclusion
Our study suggests that increased cannabinoid receptor expression contributes to thyroid carcinogenesis. The CB-2R expression level could provide additional information aiding risk management. Furthermore, the CB-1R and CB-2R antibodies might increase the accuracy of papillary thyroid carcinoma diagnosis when combined with the papillary thyroid carcinoma biomarkers assayed after fine-needle aspiration of neoplastic cells.
Introduction
The endocannabinoid system (ECS) is present throughout the human body. 1 Like the arachidonic acid metabolites, endocannabinoids are lipid-based endogenous metabolites that regulate and control many of the most critical functions such as learning and memory, emotional processing, sleep, temperature control, pain control, inflammatory and immune responses, and eating. 2 The ECS also has antiproliferative effects by the inhibition of several growth factors 3 and the deregulation of such signaling pathways like Ras-Raf-MKKK1-ERK1/2, 4 PI3K-Akt/PKB-mTOR 5 and c-Jun N-terminal kinase-MAPK. 6 The ECS effects by binding to cannabinoid-specific receptors (CBR), CB-1, and CB-2. 7 Activation of CB-1R, the dominant receptor of the central nervous system, is responsible for many central and peripheral effects.8–10 CB-2R on the other hand, is highly expressed in B and T lymphocytes, macrophages, and in tissues such as the spleen, tonsils, and lymph nodes, 8 although CB-2R has been discovered in the brainstem as well. 11 Both CB-1R and CB-2R are G-coupled receptors that are coupled through the Gi/o family of proteins to signal transduction mechanisms that include inhibition of adenylyl cyclase, activation of mitogen-activated protein kinase, regulation of calcium and potassium channels (CB-1R only), and other signal transduction pathways. 7
The endogenous ligands, agonists for cannabinoid receptors, include eicosanoids arachidonoyl ethanolamide (anandamide), 2-arachidonoylglycerol (2-AG), and 2-arachidonylglyceryl ether. 12 Exogenous ligands are the so-called phytocannabinoids, which are bioactive components of the Cannabis Sativa plant (marijuana), and synthetic cannabinoid-like compounds. More than 113 cannabinoids have been isolated from Cannabis Sativa, including delta-9 tetrahydrocannabinol (Δ9-THC) and cannabidiol (CBD), which are two of the most notable major constituents. 13 Synthetic cannabinoids consist of several molecules, which are classified by the creator of the substance (e.g., AM-xxx, CP-xxx, HU-xxx, WIN-xxx, and JWH-xxx). 14
It has been proposed that the regulation of ECS has been compromised during carcinogenesis, thus leading to a biologically more aggressive cancer behavior. Hope for new treatment modalities have emerged since the discovery of cannabinoid's anticancer mechanisms.15–17 To date, these are the most well-known anticancer effects of cannabinoids:
Cannabinoids increase the expression of intercellular adhesion molecule-1 (ICAM-1) in cancer tissue which helps tumor infiltrating lymphocytes (TIL) for increased target recognition.18,19 Activation of CBR triggers apoptosis and autophagy of neoplastic cells, and inhibits proliferation through relevant pathways.17,20,21 Cancer cells rely on the activity of matrix metalloproteinase enzyme-2 (MMP-2) in order to infiltrate surrounding tissues. CBR activation inhibits MMP-2 and activates simultaneously tissue inhibitor enzyme-1 of MMP (TIMP-1), thus decreasing the invasiveness of cancer cells.21–23 Cannabinoids are capable of blocking the Wnt/β-catenin pathway, which inhibits epithelial-mesenchymal transition resulting in migration, invasion, and angiogenesis inhibition.24–26 Cannabinoids can modulate angiogenesis via related mechanisms
27
by suppressing survival and migration properties of endothelial cells, and several angiogenetic growth factors including vascular endothelial growth factor (VEGF).27–29 Monoacylglycerol lipase (MAPL) enzyme degrades 2-AG into glycerol and free fatty acids. The activation of CB-1R results in inhibition of MAPL, which increases the level of 2-AG and TIMP-1, thus leading to a decrease of infiltration and metastasis capability.30,31
Papillary thyroid carcinoma (PTC) is the thyroid's most common malignancy, which originates from thyroid follicular epithelial cells (aka thyrocytes). PTC involves young adults and is three times more likely to develop in women. Despite its malignant nature, PTC has an excellent outcome 32 ; however, exceptional cases with certain clinical and pathological features related to poor prognosis such as older age, male gender, lymph node metastasis, distant metastasis, large tumor size, extrathyroidal extension, and certain histologic subtypes do exist. 33 Head and neck lymph node involvement of PTC are diagnosed by ultrasound, while nuclear medicine procedures are being used for regional and distant metastases. In recent years, 131I SPECT/CT has shown the most performance.34,35 With surgery and radioiodine therapy the 5-year survival rate for regional PTC is 99%, whereas in metastatic PTC the 5-year survival rate drops down to 75%. 32 In this regard the search for new therapy strategies, especially in advanced cases, is required in order to enhance current treatment modalities. The current study aims to investigate the expression of either CB-1 and CB-2 receptors in PTC and benign thyroid lesions, and compare the expression rate with pathological prognostic and clinical parameters.
Material and methods
Patient selection
In the current study, we retrospectively retrieved 100 thyroidectomy cases from the archives of Gaziantep University Hospital, Faculty of Medicine, Department of Pathology between 2017 and 2022, which were diagnosed as PTC, and 40 thyroidectomy cases diagnosed with benign thyroid nodules (BTN), including follicular adenoma and hyperplastic/colloid nodule. All patients were diagnosed and follow-ups were made in our institute. In addition to histological findings, clinical data were obtained from the medical archives and recorded. Patients that were lost to follow-up, whose clinical or histological material were not available, were excluded from the current study. Among clinical parameters serum thyroid stimulating hormone (TSH), thyroglobulin, stimulated TSH, and stimulated thyroglobulin levels were recorded during follow-up.
Histological assessment
For PTC cases, all slides and pathological reports were re-evaluated retrospectively by two pathologists for histological subtype, location of tumor (bilateral/unilateral and multifocality), tumor size (greatest diameter), thyroid capsule invasion, extrathyroidal extension, vascular invasion, and lymph node involvement. During histological assessment, representative PTC and BTN samples were selected for immunohistochemical analysis.
Immunohistochemical analysis
Immunohistochemical studies were performed on 5μm sections of formalin-fixed, paraffin-embedded tissues. CB-1R (bs-1683R, Polyclonal Rabbit IgG, 1:100, Bioss, Beijing, China) and CB-2R (bs-2377R, Polyclonal Rabbit IgG, 1:100, Bioss, Beijing, China) antibodies were performed using an automated immunohistochemistry-staining device (Ventana, Bench Mark Ultra Auto Stainer, Roche Diagnostics, Indianapolis, IN, USA).
Semi quantitative evaluation of immunohistochemical expression
Staining was evaluated and scored by using the immunoreactive score (IRS) system. 36 Cytoplasmic reaction was evaluated as positive staining for both antibodies. Appropriate external positive controls were used: placenta samples for CB-1R and lung samples for CB-2R served as positive controls.
Statistics
The defining statistics of the data obtained from this study were calculated by using mean for numerical variables, frequency and percent analysis for standard deviation and categorical variables. The Kruskal–Wallis test was used to compare clinical parameters with CB-1R and CB-2R expression. The differences between categorical variables were analyzed with the chi-square test. After variant analysis, the Tukey multiple comparison test was used to determine the differences between groups. To compare the epidemiological data with the expression level of CBR antibodies t-test and chi-square test were used. Analysis was carried out with SPSS ver. 2.0. P < 0.05 were evaluated as statistically significant.
Results
Demographic and histomorphological findings
A total of 100 thyroidectomy cases with PTC and 40 thyroidectomy cases with benign nodules were studied. Of the patients with malignant disease 82 (82%) were female, 18 (18%) were male, and the female/male ratio was 4/55; whereas of the patients with benign disease 33 (82.5%) were female, were 7 (17.5%) were male, and the female/male ratio was 4/71. The mean age of patients with malignant disease was 40.87 ± 14.99 and 4.5 ± 14.39 in the benign group. Histological subtypes of PTC consisted of 43 classic, 42 follicular, and 15 tall cell variants. The median diameter of tumors was 2.47 cm (range 1–6.5). Among all cases neither patient had distant metastasis whereas lymph node metastasis and vascular invasion was evident in 20 cases, multifocality in 43 cases, extrathyroidal extension in 21 cases, capsular invasion in 48 cases, and the presence of tumor in bilateral lobes in 55 cases (Table 1).
Table of characteristics of PTC cases.
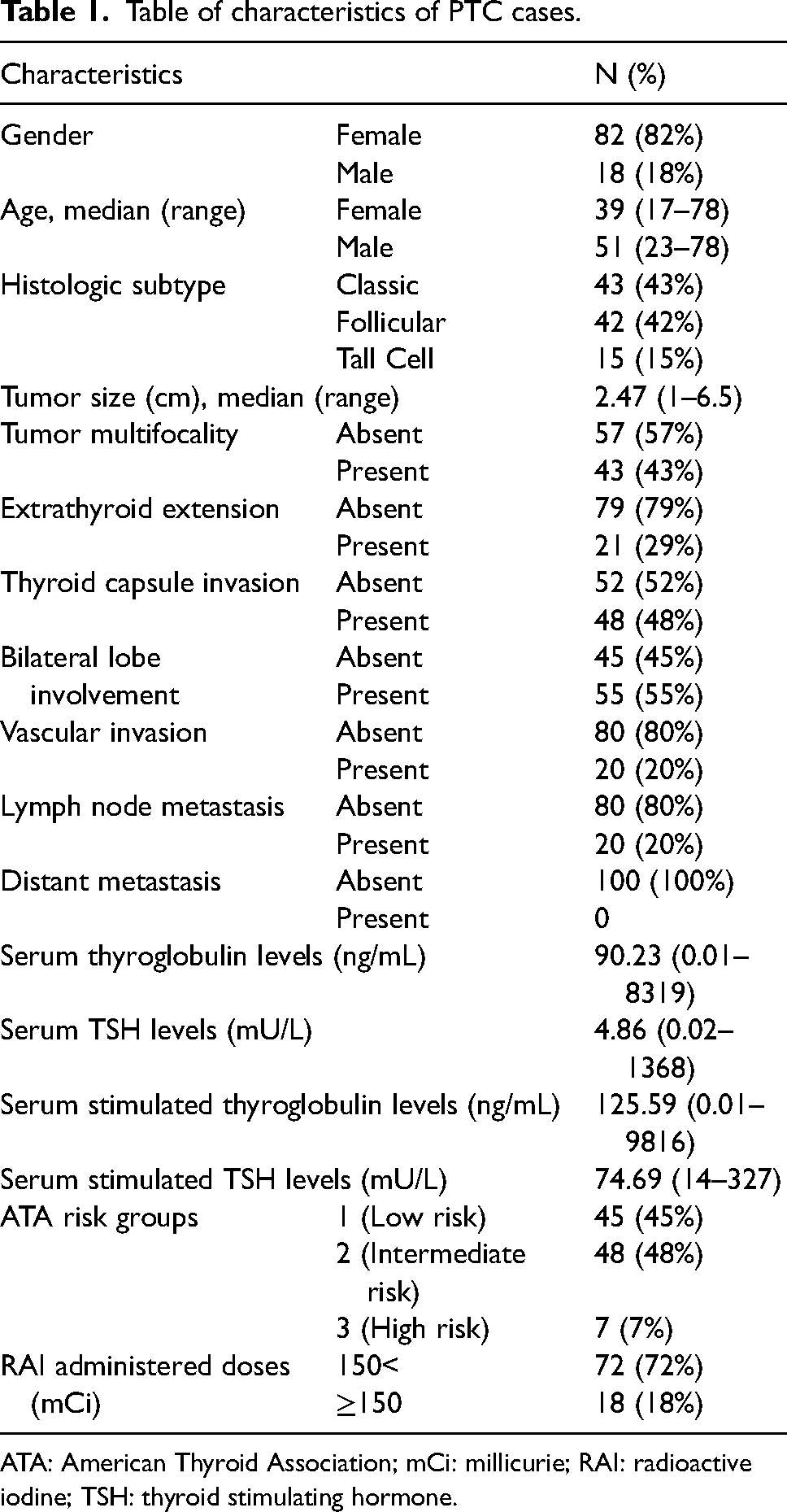
ATA: American Thyroid Association; mCi: millicurie; RAI: radioactive iodine; TSH: thyroid stimulating hormone.
Expression rate of CB-1R and CB-2R in BTN and PTC patients
The expression rate of CB-1R in BTN was negative (IRS 0) 30 cases (75%); had mild reaction (IRS 1) in 9 cases (22.5%); and moderate reaction (IRS 2) in 1 case (2.5%). The expression rate of CB-2R in BTN was negative in 28 cases (70%); had mild reaction in 11 cases (27.5%); and strong reaction (IRS 3) in 1 case (2.5%). CB-1R expression in PTC was negative in 30 cases (30%); had mild reaction in 36 cases (36%); moderate reaction in 27 cases (27%); and strong reaction in 7 cases (7%). CB-2R expression in PTC was negative in 13 cases (13%); had mild reaction in 20 cases (20%); moderate reaction in 43 cases (43%); and strong reaction in 24 cases (24%) (Table 2). Thyroid parenchyma (thyroid follicular epithelium) was negative for both biomarkers (Figure 1 and 2).
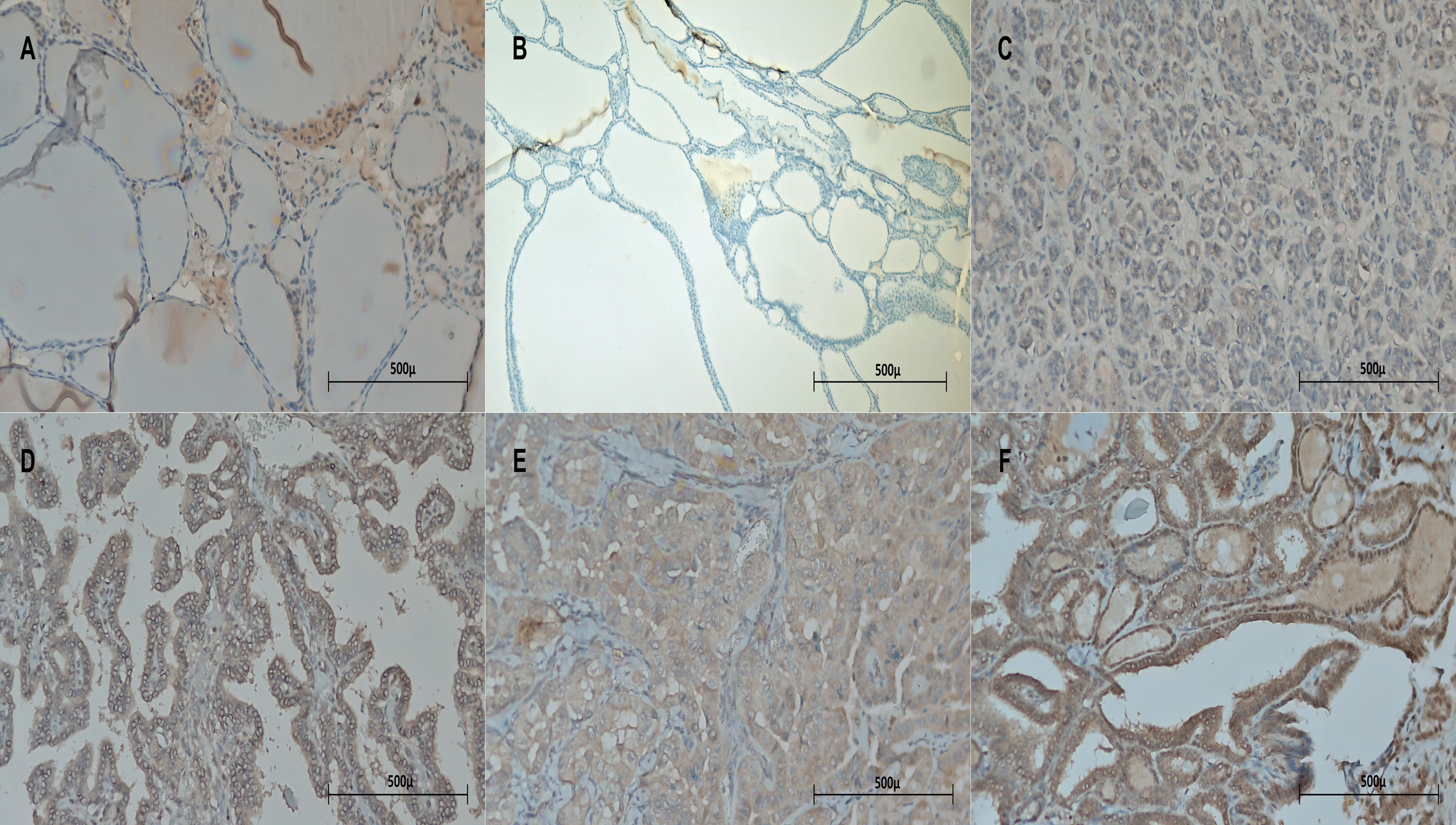
Expression of CB-1R in thyroid parenchyma, BTN, and PTC. (a) Normal thyroid follicular epithelium was negative for CB-1R (X200). (b) Benign thyroid nodule was negative for CB-1R (X200). (c) PTC was negative for CB-1R (IRS 0) (X200). (d) PTC demonstrates mild reaction for CB-1R (IRS 1) (X200). (e) PTC demonstrates moderate reactivity for CB-1R (IRS 2) (X200). (f) PTC shows strong reaction with CB-1R (IRS 3) (X200).
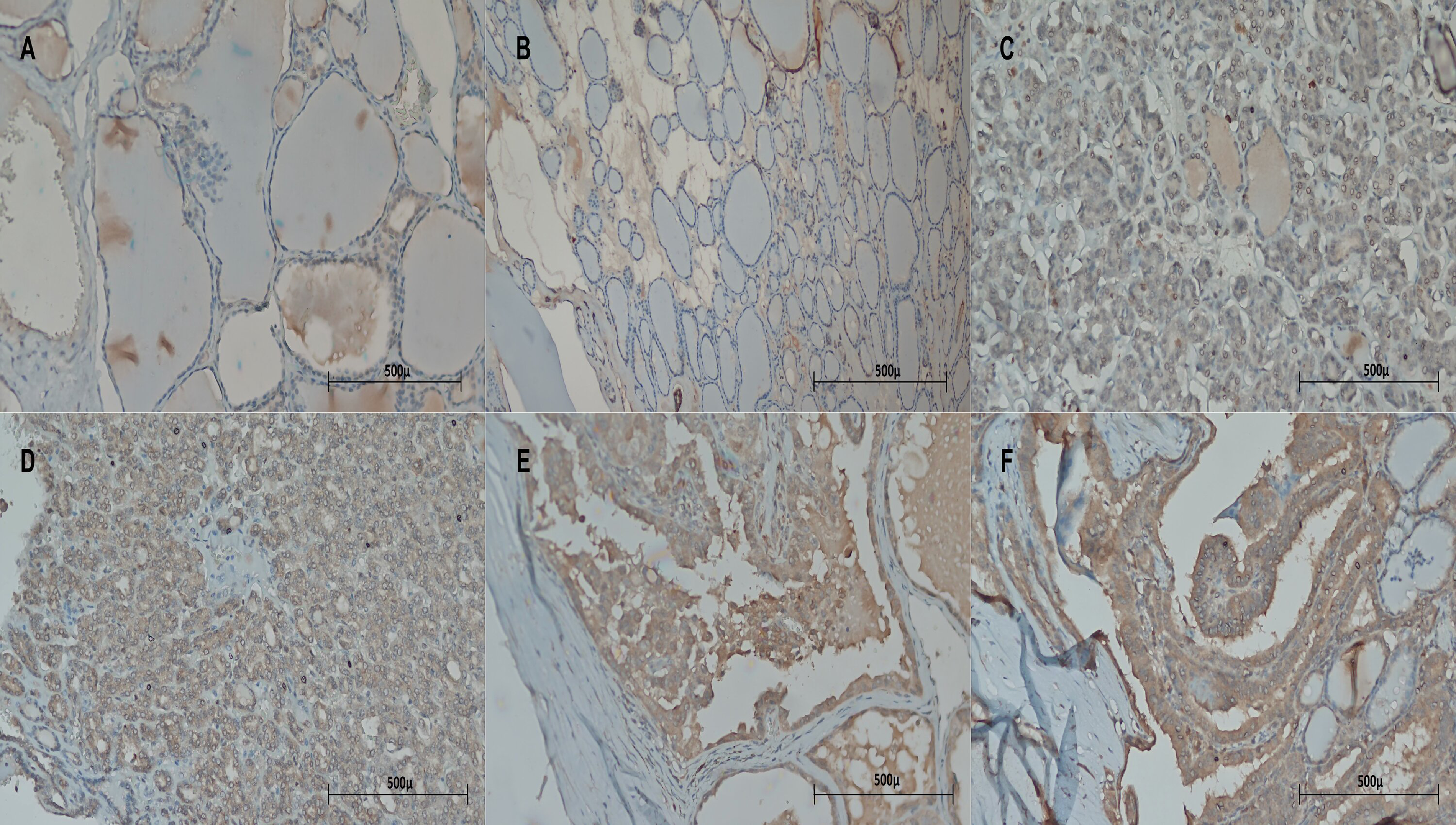
Expression of CB-2R in thyroid parenchyma, BTN, and PTC. (a) Normal thyroid follicular epithelium was negative for CB-2R (X200). (b) Benign thyroid nodule is negative for CB-2R (X200). (c) PTC is negative for CB-2R (IRS 0) (X200). (d) PTC demonstrates mild reaction for CB-2R (IRS 1) (X200). (e) PTC demonstrates moderate reactivity for CB-2R (IRS 2) (X200). (f) PTC shows strong reaction with CB-2R (IRS 3) (X200).
Comparison of CB-1R and CB-2R expression in PTC and BTN.
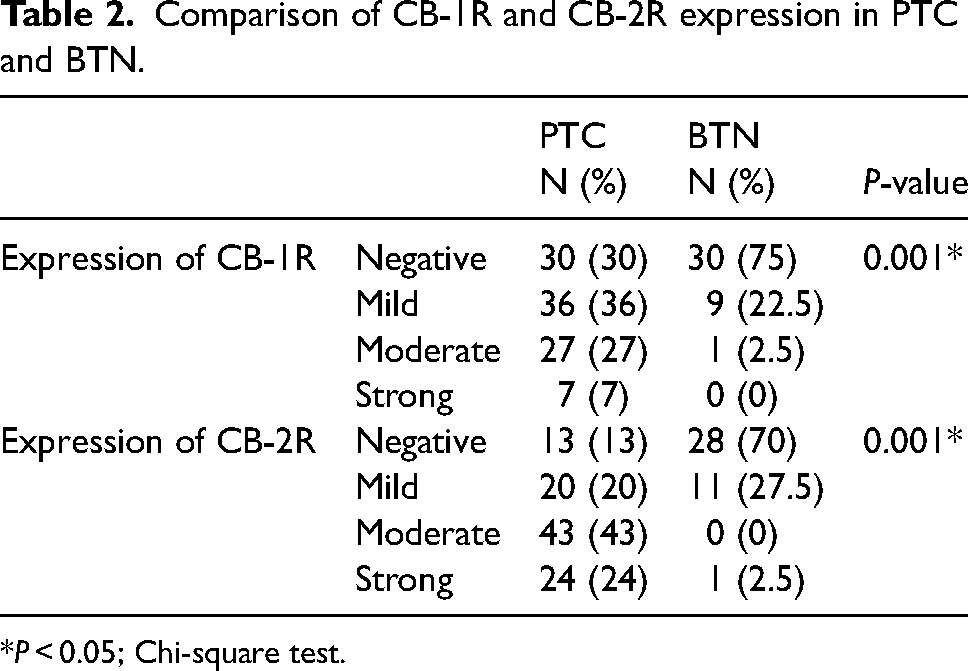
*P < 0.05; Chi-square test.
Comparison of CB-1R and CB-2R expression rate of patients with benign nodules versus papillary carcinoma
Immunoreactivity of both antibodies was evident in both benign and malignant cases, although expression of both antibodies was considerably higher in PTC compared to BTN. Comparison of CB-1R and CB-2R expression in PTC versus BTN was statistically significant (P = 0.001 and P = 0.001, respectively) (Table 2).
Comparison of CB-1R and CB-2R expression rate in PTC with clinical parameters
Comparison of tumor size, serum levels of thyroglobulin and TSH, stimulated thyroglobulin and TSH, and the radioactive iodine doses with the expression of CB-1R and CB-2R had no statistical significance except the relation between TSH level and CB-1R expression (P = 0.04) (Table 3).
Comparison of CB-1R and CB-2R expression rate in PTC with clinical parameters and tumor size.
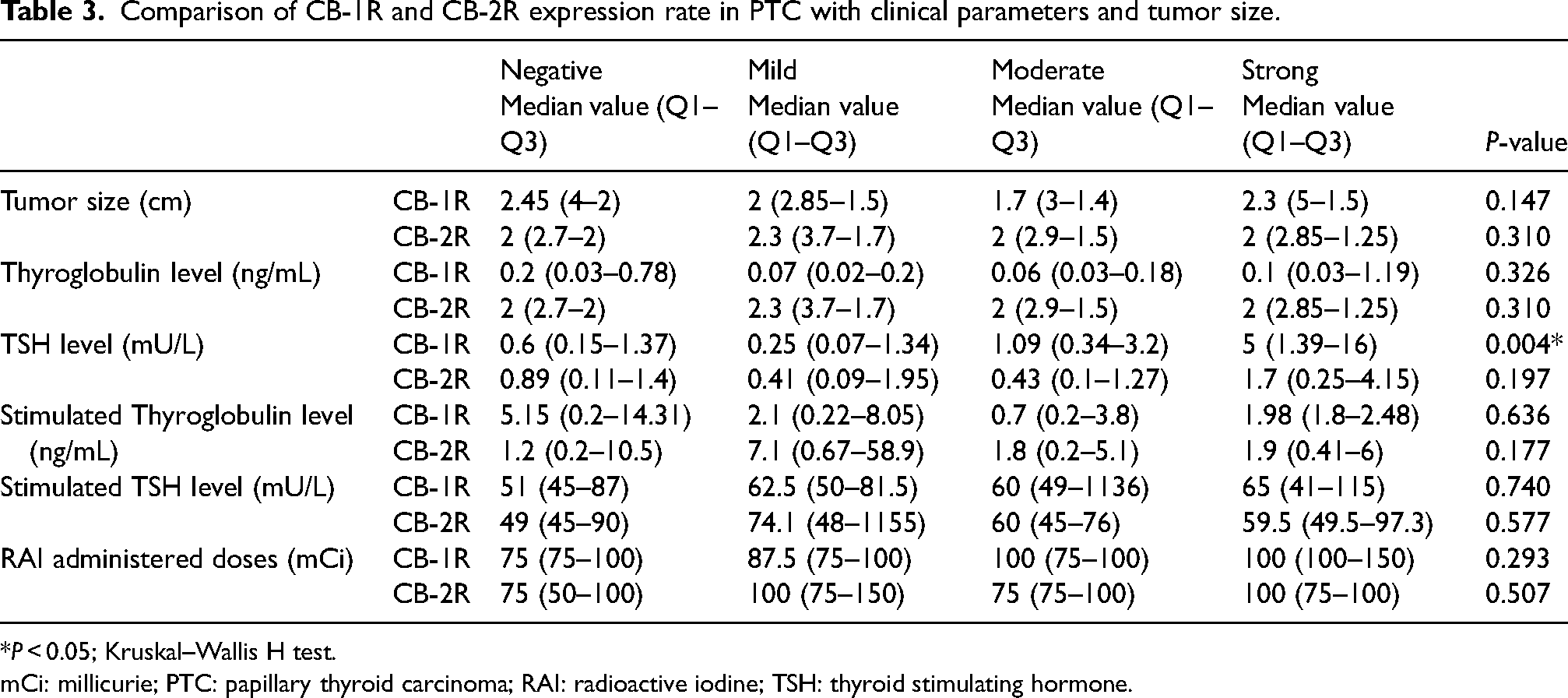
*P < 0.05; Kruskal–Wallis H test.
mCi: millicurie; PTC: papillary thyroid carcinoma; RAI: radioactive iodine; TSH: thyroid stimulating hormone.
Comparison of CB-1R and CB-2R expression rate in PTC with histomorphological parameters
Across all histological subtypes we did not observe any statistical relation with the expression of CB-1R and CB-2R (Table 4). Among histomorphological parameters including lymph node involvement, vascular invasion, multifocality, unilateral versus bilateral lobe involvement, capsule invasion, and extrathyroidal extension, the only significant relation was between CB-1R expression compared with extrathyroidal extension and capsule invasion (P = 0.022 and P = 0.001, respectively). Comparison of gender and age with both biomarker expressions was statically insignificant (Table 4).
Comparison of CB-1R and CB-2R expression with histological features, ATA risk classification, gender, and age.
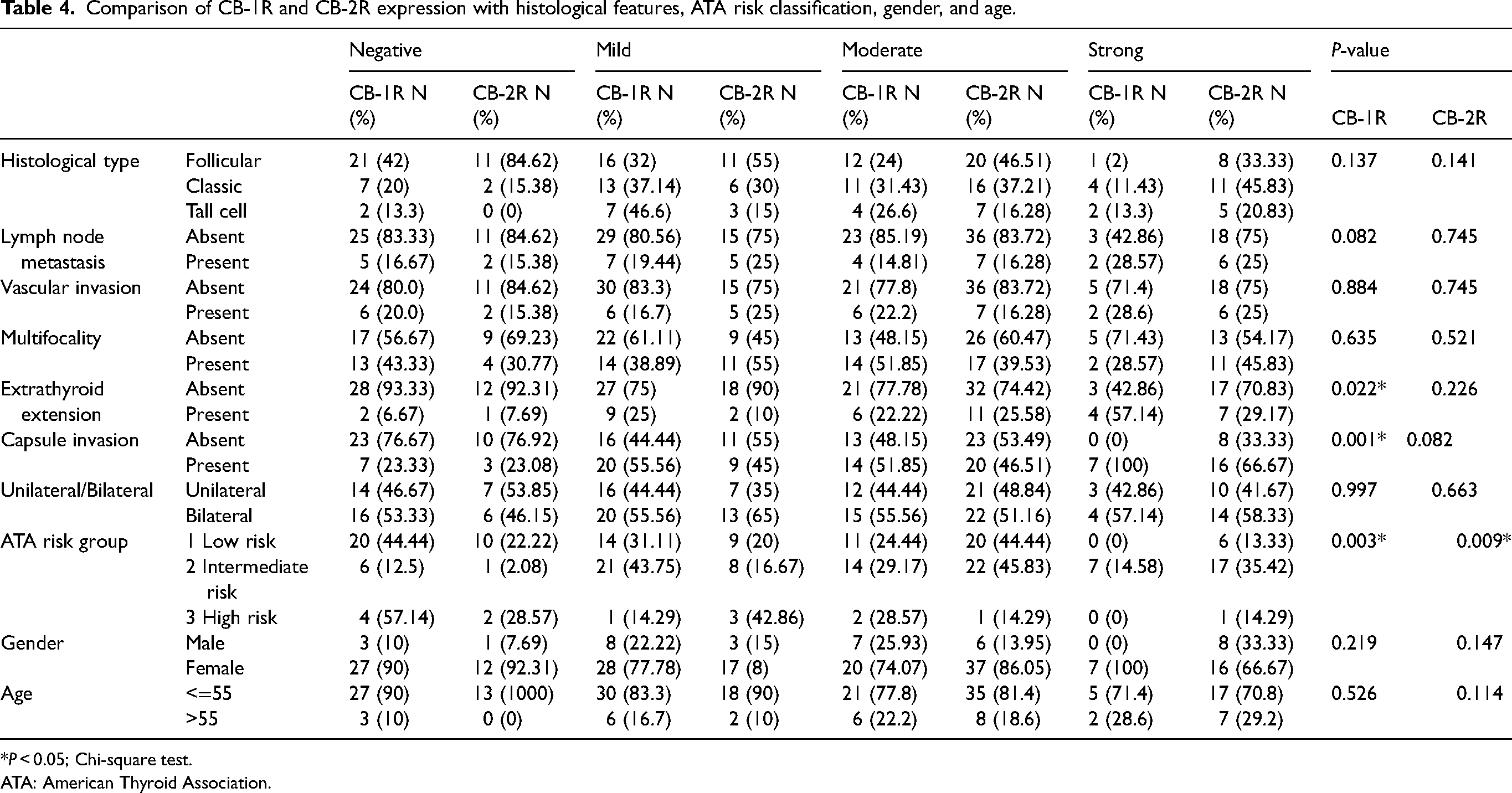
*P < 0.05; Chi-square test.
ATA: American Thyroid Association.
Comparison of CB-1R and CB-2R expression rate in PTC with American Thyroid Association risk system
Relation between CB-1R and CB-2R expression and the American Thyroid Association (ATA) risk system was statistically significant (P = 0.003 and P = 0.009, respectively) (Table 4). By regrouping the four-tier scoring system as low (negative and mild reaction) and high (moderate and strong reaction) expression of the statistical results were, this time, only relevant for CB-2R (P = 0.004) and insignificant for CB-1R (P = 0.138) (Table 5).
Comparison of CB-1R and CB-2R expression with rearranged ATA risk classification.
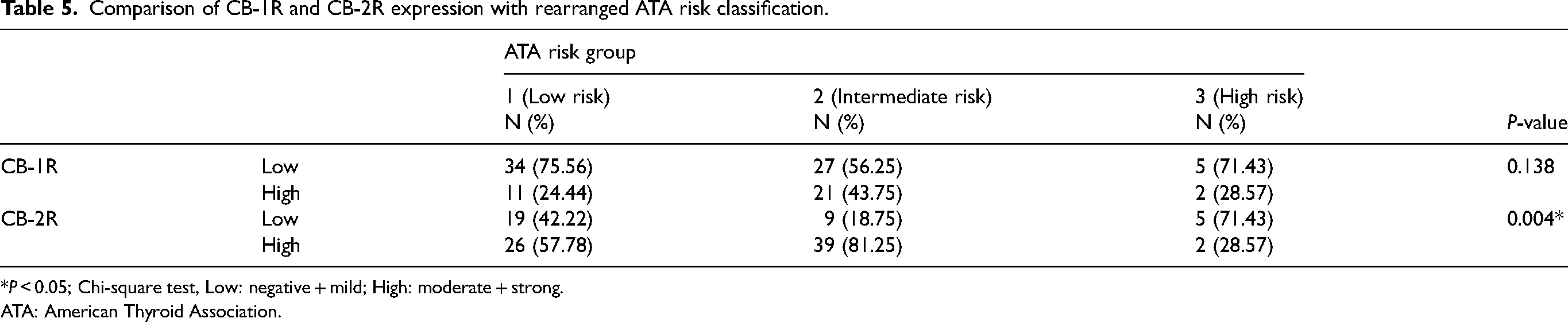
*P < 0.05; Chi-square test, Low: negative + mild; High: moderate + strong.
ATA: American Thyroid Association.
Discussion
The prognosis of PTC is generally excellent. The life expectancy is similar to that of the general population. In a minority of cases with adversely prognostic pathological features, disease management becomes more challenging. Thus, this study measured the expression levels of CB-1R and CB-2R in patients with PTC and benign conditions, explored whether such levels were prognostic, and whether they could be utilized in daily practice as predictive and/or diagnostic biomarkers.
Immunohistochemical tests revealed increased CB-1R and CB-2R expression levels in PTC tissues, lower levels in benign thyroid nodules, and no expression in normal thyroid follicular epithelium. Upregulation of CB-2R in PTC tissues was higher than that of CB-1R (87% vs. 70%). These findings correspond to the study results of Lakiotaki et al., 37 which analyzed expression rates of CB-1R and CB-2R in PTC versus benign thyroid conditions. Expression of both cannabinoid receptors was significantly higher in PTC than in benign lesions, and negative in surrounding thyroid tissue. Overexpression of cannabinoid receptors has been studied in esophageal squamous cell and renal cell carcinomas, breast cancer, endometrial carcinoma, non-Hodgkin lymphoma, and hepatocellular carcinoma.38–42 Regardless of the relationship between increased cannabinoid receptor expression and prognosis, the antineoplastic effects of cannabinoid agonists by cannabinoid receptor activation, renders cannabinoid receptors as an appropriate target for cancer treatment. Cozzolino et al. 43 showed that a stable analogue of anandamide inhibited thyroid anaplastic carcinoma and poorly differentiated PTC cells by activating apoptosis. Shi et al. 44 transfected anaplastic thyroid carcinoma cells with the gene encoding interleukin (IL)-12. This significantly upregulated CB-2R. Such cells were then treated with JWH133, a synthetic CB2 agonist that induced significant apoptosis. Activation of CB-1R in lung cancer cell lines inhibited the MAPL enzyme, reducing cancer invasion and metastasis. 31 Cannabidiol reduced the invasiveness of lung cancer cell lines by upregulating ICAM-1 and inducing TIMP-1. 19
Apart from the therapeutic utility of targeting the cannabinoid receptors, the increased levels of these biomarkers in PTC may add to the utility of fine-needle aspiration cytology. Diagnosis of PTC is based on the nuclear features of the tumor cells, although depending on the cytologic preparation method, immunohistochemical studies can be applied to aspiration cytology biopsies. The PTC immunohistochemical panel currently evaluates Hector Battifora and Mesothelioma (HBME-1), cytokeratin 19 (CK19), and galectin 3 reactivity in tumor cells. 45 The addition of cannabinoid receptor antibodies, especially CB-2R (which is expressed more highly than is CB-1R), to the PTC immunohistochemical panel can increase the diagnostic accuracy of PTC. Given the very low expression rate of CB-1R and CB-2R in benign conditions and the absence in normal follicular epithelium, the positive reactivity of either cannabinoid receptor markers combined with HBME-1, CK19, and galectin 3 would be highly beneficial in favor of PTC.
Another goal was to explore the prognostic utility of cannabinoid receptor overexpression and whether this was a valuable indicator in advanced cases. Lakiotaki et al. 37 reported a significant relationship between increased CB-2R expression and the presence of lymph node metastasis; a finding that, despite a larger number of cases with positive lymph node involvement, we did not observe in this study. Of all pathological and clinical parameters that yielded statistically significant results—the relationships between increased CB-1R expression and the TSH level, extrathyroidal extension, and capsular invasion (P = 0.004, P = 0.022, and P = 0.001, respectively)—the most useful finding was the relationship between increased CB-1R and CB-2R expression and the ATA risk score (P = 0.003 and P = 0.009). When the immune reactivity was divided into low and high, the association with CB-1R disappeared (P = 0.138) but that with CB-2R did not (P = 0.004). The ATA risk stratification system is used to manage treatment by categorizing patients into low, intermediate, and high-risk groups. 46 Elevated CB-2R expression was observed only in the intermediate-risk group, perhaps reflecting the fact that the high-risk group contained few patients. Thus, elevated CB-2R expression indicates that a patient is at least at intermediate risk. In PTC management the ATA system evaluates certain pathological features such as local and distant metastasis, complete/incomplete resection, invasion of surrounding structures, histological subtype, vascular invasion, and lymph node status. As stated earlier, most of the pathological features of this study and their relation with CB-1R and CB-2R expression were statistically insignificant, unlike the comparison of CB-2R expression and the ATA system that evaluates these features as a whole instead of individually.
In the time since the discovery of CB-1R by Matsuda et al., 8 followed by the characterization of CB-2R by Munro et al., 9 the relationship between cannabinoid agonists/receptors and malignancies have attracted the attention of researchers and continues to be subject to many studies in order to find new treatment strategies or to enhance existing protocols. Despite the abovementioned promising anti-cancer mechanisms through cannabinoid receptor activation in various organ neoplasms, currently there is no available therapy based on either cannabinoid receptor targeting or cannabinoid receptor activation via phytocannabinoid/synthetic cannabinoid agonists. The reason could be that apart from the limiting psychotic effects of cannabinoid agonists through activation of CB-1R in the test candidate's central nervous system, the results of many studies are discrepant on whether cannabinoid receptor activation inhibits or enhances tumor development and progression. Vidlarova et al. 47 found that tumors expressing CB-2R mRNA were associated with significantly longer overall, cancer-specific, and disease-free survival, whereas CB-2R protein expression was associated with a less advanced disease stage and grade. Similar results were observed in hepatocellular carcinomas; increased expression of CB-1R and CB-2R significantly related to disease-free survival and was associated with histologically well-differentiated tumors as well as less portal vein involvement. 48 However, several studies reported contrary results. High-level expression of CB-1R in renal cell carcinoma (RCC) was associated with more severe disease at diagnosis and a poorer outcome. CB-1R knockdown decreased RCC proliferation and invasion and increased apoptosis. 39 Elevated CB-2R expression was associated with human epidermal growth factor receptor 2 (HER2)-positive breast tumors with poor prognosis, thus creating greater risks of local recurrence and distant metastasis. 40 Gustafsson et al. 49 reported that CB-1R expression was associated with poorer outcomes of patients with stage 2 microsatellite-stable colorectal cancer. Another study found that colorectal cancer patients with high-level CB-1R expression experienced poorer outcomes than others. 50 Currently, it is difficult to state generally that cannabinoid receptor activation either inhibits or promotes tumor progression, which could be related to the fact that endocannabinoids might have different functions across various malignancies. Depending on the type of cancer, it may be useful to block the cannabinoid receptors or to activate them by using appropriate cannabinoid agonists.
Conclusion
The CB-1R and CB-2R expression levels in PTCs were significantly higher than in benign tissues, suggesting that receptor targeting might be beneficial. Despite our limited number of cases, the relationship between CB-2R expression and the ATA risk could provide useful prognostic information. Furthermore, the addition of CB-1R and (especially) CB-2R assays to thyroid aspiration cytology could increase the diagnostic accuracy of PTC when combined with immunohistochemical PTC markers.
Supplemental Material
sj-jpg-1-jbm-10.1177_03936155231200285 - Supplemental material for
Supplemental material, sj-jpg-1-jbm-10.1177_03936155231200285 for by Damla Zeynep Doyuran and Ömer Eronat in The International Journal of Biological Markers
Supplemental Material
sj-jpg-2-jbm-10.1177_03936155231200285 - Supplemental material for
Supplemental material, sj-jpg-2-jbm-10.1177_03936155231200285 for by Damla Zeynep Doyuran and Ömer Eronat in The International Journal of Biological Markers
Supplemental Material
sj-jpg-3-jbm-10.1177_03936155231200285 - Supplemental material for
Supplemental material, sj-jpg-3-jbm-10.1177_03936155231200285 for by Damla Zeynep Doyuran and Ömer Eronat in The International Journal of Biological Markers
Supplemental Material
sj-jpg-4-jbm-10.1177_03936155231200285 - Supplemental material for
Supplemental material, sj-jpg-4-jbm-10.1177_03936155231200285 for by Damla Zeynep Doyuran and Ömer Eronat in The International Journal of Biological Markers
Supplemental Material
sj-jpg-5-jbm-10.1177_03936155231200285 - Supplemental material for
Supplemental material, sj-jpg-5-jbm-10.1177_03936155231200285 for by Damla Zeynep Doyuran and Ömer Eronat in The International Journal of Biological Markers
Supplemental Material
sj-jpg-6-jbm-10.1177_03936155231200285 - Supplemental material for
Supplemental material, sj-jpg-6-jbm-10.1177_03936155231200285 for by Damla Zeynep Doyuran and Ömer Eronat in The International Journal of Biological Markers
Supplemental Material
sj-jpg-7-jbm-10.1177_03936155231200285 - Supplemental material for
Supplemental material, sj-jpg-7-jbm-10.1177_03936155231200285 for by Damla Zeynep Doyuran and Ömer Eronat in The International Journal of Biological Markers
Supplemental Material
sj-jpg-8-jbm-10.1177_03936155231200285 - Supplemental material for
Supplemental material, sj-jpg-8-jbm-10.1177_03936155231200285 for by Damla Zeynep Doyuran and Ömer Eronat in The International Journal of Biological Markers
Supplemental Material
sj-jpg-9-jbm-10.1177_03936155231200285 - Supplemental material for
Supplemental material, sj-jpg-9-jbm-10.1177_03936155231200285 for by Damla Zeynep Doyuran and Ömer Eronat in The International Journal of Biological Markers
Supplemental Material
sj-jpg-10-jbm-10.1177_03936155231200285 - Supplemental material for
Supplemental material, sj-jpg-10-jbm-10.1177_03936155231200285 for by Damla Zeynep Doyuran and Ömer Eronat in The International Journal of Biological Markers
Supplemental Material
sj-jpg-11-jbm-10.1177_03936155231200285 - Supplemental material for
Supplemental material, sj-jpg-11-jbm-10.1177_03936155231200285 for by Damla Zeynep Doyuran and Ömer Eronat in The International Journal of Biological Markers
Supplemental Material
sj-jpg-12-jbm-10.1177_03936155231200285 - Supplemental material for
Supplemental material, sj-jpg-12-jbm-10.1177_03936155231200285 for by Damla Zeynep Doyuran and Ömer Eronat in The International Journal of Biological Markers
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by the Scientific Investigation Project (BAP) Department of Gaziantep University, Turkey.
Supplemental material
Supplemental material for this article is available online.
Ethics approval
Ethical approval of this study was obtained by the Ethics Committee of Gaziantep University Faculty of Medicine (Number of approval decision: 2021/305).
Informed consent
All patients were informed about the study and signed the informed patient consent form.
Authors’ contribution
D. Doyuran and O. Eronat were involved in patient selection, re-evaluation of all cases, pathological data recording, CB-1R and CB-2R immune reaction interpretation, and scoring. O. Eronat retrieved clinical data. All authors contributed equally to manuscript drafting, writing, and statistics.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
