Abstract
Background
Endometrial cancer is currently the prevalent malignant cancer worldwide. Diagnostic efficiency of tumor markers is limited, and coagulation function indicators in endometrial cancer are less concerned.
Methods
This study attempted to evaluate the effects of coagulation function indicators and tumor markers on the clinical diagnosis and clinicopathological characteristics of patients with endometrial cancer. The retrospective analysis compared the differences in coagulation function indicators and tumor markers among 175 patients with endometrial cancer and 170 healthy women from January 2020 to October 2022.
Results
Compared to the healthy control, the levels of D-dimer, fibrinogen, human epididymis protein 4 (HE4), carbohydrate antigen 125 (CA125), CA153, and CA199 in patients with endometrial cancer were significantly higher (P < 0.05). Univariate and multivariate regression analyses revealed that abnormal levels of D-dimer, fibrinogen, HE4, CA125, CA153, and CA199 were related risk factors affecting the incidence of endometrial cancer. Receiver operating characteristic curve analysis exhibited that the area under the curve (0.931) and accuracy (85.2%) of combined diagnosis of coagulation function indicators (D-dimer, fibrinogen) and tumor markers (HE4, CA125, CA153, CA199) were the highest, and its sensitivity (82.3%) and specificity (88.2%) were higher than any single or combined indicators of four tumor markers. Moreover, relative expression levels of the combined indicators were significantly different among clinicopathological characteristics that had the highest predictive value in the FIGO stage (P < 0.001).
Conclusions
D-dimer and fibrinogen represent potential diagnostic factors for endometrial cancer. The combination of coagulation function indicators and tumor markers exhibited high diagnostic value in endometrial cancer, as well as predictive value for clinicopathological characteristics.
Keywords
Introduction
Endometrial cancer is the most common epithelial malignant tumor in the female reproductive system and its incidence is increasing by nearly 1–2% annually worldwide, which seriously threatens and damages women's life and health.1,2 Early-stage endometrial cancer is so insidious and asymptomatic with difficult diagnosis and poor prognosis that the mortality rate and cost are high.3,4 Therefore, clear, objective, and effective biomarkers are essential to facilitate early diagnosis and treatment of endometrial cancer, while also being utilized for preoperative evaluation to provide reference for the formulation of postoperative adjuvant therapy and prognostic judgment of patients.
Currently, the most commonly used methods for early diagnosis of endometrial cancer include hysteroscopy and diagnostic curettage, while hysteroscopy increases the probability of cancerous metastasis. As the gold standard for the pathological diagnosis of endometrial cancer, diagnostic curettage alters the thickness of the endometrium, leading to serious complications such as severe pain, discomfort, and uterine perforation. Imaging techniques, such as ultrasound and magnetic resonance imaging, have limitations in their ability to accurately diagnose and stage endometrial cancer. 5 Emerging molecular diagnosis is devoted to the accurate evaluation despite limitations in the regional economy and laboratory technology. Currently, there is no reliable, accurate, economical, and non-invasive diagnostic method for endometrial cancer. The detection of serum tumor markers offers advantages of non-invasiveness, stability, and repeatability in disease diagnosis. Human epididymal epithelial secretory protein 4 (HE4), a newfound tumor marker, is primarily expressed in epithelial cells of the reproductive system but less expressed in normal endometrium. 6 There are few recognized sensitive and specific markers currently. The diagnostic value of various potential biomarkers for endometrial cancer, such as HE4, carbohydrate antigen 125 (CA125), and CA153 has been evaluated and has not yet achieved the ideal diagnostic capacity. Specifically, the missed diagnosis rate of endometrial cancer is higher when using a single indicator; however, the combined diagnosis of multiple indicators can improve diagnostic efficiency. 7 Therefore, finding suitable early noninvasive serological diagnostic indicators is of great practical value.
Malignant tumors are closely related to thrombosis. 8 Extensive studies revealed that the first-onset indication of malignant tumors is abnormal coagulation function. Over 90% of tumor patients exhibit abnormal coagulation, and the hypercoagulable state forces further tumor development.9,10 It addresses, for instance, fibrinogen (FIB), which has proven to be closely associated with tumor metastasis and is a risk factor for poor prognosis. 11 As one of the major hydrolysates of fibrin, D-dimer (D2) inevitably participates in the coagulation and fibrinolysis system and signals activation of the coagulation system in various cancers, especially in advanced stages.12,13 Extensive evidence consistently links abnormal coagulation function, specifically FIB and D2, to the occurrence, development, and prognosis of cancers.14–17 The changes in coagulation function indicators in endometrial cancer are concerned and investigated in various research. Regrettably, there is a scarcity of reported research on the combined detection of coagulation function indicators and tumor markers in the diagnosis of endometrial cancer and its clinicopathological characteristics.
The purpose of our present study was to retrospectively analyze the levels of coagulation function indicators and tumor markers in patients with endometrial cancer to develop a convenient and accurate diagnostic workup method. This involved evaluating their differences and diagnostic efficacy between patients with endometrial cancer and healthy individuals, and intended to explore an optimal combination for the diagnosis of endometrial cancer. Simultaneously, the correlation between these indicators and the clinicopathological characteristics of endometrial cancer was analyzed. Furthermore, we aimed to evaluate the predictive value to provide a reference for early diagnosis, efficacy observation, and prognosis evaluation of endometrial cancer.
Materials and methods
Patients
A total of 175 patients who were hospitalized at Zhujiang Hospital of Southern Medical University from January 2020 to October 2022 and diagnosed with endometrial cancer by postoperative pathology were selected for retrospective analysis. Inclusion criteria: (a) those pathologically confirmed endometrial cancer by diagnostic curettage or surgery; and (b) those with complete clinical data. Exclusion criteria: (a) those with severe heart, liver, kidney, and other important organ dysfunction diseases; or (b) those with other malignant tumors or diseases affecting the level of serum tumor markers; or (c) those with severe thrombotic diseases, acute phase of infectious diseases; or (d) those with medications affecting coagulation function. Meanwhile, 170 women were selected from the physical examination as healthy controls. Both groups showed no statistically significant differences in age, body mass index (BMI), menopause, or other patient population characteristics between both groups (P > 0.05), indicating comparability, as shown in Table S1. The study protocol strictly adhered to the principles of the Declaration of Helsinki and was approved by the Ethics Committee of Zhujiang Hospital of Southern Medical University. All enrolled patients provided informed consent while their identities and medical records remained confidential, ensuring that research data are accessible solely to researchers.
Data collection
The clinicopathological data of the participants were collected by reviewing the patients’ medical records and pathological reports, including age, BMI, menopause, myometrial invasion, cervical involvement, pathologic grade, and the International Federation of Gynecology and Obstetrics (FIGO) stage, in accordance with the FIGO and the World Health Organization. Besides, the coagulation function indicators including thrombin time (TT), prothrombin time (PT), activated partial thromboplastin time (APTT), FIB, D2, and platelet (PLT), and serological tumor markers including HE4, carcinoembryonic antigen (CEA), CA125, CA153, CA199, squamous cell carcinoma-associated antigen (SCCA), and alpha-fetoprotein (AFP) were detected prior to surgery or medication.
Hematological detection
Hematological indicators were rigorously tested by professional technicians in our hospital's laboratory following the instructions and operation manual for instruments and reagents. The instruments and reagents utilized mainly consisted of the automatic blood and body fluid analyzer and its supporting reagents (XN-2000, Sysmex, Lincolnshire, IL, USA); the automatic coagulation analyzer and its supporting reagents (Sysmex, CS5100); and the automatic chemiluminescence immunoassay system and its supporting reagents (Tesmi, F3999). Assessment criteria for normal values in each indicator were: TT 14–21 s; PT 9.8–12.1 s; APTT 25.0–31.3 s; D2 < 0.55 mg/L; FIB 1.80–3.50 g/L; PLT (125–350) × 109/L; HE4≤70 in premenopause, ≤140 pmol/L in postmenopause; CEA≤5.0 μg/L; CA125≤35 kU/L; CA153≤28.5 kU/L; CA199≤34 kU/L; SCCA≤2.7 μg/L; and AFP≤7.0 μg/L. A result higher than the critical value was considered to be positive.
Study design
The differences in serum levels of coagulation function indicators and tumor markers between patients with endometrial cancer and healthy controls were compared. The correlation between coagulation function indicators and tumor markers in endometrial cancer was analyzed using the Spearman correlation analysis. Related factors in endometrial incidence were carried out through univariate analysis and multivariate logistic regression analysis. Furthermore, the clinical diagnostic value of single or combined detection of coagulation function indicators and tumor markers in endometrial cancer was evaluated. The levels of these two indicators and the clinicopathological characteristics of patients with endometrial cancer were analyzed. Ultimately, the correlation between the combined indicators and clinicopathological characteristics of patients with endometrial cancer was analyzed to evaluate the predictive values in relation to the clinicopathological characteristics of patients with endometrial cancer.
Statistical analysis
Statistical processing was performed using the Statistical Package for the Social Sciences (SPSS), Version 23.0 (Chicago, IL, USA) SPSS23.0 software. The Kolmogorov–Smirnov test was used to ascertain whether the data were normal distribution. For data with non-normal distribution, descriptive statistics were represented by the median (quartile interval) (M (Q)); the Mann–Whitney U test was adopted for the evaluation of two-group comparisons; and the Kruskal–Wallis test was used for multiple-group comparisons. For data with normal distribution, the mean value and standard deviation were used. The count data were displayed as n (%), and were evaluated using x2 test. The correlation between coagulation function indicators and tumor markers in endometrial cancer patients was determined using the Spearman rank correlation coefficient. Univariate and multivariate logistic regression analyses were conducted on the related risk factors affecting the occurrence of endometrial cancer to construct an optimally combined diagnosis. The diagnostic efficacy was evaluated by drawing the receiver operating characteristic (ROC) curve and calculating the area under the curve (AUC). P < 0.05 was considered statistically significant.
Results
Comparisons of coagulation function indicators and tumor markers between patients with endometrial cancer and healthy controls
A total of 345 women were enrolled in the study (175 patients with pathologically proven endometrial cancer and 170 healthy controls). Compared with the healthy control group, the overall levels of coagulation function indicators and tumor markers in the endometrial cancer group demonstrated an increasing trend, among which the levels of D2, FIB, HE4, CA125, CA153, and CA199 were decidedly elevated (P < 0.05) (Table 1). These results show that these differential indicators may be helpful and beneficial in assisting the diagnosis of endometrial cancer.
Comparisons of coagulation function indicators and tumor markers between patients with endometrial cancer and healthy controls (M(Q)).
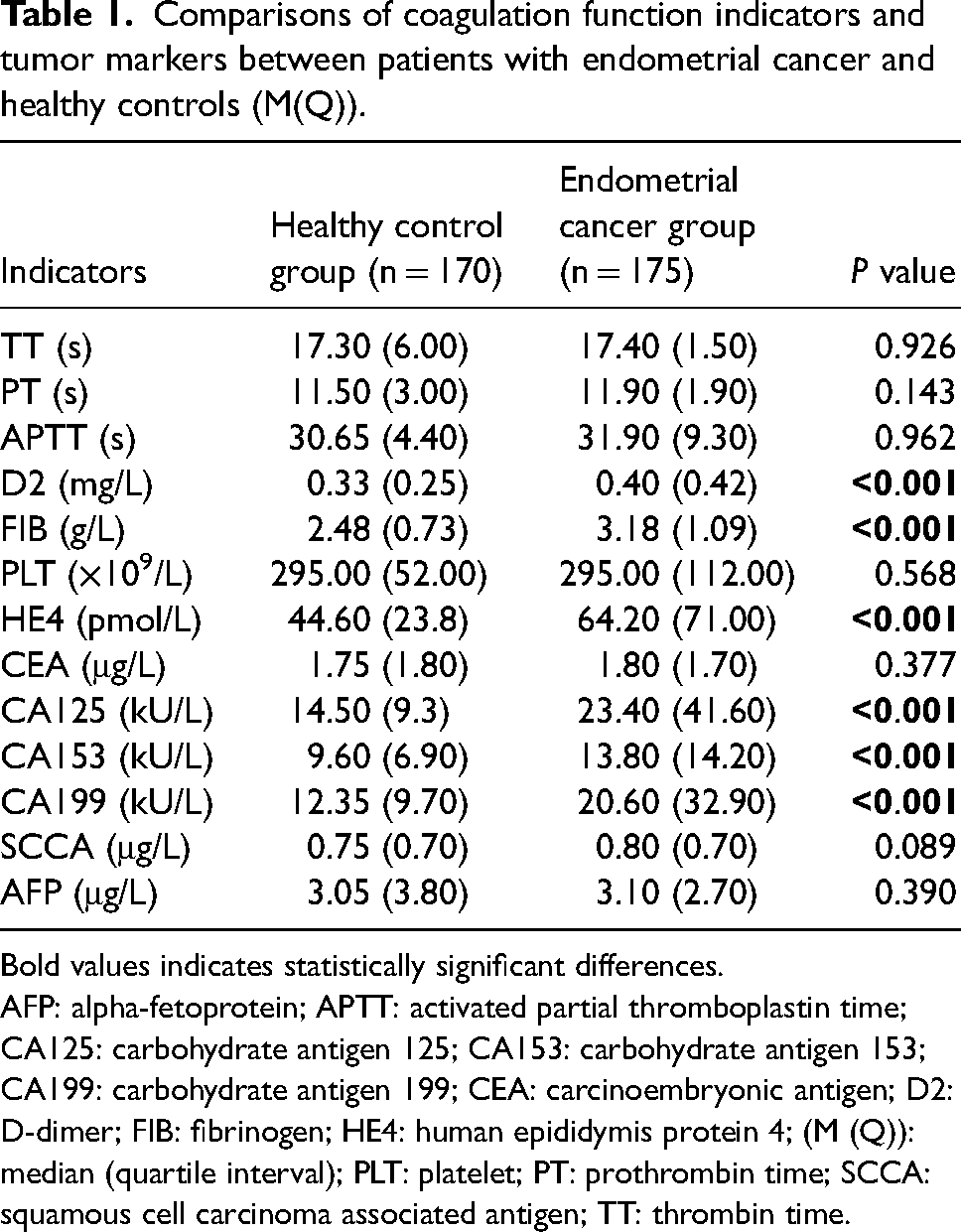
Bold values indicates statistically significant differences.
AFP: alpha-fetoprotein; APTT: activated partial thromboplastin time; CA125: carbohydrate antigen 125; CA153: carbohydrate antigen 153; CA199: carbohydrate antigen 199; CEA: carcinoembryonic antigen; D2: D-dimer; FIB: fibrinogen; HE4: human epididymis protein 4; (M (Q)): median (quartile interval); PLT: platelet; PT: prothrombin time; SCCA: squamous cell carcinoma associated antigen; TT: thrombin time.
Correlations between coagulation function indicators and tumor markers in patients with endometrial cancer
Correlations between coagulation function indicators and tumor markers in patients with endometrial cancer were analyzed using the Spearman correlation analysis. The results showed that TT was positively correlated with CA199 in the serum level of EC patients (r = 0.163; P = 0.031). PT was positively correlated with CEA, CA125, and CA153, respectively (r = 0.150, 0.178, 0.211; P = 0.047, 0.019, 0.005), and negatively correlated with SCCA (r = −0.238; P = 0.002). APTT was negatively correlated with HE4 and SCCA, respectively (r = -0.177, −0.212; P = 0.019, 0.005. D2 was positively correlated with HE4, CA125, and CA199, respectively (r = 0.173, 0.230, 0.255; P = 0.022, 0.002, 0.001). FIB was positively correlated with CA125 and CA153, respectively (r = 0.169,0.197). No significant correlations were observed between the other indicators of these two categories (P > 0.05). Moreover, there was an obvious correlation among tumor markers, suggesting that multiple tumor markers could act synergistically for disease diagnosis (Figure 1).
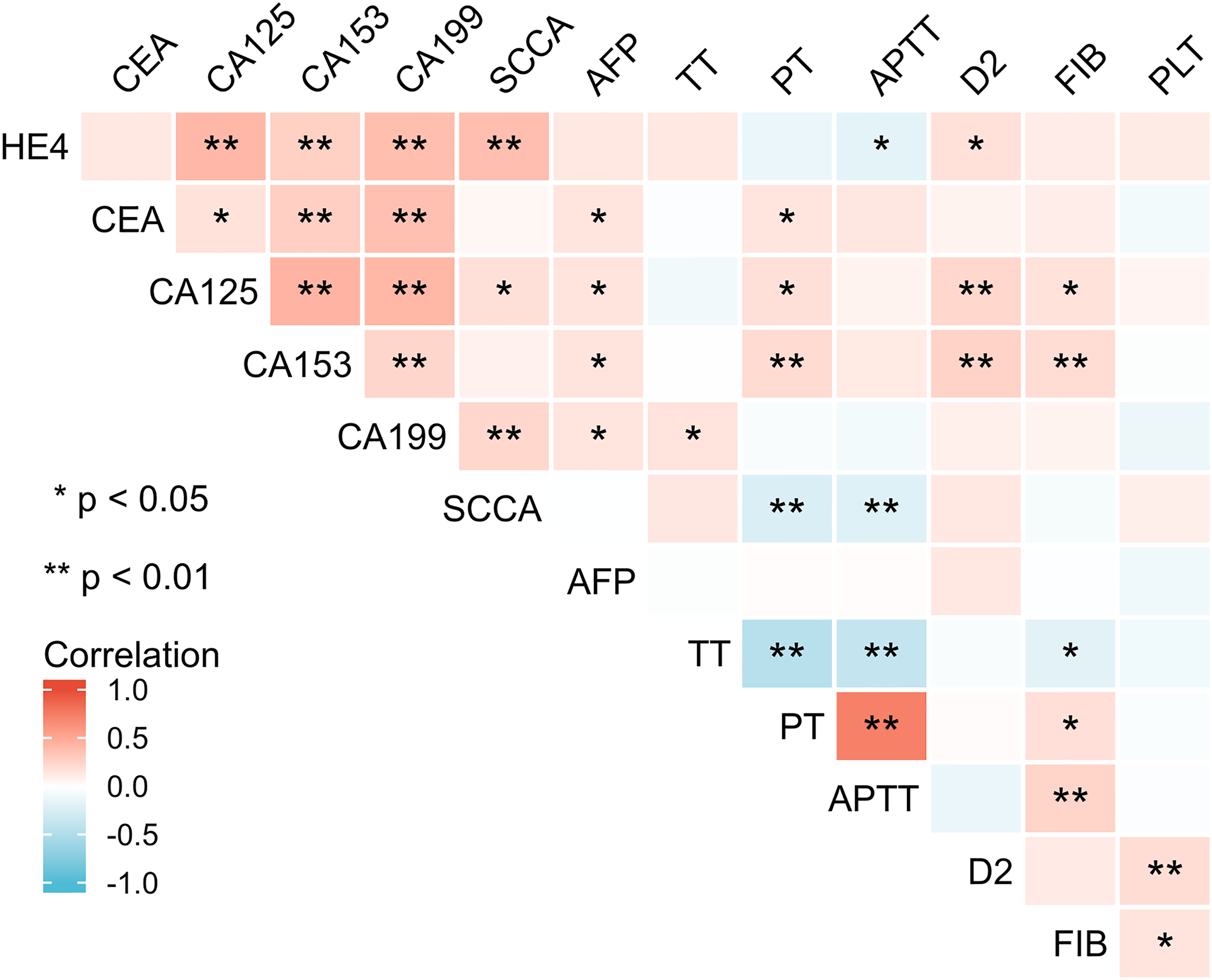
Correlation heat map analysis of coagulation function indicators and tumor markers in patients with endometrial cancer.
Diagnostic efficacy of single or combined diagnosis of coagulation function indicators and tumor markers for endometrial cancer
Subsequently, we evaluated the clinical diagnostic value of single indicators or combinations of coagulation function and tumor markers in endometrial cancer. It was distinctly revealed through univariate analysis that abnormally increasing levels of D2, FIB, HE4, CA125, CA153, and CA199 were related risk factors affecting the incidence of endometrial cancer (Table S2). The aforementioned high-risk factors with statistically significant differences, namely D2, FIB, and tumor markers (HE4, CA125, CA153, and CA199), were included in multivariate logistic regression analysis to explore the related risk factors of endometrial cancer. Based on the results, HE4 (OR = 75.421; 95% CI 10.292-5, 52.716; P < 0.001), CA125 (OR = 1.071; 95% CI 1.028, 1.116; P = 0.001), CA153 (OR = 1.067; 95% CI 1.002, 1.136; P = 0.043), CA199 (OR = 1.046; 95% CI 1.008, 1.085; P = 0.018), D2 (OR = 8.173; 95% CI 1.813, 36.836; P = 0.006), and FIB (OR = 8.156; 95% CI 4.287, 15.516; P < 0.001) were evidently associated with the incidence of endometrial cancer (Table S3). Thereafter, based on the aforementioned result, we constructed combinations of several valuable indicators to explore the diagnostic value of coagulation function indicators and tumor markers for endometrial cancer. Using healthy subjects as the reference population, the ROC curve was drawn to access the single and combined diagnostic efficacy of HE4, CA125, CA153, CA199, D2, and FIB for endometrial cancer. We found that the AUC values for these curves in single indicators ranged from 0.658 to 0.805 and the corresponding accuracy rates varied from 63.2% to 73.9%. The AUC (0.852), sensitivity (77.7%), and accuracy (82.3%) of the combined detection of four tumor markers (HE4, CA125, CA153, CA199) were higher than any single detection, which suggested that the combination of specific tumor markers facilitated the diagnosis of endometrial cancer. The AUC (0.931) and accuracy (85.2%) of combined detection with four tumor markers (HE4, CA125, CA153, CA199) and coagulation function indicators (D2, FIB) were obviously the highest, and its sensitivity (82.3%) and specificity (88.2%) were higher than those of the four tumor markers (87.6%, 82.3%). Therefore, combined associated tumor markers had unique advantages in the diagnosis of endometrial cancer (P < 0.001). (Table S4 and Figure 2).
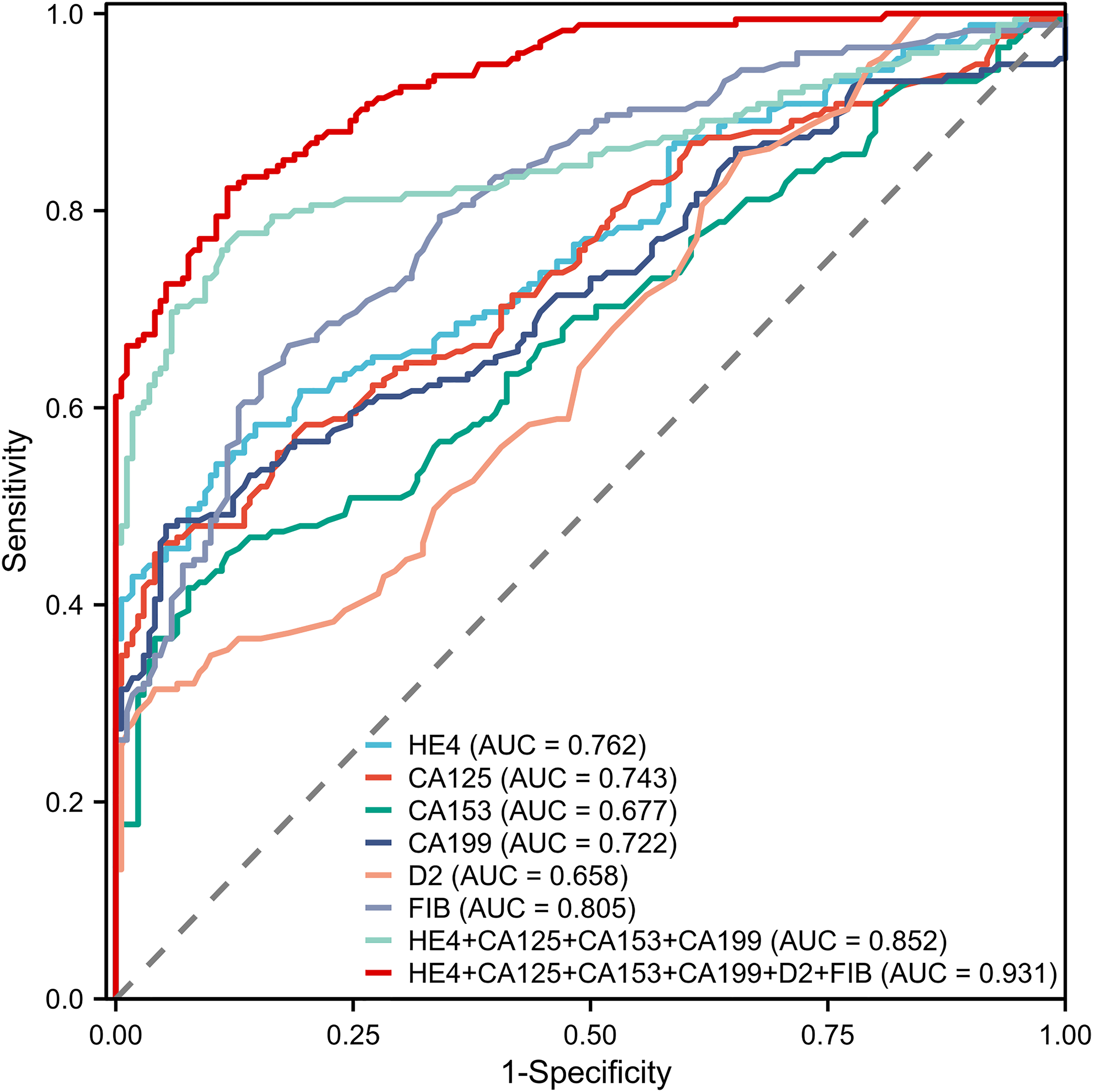
Receiver operating characteristic curves of single or combined diagnosis of coagulation function indicators and tumor markers for endometrial cancer.
Comparison of coagulation function indicators and tumor markers in clinicopathologic characteristics of endometrial cancer
To identify the correlations between coagulation function indicators or tumor markers and clinicopathologic characteristics in endometrial cancer. Serum FIB, HE4, CA125, and CA199 levels were observably higher in myometrial invasion depth (≥1/2) than in myometrial invasion depth (<1/2). Otherwise, the expression levels of HE4 and CA125 in cervical involvement were significantly higher than in cervical noninvolvement (P < 0.05). In pathologic grades, G1/G2 were highly and moderately differentiated, while G3 was poorly differentiated and highly malignant. APTT, HE4, CEA, and CA125 exhibited significantly prolonged or increased levels in G3 and G1/G2 (P < 0.05). In the FIGO stage, D2, FIB, HE4, CA125, CA153, and CA199 levels in patients with advanced-stage (III/IV) endometrial cancer were clearly higher than those with early-stage (I/II) (P < 0.05). No statistically significant variations were observed in the expression levels of other coagulation function indicators and tumor markers not mentioned in the groups of myometrial invasion, cervical involvement, pathologic grade, and FIGO stage (P > 0.05). (Table S5)
Correlation between the combined diagnosis and clinicopathological characteristics of endometrial cancer
To determine the clinical correlation between the above-combined diagnosis of coagulation function indicators and tumor markers and clinical pathological characteristics such as myometrial invasion, cervical involvement, pathologic grade, and FIGO stage. There were significant differences in the relative expression levels of combined diagnosis based on Logistic regression analysis in terms of myometrial invasion (P < 0.001), cervical involvement (P = 0.015), pathologic grade (P = 0.011), and FIGO stage (P < 0.001) (Table 2). Therefore, the predictive value of the combined diagnosis of coagulation function indicators and tumor markers in different clinicopathological characteristics of patients with endometrial cancer was analyzed. ROC curve analysis revealed the following results: the AUC of the combined diagnostic model in the myometrial invasion, cervical involvement, pathologic grade, and FIGO stage of endometrial cancer was 0.697, 0.635, 0.668, and 0.762, respectively; the sensitivity was 58.5%, 55.9%, 68.2%, and 73.9%, respectively; the specificity was 78.4%, 77.3%, 69.3%, and 76.3%, respectively; and the accuracy was 73.7%. 73.1%, 69.1%, and 76.0%, respectively. These results suggested that the auxiliary predictive value of the combined diagnostic model was highest in the FIGO stage (Table 3).
Correlation between the combined diagnosis and clinicopathological characteristics of endometrial cancer.

Bold values indicates statistically significant differences.
BMI: body mass index; FIGO: Federation of Gynecology and Obstetrics.
The relative expression levels of combined diagnosis were based on Logistic regression analysis.
Predictive values of combined diagnosis in clinicopathological characteristics of endometrial cancer.
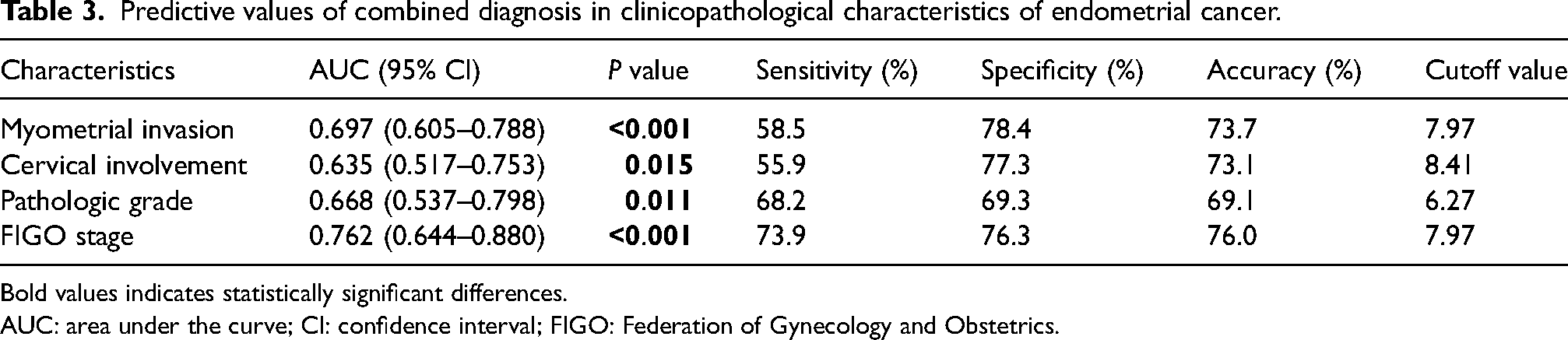
Bold values indicates statistically significant differences.
AUC: area under the curve; CI: confidence interval; FIGO: Federation of Gynecology and Obstetrics.
Discussion
According to the statistics of the GLOBOCAN database, the incidence of endometrial cancer ranks sixth among female tumors worldwide. 18 Early-onset endometrial cancer has no specific symptoms, with the most common manifestations being vaginal bleeding, vaginal discharge, abdominal pain, etc., which are easily confused with other gynecological diseases; therefore, the best treatment time is delayed, seriously threatening women's health and safety and affecting their quality of life. Early diagnosis forms the basis of endometrial cancer treatment. At present, diagnostic curettage pathological examination is considered the gold standard for endometrial cancer. Diagnostic curettage is an invasive examination method, which has high requirements for the operator and the operating environment, and there are still missing lesions due to the location of the material. Imaging examinations offer clear and visual advantages in the diagnosis of endometrial cancer, although their usefulness is limited to tumor staging and the early detection of small lesions. Serological detection offers several advantages, including minimal trauma, convenient sampling, good repeatability, and quantitative diagnosis. It is well-known that tumor markers can be widely used in the early diagnostic workup of malignant tumors. Serum tumor markers included HE4, CA125, CA153, CA199, etc., which have the utility as potential diagnosis markers for patients with endometrial cancer. Recently, it was found that malignant tumors are associated with thrombosis. 19 Patients with malignant tumors exhibited abnormal coagulation function, even in the hypercoagulable state that promotes further tumor development. Consequently, coagulation function indicators received significant privileged attention in cancer diagnosis. This article focused on the levels of coagulation function indicators and tumor markers in patients with endometrial cancer to discover the diagnostic value of these indicators, as well as their relationships with different clinicopathological characteristics.
In our study, compared with the healthy controls, the overall levels of coagulation function indicators and tumor markers in endometrial cancer stayed at an increasing trend, among which the levels of D2, FIB, HE4, CA125, CA153, and CA199 were significantly increased (P < 0.05). On the one hand, it suggested that there were changes in coagulation function in patients with endometrial cancer. We detected the increase of D2 and FIB levels, suggesting abnormal coagulation function after tumor occurrence, and the coagulation and fibrinolysis system was overactivated in patients with endometrial cancer, leading to a hypercoagulable state of the blood. Coagulation or fibrinolysis activation was frequently observed in numerous tumors. 20 Coagulation is the conversion of liquid blood into gel, ultimately leading to the formation of a clot. Fibrinolysis prevents thrombosis by destroying fibrin. D2 consists of two cross-linked D fragments of fibrin, which is the product of fibrin degradation. 21 Recently, elevated D2 levels have been identified to be favorable in the diagnosis and prognosis of many malignancies; for example, lung,22,23 pancreatic, 24 breast, 25 ovarian, 26 endometrial.27–29 FIB serves as a precursor of fibrin, and during the final stages of clotting, soluble fibrinogen converts into insoluble fibrin, leading to blood clot formation. It is known to be involved in hemostasis, acting as a key component within the clotting cascade. 30 Additionally, FIB functions as a significant inflammatory stress protein in the acute phase, with its synthesis being up-regulated under the stimulation of inflammatory mediators. FIB constitutes the predominant component of the tumor matrix, forming a protective barrier around malignant tumor cells, thereby enabling them to escape their evasion from endogenous defense mechanisms. Thus, its elevation may indicate a suggestive role in tumorigenesis. Conversely, the elevations of HE4, CA125, CA153, and CA199 in patients with endometrial cancer indicated their potential diagnostic specificity. Tumor markers could be widely used in early diagnostic workup of malignant tumors; especially, the combined detection of tumor markers could effectively improve the specificity and sensitivity of early cancer diagnosis. HE4, a novel serum tumor marker, is low in human healthy tissues and benign tumors, but is abnormally elevated in both tissues and serum of patients with endometrial cancer. HE4 exhibits high diagnostic specificity for endometrial cancer. 31 CA125 is a high polymer carbohydrate antigen commonly expressed in malignant tumor cells, and accumulating studies have shown that CA125 can be effectively screened for endometrial cancer by blood sampling tests. 32 CA153 is a glycogen protein primarily synthesized on the surface tissue of the breast, which is mainly used in the clinical diagnosis of breast cancer. It is abnormally expressed in a variety of gynecological benign and malignant tumors such as endometrial cancer. 33 CA199 is a high polymer mucin glycoprotein present in malignant tumor cells. 34 Also, we analyzed the correlation between coagulation function indicators and tumor markers in patients with endometrial cancer and found that D2 was positively correlated with HE4, CA125, and CA199. Similarly, FIB was positively correlated with CA125 and CA153, indicating that the higher the level of these tumor markers, the more hypercoagulable the patient was. Notably, D2 and FIB demonstrate essential clinical significance in the auxiliary diagnosis of endometrial cancer. In addition, we analyzed the risk factors related to coagulation function indicators and tumor markers in the incidence of endometrial cancer, encompassing D2, FIB, HE4, CA125, CA153, and CA199, to evaluate the efficacy of their single or combined diagnosis of endometrial cancer, and to identify the preferred combined diagnostic indicators. Through ROC curve analysis, compared with a single tumor marker, the combined detection of four tumor markers (HE4, CA125, CA153, CA199) had improved clinical diagnostic efficacy for endometrial cancer; also, the AUC, accuracy, sensitivity, and specificity of the combined detection with coagulation function indicators (D2, FIB) and four tumor markers were higher than those of the combined detection of the four tumor markers. Therefore, the usefulness of combined indicators in diagnosing endometrial cancer deserves positive recognition. In our study, it was confirmed that the detection of a single tumor marker had certain limitations. While it was highly expressed in gynecological tumors, its sensitivity and specificity were inadequate, and the combined detection of multiple tumor markers was needed to improve the detection accuracy of endometrial cancer. Moreover, coagulation function indicators are closely linked to the occurrence and development of endometrial cancer, especially D2 and FIB, which cooperate with tumor markers to improve the diagnostic accuracy of endometrial cancer.
According to the National Comprehensive Cancer Network guidelines for the diagnosis and treatment of endometrial cancer, clinicopathologic characteristics including age, obesity, menopause, muscular infiltration, cervical involvement, pathologic grade, and FIGO stage serve as important prognostic factors. In this study, the correlation between coagulation function indicators and tumor marker levels and the clinicopathological characteristics of patients with endometrial cancer was analyzed. The results showed that FIB, HE4, CA125, and CA199 levels correlated with the depth of myometrium infiltration; the deeper the myometrium infiltration, the higher the serum level. The levels of HE4 and CA125 exhibited significant elevation in patients with endometrial cancer with cervical involvement. In terms of pathologic grade, APTT, HE4, CEA, and CA125 showed noticeable prolongation or increase in G3 compared with G1/G2. Interestingly, the incidence of the advanced stage (III/IV) was highly correlated with D2, FIB, HE4, CA125, CA153, and CA199, which facilitated the establishment of risk stratification for recurrence through preoperative staging, determination of both the most suitable surgical treatment and the postoperative management. 35 Furthermore, the relative expression level of combined diagnosis based on coagulation function indicators and tumor markers was significantly correlated with the clinical characteristics of endometrial cancer, such as muscular infiltration, cervical involvement, pathologic grade, and FIGO stage—with the FIGO stage demonstrating the highest predictive value. The coagulation function indicators combined with the tumor markers can provide a basis for evaluating disease severity and prognosis.
This study has certain limitations, as it is a retrospective study designed with a limited sample size and conducted at a single medical center. Therefore, uncontrollable selection and monitoring biases will have inevitably occurred in our analysis, necessitating further verification through large-scale research with a large sample size collaboration among multiple medical centers.
Conclusion
Among the coagulation function indicators, D2 and FIB have positive diagnostic values for endometrial cancer and can serve as auxiliary markers for diagnosis. The combined diagnostic efficacy of these coagulation function indicators and tumor markers is higher than that of single detection, providing a convenient diagnostic workup method and guidance for endometrial cancer. Moreover, there is an intimate correlation between the coagulation function indicators and tumor markers in patients with endometrial cancer, and those levels have different relationships with clinicopathological characteristics, which is conducive to clinicians judging the patient's status with endometrial cancer in preoperative evaluation.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155231196253 - Supplemental material for Effects of coagulation function indicators and tumor markers on diagnosis and clinicopathological characteristics of endometrial cancer
Supplemental material, sj-docx-1-jbm-10.1177_03936155231196253 for Effects of coagulation function indicators and tumor markers on diagnosis and clinicopathological characteristics of endometrial cancer by Huayan Li, Huifeng Liao, Bilin Jing and Yifeng Wang in The International Journal of Biological Markers
Footnotes
Author contributions
HYL and HFL designed, analyzed the data, and wrote the manuscript. HYL and BLJ collected clinical data and generated the tables. YFW conceptualized and revised the manuscript. All authors contributed to the article and approved the submitted version.
Data availability
Data will be made available on request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Zhujiang Hospital of Southern Medical University. Informed consent was provided to all enrolled patients.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
