Abstract
Background
The acid glycoprotein 1 (AGP1) is downregulated in lung cancer. However, the performance of AGP1 in distinguishing benign from malignant lung lesions is still unknown.
Methods
The expression of AGP1 in benign diseases and lung cancer samples was detected by Western blot. The receiver operating characteristic curves, bivariate correlation, and multivariate analysis was analyzed by SPSS software.
Results
AGP1 expression levels were significantly downregulated in lung cancer and correlated with carcinoembryonic antigen (CEA), CA199, and CA724 tumor biomarkers. The diagnostic performance of AGP1 for distinguishing malignant from benign pulmonary lesions was better than the other four clinical biomarkers including CEA, squamous cell carcinoma-associated antigen, neuron-specific enolase, and cytokeratin 19 fragment 21-1, with an area under the curve value of 0.713 at 88.8% sensitivity. Furthermore, the multivariate analysis indicated that the variates of thrombin time and potassium significantly affected the AGP1 levels in lung cancer.
Conclusions
Our study indicates that AGP1 expression is decreased in lung cancer compared to benign samples, which helps distinguish benign and malignant pulmonary lesions.
Introduction
Lung cancer is a type of malignant tumor with high morbidity and mortality worldwide 1 and includes non-small-cell lung cancer (NSCLC) and small cell lung cancer. NSCLC can be classified as lung adenocarcinoma (LUAD), lung squamous cell carcinoma (LUSC), and large cell carcinoma based on histopathology. Early detection can significantly improve the survival rate of lung cancer patients, and the 5-year survival rate can increase above 85% for patients diagnosed with stage IA NSCLC. 2 Therefore, it has important clinical significance for early detection and treatment. In the clinic, the common biomarkers for lung cancer screening are carcinoembryonic antigen (CEA), cytokeratin 19 fragment (CYFRA21-1), squamous cell carcinoma-associated antigen (SCCA), and neuron-specific enolase (NSE), which have low sensitivity and specificity and have limitations for use in early detection of lung cancer, especially in distinguishing benign nodules from malignant tumors in the lung. Approximately 30% of patients with uncertain lung diseases are diagnosed with benign disease by biopsy after surgical resection. 3 Therefore, it is urgent to screen effective methods for the diagnosis of early lung cancer from benign lung lesions.
At present, according to radiological technology, the most common instrumental diagnostic methods for differentiating malignant from benign lung lesions mainly include low-dose computed tomography (CT), positron emission tomography (PET), and magnetic resonance imaging, which help to improve the survival rate. 4 Bianconi et al. 5 reported that deep learning was a viable approach for semiautomated segmentation of pulmonary nodules on CT scans and further found that the four form factors, ACO, AFL, KFL, and MPS, could be evaluated on lung nodules on CT scans and can improve the differential diagnosis between benign and malignant lesions. 6 Dual-energy spectral CT also has high accuracy in the differential diagnosis of benign and malignant pulmonary necrotic lesions. 7 Palumbo et al. 8 showed that shape and texture features from 18F-FDG PET/CT could better discriminate benign from malignant lung nodules based on accurate segmentation.
On the other hand, the development of lung cancer is closely associated with the immune system, especially in the early stage. 9 It was found that immune-related proteins also have the potential to improve the performance of lung cancer screening, 10 for example, complement factor B, 11 tumor-associated autoantibody, 12 and the checkpoint molecule B7-H3, and can be used as promising biomarkers for the diagnosis of lung cancer. 13 As previously described, we found that an immune-related molecule, namely, acid glycoprotein 1 (AGP1), was reduced in sera and tissues of early NSCLC, and the AGP1 expression level also decreased in early-stage tumors with the established mouse model of lung cancer. 14 Furthermore, we also found that AGP1 could distinguish healthy samples from lung cancer patients and be a biomarker for diagnosing early-stage lung cancer. 15
However, the performance of AGP1 in distinguishing benign and malignant lung lesions and its clinical significance is unknown. In this study, we intend to analyze the diagnostic performance of AGP1 in lung cancer and explore the correlation between clinical parameters and AGP1 in lung cancer, which will provide a candidate biomarker for differentiating benign from malignant pulmonary lesions.
Methods
Experimental subjects
A total of 234 patients with pulmonary disease were collected from Suzhou Municipal Hospital (Suzhou, China), 63 of whom had benign pulmonary lesions and 171 had lung cancer. All patients were diagnosed upon CT and histopathological analyses. The lung cancer patients were graded using the eighth edition of the tumor node metastasis (TNM) classification of “The International Association for the Study of Lung Cancer.” The serum from patients was collected at a preoperative stage and in peripheral blood. All data from laboratory tests were collected from the electronic patient record (Table S1).
Western blot analysis
Equal amounts of protein were loaded on 12% SDS-PAGE and transferred to PVDF membranes. The membranes were blocked with 5% BSA in TBS containing 0.1% Tween-20, then incubated with primary antibody against AGP1 (1:2000, Proteintech, Wuhan, China), washed with TBST and incubated with secondary antibody against HRP-IgG (1:10000, Proteintech). After washing with TBST, the immunoreactivity was detected using the ClarifyTM Western ECL Substrate (Bio-rad). AGP1 expression levels were quantified by Image J and analyzed by Prism Graphpad v8.0.
Statistics analysis
The messenger RNA (mRNA) expression level was analyzed by The HUMAN PROTEIN ATLAS database (https://www.proteinatlas.org/ENSG00000229314-ORM1). The correlation between AGP1 expression and tumor stage was investigated by the GEPIA database (http://gepia.cancer-pku.cn/). Receiver operating characteristics (ROC) curves were used to define sensitivity and specificity, and the differences in the area under the curves (AUC) were detected. The correlation of variates was analyzed using bivariate correlation analysis by Spearman test (r) with a two-tail test. In the multivariate analysis, the factors associated with AGP1 were determined by a linear regression model with a backward stepwise. Calculations were performed using IBM SPSS statistics software (v26.0). A P-value <0.05 was considered statistically significant.
Results
AGP1 expression was reduced in lung cancer at the serological level
AGP-1 is mainly synthesized in the liver, and the Human Protein Atlas database showed that the mRNA level of AGP1 was mainly expressed in liver and liver cancer cells, and was expressed at lower levels in lung cancer cell lines (Figure 1(a)). We collected 234 cases to detect the serological expression level of AGP1 in lung cancer. Clinical characteristics of general information with these 234 cases, such as age, sex, and disease types, are listed in Table 1. The mean age was approximately 55 years in all patients. There were 34 female and 29 male patients in the benign group, and 105 female and 66 male patients in the lung cancer group, which was not a significant difference between benign and malignant lung lesion patients. The protein expression level of AGP1 was detected by Western blot in 234 cases (Figure 1(b)), and dot plot analysis is shown in Figure 1(c), which indicated that the expression level of AGP1 protein in the serum of lung cancer patients was decreased compared with that in benign patients.
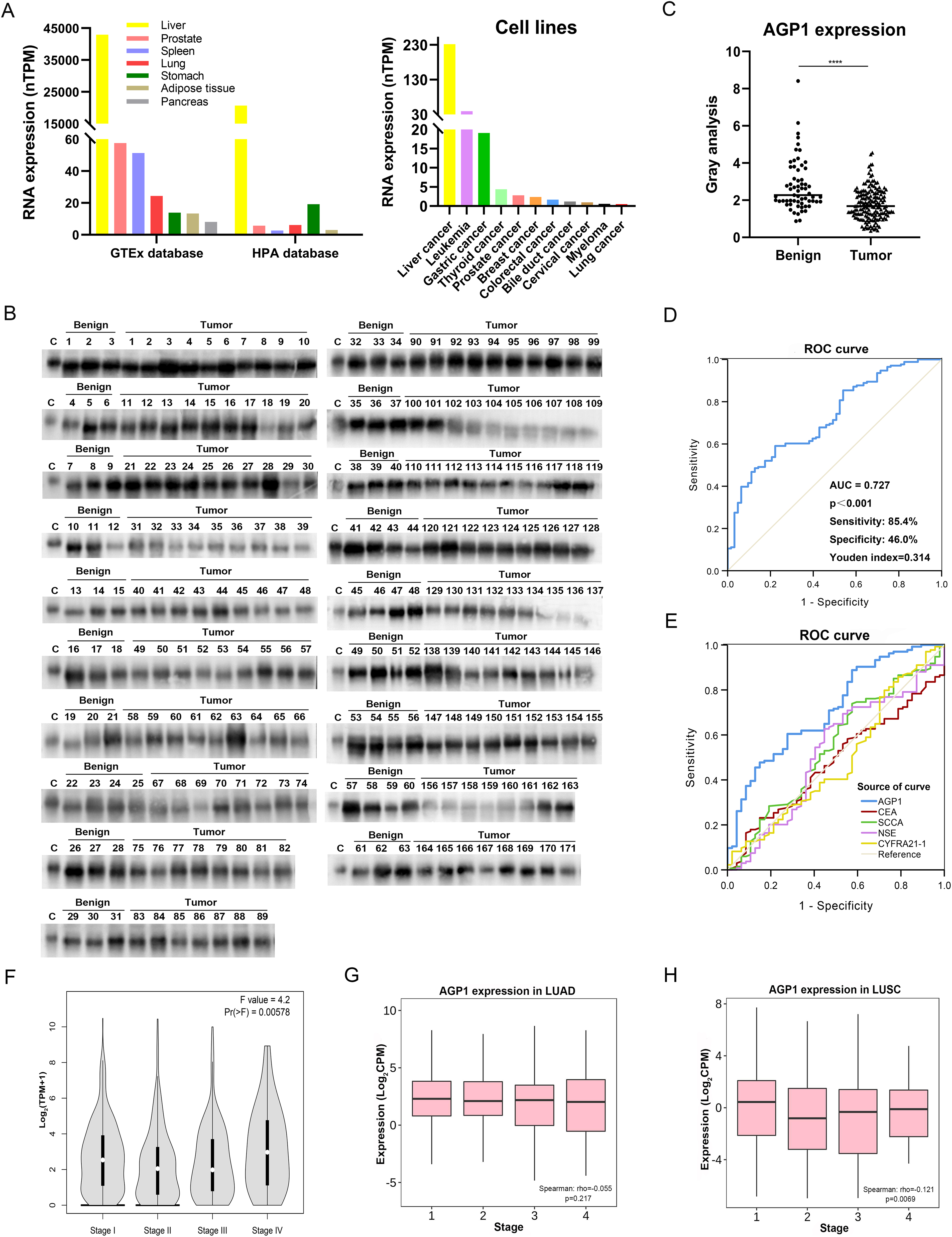
Analysis of AGP1 expression between benign lung lesions and lung cancer patients. (a) The AGP1 mRNA expression in tissues and tumor cell lines. (b) Detection of AGP1 expression by Western blot. (c) The dot plot of AGP1 expression between benign lung lesions and lung cancer patients was analyzed by GraphPad Prism 8.0 software. (****presents a P-value <0.0001.) (d) ROC curve analysis by SPSS 26.0 software. (e) ROC curve analysis by SPSS 26.0 software. (f) Analysis of the correlation between AGP1 expression and tumor stage in lung cancer. (g) The correlation analysis of AGP1 expression and tumor stage in LUAD. (h) The correlation analysis of AGP1 expression and tumor stage in LUSC.
Baseline characteristics in benign lung lesions and lung cancer patients.
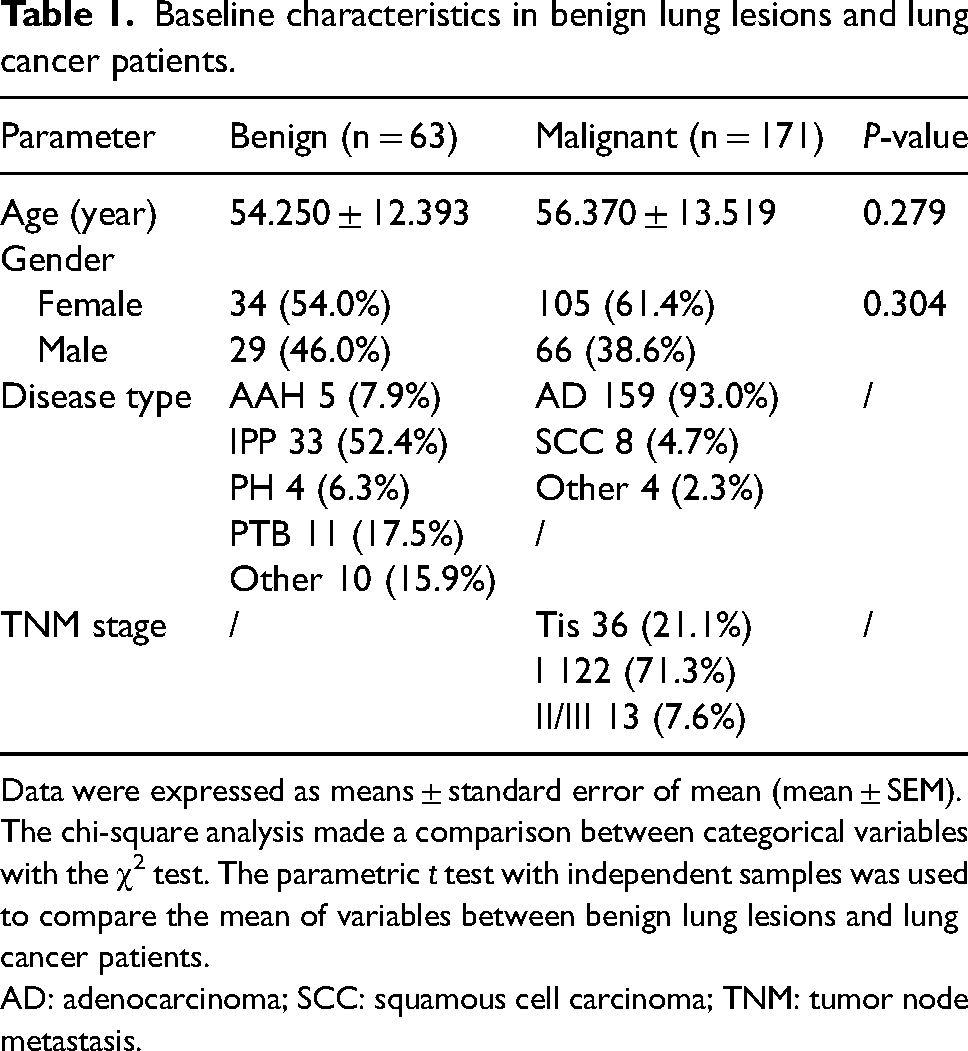
Data were expressed as means ± standard error of mean (mean ± SEM). The chi-square analysis made a comparison between categorical variables with the χ2 test. The parametric t test with independent samples was used to compare the mean of variables between benign lung lesions and lung cancer patients.
AD: adenocarcinoma; SCC: squamous cell carcinoma; TNM: tumor node metastasis.
The clinical significance of AGP1 in lung cancer
A ROC curve was constructed to detect the diagnostic performance of AGP1, which showed that AGP1 could differentiate benign samples from lung cancer with an AUC value of 0.727 at a sensitivity of 85.4% (Figure 1(d)). We also analyzed the correlation between AGP1 expression levels and tumor biomarkers in 234 patients, as shown in Table 2, and found that AGP1 was significantly associated with CEA, carbohydrate antigen 199 (CA199), and carbohydrate antigen 724 (CA724). Furthermore, ROC analysis showed that AGP1 had better diagnostic performance in distinguishing lung cancer from benign lung lesions than four common clinical biomarkers: CEA, SCCA, NSE, and CYFRA21-1 (Table 3). The AUC values were 0.713 with 88.8% sensitivity and P-value less than 0.05 (Figure 1(e)). The GEPIA database indicated that AGP1 expression was associated with tumor stage in lung cancer (Figure 1(f)), and it was not significantly correlated with LUAD (Figure 1(g)) but was correlated with LUSC (Figure 1(h)).
Correlation analysis between AGP1 expression and tumor biomarkers in 234 cases.
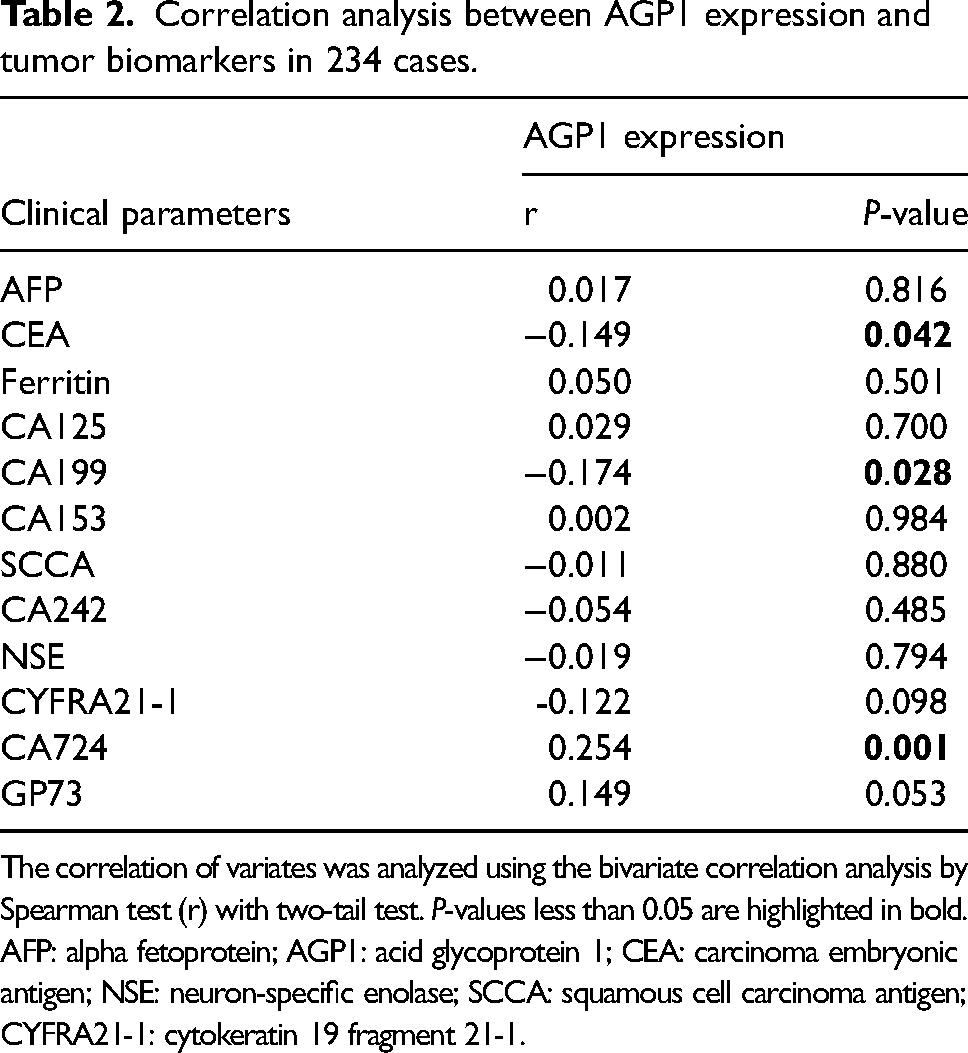
The correlation of variates was analyzed using the bivariate correlation analysis by Spearman test (r) with two-tail test. P-values less than 0.05 are highlighted in bold.
AFP: alpha fetoprotein; AGP1: acid glycoprotein 1; CEA: carcinoma embryonic antigen; NSE: neuron-specific enolase; SCCA: squamous cell carcinoma antigen; CYFRA21-1: cytokeratin 19 fragment 21-1.
Performance of variates in distinguishing lung cancer from benign lung lesions.
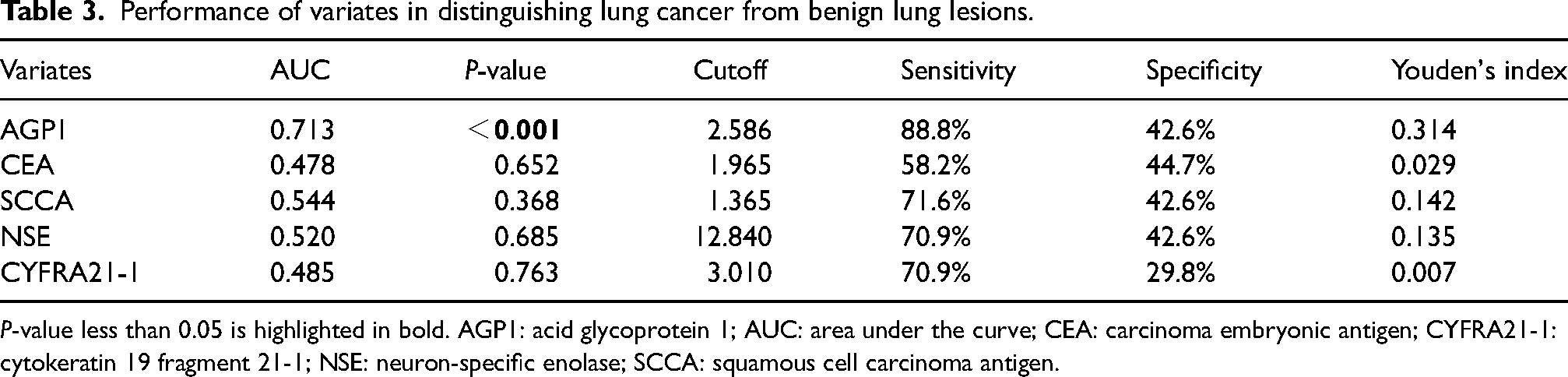
P-value less than 0.05 is highlighted in bold. AGP1: acid glycoprotein 1; AUC: area under the curve; CEA: carcinoma embryonic antigen; CYFRA21-1: cytokeratin 19 fragment 21-1; NSE: neuron-specific enolase; SCCA: squamous cell carcinoma antigen.
Comparison of the AGP1 with clinicopathologic parameters
The correlation between AGP1 expression with clinicopathologic parameters was also analyzed, which indicated that AGP1 expression was significantly associated with thrombin time (TT), D-dimer, potassium (K), and the ratio of albumin (ALB) to globulin (GLB) (Table S2). In addition, the independent factor associated with AGP1 was estimated by multivariate analysis with a linear regression model, which showed that the variates of TT and K were significantly negative correlated with AGP1 expression levels in lung cancer (Table 4), as shown in Figure S1.
Factors associated with AGP1 in lung cancer by multivariate analysis.

The multivariate analysis was determined by a linear regression model with backward stepwise. P-values less than 0.05 are highlighted in bold.
AGP1: acid glycoprotein 1; ALB/GLB: albumin/globulin; B: regression coefficient; CI: confidence interval; K: potassium; TT: thrombin time.
Discussion
AGP1 is a serine protease inhibitor and a major acute-phase protein that plays a role in inflammation and immunity. 16 AGP1 protein can regulate the proliferation of lymphocytes, aggregation of platelets, chemotaxis, peroxidation reaction, and so on. 17 Moreover, different glycoforms of AGP1 affect the activity of platelets and neutrophils in acute inflammation. 18 The abnormal expression of AGP1 was found to be associated with cardiovascular diseases, 19 primary aldosteronism, 20 and tumorigenesis of cancers. 21 Recently, more evidence has demonstrated that AGP1 could be a diagnostic or prognostic biomarker in tumors such as laryngeal 17 and pancreatic cancer. 22 It has been found that AGP1 expression is decreased in the early stage of NSCLC compared to healthy subjects, 15 which could be used as a biomarker for the early diagnosis of NSCLC.
This study first analyzed the AGP1 protein level in the serum of patients with benign and malignant lung lesions. We found that downregulated AGP1 in serum showed good performance in lung cancer, and the AUC value was 0.713 with a better sensitivity of 88.8%, which indicated that AGP1 is more helpful for the clinical diagnosis of lung cancer and yields a higher AUC value with better sensitivity than the other four clinical tumor biomarkers. Therefore, our study shows that AGP1 is an effective diagnostic biomarker for distinguishing benign from malignant pulmonary lesions.
In this study, we found that the number of patients was more female than male patients from Suzhou Municipal Hospital. The incidence rates of lung cancer in females are now comparable to or higher than those in males and are rising alarmingly in many parts of the world. 23 LUAD is the most common subtype of NSCLC and is more common among females than males. 24 The reason is that a sex-specific difference was exhibited in the occurrence of lung cancer, 25 including molecular abnormalities, growth factor receptors, hormonal influences, DNA repair capacity, and differences in female histology.26,27 Females may be more susceptible to the effects of carcinogens in tobacco due to hormonal, genetic, and metabolic differences between the sexes. Compared with males, females had a higher risk of LUSC and LUAD at the same smoking level. 28 In addition, genetic factors such as EGFR mutation were different in female and male lung cancer patients; the females with genetic mutations that are never-smokers are frequently at risk of carcinogenesis in lung cancer. 29
TT is a coagulation indicator that reflects the conversion of fibrinogen to fibrin after adding a thrombin reagent. TT is used as a screening test for detecting fibrinogen abnormalities and evaluating fibrin formation disorders. 30 Thrombin levels are high in cancer patients. 31 Lung cancer is associated with hypercoagulability, 32 and coagulation factors are abnormally increased in lung cancer patients, which might be used for the diagnosis of early-stage lung cancer or as a prognostic biomarker for lung cancer.33,34 It was found that AGP1 plays a role in coagulation and is a novel gene that potentially controls thrombin generation. In an experimental model of peritonitis in rats, high doses of AGP1 normalized platelet aggregation, blood clotting parameters, and antithrombin activity, which suggests that AGP1 can act as a coagulation inhibitor delaying thrombin generation. 35 In our study, we found that TT negatively correlated with AGP1 levels in lung cancer, as Rocanin-Arjo et al. 35 reported. However, the correlation between coagulation factor TT and AGP1 expression in lung cancer remains unclear and needs further research.
K mainly maintains cell metabolism, regulates osmotic pressure, and maintains primary neuromuscular irritability and normal cardiac function. The change in K concentration will affect hypotension or cardiovascular disease. It was reported that potassium intake was correlated with tumorigenesis in lung cancer; appropriate potassium intake was associated with a decreased risk of lung cancer; and increasing potassium intake could decrease the incidence of lung cancer. 36 The dysregulated expression of the potassium channel was associated with cell proliferation and migration to promote tumor growth in lung cancer. 37 For example, potassium voltage-gated channel subfamily D member 2 (KCND2) promotes the aggressiveness of LUAD. 38 Our study found that K was negatively correlated with AGP1 levels. Nevertheless, the relationship between electrolyte K and the expression of AGP1 is unknown and needs to be explored in the future.
In summary, our findings show that the expression of AGP1 in lung cancer patients is decreased and performs well for distinguishing malignant from benign pulmonary lesions, which will serve as an effective diagnostic biomarker for lung cancer.
Supplemental Material
sj-tif-1-jbm-10.1177_03936155231192672 - Supplemental material for A diagnostic biomarker of acid glycoprotein 1 for distinguishing malignant from benign pulmonary lesions
Supplemental material, sj-tif-1-jbm-10.1177_03936155231192672 for A diagnostic biomarker of acid glycoprotein 1 for distinguishing malignant from benign pulmonary lesions by Ying Chen, Yueyang Zhang, Ankang Huang, Yongsheng Gong, Weidong Wang, Jicheng Pan and Yanxia Jin in The International Journal of Biological Markers
Supplemental Material
sj-xlsx-2-jbm-10.1177_03936155231192672 - Supplemental material for A diagnostic biomarker of acid glycoprotein 1 for distinguishing malignant from benign pulmonary lesions
Supplemental material, sj-xlsx-2-jbm-10.1177_03936155231192672 for A diagnostic biomarker of acid glycoprotein 1 for distinguishing malignant from benign pulmonary lesions by Ying Chen, Yueyang Zhang, Ankang Huang, Yongsheng Gong, Weidong Wang, Jicheng Pan and Yanxia Jin in The International Journal of Biological Markers
Supplemental Material
sj-docx-3-jbm-10.1177_03936155231192672 - Supplemental material for A diagnostic biomarker of acid glycoprotein 1 for distinguishing malignant from benign pulmonary lesions
Supplemental material, sj-docx-3-jbm-10.1177_03936155231192672 for A diagnostic biomarker of acid glycoprotein 1 for distinguishing malignant from benign pulmonary lesions by Ying Chen, Yueyang Zhang, Ankang Huang, Yongsheng Gong, Weidong Wang, Jicheng Pan and Yanxia Jin in The International Journal of Biological Markers
Footnotes
Acknowledgments
We thank nurses and patients from Suzhou Municipal Hospital of Nanjing Medical University.
Abbreviations
Authors’ contributions
YXJ designed the project and revised the manuscript. YXJ, YC, and YYZ performed the experiments, analyzed the data, drew the figures, and wrote the manuscript. AKH collected the data from clinical patients. YSG, WDW, and JCP helped to supervise the project and revised the manuscript. All authors have read and approved the published version of the manuscript.
Ethics approval and consent to participate
The study was conducted by the Declaration of Helsinki and approved by the institution's Research Ethics Board of Nanjing Medical University and all methods were carried out according to relevant guidelines. The informed consents were obtained from all participants.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Natural Science Foundation of China (NSFC) program (No. 32000908), the Natural Science Foundation of Hubei Province program (No. 2020CFB417), and the Hubei Key Laboratory of Edible Wild Plants Conservation and Utilization (No. EWPL202002, No. EWPL202106, No. EWPL202211).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
