Abstract
Background
Epithelial ovarian cancer (EOC) is the leading cause of death associated with gynecologic tumors. EOC is asymptomatic in early stages, so most patients are not diagnosed until late stages, highlighting the need to develop new diagnostic biomarkers. Mediators of the tumoral microenvironment may influence EOC progression and resistance to treatment.
Aim
To analyze immune checkpoints to evaluate them as theranostic biomarkers for EOC.
Patients and methods
Serum levels of 16 immune checkpoints were determined in EOC patients and healthy controls using the MILLIPLEX MAP® Human Immuno-Oncology Checkpoint Protein Magnetic Bead Panel.
Results
Seven receptors: BTLA, CD40, CD80/B7-1, GITRL, LAG-3, TIM-3, TLR-2 are differentially expressed between EOC and healthy controls. Serum levels of immune checkpoints in EOC patients are positively significantly correlated with levels of their ligands, with a higher significant correlation between CD80 and CTLA4 than between CD28 and CD80. Four receptors, CD40, HVEM, PD-1, and PD-L1, are positively associated with the development of resistance to Taxol-platinum-based chemotherapy. All of them have an acceptable area under the curve (>0.7).
Conclusion
This study has yielded a first panel of seven immune checkpoints (BTLA, CD40, CD80/B7-1, GITRL, LAG-3, TIM-3, TLR-2) associated with a higher risk of EOC and a second panel of four immune checkpoints (CD40, HVEM, PD-1, PD-L1) that may help physicians to identify EOC patients who are at high risk of developing resistance to EOC chemotherapy.
Introduction
The most common subtype of ovarian cancer (OC) is epithelial ovarian cancer (EOC), accounting for 90% of all ovarian cancer. It remains one of the most important diagnostic and therapeutic issues in contemporary gynecologic oncology, with a life expectancy of 25–35% among these patients. 1 The low long-term survival rates are due, in part, to the lack of effective screening technologies. By the time symptoms appear, most patients are already in an advanced stage of disease (more than 60% of ovarian cancer patients are diagnosed after distant metastasis). 2 In addition, 20% of EOC patients do not respond to first-line chemotherapy (CT) with carbo-platinum, and treatment of resistant and recurrent EOC is currently difficult because there are few therapeutic options that can significantly improve overall survival once the cancer becomes chemoresistant. 2
One of the most widely used biomarkers for EOC is CA125, a protein found in elevated levels in the blood of many women with EOC. 3 However, it is well established that relying on a single biomarker is improper for accurate ovarian cancer diagnosis and prognosis. Other comorbidities can influence the expression levels of a single biomarker, making it difficult to distinguish cancer from other conditions. 3 However, by using multiple biomarkers in a panel, the chances of finding the same up or down regulation of expression levels in two different diseases is low, which increases the specificity and accuracy of cancer diagnosis and prognosis.
In this context, several biomarkers have been investigated to improve early detection and diagnosis of EOC, including the immune checkpoint (IC) molecules. 4 These molecules play an important role in immune response regulation, either enhancing or inhibiting it. 5 They are regulatory pathways that act as natural mechanisms to prevent excessive immune activation and maintain immune homeostasis (Figure 1). 5 However, some cancer cells hijack these pathways to evade immune surveillance, allowing the growth and spread of cancer cells.
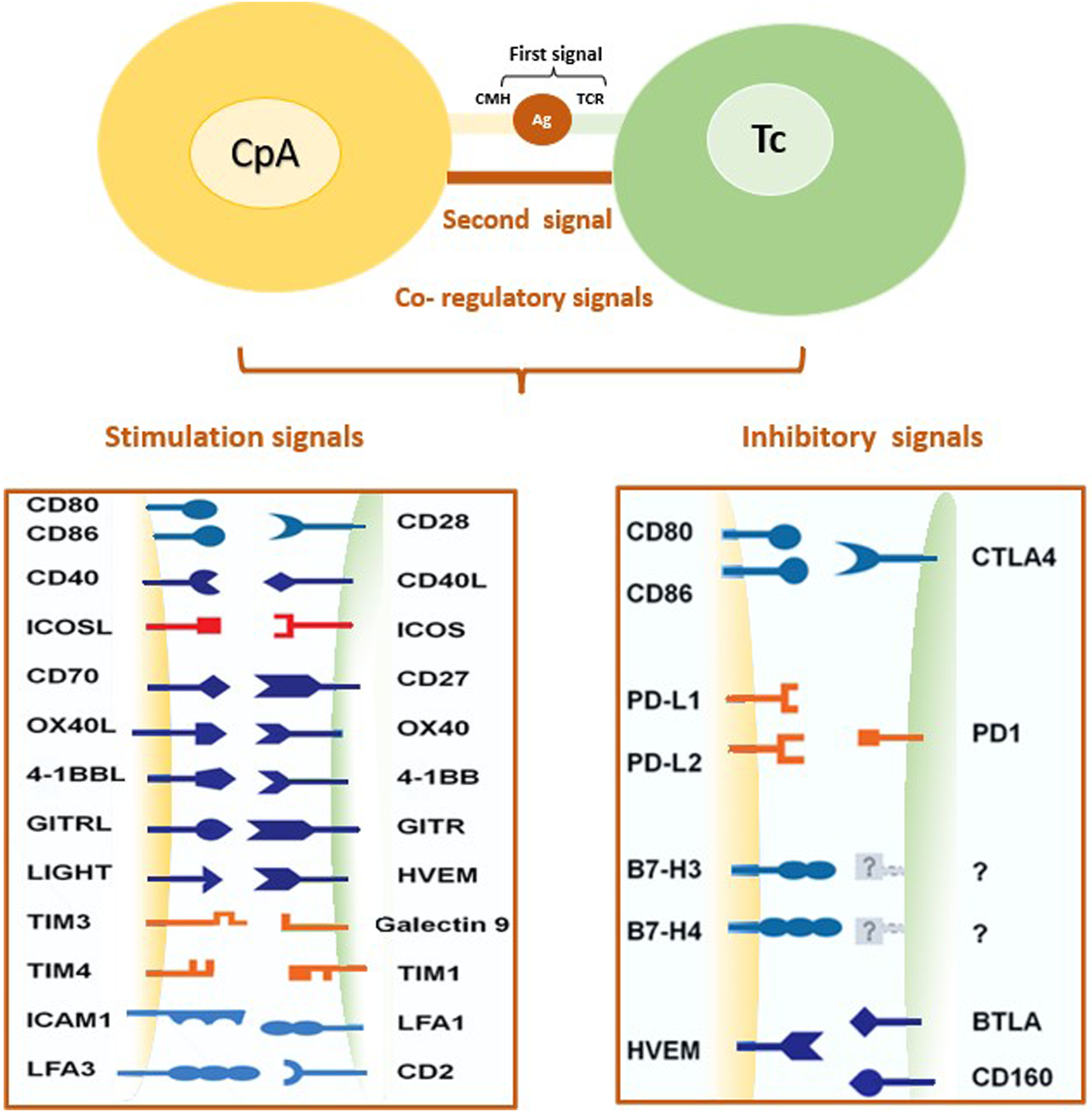
Immune checkpoints: co-inhibitory and co-stimulatory signaling.
Several studies have investigated the potential value of IC molecules as biomarkers for EOC, by focusing on a wide range of IC molecules, including BTLA, CD27, CD28, CD40, CD80/B7-1, CD86/B7-2, CTLA-4, glucocorticoid-induced TNFR-related protein (GITR), GITRL, herpesvirus entry mediator (HVEM), ICOS, LAG-3, programmed death-1 (PD-1), programmed death-ligand 1 (PD-L1), TIM-3, and TLR2.4–6 For example, one study discovered that PD-L1 expression in tumor cells was associated with a worse prognosis in EOC patients. 7 Another study discovered that high LAG-3 and TIM-3 expression on tumor-infiltrating lymphocytes was associated with better overall survival in EOC patients. 6 In addition, other studies have looked into the use of IC molecules as therapeutic targets in EOC.6–9 For example, clinical trials with checkpoint inhibitors targeting PD-1 and CTLA-4 have yielded promising results for EOC.8,9
These findings suggest that targeting these molecules may hold promise in improving outcomes for EOC patients and more researches are needed to fully understand ICs molecules’ potential as biomarkers and therapeutic targets in EOC,
In this context we investigate the serum levels of six co-inhibitory factors (BTLA, TIM3, LAG-3, CTLA-4, PD-1, and PD-L1) and 10 co-stimulatory factors (CD27, CD28, CD40, HVEM, TLR-2, GITR, GITRL, ICOS, CD80, CD86) as potential theranostic biomarkers for EOC.
Patients and methods
Patients
Between March 2019 and November 2020, 57 female EOC patients were recruited from the outpatient surgical and oncology service of Salah Azaeiz Institute (SAI), Tunisia. All female EOC patients had been newly diagnosed with confirmation by histopathology. They had not received therapy at the time of recruitment, and their medical records were used to follow them for at least 6 months after treatment. The patients’ clinical and pathologic characteristics were obtained from their medical records and structured questionnaire interviews. Patients’ medical records were also reviewed 6 months after sampling to assess their response to first-line Taxol-platinum-based chemotherapy (CT).
A total of 49 age-matched healthy women who did not have EOC or any other cancer were included as healthy controls. Written informed consent was obtained from all patients and controls. The SAI Research and Ethics Committee (registration number: ISA/2019/01, granted February 22, 2019) reviewed and approved all documents and procedures. The research was conducted in accordance with the ethical standards of the 1964 Declaration of Helsinki.
Collection of blood
In a sterile serum coagulation tube, 5 mL of venous blood was collected from all study patients and controls. Within 2 h of collection, serum samples were separated by centrifugation at 2500 rpm for 20 min. They were transferred to a clean microcentrifuge tube and centrifuged for 10 min at 14,000 rpm for 10 min to remove cell debris and fragments. Aliquots of the serum were prepared and stored at −80 °C until analysis.
Multiplex immunoassay
The concentrations of sixteen IC proteins: BTLA, CD27, CD28, CD40, CD80/B7-1, CD86/B7-2, CTLA-4, GITR, GITRL, HVEM, ICOS, LAG-3, PD-1, PD-L1, TIM-3, and TLR2) were determined in EOC samples using the MILLIPLEX MAP® Human Immuno-Oncology Checkpoint Protein Magnetic Bead Panel (Millipore, Billerica, MA, USA) according to the manufacturer's protocol. Data were collected using a Bio-Plex® 200 instrument and analyzed using Manager 5.0 software (Bio-Rad, Hercules, CA, USA).
Statistical analysis
SPSS 24.0 software (IBM, Armonk, NY, USA) was used for statistical analysis, and graphs were generated using GraphPad Prism 7.0 (GraphPad Software, San Diego, CA, USA). Statistical significance was defined as a P-value < 0.05. Data are presented as mean SD. The Mann–Whitney test was used to compare differences between groups (EOC patients and healthy controls) and EOC patients according to clinicopathological parameters. An analysis of the receiver operational characteristics was performed for molecules that differed significantly between the compared groups; area under the curves (AUC), sensitivity for 95% specificity.
Results
Study subjects
We compared serum protein expression profiles of 16 IC proteins between EOC patients (n = 57) and healthy controls (n = 49) to identify biomarker signatures in serum associated with EOC. The mean age of EOC patients was 56 ± 13 years with a mean body mass index (BMI) of 27.7 ± 4.5. According to the International Federation of Gynecology and Obstetrics (FIGO), 10 patients (17.6%) were diagnosed with early stage EOC (stage I and stage II), 47 (82.4%) were diagnosed with late stage EOC (stage III and stage IV), and 30 (52.7%) had distant metastases. All patients were treated with six doses of Taxol in combination with carboplatin CT, which can be used before or after surgery depending on the size, grade, and type of tumor. Twelve cases (21%) were resistant to CT (Taxol-platinum), and 14 experienced a recurrence of the disease after 6 months of the CT treatement (Table 1).
Clinical data of EOC patients and healthy controls.
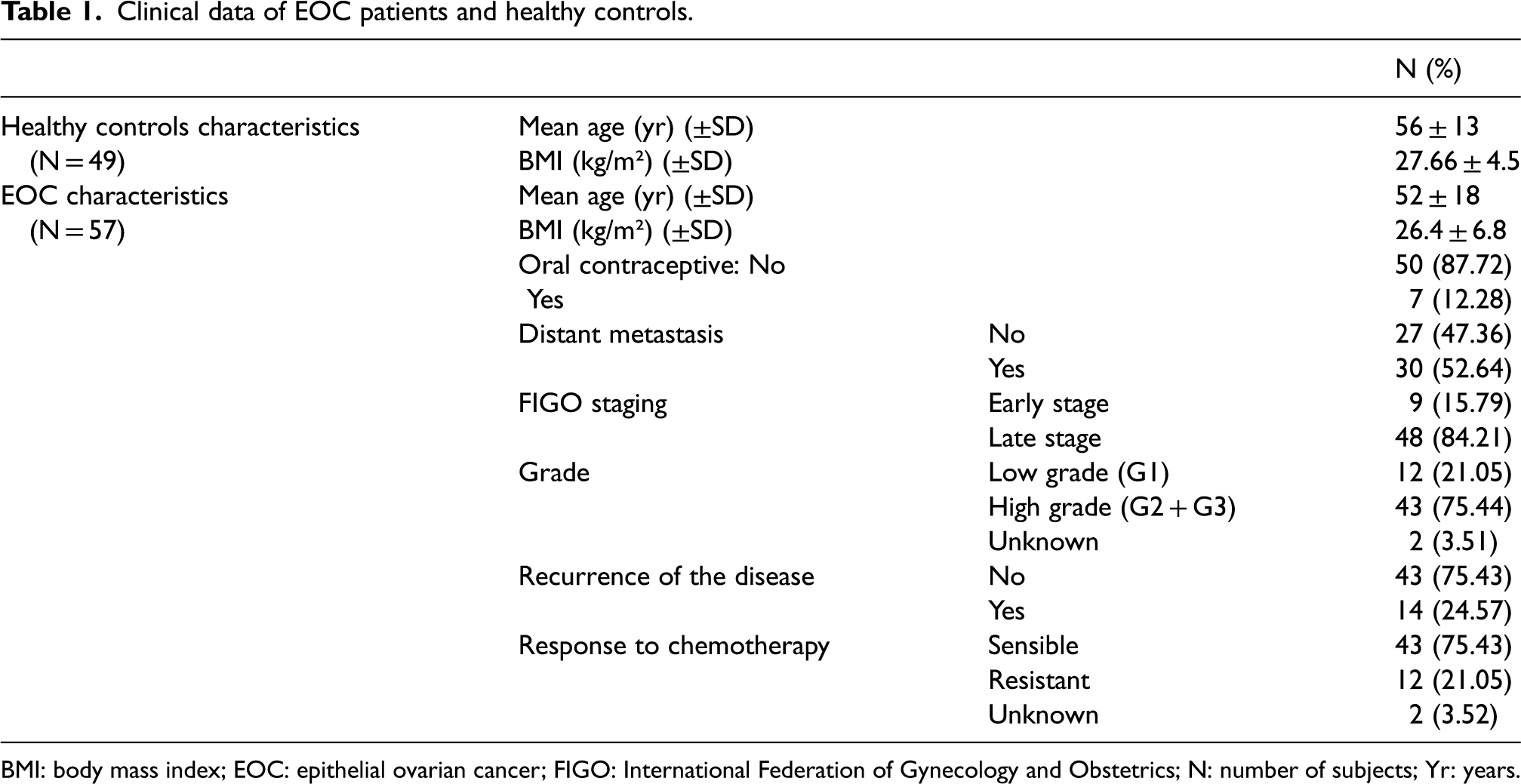
BMI: body mass index; EOC: epithelial ovarian cancer; FIGO: International Federation of Gynecology and Obstetrics; N: number of subjects; Yr: years.
Analytes associated with the development of EOC
Seven receptors were differentially expressed between EOC and healthy controls (BTLA, CD40, CD80/B7-1, GITRL, LAG-3, TIM-3, TLR-2). Of the seven dysregulated receptors, three were upregulated (CD40 (0.004**), LAG3 (<0.0001***) and TIM3 (0.001**), while BTLA (0.05*), CD80/B7-1 (0.006**), GITRL (0.01*), and TLR2 (0.004**) were down-regulated (Figure 2, Table 2).
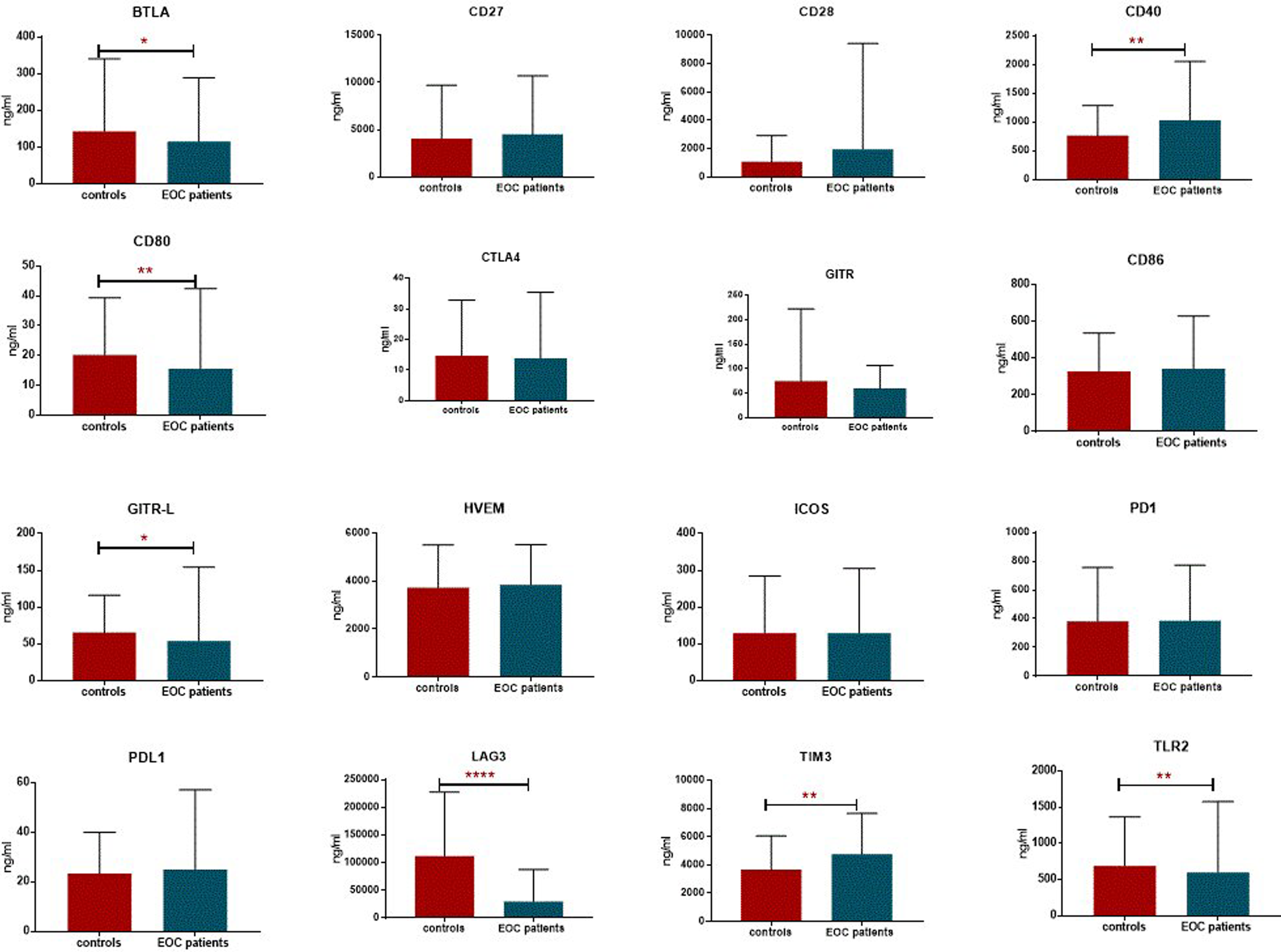
Serum immune checkpoints levels in EOC and healthy controls / ***P < 0.0001; **P < 0.005; *P < 0.05.
Serum immune checkpoints levels in EOC and healthy control.
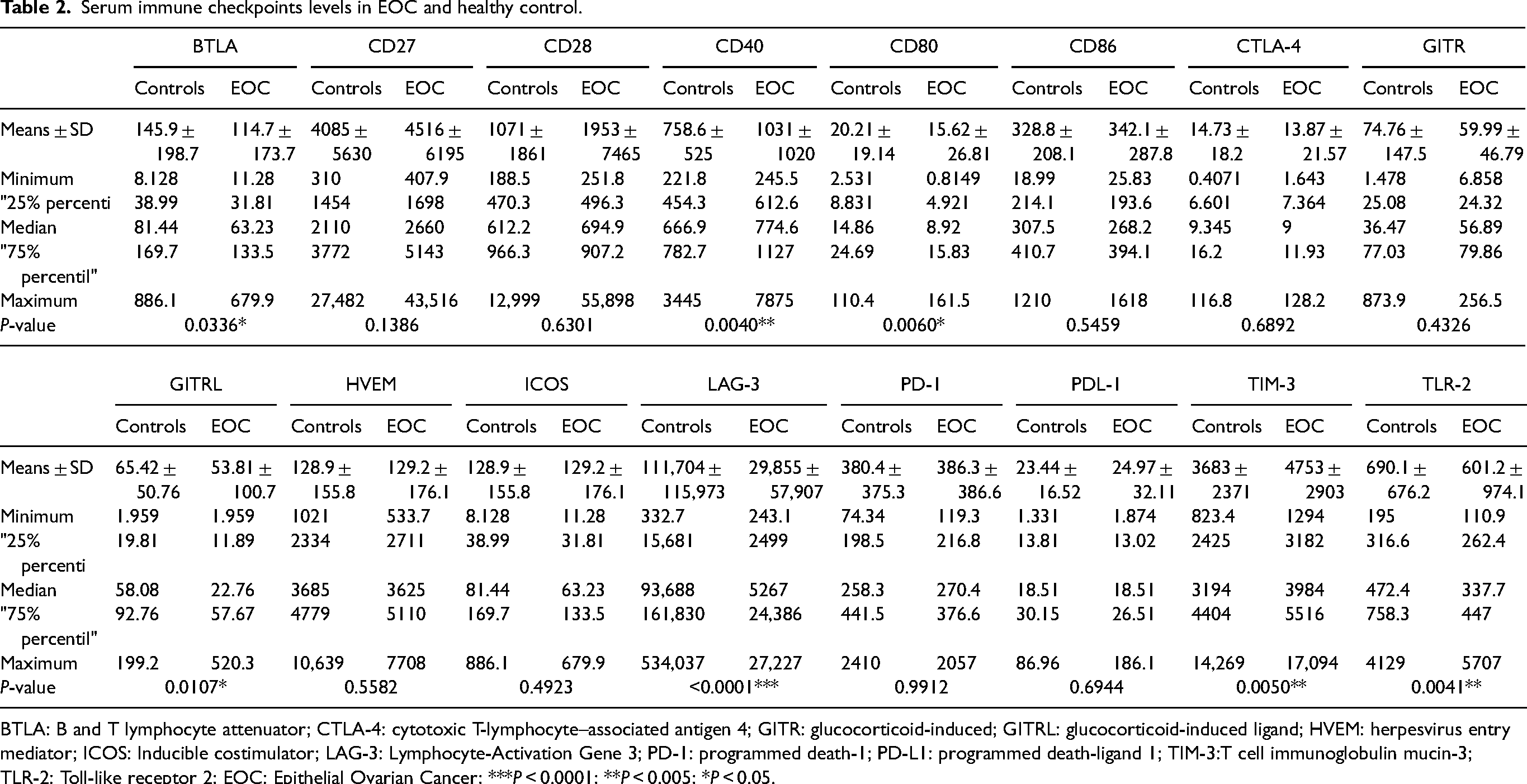
BTLA: B and T lymphocyte attenuator; CTLA-4: cytotoxic T-lymphocyte–associated antigen 4; GITR: glucocorticoid-induced; GITRL: glucocorticoid-induced ligand; HVEM: herpesvirus entry mediator; ICOS: Inducible costimulator; LAG-3: Lymphocyte-Activation Gene 3; PD-1: programmed death-1; PD-L1: programmed death-ligand 1; TIM-3:T cell immunoglobulin mucin-3; TLR-2: Toll-like receptor 2; EOC: Epithelial Ovarian Cancer; ***P < 0.0001; **P < 0.005; *P < 0.05.
To identify a potential biological network between IC proteins, we calculated a correlation of ICs in all patients and calculated the Spearman coefficient and heat map for each pair of IC proteins. The results showed that groups of proteins belonging to both inhibitory and co-stimulatory pathways were significantly and positively correlated with each other, such as CD28, CD80, CD86, CTLA4, GITR, GITRL, PD-1, and PD-L1 (Figure 3).
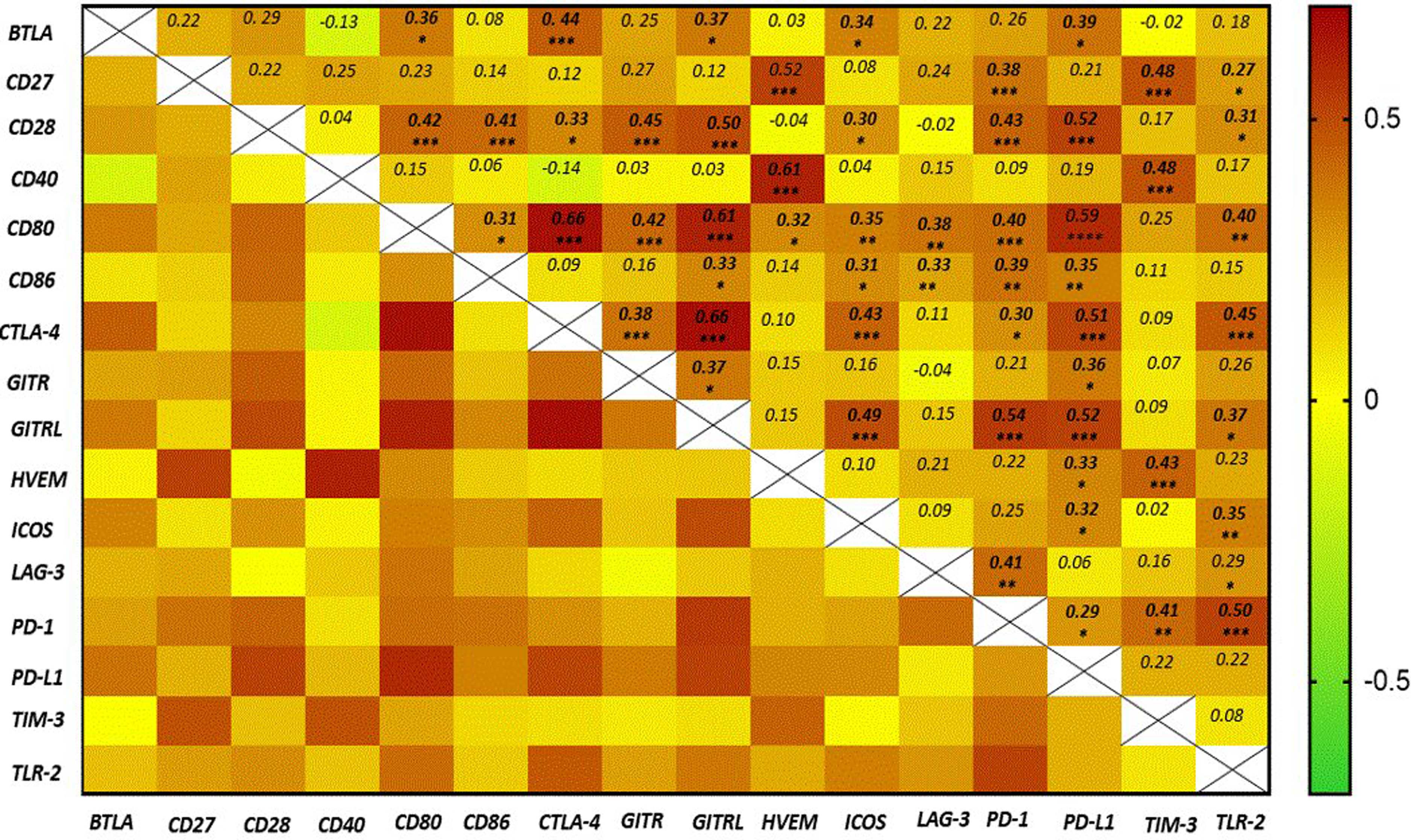
Heatmap generated by spearman rank correlation / ***P < 0.0001; **P < 0.005; *P < 0.05.
Moreover, serum levels of IC molecules in EOC patients correlated significantly and positively with the levels of their ligands; for example, CD28 levels correlated with CD80 levels (ρ = 0.42, P < 0.0001) and CD86 levels (ρ = 0.41, P = 0.001); PD-1 levels correlated with PD-L1 levels (ρ = 0.286, P = 0.031); and GITR levels correlated with GITRL levels (ρ = 0.371, P = 0.023). These data suggest that the production and/or secretion of ICs and their corresponding ligands are interdependent and that these molecules form a biological complex involving both inhibitory and co-stimulatory pathways (Figure 3).
Associated analytes with features of EOC
We investigated the possible association between the tested receptors and EOC features.
Serum levels were compared between EOC patients stratified by age, BMI, oral contraceptive use, stage, grade, development of metastases, disease recurrence, and resistance to first-line chemotherapy (Taxol-platinum).
Results show that the 16 ICs (BTLA, CD27, CD28, CD40, CD80/B7-1, CD86/B7-2, CTLA-4, GITR, GITRL, HVEM, ICOS, LAG-3, PD-1, PD-L1, TIM-3, TLR-2) were not associated with patient age and BMI (supplementary material).
Four proteins: CD86/B7-2 (360 ± 297.5 vs. 160.6 ± 99.23 P = 0.0178), GITR (63.88 ± 48.2 vs. 26.66 ± 16.58 P = 0.0370), GITRL (58.87 ± 107.2 vs. 10.22 4.412 P = 0.0202), and HVEM (4366 3150 vs. 2276 ± 1331 P = 0.0096) were significantly down-regulated in patients who took the oral contraceptive pill compared to EOC patients who did not (supplementary material). PD-1 (203 ± 46.58 vs. 381.6 ± 354.4 P = 0.0136) was upregulated in patients with late-stage disease compared to those with early-stage disease (supplementary material). Two receptors were differentially expressed in patients with high-grade and low-grade disease. CD28 (2388 ± 9186 vs. 613.1 ± 257.9 P = 0.0342) was significantly more expressed and TIM3 (5411 ± 3314 vs. 3625 ± 1503 P = 0.0108) was downregulated in serum levels of patients with high-grade cancer.
Serum levels were compared between EOC patients stratified by the development of distant metastases. Both GITR and TLR2 proteins (947.7 ± 1403 vs. 320.8 ± 97.77 P = 0.0411) were significantly down-regulated in patients who developed distant metastases. In EOC patients stratified by disease recurrence, TLR2 was found to be significantly down-regulated in patients who had relapsed (supplementary material).
Analytes associated with resistance to CT
Serum levels of the analyzed IC molecules were compared between two groups of EOC patients stratified by resistance to first-line chemotherapy. The results show that four receptors: CD40 (0.0169), HVEM (0.0073), PD-1 (0.0139), and PD-L1 (<0.0001) are positively associated with the development of resistance to Taxol-platinum-based chemotherapy (Figure 3). They all have an acceptable AUC (>0.7). The highest was for PD-L1 (0.9), followed by HVEM (0.75), PD-1 (0.73), and finally CD40 (0.72) (Figure 4).
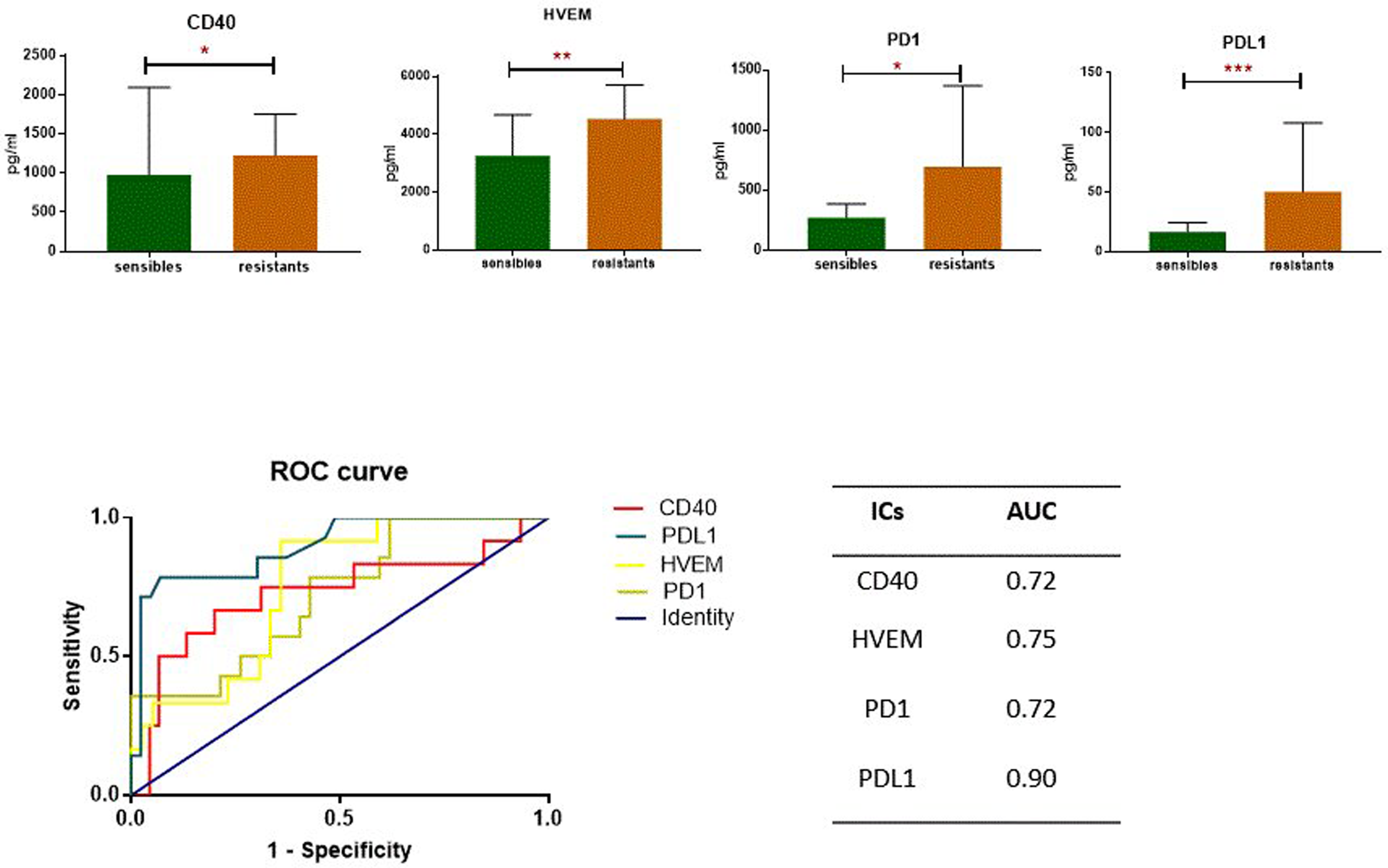
ICs significantly expressed between between Chemotherapy Sensitive and Resistant Patients.
Discussion
Currently, scientific researchers are continuously searching for new theranostic indicators able to predict cancer patient survival and improve the efficacy of therapy in tumor diseases with poor prognosis and high morbidity, including EOC.
Since it is difficult to detect EOC at early stages, identifying specific biomarkers could improve the treatment of EOC management and provide helpful information for predicting prognosis and response to CT. 10
Among the many biomarkers investigated, until now, the carbohydrate antigen-125 (CA-125) has been the most important indicator for screening, detection, treatment, and survival of EOC patients. 3 However, at diagnosis, only 80% of women with EOC had a high serum level of CA-125. 11 In addition, several physiological and non-physiological factors may influence normal CA-125 serum levels. 3 Therefore, characterization of novel, more specific theranostic biomarkers for EOC is an emergency.
Recent studies reported that EOC is associated with immune dysfunction that fails to recognize malignant cells and prevents successful elimination during apoptosis,12,13 thus implicating specific immune receptors and their ligands. Recent studies have examined a panel of ICs as novel theranostic biomarkers for neuroblastoma 14 and cervical cancer. 15
However, in a recent study that highlighted BTLA and CD27 as unfavorable prognostic factors for ovarian cancer, only 5 ICs were considered for ovarian cancer. 16 In the present study, 16 ICs were evaluated as theranostic biomarkers for EOC.
The major systems involved in the cytotoxic effects responsible for the development of cancers including EOC include CD40-CD40L, CD28-CD80, CTLA4-CD86, PD-1-PDL1, GITR-GITRL, and many others. Both these receptors and their ligands can be secreted into body fluids where they appear in soluble form. They have a major impact on the communication between immune and tumor cells leading to control, maturation, and apoptosis.
However, the interactions between the immune system and the microenvironment during ovarian carcinogenesis are still not well understood. In this study, we examined a broad range of IC proteins secreted in the serum of EOC women and investigated the relationships between these key immunoregulatory proteins and the development, the aggressivity of the disease, and the resistance to first line chemotherapy.
Two immune inhibitory checkpoint molecules LAG-3 and TIM-3, and one immune co-stimulatory checkpoint molecule CD40, are significantly increased in patients with EOC. The immunoinhibitory checkpoint BTLA, as well as three immunostimulatory checkpoints molecules (CD80, GITRL, and TLR2), showed significantly lower expression in the serum of patients with EOC compared to healthy controls.
Previous studies have also shown that overexpression of LAG-3 and TIM-3 can be associated with tumor progression in various tumors, including ovarian cancer,17,18 highlighting their potential value as diagnostic biomarkers for ovarian cancer. Previous finding has pointed out that co-expression of PD-1 with TIM-3 or LAG-3 on T cells from patients with solid tumors, including ovarian cancer, is associated with human exhaustion,17,18 a chronic antigen stimulation causes T cell dysfunction and is one of key aspect of the lack of sustained immune response to cancer. 18
LAG-3 binds to MHC-II with higher affinity than CD4 and disrupts CD4-MHC-II interactions, 19 whereas binding of TIM-3 to GAL-9 has been described as inducing apoptosis in T helper 1 effector cells and decreasing IFN-γ production. 20 Up-regulation of LAG-3 and TIM-3 mediates anti-tumor immunity tolerance and promotes tumor escape. 6
BTLA is one of the most important co-signaling molecules. It belongs to the CD28 superfamily and is similar in structure and function to PD-1 and CTLA-4. The association of BTLA with its ligand HVEM directly bridges the CD28 and TNFR families and mediates broad and potent immune effects. There are limited data on the use of BTLA as a diagnostic and prognostic biomarker in ovarian cancer. 4 In contrast to our findings, previous studies have shown that BTLA is upregulated in several cancers such as gallbladder cancer, 21 hepatocellular carcinoma, 22 lung cancer, 23 and ovarian cancer. 4 Thus, additional well-designed studies must be enrolled to better evaluate BTLA as novel biomarkers for ovarian cancer.
GITR is triggered by its ligand, GITRL, which is mainly expressed on B cells, dendritic cells, macrophages, and endothelial cells. 24 Activation of GITR on effector T cells can produce a positive costimulatory signal and promote T-cell activation and proliferation. In contrast, activation of GITR on Tregs abrogates their suppressive function. 25 This is consistent with the present results showing a significant association between downregulation of GITRL and the development of EOC.
TLR2 belongs to the superfamily of toll-like receptors (TLRs) that recognize microbe- or pathogen-associated molecular patterns (PAMPs) expressed by bacteria, viruses, and host-derived PAMPs such as stress proteins. 26 TLR2 has been shown to be expressed in normal ovarian epithelium but is also involved in the pathogenesis of ovarian cancer. 27 Although TLR2 is one of the most studied members of the TLR family, there are still no conclusive studies on the effects of TLR2 expression in ovarian cancer. 16 The present study shows that TLR2 is downregulated in EOC patients compared with healthy controls. In contrast, Zhang et al. 28 and Sobstyl 16 have documented significantly increased TLR2 expression in women with ovarian cancer. Our finding seems to be consistent with the function of TLR2 in recognizing danger signals released by cancer cells and its involvement in triggering the mechanisms of innate immunity and specific anti-tumor response, which has been documented by Sasai and Yamamoto. 26
The two clusters of differentiation—CD40 and CD80—are costimulatory molecules that play a key role in the activation of antigen-presenting cells, other innate immune cells, and anti-tumor immunity. 29
In accordance with previous studies that show an overexpression of CD40 in several malignancies including colorectal, 30 breast, 31 prostate, 32 nasopharyngeal carcinoma, 33 and ovarian cancer, 34 the present results revealed an up-regulation of this IC, whereas CD40L expression is not associated with cancers as documented by other studies.1,30
The CD80 IC can interact with both co-stimulatory molecules (CD28) and co-inhibitory molecules (CTLA-4, PD-1, and PD-L1) to influence T-cell activation.35–38 The binding of CD28 and CTLA-4 is competitive, and CTLA-4 has been shown to have a higher affinity for CD80 than for CD28.35,36 These findings are consistent with the present results showing that CD80-CTLA4 has a significantly stronger correlation compared with CD28-CD80. CD80 is significantly less expressed in EOC patients compared with healthy controls. Previous studies reported that the low expression of CD80 serves as a mechanism for tumor escape from immune surveillance due to its higher affinity and preferential binding to CTLA-4. 36
The variability in signaling pathways and IC expression levels between different types of cancer underscores the importance of identifying a specific biomarker panel for ovarian cancer. In our case, this panel includes BTLA, CD40, CD80, GITRL, LAG3, TIM3, and TLR2, which could serve as promising candidates for further investigation as potential theranostic biomarkers for ovarian cancer.
Furthermore, we investigated the association between the 16 IC proteins and EOC severity. Our findings suggest that only 1 or 2 of the 16 ICs showed significant association with disease severity in our cohort. For instance, PD-1 was found to be upreagulated in the late stage, which is consistent with previous studies demonstrating its overexpression in other cancer types, including lung cancer 37 and melanoma. 38 Furthermore, the downregulation of TLR2 in patients who developed distant metastasis has also been shown in various cancer types. 39 Therefore, further investigations are needed to identify the specific signaling pathways of individual ICs in EOC and to develop more effective biomarkers for predicting EOC severity.
Platinum resistance in EOC patients is associated with poor prognosis and limits treatment options. There appears to be an overlap between molecular mechanisms responsible for platinum resistance and the immunogenicity of EOC.
To the best of our knowledge—and, except for PD-1 and its ligand PD-L1—this is the first time that serum levels of the 16 remaining ICs have been measured simultaneously in EOC patients stratified by their response to chemotherapy.
Overexpression of four ICs—CD40, HVEM, PD-1, PD-L1—was found in EOC patients who developed resistance to chemotherapy, and thus they may represent future biomarkers useful to identify EOC patients prone to develop chemotherapy resistance and thus improve their therapeutic management. Previous results demonstrated the increased expression of PD-1/PD-L1 on tumor and immune system cells in ovarian cancer patients who developed resistance to the standard chemotherapy carbo-platinum.40,41 The interaction between PD-1 and its ligands limits the inflammatory response by inhibiting the function of cytotoxic T cells,18,41 leading to tolerance of the chemotherapy and a decreased immune response of the neoplastic process. 42
There have been several clinical studies investigating the use of ICs as potential targets for therapy in patients who have developed resistance to chemotherapy for EOC.9,41–43 For instance, a phase II clinical trial evaluated the efficacy and safety of the anti-PD-1 antibody pembrolizumab in combination with chemotherapy in patients with recurrent platinum-resistant EOC and demonstrated a promising result, with an overall response rate of 18.5% and a disease control rate of 66.7%. 9 Another study by Matulonis et al. 43 investigated the use of a dual PD-1 and PD-L1 blockade in patients with recurrent ovarian cancer who had progressed on prior chemotherapy. The study showed promising clinical activity, with an overall response rate of 18%.
The present study is the first to demonstrate a potential correlation between the expression of HVEM and CD40 co-stimulatory factors and the development of treatment resistance. However, further well-designed studies are necessary to confirm or refute these findings and determine the underlying mechanisms of this association.
The present study has several strengths. First, EOC cases and controls were matched according to their age and ethnicity (only Arab Tunisian-born women included), which minimizes the problems of ethnic differences inherent in genetic-association studies.
Second, the 16 ICs analyzed are the most studied in cancers with the possibility of their use as a personalized treatment in immunotherapy. However, this study also has shortcomings: the relatively small number of the cohort and the fact that these results are not confirmed in peritoneal ascites and tumoral tissues.
As a potential perspective for the future, investigating EOC cell lines and confirming our results in peritoneal ascites and tumor tissues could provide valuable insights into the mechanisms of immune response in EOC and aid in the development of targeted therapies.
Conclusion
In the present study, it can be concluded that two panels of IC biomarkers could be useful for early detection and prediction of chemotherapy resistance in patients with EOC. The first panel, consisting of BTLA, CD40, CD80/B7-1, GITRL, LAG-3, TIM-3, and TLR-2, could be potentially used as a tool for early diagnosis of EOC. The second panel, including: HVEM, CD40, PD-1, and PD-L1, could be helpful for clinicians to identify EOC patients who are at high risk of developing resistance to the chemotherapy of EOC.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155231186163 - Supplemental material for Immune checkpoints as potential theragnostic biomarkers for epithelial ovarian cancer
Supplemental material, sj-docx-1-jbm-10.1177_03936155231186163 for Immune checkpoints as potential theragnostic biomarkers for epithelial ovarian cancer by Azza Habel, Xu Weili, Mariem Hadj Ahmed, Mouna Stayoussef, Hanen Bouaziz, Mouna Ayadi, Amel Mezlini, Anis Larbi and Basma Yaacoubi-Loueslati in The International Journal of Biological Markers
Footnotes
Acknowledgements
We thank all blood donors and patients with EOC who volunteered to participate in the present study. We thank the staff of the Salah Azaiez Oncology Institute for their help in collecting blood samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported in part by the Tunisian Ministry of Higher Education and Scientific research, the Agency for Science Technology and Research (A*STAR) and the PRFD4-P1-ANPR project.
Ethics approval
The study was done per Helsinki II declaration and was approved by the Ethics Committee of SAI and that all participants have provided informed consent, and the approval code is: ISA/2019/01, granted in February 22, 2019.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
