Abstract
Background
Liver cancer seriously threatens human health. Natural killer (NK) cells are an important part of the innate immune system and have strong anti-tumor ability. Immunotherapy based on NK cells has become a hot topic in the treatment of liver cancer.
Methods
In this study, we checked the serum DKK3 (sDKK3) and circulating CD56bright NK cells using ELISA and flow cytometry, respectively, in the blood of liver cancer patients. The effect on recombinant human DKK3 (rhDKK3) on CD56bright NK cells was analyzed in vitro.
Results
We found low levels of sDKK3 in liver cancer patients and a negative correlation between sDKK3 and circulating CD56bright NK cells. In addition, we found that DKK3 induced the differentiation and improved the cytotoxicity of CD56bright NK cells for the first time. It could be used as an agonist for NK cell-based immunotherapy.
Conclusions
Improving the clinical efficacy of NK cells through DKK3 will become a new strategy for cancer immunotherapy.
Introduction
The incidence rate and mortality of liver cancer are increasing year by year, which seriously threatens human health and life. 1 Primary liver cancer, including hepatocellular carcinoma and intrahepatic cholangiocarcinoma, is related to alcohol consumption, viral hepatitis, food, and other factors. 2 The application of ultrasound in the diagnosis of liver diseases and the differentiation of liver cirrhosis and tumor is conducive to early detection of tumors; however, treatment resistance is always the bottleneck of liver cancer treatment. 3 Natural killer (NK) cells are an important part of the innate immune system and the first line of defense against invasive pathogens and tumor cells. 4 Due to the change of the immune microenvironment in the process of hepatocarcinogenesis, the immune function of cytotoxic T lymphocytes (CTL) and NK cells is inhibited, which provides the possibility for tumor cell immune escape. 5 NK cell killing target cells are not limited by the major histocompatibility complex (MHC), and can directly and rapidly kill the diseased cells, and perform immune clearance and immune surveillance functions. 6 NK cells account for 10%∼20% of peripheral blood lymphocytes. 7 According to the expression of CD56, they can be divided into two subgroups: CD56dim and CD56bright. 8 The CD56dim subgroup accounts for about 90%. 8 It is a fully mature NK cell, and plays a cytotoxic role through antibody-dependent cell-mediated cytotoxicity (ADCC). 8 CD56bright subtype, accounting for about 10% of the total number of NK cells in peripheral blood, is an immature NK cell with weak cytotoxicity, and regulates immune function by secreting cytokines. 9 Immunotherapy based on NK cells has become a promising treatment for liver cancer. 10 Promoting the transformation of CD56bright NK cells to CD56dim NK is beneficial for patients to fight tumors through their own immune systems. 10
The Wnt signaling pathway is involved in the process of embryonic development and maintains the differentiation and self-renewal of stem cells. 11 Abnormal activation of the Wnt signaling pathway leads to hepatocellular carcinogenesis and promotes the development of liver cancer. 12 DKK3, a member of the Dickkopf family, is an antagonist of the Wnt signaling pathway. 13 Previous studies have shown that the downregulation of DKK3 expression in cancer tissues of the patients with hepatocellular carcinoma (HCC) and indicated a poor survival of these patients.14–16 The serum DKK3 protein level could be used as a prognostic marker for the patients with HCC. 17 However, to our knowledge, no previous study showed the effect of DKK3 on the cytotoxicity of NK cells.
In this study, we compared the serum levels of DKK3 with the percentage of CD56bright/CD56dim NK cells in liver cancer patients and healthy volunteers. The roles of DKK3 in the differentiation of CD56bright NK cells were analyzed in vitro.
Materials and methods
Blood samples
Blood samples of the patients with liver cancer (n = 82) were collected in the Center Hospital of Handan, between September 2017 and June 2020. Control blood samples were obtained from 30 healthy individuals with similar age and daily lifestyle as the patients. The details can be found in our previous study. 18 All patients provided informed consent. This study was conducted according to the Helsinki Declaration of 1975 and approved by the Ethics Committee of Liaoning Medical University (20170013).
Serum DKK3 level determination
We used the same method as in our previous study 19 ; serum DKK3 levels were measured by enzyme-linked immunosorbent assay (ELISA), according to the manufacturer's instructions (R&D Systems, Shanghai, China).
Proportion of circulating CD56bright and CD56dim NK cells
CD56bright and CD56dim NK cells from peripheral blood were enriched using a negative selection MACS NK column as recommended by the manufacturer (Miltenyi Biotec, Shanghai, China). The purity of sorted cells was evaluated using flow cytometry (Becton Dickinson Medical Devices, Shanghai, China). 20
Cytotoxicity assays
CD56bright or CD56dim NK cells (1 × 103) pretreated with recombinant human DKK3 (rhDKK3, 200 ng/mL) (R&D Systems) 24 h and target cells (HepG2) were separately seeded into each well of 96-well plates and allowed to adhere (effector-target ratio of 5:1). The supernatant of cells after co-culture was used to check the lactate dehydrogenase (LDH) release. Killing activity % = (corrected experimental LDH release - effector spontaneous LDH release) / (target cell maximum LDH release - target cell spontaneous LDH release) × 100%.21,22
Flow cytometry
As the method in our previous study, 23 CD56bright NK cells with rhDKK3 treatment were washed with phosphate-buffered saline (PBS) and stained with allophycocyanin (APC)-conjugated anti-human NKG2D (eBioscience, Shanghai, China) for 30 min in the dark at 20 °C. The cells were analyzed by flow cytometry (Becton Dickinson).
Western blot
Cellular proteins were collected from CD56bright or CD56dim NK cells that collected from the patients with or without rhDKK3 treatment and separated by 10% SDS-polyacrylamide gel electrophoresis and transferred to nitrocellulose membranes. The nitrocellulose (NC) membranes were incubated with primary antibody for CD56 (sc-7326), p-GSK-3β (sc-81496), GSK-3β (sc-7291), GAPDH (sc-47724) (Santa Cruz, CA, USA), p-NF-κB p65 (ab239882), and NF-κB p65 (ab32536) (Abcam, Shanghai, China) overnight at 4°C. The reaction was followed by probing with peroxidase-coupled secondary antibodies (KeyGEN, Nanjing, China), and binding results were visualized via enhanced chemiluminescence (KeyGEN).
Statistical analysis
The results were analyzed using the Mann-Whitney U-test and Student's t-test. A threshold of P < 0.05 was used to determine statistically significant differences. GraphPad Prism 5 software (GraphPad Software Inc., San Diego, CA, USA) was used to analyze all data.
Results
Circulating NK cells and serum DKK3 from liver cancer patients
Serum DKK3 levels were lower in the liver cancer patients compared to that in healthy controls (P < 0.05, Figure 1(a)). The sDKK3 levels in the 82 patients ranged from 5 ng/mL to 64 ng/mL, with a median of 27 ng/mL. A higher percentage of circulating CD56bright NK cells were observed in the blood samples from liver cancer patients compared with those from healthy controls; however, the data were not significant (P > 0.05, Figure 1(b)). The sDKK3 levels were significantly negatively correlated with the number of circulating CD56bright NK cells in these patients (r = −0.2451) (P < 0.01, Figure 1(c)). In addition, circulating CD56bright NK cells were associated with tumor number (P = 0.0001), differentiation (P = 0.0116), and tumor size (P = 0.0132). sDKK3 was associated with tumor number (P = 0.0001), differentiation (P = 0.0092), tumor size (P = 0.0001), and lymphatic invasion (P = 0.0001). The association between sDKK3 or circulating CD56bright NK cells with the clinicopathological data of these patients is summarized in Table 1.
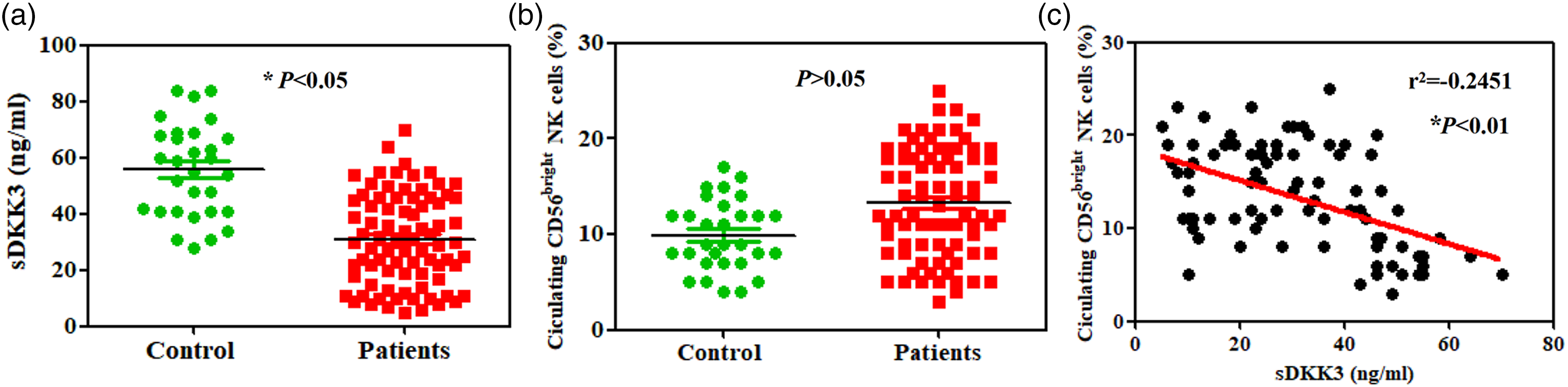
Circulating CD56bright NK cells and serum DKK3 in liver cancer patients’ blood samples. (a) Results of ELISA showing the serum levels of DKK3 in the patients and healthy controls. (b) Percentage of circulating CD56bright NK cells were determined using flow cytometry in patients and healthy controls. (c) The negative correlation between sDKK3 and circulating CD56bright NK cells in the patients.
Relationship between sDKK3 or circulating CD56bright NK cells and the clinicopathological parameters of patients with liver cancer.
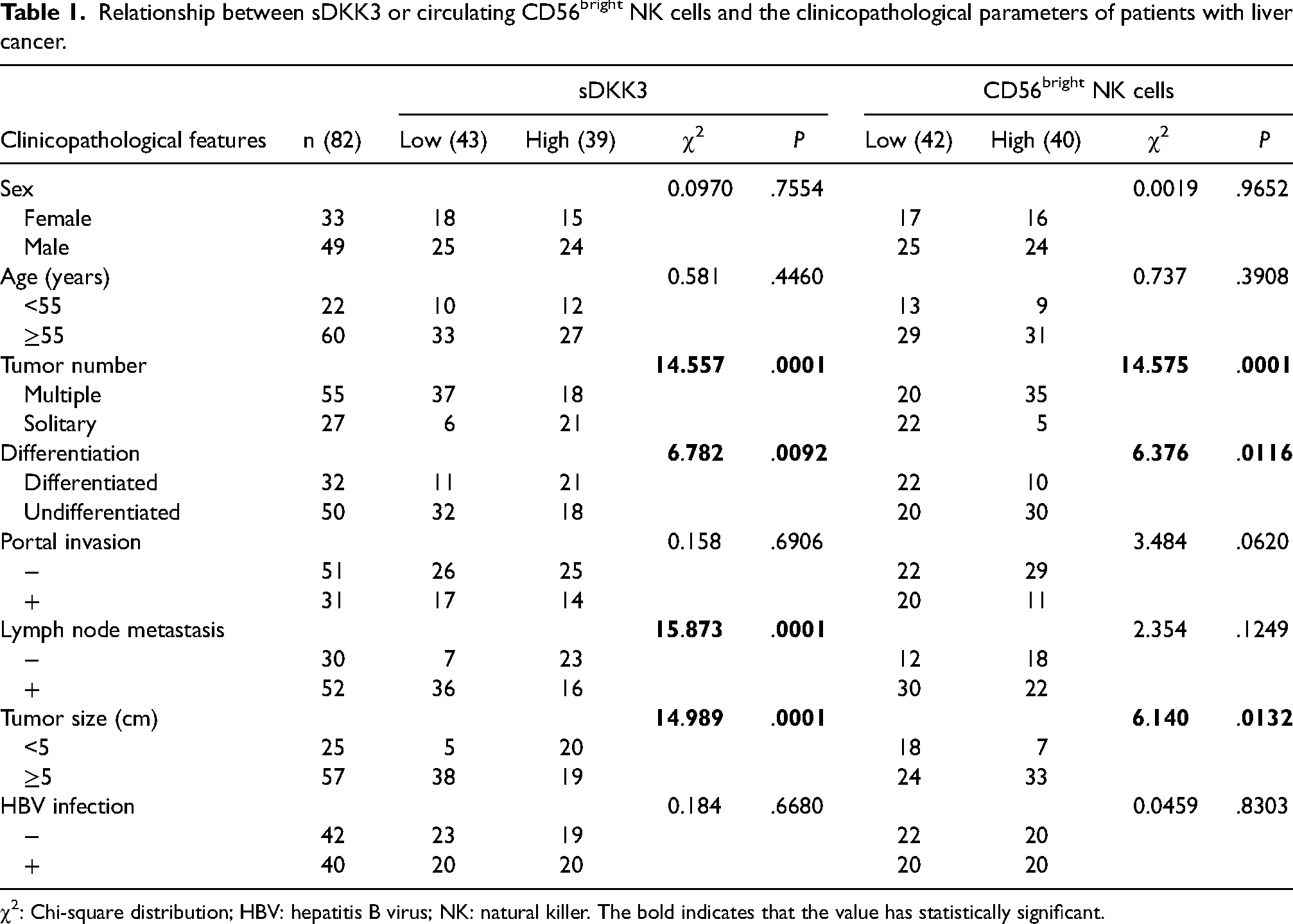
χ2: Chi-square distribution; HBV: hepatitis B virus; NK: natural killer. The bold indicates that the value has statistically significant.
DKK3 improved the cytotoxicity and differentiation of CD56bright NK cells
Based on the clinical data shown above and the bioinformatic data (Supplementary Figure 1(a)), we checked the cytotoxicity and differentiation of CD56bright NK cells with rhDKK3 treatment. CD56 expression was downregulated and NKG2D expression was upregulated in CD56bright NK cells with DKK3 treatment (Figure 2(a) and (b)). It means DKK3 induced transformation of CD56bright NK cells to CD56dim NK cells. p-GSK-3β and p-NF-κB p65 were downregulated in CD56bright NK cells and CD56dim NK cells after DKK3 treatment (P < 0.01, Figure 2(a)). Interestingly, both of them decreased more in CD56dim NK cells than in CD56bright NK cells (P < 0.01, Figure 2(a)). The levels of total GSK-3β and NF-κB p65 protein did not change (P < 0.01, Figure 2(a)). Accordingly, the cytotoxicity of CD56bright NK cells was also upregulated after DKK3 treatment (P < 0.01, Figure 2(c)).
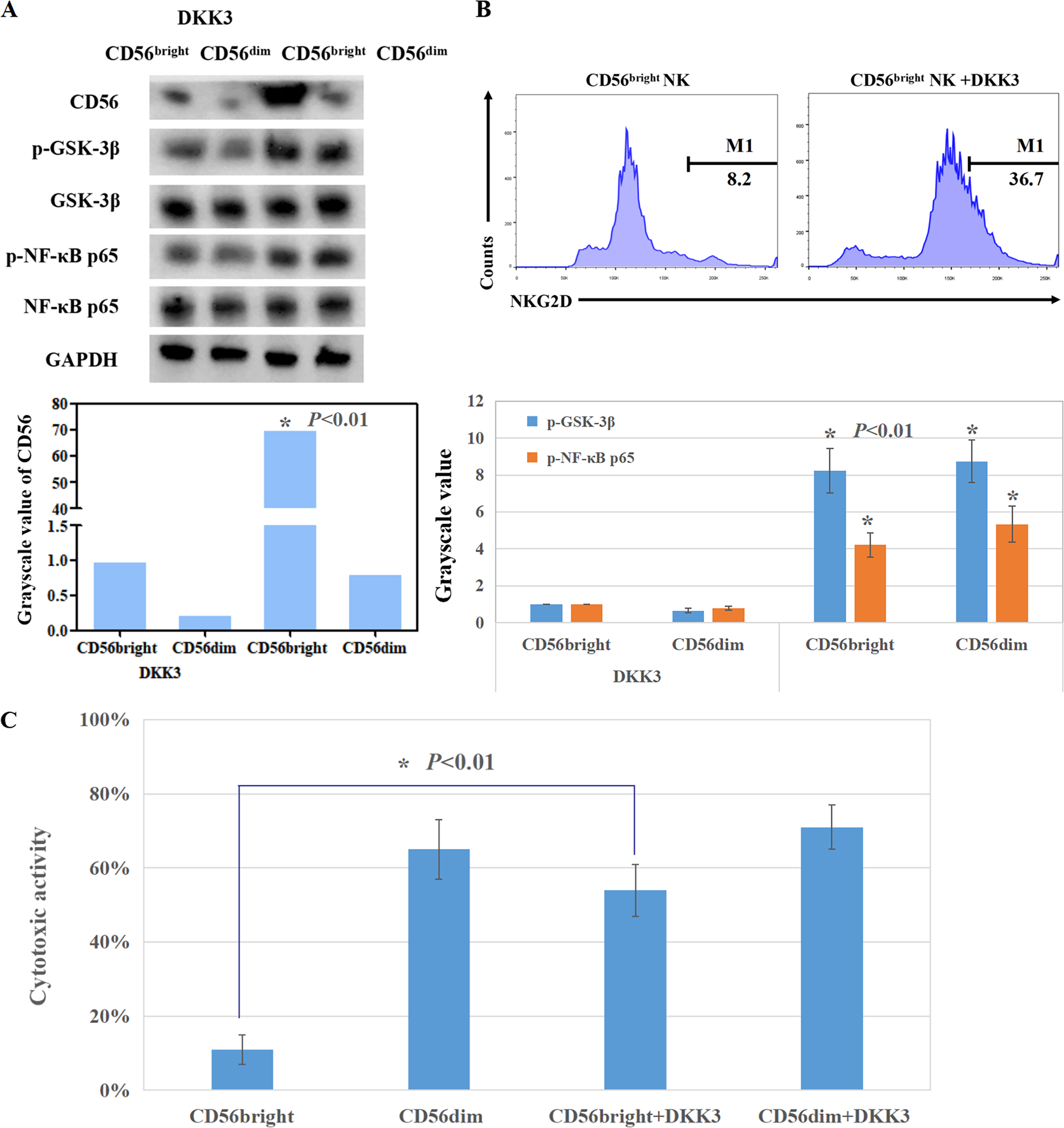
The roles of recombinant human DKK3 in the cytotoxicity and differentiation of CD56bright NK cells in vitro. (a) CD56, phosphorylation and total GSK-3β, and NF-κB p65 of CD56bright NK cells was detected using western blot. (b) NKG2D expression of CD56bright NK cells was detected using flow cytometry. (c) The cytotoxicity of CD56bright NK cells was determined using LDH release assay.
Discussion
Low expression of DKK3 has been confirmed in liver cancer tissues, which directly leads to an unfavorable outcome for liver cancer patients.14–16 Patients with a high serum DKK3 level have a favorable disease-free survival. 17 A combination of serum DKK1 and alpha fetoprotein (AFP) levels could provide more accurate diagnostic results than serum DKK1 or AFP levels alone. 24 AFP is the first marker for liver cancer; however, it is still used in routine practice. 25 In this study, we confirmed a low DKK3 level in the blood samples of liver cancer patients, but there was no significant difference of circulating CD56bright NK cells compared to healthy volunteers. Jin et al. 26 also found no significant difference of circulating CD56bright NK cells between liver cancer patients and healthy controls. However, we confirmed that circulating CD56bright NK cells were associated with tumor number, size, and differentiation. In addition, there was a negative correlation between sDKK3 levels and circulating CD56bright NK cells. CD56bright NK cells fight tumor cells by secreting cytokines, 5 and these cells are generally considered to be less cytotoxic than CD56dim NK cells. 5 Infiltrating CD56bright NK cells exhibited an exhausted phenotype in liver cancer tissues and played a protective role for metastatic liver cancer cells. 7 Therefore, how to improve the cytotoxic activity of CD56bright NK is a hot topic. The cytotoxicity was affected by cytokines in the tumor microenvironment (TME).27,28 Our bioinformatic analysis also showed that DKK3 is a regulator for NK cell resting based on The Cancer Genome Atlas TCGA database. Both our clinical data and bioinformatic data indicated DKK3 could be used to upregulate the antitumor immune function of NK cells.
With the evidence above, we naturally checked the roles of DKK3 in CD56bright NK cells. Interestingly, CD56 expression decreased in CD56bright NK cells after DKK3 treatment. NKG2D—a major activated receptor on the surface of NK cells—was upregulated after DKK3 treatment. This means that DKK3 induced the differentiation and improved the cytotoxicity of CD56bright NK cells. Our previous study found that DKK3 overexpressed in NK cells could inhibit its activity by suppressing immunological synapse (IS) formation. 23 In the DKK3 knockout mouse model, a slight increase in the frequency of CD49+ NK cells was detected; however, there was no functional checking for these cells. 29 To our knowledge, no previous study has shown the regulatory roles of secretory DKK3 in NK cells. This study shows that recombinant DKK3 could be used as a NK cell agonist for the first time. The molecular mechanism is the limitation of our study. The Wnt signaling pathway is known to take part in NK cell differentiation. 30 A blockade of the Wnt signaling pathway via DKK1 or DKK2 will lead to a significant decrease in NK cell generation and activation.31,32 In this study, we confirmed that p-GSK-3β and p-NF-κB p65 were suppressed by DKK3 in both CD56bright and CD56dim NK cells. GSK-3β is a key member of the Wnt and NF-κB signaling pathways. 33 Inhibition of GSK-3β could enhance NK cell activity against cancer cells. 34 Therefore, DKK3 can be used to promote CD56bright NK cell toxicity; however, the mechanism needs to be further studied.
In this study, we have confirmed the negative correlation between serum DKK3 and circulating CD56bright NK cells in liver cancer patients. Furthermore, we have demonstrated that DKK3 could improve the toxicity of NK cells for the first time. Improving the clinical efficacy of NK cells through DKK3 will become a new strategy for cancer immunotherapy.
Supplemental Material
sj-doc-1-jbm-10.1177_03936155231169796 - Supplemental material for Effect of secretory DKK3 on circulating CD56bright natural killer cells in patients with liver cancer
Supplemental material, sj-doc-1-jbm-10.1177_03936155231169796 for Effect of secretory DKK3 on circulating CD56bright natural killer cells in patients with liver cancer by Da-Hua Liu, Gui-Min Wen, Chang-Liang Song and Pu Xia in The International Journal of Biological Markers
Footnotes
Acknowledgements
Not applicable.
Author contributions
Conception: Pu Xia. Interpretation or analysis of data: Da-Hua Liu, Gui-Min Wen, Chang-Liang Song, Pu Xia. Preparation of the manuscript: Da-Hua Liu, Pu Xia. Revision for important intellectual content: Da-Hua Liu, Pu Xia. Supervision: Pu Xia. Da-Hua Liu, Gui-Min Wen contributed equally to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Scientific Foundation of China (No.81972784) and “Double First-Class” Disciplinary Construction Project of Jinzhou Medical University.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
