Abstract
Background
Advanced intercellular communication is a known oncogenic factor. In the central nervous system, Connexin-43 (Cx43) forms this junctional networking. Moreover, it correlates with the proliferation rate, and thus behavior, of gliomas. We assessed the expression of Cx43 and its relationship to Ki67 in other common central nervous system tumors.
Methods
The expression of Cx43 and Ki67 were assessed in formalin-fixed paraffin embedded samples of human brain metastases, meningiomas, and neurinomas using immunohistochemistry. Neurinomas and meningiomas were jointly evaluated due to similar non-malignant behavior.
Results
A total of 14 metastases of different extracerebral carcinomas, 6 meningiomas, and 10 neurinomas were evaluated. Five (36%) metastases and 5 (31%) meningiomas/neurinomas showed minor expression, whereas 6 (43%) metastases and 2 (13%) meningiomas/neurinomas showed no Cx43 expression at all. In 3 (21%) metastases and 9 (56%) meningiomas/neurinomas, moderate or strong expression of Cx43 was identified. The higher expression of Cx43 in meningiomas and neurinomas directly correlated with Ki67, r = 0.53 (P = 0.034). For metastases no significant correlation was found. Mitotic index in meningiomas/neurinomas correlated with Ki67 expression, r = 0.74 (P < 0.001), but did not show statistically significant correlation with Cx43 expression in these tumors.
Conclusions
The expression of Cx43 as a marker of cell-to-cell networking exposed a significant correlation with the Ki67-defined proliferation index in case of primary central nervous system neuroectodermal neoplasms. However, it does not seem to play a comparable role in metastases with extracerebral origin.
Introduction
The incidence of newly diagnosed central nervous system (CNS) neoplasms of different etiology has increased steadily over time. 1 This tendency will be continued due to improved resolution and simplified access to imaging, particularly magnetic resonance imaging. 2
Gap junctional mediated intercellular communication plays an important role in the exchange of small molecules and ions, which serve not only to maintain homeostasis, but also to influence cell proliferation and differentiation.3–5 The protein connexin-43 (Cx43) is involved in the formation of these gap junctional channels and hemichannels. 6 In the CNS, it is expressed not only by regular astrocytes,7,8 but also by neural precursor cells. 9 In malignant gliomas with higher proliferation index, the expression of Cx43 is reduced.10–13 Thus, Cx43 may have a role as a tumor suppressor, while increased expression is associated with a prolonged transition of G1 to S phase (G1 delay), and has effects on the expression of other cell-cycle-related genes.14–16 At the same time, upregulated Cx43-mediated gap intercellular communication protects tumor cells against temozolomide and radiotherapy, possibly providing resistance against specific treatment in lower-malignant gliomas.17,18
Ki67 protein is only expressed by proliferating cells, therefore it is used as a marker to evaluate the biology of a neoplasm.19,20 The activity of Ki67 is reported as a percentage and indicates the number of cells currently undergoing mitosis. Generally, an increased proliferation rate is associated with a more malignant behavior and consequently worse outcome in intracranial tumors. The evaluation of Ki67 is usually performed using immunohistochemistry (IHC) for MIB-1, which is a widely used monoclonal antibody that recognizes the nuclear antigen expressed in all non-G0 phase cells. 21 Consequently, the expression of MIB-1 and Ki67 can be considered equivalently. However, due to simpler handling and staining, MIB-1 is mostly used in clinical practice.22,23
It remains unclear whether a correlation between Cx43 and MIB-1 can be found in gliomas only. The IHC evaluation of an independent marker in a restricted number of cases gathered from corresponding groups can provide this overview and could show further way for investigation.24–26 The aim of this study was to summarize the potential expression of Cx43 in the common non-glial CNS tumors—metastases, meningiomas, and neurinomas—and to explore a potential correlation between aberrant expression of Cx43 and malignancy in terms of an increased proliferation index.
Materials & methods
Formalin-fixed paraffin embedded tissue samples of adult patients with 14 brain metastasis, 6 meningiomas, 10 neurinomas were randomly selected from the institutional tissue bank and analyzed. These specimens were obtained during surgery performed in our institution between 2017 and 2019 according to clinical indication, and are included in our prospective tumor database. The neuro-oncological database and tissue bank are approved by the institutional ethics committee (AN5220 329/4.4). The neuropathological examination and tumor grading was routinely performed after surgery, and diagnosis was based on the revised Fourth World Health Organization (WHO) Classification of Central Nervous System Tumors. 27
Cx43 was determined by IHC using commercial specific antibodies (Abcam ab11370 anti-connexin-43). Its expression in the most positive tumor cellular fraction was evaluated and semi-quantitatively classified into categories: 0 = no expression at all; 1 = minimal expression 2 = moderate expression (more than minimal but less than strong); 3 = strong expression (comparable to cortex). Using MIB-1 antibodies (Dako Omnis GA626), Ki67 antigen was stained. To obtain the proliferation index, the maximal proportion of nuclei with MIB-1 expression in relation to all cells in the same field was marked.28,29 Therefore, a hematoxylin and eosin-stained slide was used to ensure the neoplastic origin of evaluated tissue and to reveal mitotic figures for the mitotic index determination. The assessment of Ki67 and mitotic figures was performed using 10 high-power field (HPF, 40× magnification, 1.6 mm2 in total).30,31 The evaluation was performed by an experienced neuropathologist.
Statistical processing and analysis were performed using IBM SPSS Statistics (IBM SPSS Statistics for Mac OS, Version 26.0. Armonk, NY, USA: IBM Corp.). In analytical evaluation, meningiomas and neurinomas were stratified together due to their similar non-malignant behavior and clinical course and compared with metastases.
Normal distribution of scale parameters was checked by the Kolmogorov–Smirnov test correlations for non-parametric data and were exposed according to Spearman's method. Mann–Whitney U-test for rank or non-parametric scale data was applied. The Benjamini–Hochberg correction was respected for multiple hypothesis.32,33 The confidence interval and 1 – α were defined as 95%.
Results
Samples of 14 (47%) men and 16 (53%) women were evaluated in this study. The median MIB-1 expression was 10% (interquartile range (IqR): 3–29, absolute range: 0–70) in the entire series. Fourteen (47%) metastases showed median MIB-1 expression of 28% (IqR: 5–40), whereas the six meningiomas included showed median MIB-1 expression of 11% (IqR: 9–16). Five were classified as WHO grade I, while one presented with WHO grade II. Ten neurinomas were included, with a median MIB-1 expression of 3% (IqR: 1–7). The detailed tumor stratification and proliferation index according to Ki67 expression is listed in Table 1. The median mitotic index according to the number of mitotic figures in 10 consecutive HPF within the group of meningiomas and neurinomas was 1 (IqR: 0–2), whereas it was 1 (range: 1–3) in WHO °I meningiomas and 11 in the case of WHO °II meningioma. In neurinomas, the median mitotic index was 0 (range: 0–3).
Ki67-defined proliferation index in metastases, neurinomas and meningiomas. Expression of MIB-1 is showed as median and absolute range.
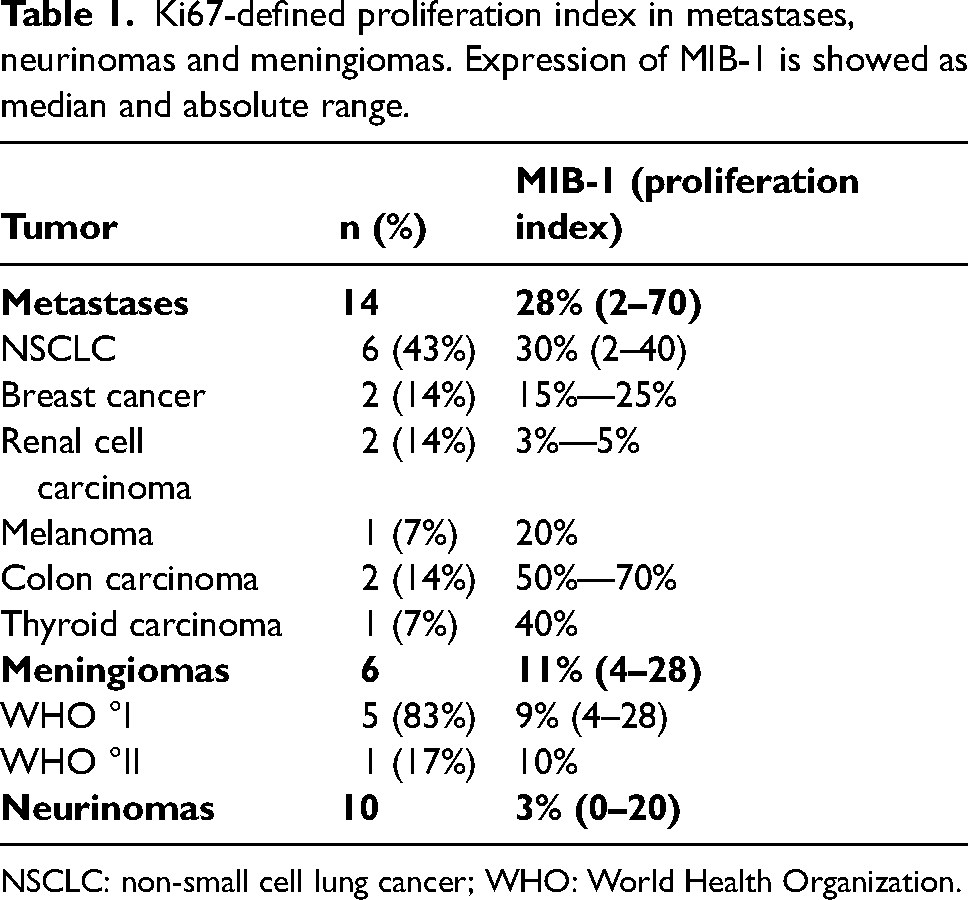
NSCLC: non-small cell lung cancer; WHO: World Health Organization.
In total, 8 samples (27%) showed no expression of Cx43, while 10 samples (33%) showed only minimal expression of Cx43. In 6 tumors (20%), moderate expression of Cx43 was found, whereas a strong signal of Cx43 was detected in another 6 tumors (20%). The Cx43 expression rates in our tumor groups are shown in Table 2 and examples in Figure 1. The Cx43 expression pattern was similar in all the types of tumors: cytoplasmatic without any specific nuclear staining. The Cx43 expression was distributed heterogeneously and the position of elevated Ki67 did not certainly match the localization of the elevated Cx43 expression in all types of tumors. High-power examples are provided in Figure 2.
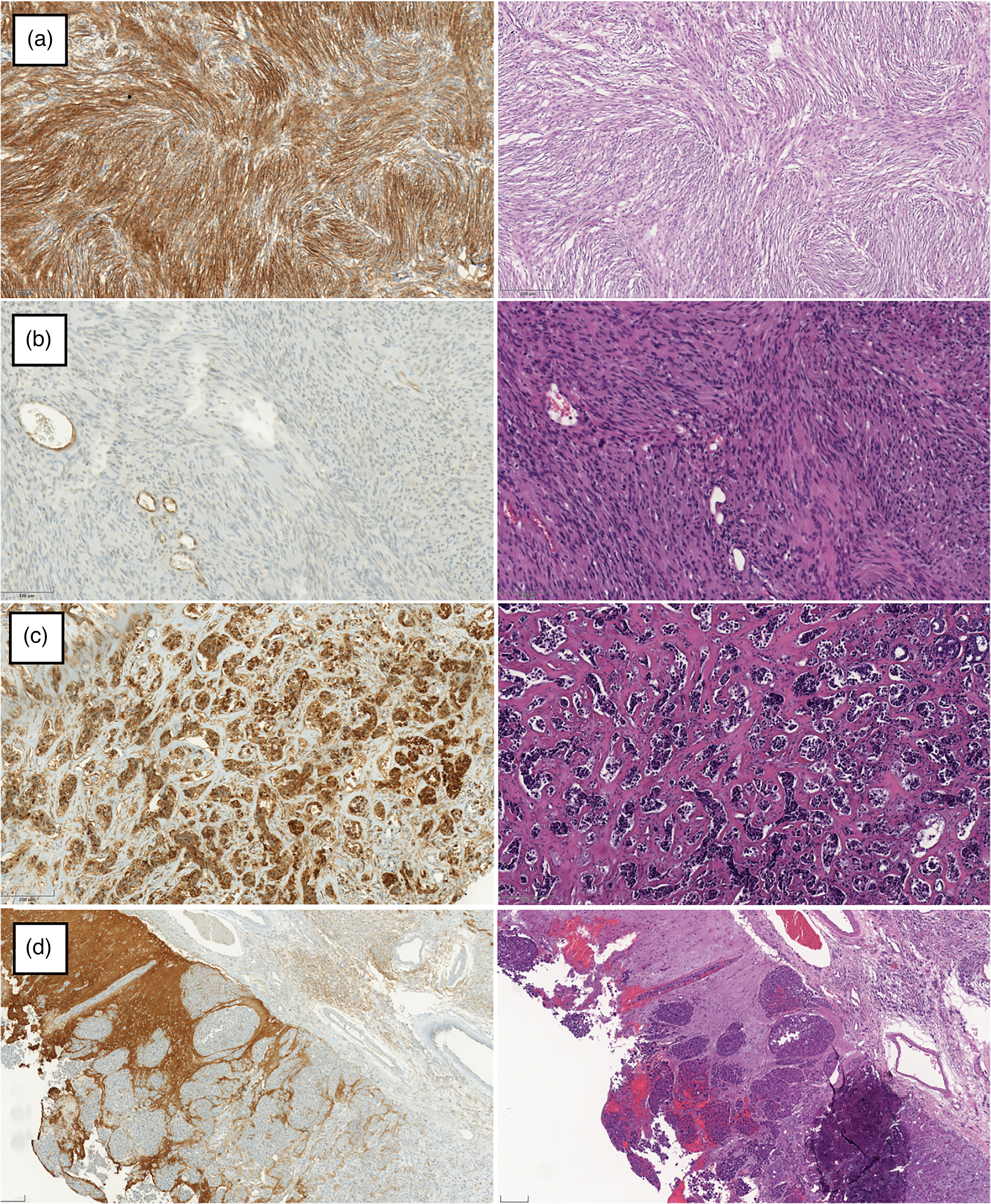
Cx43 IHC with corresponding HE-stained slides. (a) Meningioma WHO °I showing strong Cx43 expression, 200× magnification. (b) Vestibular schwannoma appears without relevant Cx43 expression in contrast to vascular endothelium, 400× magnification. (c) Metastasis of breast adenocarcinoma demonstrates strong Cx43 expression in contrast to fibrous trabeculae, 200× magnification. (d) Metastasis of NSCLC does not express Cx43 in contrast to invaded cortex, 200× magnification. (M8 Slide-scanner (PreciPoint, Germany).)
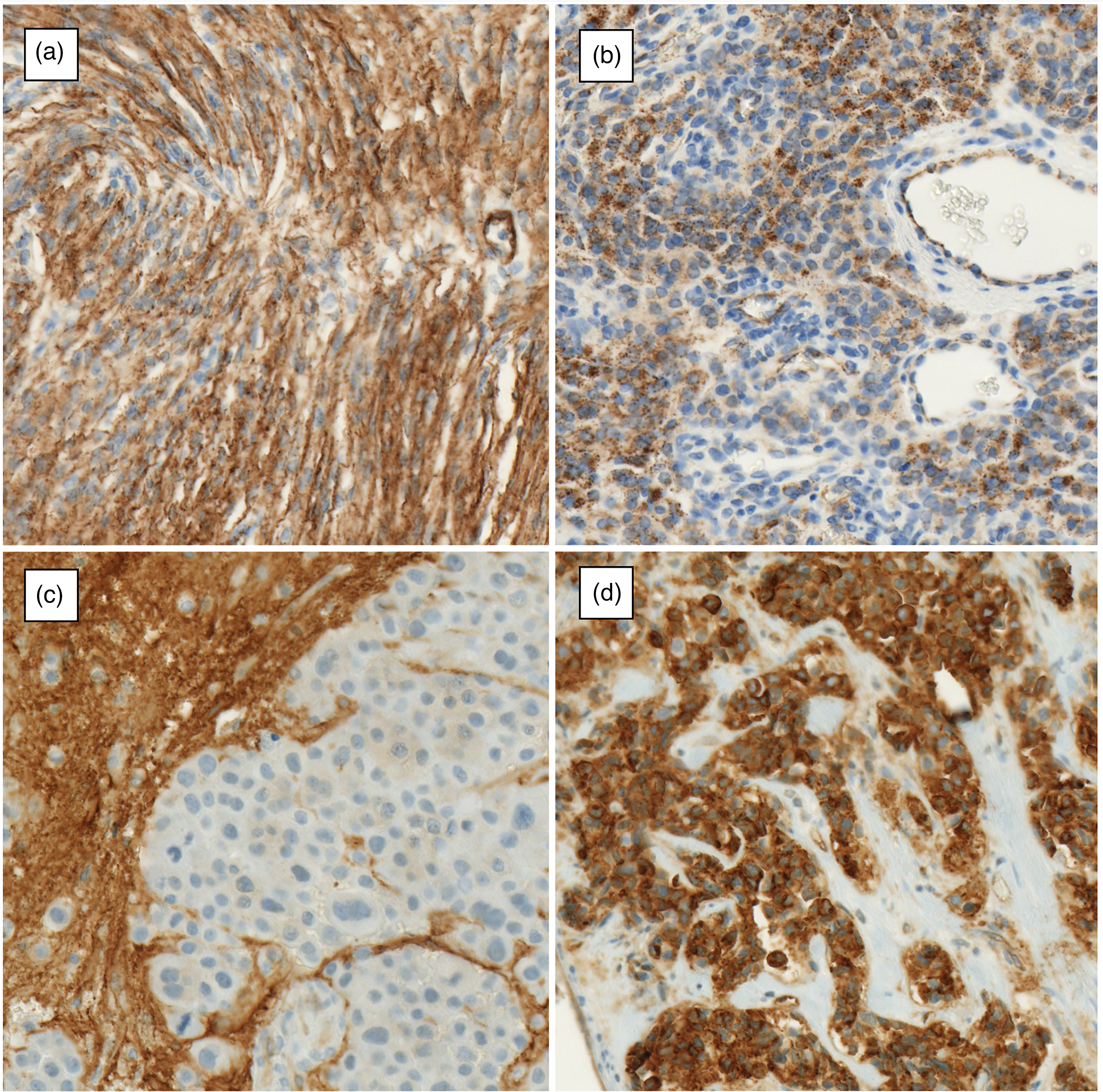
Cx43 IHC examples in a high-power field. (a) Meningioma WHO °I with strong Cx43 expression. (b) Meningioma WHO °I with heterogenous focal Cx43 expression. (c) Metastasis of colon carcinoma without Cx43 expression in contrast to specifically stained cortical tissue. (d) Metastasis of breast adenocarcinoma with strong Cx43 expression in contrast to fibrous trabeculae. Notably, in all cases, the cellular nuclei remain unstained. 600× magnification. (M8 Slide-scanner (PreciPoint, Germany).)
Expression of Cx43 in metastases, neurinomas, and meningiomas evaluated semi-quantitatively.

Cx43: Connexin-43.
In all tumors of our cohort, MIB-1 did not correlate with Cx43 expression (P = 0.964). Within the group of meningiomas and neurinomas, a statistically significant direct correlation between Ki67-defined proliferation index and expression of Cx43 could be identified (Spearman r = 0.53, P = 0.034). In metastases, no statistically significant correlation between Cx43 and MIB-1 expression could be found (P = 0.278). No statistical differences in the comparison of Cx43 expression between the group of meningiomas and neurinomas to metastases, independently of proliferation index, were identified (P = 0.128).
The mitotic index in meningiomas and neurinomas correlated with the Ki67-defined proliferation index (Spearman r = 0.74, P < 0.001). Whereas the mitotic index did not show statistically significant correlation with Cx43 expression in these tumors (P = 0.273).
Discussion
This overview study revealed a significant direct correlation between the expression of Cx43 and the Ki67-defined proliferation index in meningiomas and neurinomas. No significant correlation was found in metastases of extracranial origin.
Upregulated proliferation with high expression of MIB-1 was detected in metastasis and a low proliferation rate in meningiomas and neurinomas. This can be explained by the increased growth of malignant neoplasms and is in accordance with existing experience.34,35
Cx43 has a known important but controversial role in glial CNS tumors: gap junctional intercellular communication between glioma cells promotes resistance to chemotherapy and radiotherapy in lower-grade tumors.17,18 At the same time, Cx43 suppression leads to a failure of delay of G1 in mitosis and, thus, an advanced proliferation index, which is observed in high-grade gliomas.11,12,14–16 There is a correlation between Cx43 expression and proliferation rate in gliomas: decreased expression of Cx43 correlates with an increased proliferation.11,36
Data considering Cx43 expression in non-glial tumors of the CNS are limited. However, as these tumors originate from neural ectoderm like glial cells, which are expressing Cx43, 9 the gap junctional cell-to-cell connection should play a role. In meningiomas, the influence of Cx43 has been less understood. It has been mentioned that a lower expression of Cx43 has been associated with an increased risk of malignancy. 37 The influence of Cx43 on the development or progression of neurinomas remains largely unknown. 38
Regarding the role of Cx43 in metastasis, no clear answer has yet been found.18,39 In brain metastases, especially in breast cancer, Cx43 overexpression may facilitate distant progression. 39 Moreover, brain metastasis is accelerated via formation of carcinoma-astrocyte gap junctions, where Cx43 plays a fundamental role. 40 The resistance and progression of brain metastasis is further supported by normal astrocytes using direct intercellular gap connection. 41 The relationship between connexins and MIB-1 remains unclear in intracranial metastases, whereas some reports exist for a direct correlation in the primary lesion of, for example, lung tumors 42 or breast cancer. 43
In our study, a direct correlation between the expression of Cx43 and Ki67 was demonstrated for primary non-glial CNS neoplasms: meningiomas and neurinomas. Increased expression of Cx43 was associated with an increased proliferation index and, consequently, faster growth. Thus, these results stand in contrast to gliomas, where increased Cx43 expression correlated with decreased proliferation.11,36 It could be explained that the Cx43-determined oncoprotective effect of G1 delay in mitosis6,44–46 is possibly relevant only in high-grade gliomas due to their extreme proliferation rates. Moreover, in case of neural tumors with low malignancy, more Cx43 could be produced due to a longer interphase and an upregulated metabolic activity in oncological cells.
There was no correlation between Cx43 expression and a proliferation index in metastatic carcinomas of extracranial origin. In accordance with the correlation in meningiomas, neurinomas, and lower-grade gliomas, the relationship between Cx43 and proliferation may be limited to the tumors of neuroectodermal origin. Still, the interaction between metastasis and glial environment, where Cx43 expression plays a significant role, could stay important. It was described, that the resistance against oncologic therapy of brain metastasis is supported by normal brain astrocytes with direct intercellular gap connection. 41
In metastases of extracranial origin, Cx43 expression showed no correlation with Ki67. Even, if the role of Cx43 in neurinomas and meningiomas is still a matter of uncertainty, our data shows that, in contrast to gliomas, it does not play a significant role as a tumor suppressor but may promote more rapid growth. As Cx43-mediated gap intercellular communication protects lower-grade glioma cells against radio- and chemotherapy, it would be interesting to explore the same features in other neural tumors like neurinomas and meningiomas.
The sample size in our cohort remains limited. Even if the statistically significant correlation between Cx43 and Ki67 expression was achieved, further validation studies with larger and/or external cohorts is necessary. IHC as an evaluation method has limitations as it is examiner-dependent and semi-quantitative. To minimize these restrictions, we used the same protocol within the cohort, and the evaluation was performed by one experienced neuropathologist. Moreover, IHC allowed us to reveal the exact protein position in the tissue.
In conclusion, increased expression of Cx43 correlates with an increased Ki67-defined proliferation index in meningiomas and neurinomas. This supports the idea that Cx43 does not play a role as a tumor suppressor and may stimulate treatment resistance to the effect seen in low-grade gliomas; broad investigation of this aspect looks promising. In metastases, Cx43 expression had no influence on the proliferation index.
Footnotes
Author contributions:
Conceptualization, AK and CFF; methodology, AK, PM, and CFF; validation, AK, PM. and HF; formal analysis, AK and PM; investigation, AK and PM; resources, JK, PM, HF, KRB, CT, and CFF; data curation, AK and MD; writing—original draft preparation, AK; writing—review and editing, HF, CFF, and CT; supervision, CFF and CT; project administration, AK. All authors have read and agreed to the published version of the manuscript.
Data availability
The raw data was generated in authors’ institution. The data that support the findings of this study are available on reasonable request from the corresponding author. The data are not publicly available due their containing information that could compromise the privacy of research participants.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Institutional Review Board statement
The study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The database and tissue bank are approved by the ethics committee of Medical University of Innsbruck (AN5220 329/4.4).
