Abstract
Breast cancer is one of the most threatening malignant tumors in women worldwide; hence, investigators are continually performing novel research in this field. However, an accurate prediction of its prognosis and postoperative recovery remains difficult. The severity of breast cancer is patient-specific and affected by several health factors; thus, unknown mechanisms may affect its progression. This article analyzes existing literature on breast cancer, ranging from the discovery of ghrelin to its present use, and aims to provide a reference for future research into breast cancer mechanisms and treatment-plan improvement. Various parts of ghrelin have been associated with breast cancer by direct or indirect evidence. The ghrelin system may encompass the direction of expanding breast cancer treatment methods and prognostic indicators. Therefore, we compiled almost all studies on the relationship between the ghrelin system and breast cancer, including unacylated ghrelin, its GHRL gene, ghrelin O-acyltransferase, the receptor growth hormone secretagogue receptor, and several splice variants of ghrelin to lay the foundation for future research.
Introduction
Breast cancer is one of the most threatening malignant tumors in women worldwide, and causes more than 2.1 million cases every year. 1 In 2019, there were an estimated 270,000 breast cancer cases and over 40,000 deaths in the United States. 2 The diagnosis and treatment of breast cancer remain a global dilemma. From 2011 to 2017 in the United States, the relative survival rates of patients with local, regional, distant, and unstaged breast cancer following diagnosis were approximately 99%, 85.8%, 29%, and 57.8%, respectively. 3
Ghrelin is a natural ligand of the growth hormone secretagogue receptor type 1a (GHSR1a), which can regulate the secretion of growth hormone (GH).4–6 This 28-amino-acid peptide stored in the secretory vesicles of the endocrine cells is produced by processing 117 amino acids by the propeptide proghrelin.4–7 Ghrelin is predominantly secreted in the stomach, despite its occurrence in several tissues and organs, including the kidneys, heart, and lungs.8–11The ghrelin system is a complex that consists of several splice variants, receptors, genes, and functions in the proteolytic process of pre-propeptide to produce different hormones 12 or specific post-transcriptional modifications 13 (Figure 1).
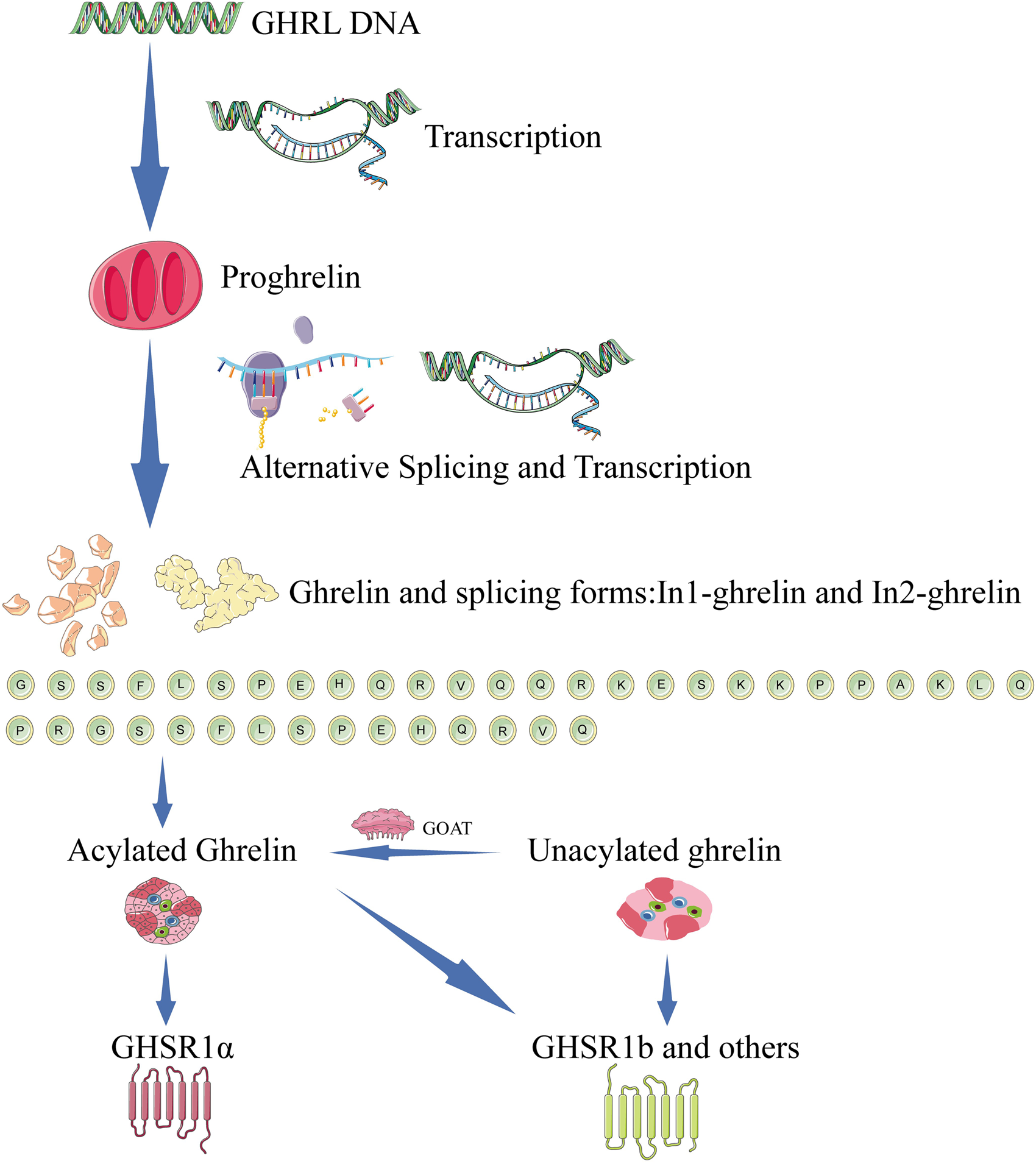
Proghrelin is transcribed from the GHRL gene, and spliced into ghrelin and its splice variants. Under the action of the GOAT enzyme, ghrelin changes from unacylated ghrelin to acylated ghrelin, which plays a physiological role. Unacylated ghrelin does not act on GHSR1a; however, ghrelin itself acts on GHSR1a and other receptors.
In recent years, numerous studies have focused on ghrelin and breast cancer. A study on the survival of patients with breast cancer through exercise and diet demonstrated increased ghrelin levels in survivors, 14 thus illustrating the potential of ghrelin as a prognostic marker for breast cancer. Aromatase is expressed in adipose stromal cells, and is an important factor in inducing postmenopausal breast cancer. 15 Ghrelin reduces intracellular cAMP levels, thereby acting on the alternative receptors of adipose stromal cells.16,17 In addition, ghrelin and unacylated-ghrelin (UAG) inhibit the expression of aromatase in adipose stromal cells,16,18–20 which may be useful for the treatment of estrogen-dependent breast cancer. A novel discovery with the potential of a prognostic marker and drug development is undoubtedly valuable for patients with breast cancer. However, some studies present contradictory results: one study demonstrated that ghrelin can inhibit cancer cell prolieferation, 21 whereas another mentioned that it can accelerate the process. 12 In other words, the effect of ghrelin may be bidirectional and complex, considering the ghrelin system includes unacylated forms, receptors, splice variants, and genes with multiple functions, in addition to acylated ghrelin involved in daily digestive and feeding activities. Therefore, this review aimed to elucidate the differences in current research on the relationship between ghrelin and breast cancer. We intended to comprehensively present the existing results of ghrelin to determine the complex clues and to explore the research of breast cancer and ghrelin.
UAG
The physiological level of UAG, which is more abundant than ghrelin in circulation, ranges between 0.1 nM and 0.5 nM. 22 The UAG has biological activity 23 and does not bind to GHSR1a at physiological concentrations; nonetheless, it regulates the differentiation of adipocytes, reduces adipose tissue inflammation, inhibits skeletal muscle atrophy, promotes glucose uptake, and stimulates osteoblast proliferation and other biological effects. 22 In addition, it prevents glucose metabolism disorders in obese mouse models. 24 Notably, a study demonstrated that UAG inhibits aromatase expression, 16 whereas others mentioned that it inhibits the levels of macrophage-derived factors, which can promote the increase of aromatase expression in patients with obesity, thus indirectly causing breast cancer. 18 UAG may promote aromatase expression; however, its effect is approximately 1000 times weaker than that of ghrelin, unlike the effect of ghrelin on GHSR1a.4,25,26 It may act through an unknown alternative growth protein receptor.16,22,27 Moreover, a study using GHSR1a knockout mice indicated that the effects of UAG are independent of GHSR1a,28,29 thereby suggesting the involvement of other receptors. Recently, a study demonstrated that UAG is a potent suppressor of breast cancer cell growth, independent of the effects on the stroma, and provides a novel mechanism of action via the activation of Gai, the suppression of cAMP production, and the inhibition of MAPK and Akt signaling. 30 In addition, UAG and its analogues are potential drugs for breast cancer treatment. Further research into the different ghrelin receptors may elucidate the mechanism underlying the impact of ghrelin and UAG on breast cancer.
The ghrelin receptor in breast cancer
GHSR
The GHSR was discovered by Howard et al. in 1996. 27 It belongs to the G-protein coupling group, and acts as a binding growth hormone secretagogue found in the pituitary gland and hypothalamus receptor.
The GHSR is complex, comprising GHSR1a and GHSR1b, and some of its functions, remains unknown. It is expressed in renal cell carcinoma and lung, colorectal, and ovarian cancers, particularly breast cancer.11,21,26,31–34 GHSR1a and GHSR1b appear to be insufficiently directly related to breast cancer in existing studies; however, based on the high expression of GHSR in tumors, we have listed relevant studies with the aim of inspiring readers and conducting follow-up studies. An improved understanding of GHSR1a and GHSR1b may possibly clarify the relationship between GHSR and breast cancer.
GHSR1a
GHSR1a is a G protein-coupled receptor with seven transmembrane domains, and leads to the release of intracellular Ca2+. It is a molecularly defined receptor with a high affinity for ghrelin.25,27,35 In addition, GHSR1a enables ghrelin to act on the hypothalamus.
GHSR1a exists among endocrine and non-endocrine tumors. 34 It has a wide functional range, including regulating GH expression in pituitary tumors and prostate cancer, 36 breast hyperplasia, 37 and follicular thyroid carcinoma. 38 Ghrelin mediates the effects of hypothalamus through GHSR1a. 28 However, some studies have demonstrated that GHSR1a does not bind to UAG. Moreover, GHSR1a does not exist in cells, such as Michigan Cancer Foundation-7 (MCF-7) and MDA-MB-231 cells and other breast cancer cell lines,21,22 which respond to UAG in vitro. 21 The efficacy of UAG binding to GHSR1a is approximately 1000 times weaker than that of ghrelin.4,25,26 Also, ghrelin and de-acylated ghrelin inhibit aromatase expression in the absence of GHSR1a, thereby indicating that ghrelin may act through an unknown alternative receptor in the breast. 16 These studies suggest that GHSR1a does not have an obvious direct relationship with breast cancer; nonetheless, the presence of an indirect effect is unknown.
GHSR1b
GHSR1b is a GHSR isoform previously considered to have no endocrine function in the early stage, and cannot bind to any secreted GH. Moreover, it has only five transmembrane domains. 35 GHSR1b is not activated by ghrelin; however, its expression has been recently identified in several tumors, including prostate, adrenal, and lung cancers. The expression level of GHSR1b is usually higher than that of GHSR1a.10,36,39–41 GHSR1b plays a neuromedin U-receptor role in lung cancer by heterodimerizing with neurotensin receptor 1. 41 These studies contrast previous conclusions that GHSR1b does not play a role in cancer. Studies on GHSR1a-knockout mice have demonstrated that ghrelin and de-acylated ghrelin exert GHSR1a-independent effects,29,42,43 thus suggesting the involvement and role of other receptors in breast cancer. 37 The potential pathophysiological role of GHSR1b overexpression is unknown. GHSR1b does not bind to ghrelin; thus, it may primarily act as a modulator of other G protein-coupled receptors through heterodimerization, besides increasing the internalization of GHSR1a.41,44 Furthermore, the upregulation of GHSR1b level is positively correlated with In1-ghrelin expression. 45 In other words, GHSR1b may play a significant role in breast cancer, despite unknown direct mechanisms.
Ghrelin variants
In1-ghrelin
In1-ghrelin variants have always received special attention of all ghrelin gene splicing products. 45 It is a splicing-derived variant produced by the retention of intron 1, and is highly correlated with ghrelin O-acyltransferase (GOAT) messenger RNA (mRNA) levels in human tissues. 45 In1-ghrelin variants are overexpressed in the pathology of breast cancer, 45 plus pituitary 46 and neuroendocrine tumors, 47 and appear to be related to the malignant degree of tumors. Notably, a study on male breast cancer mentioned that In1-ghrelin was overexpressed in the tissues and promoted the basic proliferation of breast cancer cells; that is, MDA-MB-231. 47 In1-ghrelin mRNA expression levels are correlated with breast cancer, thus suggesting the association among Ki-67, cyclin D3, and other proliferation markers. 48 The overexpression of In1-ghrelin induces increased cell viability and inhibits the apoptotic process,46,49 besides increasing the hormones of the pituitary gland and neuroendocrine tumor secretion.16,17
In1-ghrelin expression is associated with lymph node metastasis and lower disease-free survival in patients with breast cancer. Moreover, In1-ghrelin promotes cell proliferation and migration, whereas its down-regulation reduces cell proliferation and migration. 48 Another study 45 reported on no difference in the expression level of ghrelin between normal breast and breast cancer tissues; nevertheless, In1-ghrelin level in breast cancer tissues was eight times than that in normal breast tissues. In1-ghrelin promotes breast cancer by increasing mammosphere formation owing to increased mRNA levels of Jagged 1. 50 The Jagged Canonical Notch Ligand 1 gene is one of the important genes that causes breast cancer. 47 Simultaneously, In1-ghrelin activates β-catenin and is the primary activator of Notch signaling. 50 It is expressed in MCF-7 cells and causes changes in mammospheres that induce breast cancer.
In2-ghrelin
Researchers have identified a novel splice variant of pre-proghrelin, termed In2-ghrelin (intron 2-implicit), which is a novel exon sequence from intron 2 of the ghrelin gene.51,52 This transcript is overexpressed in 22Rv1 and lymph node carcinoma (LNCaP) prostate cancer cell lines, and plays a role in the progression of prostate cancer. 53 Notably, the LNCaP cell line derived from lymph node metastasis expresses high levels of In2-ghrelin. 54
The strong link between male breast cancer survival rate has been demonstrated.47,55 Despite weak evidence, it is plausible to suspect that In2-ghrelin plays a role in breast cancer; however, it requires further confirmation. Such evidence will expand an understanding of the relationship between the ghrelin family and breast cancer.
GOAT
GOAT is a membrane-bound enzyme that specifically attaches an n-octanoyl fatty acid side chain off the serine residue in the position 3 of ghrelin.5,6,56 Membrane Bound O-Acyltransferase Domain Containing 4 encodes the GOAT enzyme, which can acylate UAG,56–58 thereby exerting endocrine effects. Moreover, it is associated with obesity and insulin resistance, 4 which are substantial risk factors for breast cancer.56,57,59 The risk of obesity and breast cancer is complex, which is related to differences reported by menopausal status, tumor hormone receptor status, and race.
Physicians can use GOAT as an important target for the treatment of obesity and insulin resistance.
57
However, there are few studies on the direct correlation between GOAT and breast cancer. Currently, GOAT only demonstrates an indirect value for indicating the prognosis of breast cancer. However, compared with the monitoring model and considering the economic costs, it has limited value in assessing the prognosis of breast cancer.
Ghrelin gene (GHRL)
GHRL is a pleiotropic hormone predominantly produced in the stomach. It is an endogenous ligand of GHSR. Its primary function is to stimulate the production of GH and food intake. Human GH is encoded in the GHRL gene. Its transcription produces an immature propeptide of 117 residues, and can be acylated by GOAT. Moreover, it can be processed by prohormone-converting enzymes (PC1/32and PC2) 60 to produce acyl ghrelin or des-acylated ghrelin.13,61
GHRL stimulates the production of GH and increases the appetite by activating GHSR-1a in the hypothalamus. Moreover, it plays a role in cell proliferation;16,61–63 this combined function of stimulating GH secretion in the anterior pituitary makes GHRL a potential factor for tumorigenesis. 61 Research on premenopausal women of low native descent in the United States confirmed that GHRL increases the risk of breast cancer.61,62,64,65 Moreover, it increases the intake of foods unrelated to GHSR. In addition to orexigenic functions, GHRL promotes cell proliferation, 63 which triggers tumorigenesis, together with the effects of stimulating GH secretion in the anterior pituitary. 66
GHRL polymorphism is closely associated with the risk of breast cancer. Single nucleotide polymorphisms of the GHRL gene are associated with increased cancer risk. 67 A study involving more than 1000 volunteers from Europe demonstrated that the rs2075356GHRL polymorphism can reduce the risk of breast cancer. 67 Another study mentioned that the 4684677GHRL polymorphism may increase the risk of breast cancer in carriers. 68 In addition, insulin levels increased and obesity contributed to the increased risk of breast cancer caused by GHRL polymorphisms. 67
Notably, GHRL is related to the incidence and poor prognosis of breast cancer. Patients with high GHRL expression display low survival rates.65,69 The mechanism by which GHRL promotes the occurrence of cancer and reduces the survival rate has been studied in other types of cancers. GHRL is highly expressed in prostate cancer and initiates a cross-talk with the MAPK signaling cascade. Subsequently, it activates the ERK1/2MAPK pathway. Moreover, it promotes cell proliferation through the p38 (MAPK14) pathway. 68 The components of MAPK pathways are not directly expressed in breast cancer; however, they are related to the survival rate of some patient populations, 70 thus indicating the effect of GHRL on the survival rate. However, there is a lack of reliable evidence for the association between GHRL polymorphism and the survival rate of patients with breast cancer; existing studies only have a small sample size. Furthermore, GHRL polymorphisms are associated with obesity, 67 which contributes to the decreased survival rate of patients with breast cancer,71–73 despite these studies being controversial. 74 Concurrently, a meta-analysis study demonstrated that GHRL may be an indicator of the risk of breast cancer, despite a weak association. However, researchers should increase the sample size and conduct prospective studies to confirm this relationship. 75
Discussion
Breast cancer has become one of the leading causes of death in women, and its incidence is gradually increasing. There is a lack of sufficient comprehensive scientific methods for physicians assessing patient prognosis to completely understand their condition and the involved molecular mechanisms. This has implications for the development of accurate and patient-specific treatment usually required in breast cancer cases.
Researchers are gradually discovering the relationship between ghrelin and cancer. Recently, a study demonstrated that ghrelin promotes colon cancer by inducing HT-29 cell proliferation through the GHSR and Ras/PI3K/AKT/mTOR pathways. 76 The study confirmed ghrelin as the etiopathogenic factor of cancer. The ghrelin system is complex and evidence for its role in cancer is contradictory—particularly its involvement in breast cancer (Table 1).
List of evidences for the relationship between ghrelin and breast cancer.

GHSR1A: growth hormone secretagogue receptor type 1a; GHSR1B: growth hormone secretagogue receptor type 1b; GOAT: ghrelin O-acyltransferase; In2-ghrelin: intron 2-implicit; UAG: unacylated-ghrelin.
The research results in Table 1 separately summarize the exact evidence, the relevant possible clues, and the properties of each part of the ghrelin system. UAG exerts a positive effect on breast cancer. GHSR1a is expressed in numerous tumors and is related to breast hyperplasia, despite the lack of further evidence. GHSR1b acts as a possible receptor of UAG, whereas it up-regulates In1; therefore, ghrelin expression is neutral. In1-ghrelin is negative. In2-ghrelin is associated with male breast cancer, but this is understudied. GOAT is associated with negative prognostic factors in breast cancer, obesity, and insulin resistance; however, researchers have not demonstrated its direct relationship with breast cancer. The GHRL gene has multi-faceted roles, and those of different genes may be opposite.
GHRL at the genetic level, ghrelin and UAG as hormone products, GHSR1a and GHSR1b in the ghrelin receptor, or In1-ghrelin and In2-ghrelin as ghrelin splice variants exert positive and negative effects on breast cancer. This necessitates future research to clarify the mentioned association, thus providing a novel method for the predictive diagnostic and prognostic assessment of breast cancer. Moreover, it will suggest a novel way to investigate the mechanism of breast cancer. We have tabulated the research involving the ghrelin system to elucidate our findings. This review will be a promising avenue for an accurate and targeted treatment in patients with breast cancer worldwide.
Breast cancer is a heterogeneous disease, and consists of several specific subtypes. Unfortunately, existing research has not investigated the use of ghrelin in patients with specific types of breast cancer. This may be attributed to three reasons. First, the research on ghrelin and breast cancer is in its infancy, and current research generally acknowledges the relationship between hormones and breast cancer. The prognosis of patients with cancer is related to a part of its pathogenesis. Researchers have not sufficiently investigated ghrelin. Second, affected by factors such as the number of cases included in different regions and races, existing studies have not yet reflected the classification rules related to ghrelin. Third, investigating the relationship between ghrelin and breast cancer subtypes will substantially promote the development of breast cancer screening and prognostic detection methods. Moreover, it will provide directions for precision medicine for patients with different types of breast cancer.
Conclusion
The ghrelin system can regulate breast cancer cell proliferation and it plays a significant role in some key pathways of breast cancer. However, the detailed mechanism is unclear and warrants further investigation. This study had limitations, such as the amount of research, the number of study samples, and the level of insufficient evidence. Researchers have investigated the exact influence of ghrelin on breast cancer pathways only from the PI3K/AKT/mTOR signaling pathways. Other pathological mechanisms, such as oxidative stress and inflammatory mechanism, were unclear. A sufficient number of studies have demonstrated the close association between the ghrelin system and breast cancer; nonetheless, the role of specific parts of ghrelin in breast cancer is controversial. Thus, follow-up studies necessitate larger sample sizes and multi-center clinical research to make observations. In addition, existing studies have limited understanding of the ghrelin system, and several mechanisms cannot be explained. In addition, the factors that ghrelin affects breast cancer may not stop there. There are presumably undiscovered ghrelin receptors or splice variants involved in the course of breast cancer.
Footnotes
Author contributions
YDC, QC, and XKH designed the study. LW and QW contributed to the collection and the analysis of the literature. LFL and LSS check all statistical analyses and made some corrections. YDC and LW interpreted the data. YDC and XKH wrote the initial draft of the manuscript. QW and QC edited the manuscript. All authors reviewed the final manuscript. YDC, XKH, and LW contributed equally to the article.
Data availability
All data used in this review are fully available in the public domain.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclosure
The authors have no conflicts of interest to declare.
Funding
The study was partly supported by grants from the Chengdu Science and Technology Project (No 2019-YF09-00094-SN) and Medical Service and Guarantee Capacity Improvement Subsidy Funds (Major and difficult diseases-No CYW2019079).
Supplemental material
Supplemental material for this article is available online.
